A transmission-virulence evolutionary trade-off explains attenuation of HIV-1 in Uganda
Figures
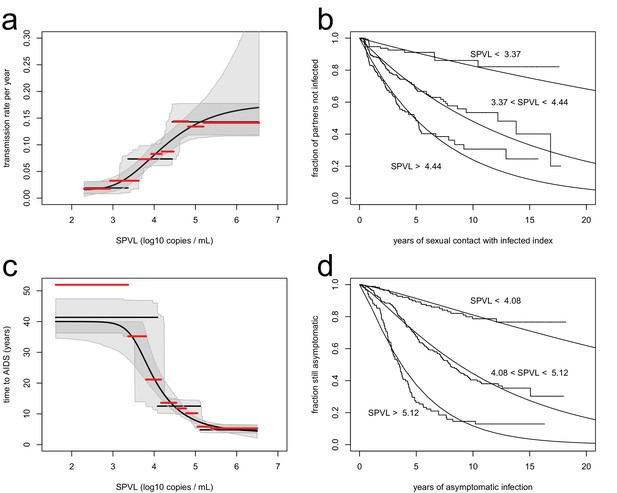
Inferred relationships between SPVL and transmission rate (a, b) and time to AIDS (b, d).
On the left panels, black lines show the maximum likelihood relationships and shaded areas the bootstrap 95% confidence intervals. Both the step function (horizontal lines) and the generalised Hill function (curved line) are shown. The red lines show a non-parametric estimation of the transmission rate (a) and the time to AIDS (c) curves, when the data is stratified by SPVL in 8 bins of equal size. The right panels show Kaplan Meier plots when the data is partitioned in three SPVL groups defined by the maximum likelihood relationships. There was good agreement between the data (step functions) and the maximum likelihood function (smooth functions).
-
Figure 1—source data 1
Data file for Figure 1.
(A) Model comparison for the transmission rate as a function of SPVL and other covariates, based on the Akaike Information Criterion. d.f. are the degrees of freedom, N is the sample size.(B) Model comparison for the time to AIDS as a function of SPVL and other covariates, based on the Akaike Information Criterion. d.f. are the degrees of freedom, N is the sample size.
- https://doi.org/10.7554/eLife.20492.005
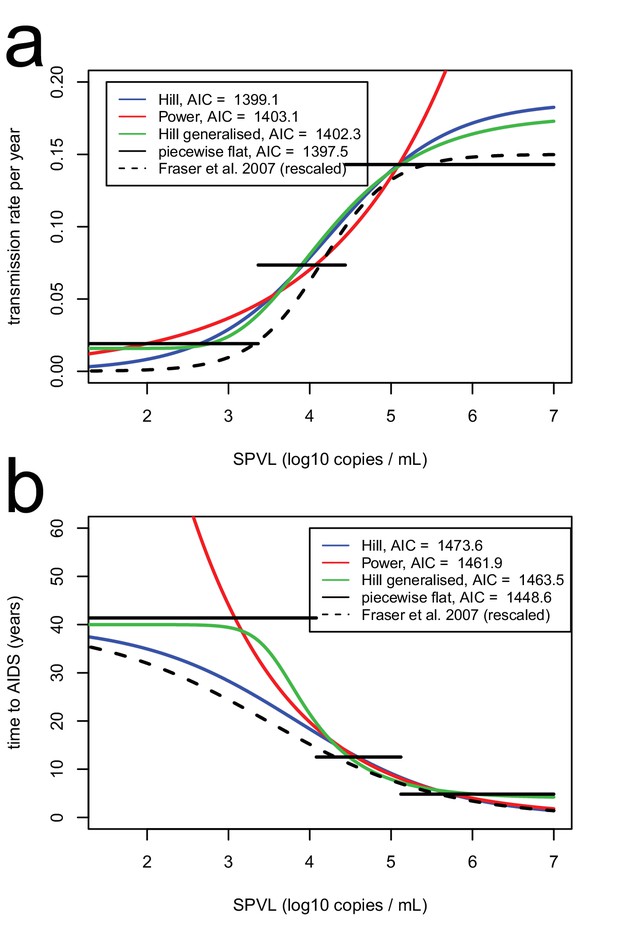
Functional forms for time to AIDS (a), and transmission rate (b), as a function of SPVL..
Functional forms include power (red), Hill (blue), generalised Hill (green), step function with three steps (black). The equivalent relationships as inferred in Fraser et al. (2007) are shown for comparison (black, dashed line).
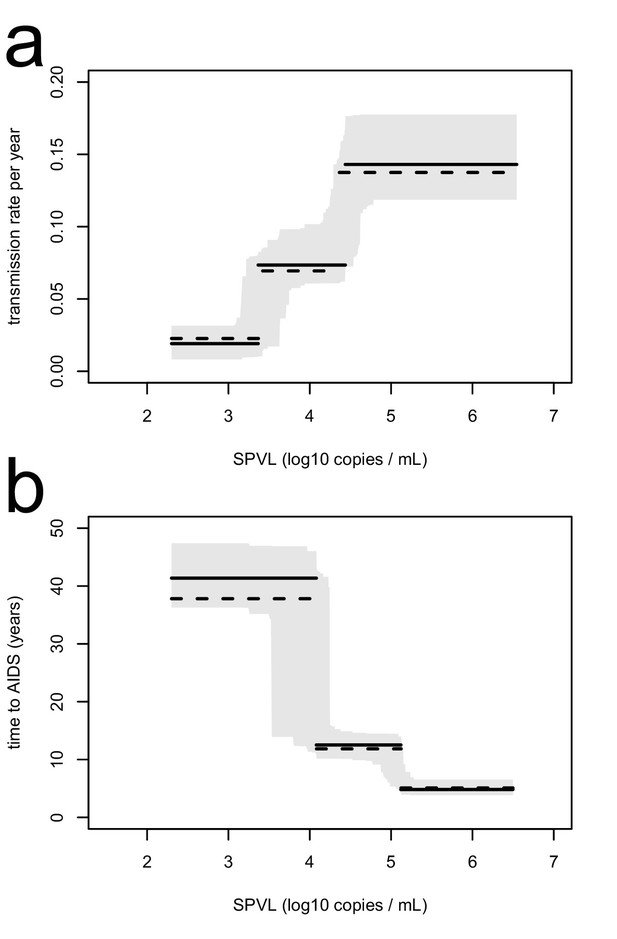
The inferred transmission rate (a) and time to AIDS (b), as a function of SPVL, are similar when removing undetectable SPVL values from the analysis.
In each panel, the maximum likelihood step function (black line) with bootstrap confidence intervals (grey) is shown together with the maximum likelihood function when undetectable SPVL values are removed (dashed line).
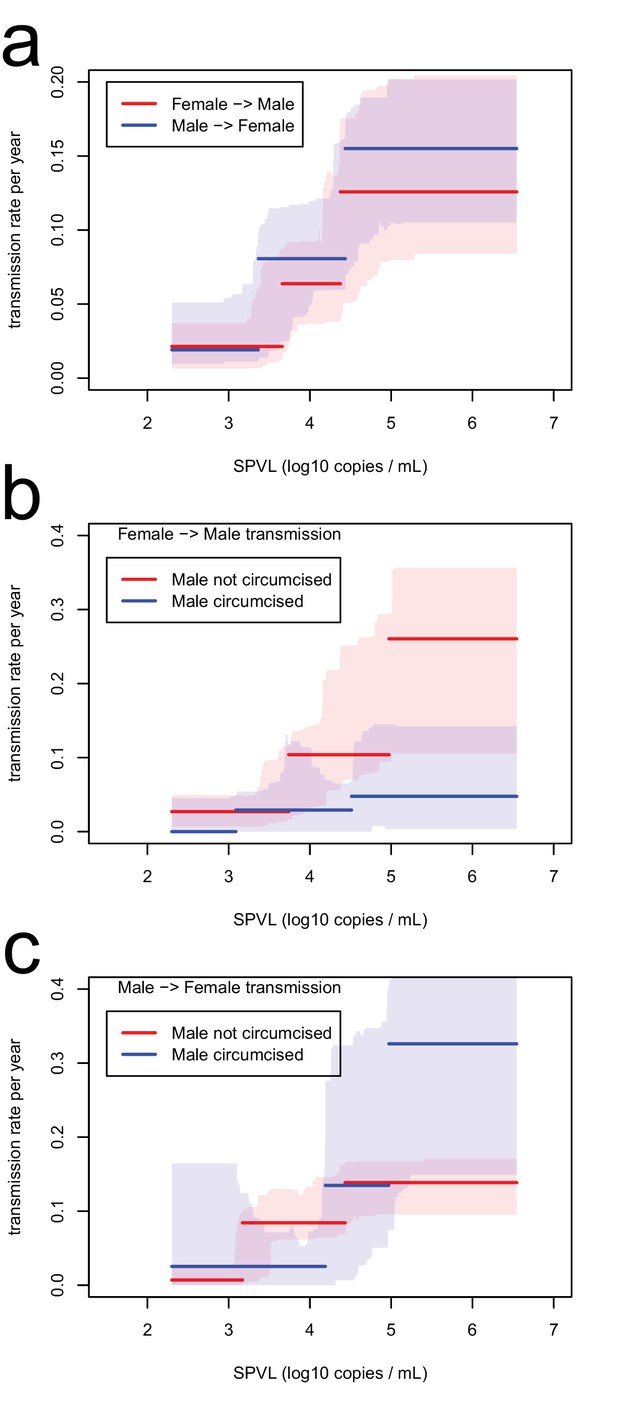
Transmission rate as a function of SPVL, stratified by gender (a) and by circumcision status (b, c).
Lines are the maximum likelihood functions; shaded intervals are the bootstrap confidence intervals.
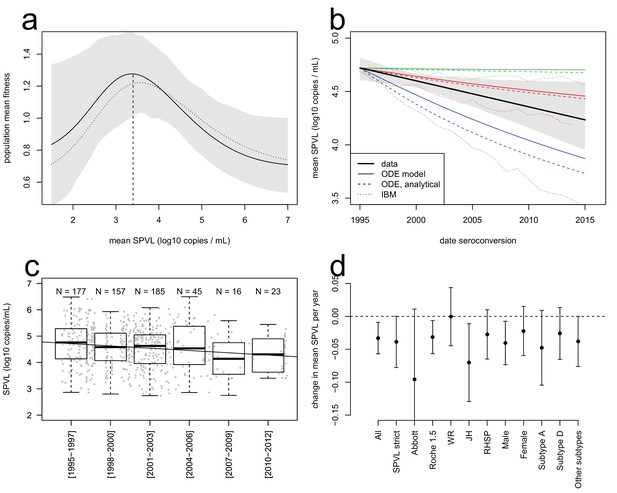
Evolutionary dynamics of SPVL.
(a), mean fitness of the viral population as a function of mean SPVL when transmission and time to AIDS are fitted as step functions (solid line; shaded area shows the 95% C.I.) or generalised Hill functions (dashed line). (b), evolutionary predictions for the temporal dynamics of mean SPVL given by the ODE model (thin solid and dashed lines), and the stochastic IBM (dotted lines), under three scenarios for the impact of within-host evolution (biased mutation) on SPVL in blue (1, α = −0.47 log10 copies/mL), red (2, α = −0.093 log10 copies/mL) and green (3, α = +0.057 log10 copies/mL). The thick line is the data, showing the linear regression of SPVL on date of seroconversion, with 95% bootstrap confidence intervals shown as a shaded area. (c), distribution of SPVL in the population over time; grey points show the data, and the line is the unadjusted regression of SPVL over time. (d) coefficient of regression of SPVL over time in the adjusted linear regression, with confidence intervals, in various subsets of the data (Material and methods). All data; SPVL strict definition; SPVL measured with Abbott assay and Roche 1.5 assay; SPVL measured at Walter Reed (WR), John Hopkins (JH) and RHSP laboratories; SPVL in males and females; subtype A, subtype D, and other/unknown subtype viruses.
-
Figure 2—source data 1
Data file for Figure 2.
Summary of adjusted effects for the linear model explaining SPVL as a function of epidemiological covariates and date of seroconversion. The linear models included all the covariates listed. Effects significant in the whole dataset are in bold ‘p<0.1, *p<0.05, **p<0.01, ***p<0.001.
- https://doi.org/10.7554/eLife.20492.010
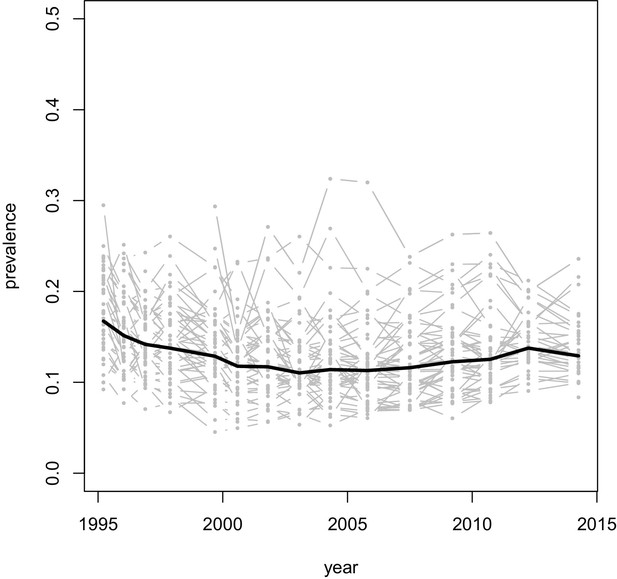
Prevalence of HIV over time, in the Rakai communities (gray lines), and on average across all communities (thick black line).
https://doi.org/10.7554/eLife.20492.011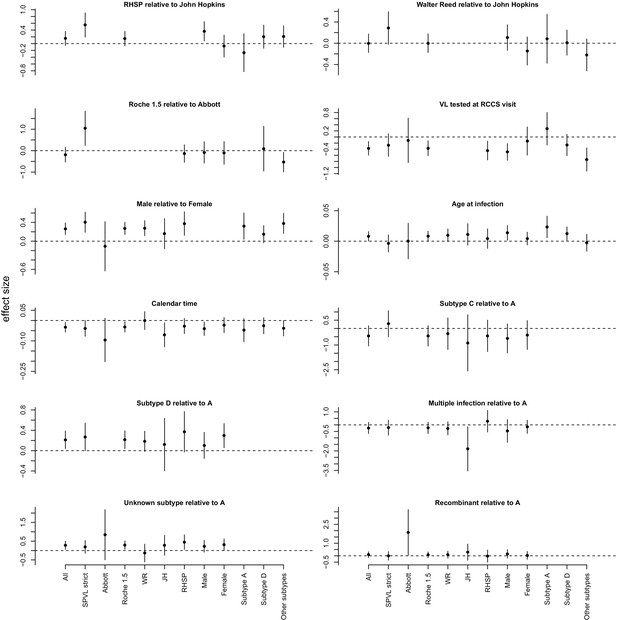
Summary of effects for the multivariate linear model explaining SPVL (Figure 2—source data 1).
This is shown for the full dataset ('All') and several subsets of data. Confidence intervals are determined assuming normality of the coefficients.
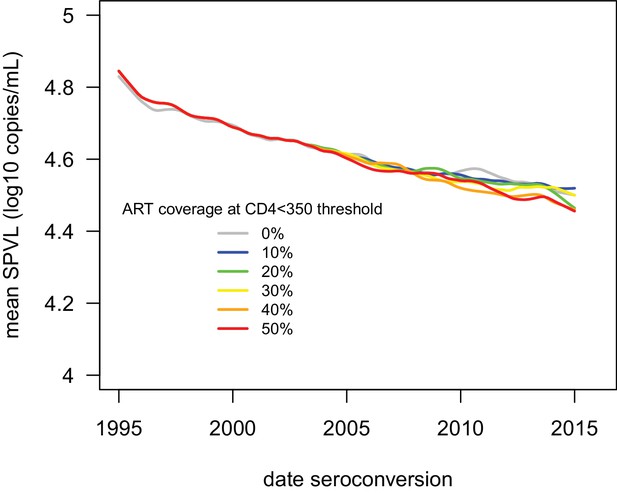
ART had little impact on the evolution of SPVL under the virulence-transmission trade-off.
Mean SPVL as a function of date of infection in the IBM including ART treatment, for heritability h2=0.36 and no biased mutation. ART treatment started in 2004. Individuals with a CD4 count below 350 cells/mm3 are eligible for treatment, and we varied coverage (the probability to receive treatment when eligible) from 0 to 50%. Treatment started 1 year after eligibility, and complete adherence was assumed. Upon treatment, the viral load is assumed to drop at 50 copies/mL.
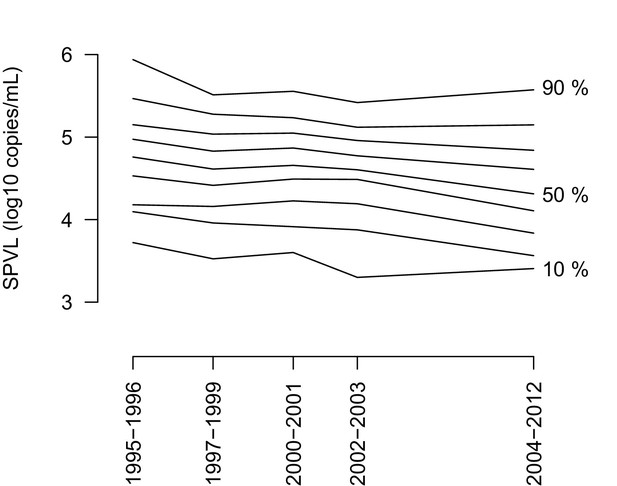
The entire distribution of SPVL shifts downwards with time.
The figure shows the 10% to 90% percentiles of the SPVL distribution as a function of time.
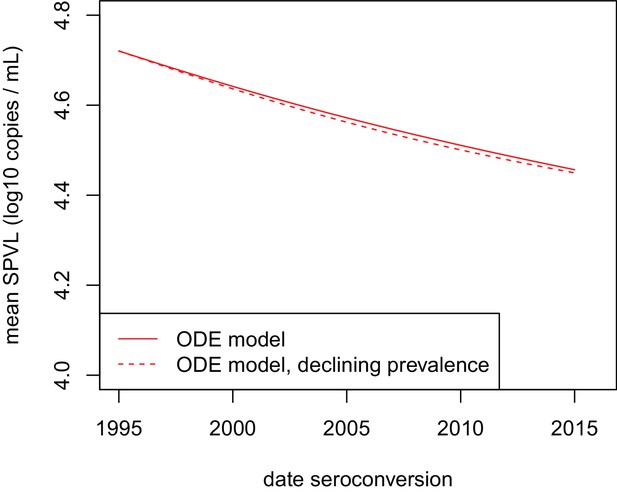
Declining prevalence had little impact on the evolution of SPVL under the virulence-transmission trade-off.
Mean SPVL as a function of date of seroconversion in the ODE model, for heritability h2=0.36 and biased mutation α = −0.093 log10 copies/mL (scenario 2). The model with approximately stable prevalence at 14% (red plain line, same as on Figure 2) is shown together with a simulation of the ODE model where initial prevalence is 20%, and the baseline transmission rate is set such that prevalence decreases to 5% over the 20 years of the simulation.
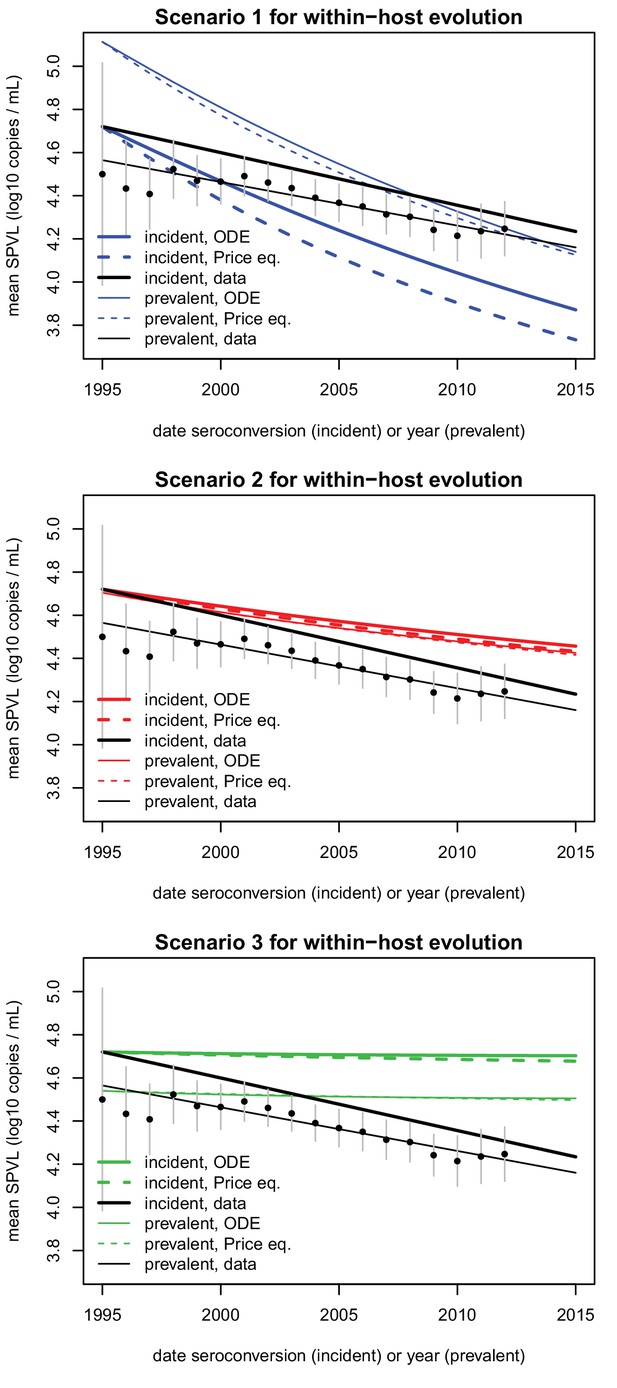
Comparison of SPVL trends in incident cases and prevalent cases.
Mean SPVL is shown as a function of date of seroconversion (for incident cases) and year (for prevalent cases). The data is shown in black, for incident cases (regression line, same as in Figure 2B) and prevalent cases (points are average SPVL each year with 95% CI, line is the regression line). Simulations of the ODE model and predictions from the Price equation show the mean genetic effect as coloured lines, for heritability h2=0.36 and three scenarios for biased mutation shown in the three panels.
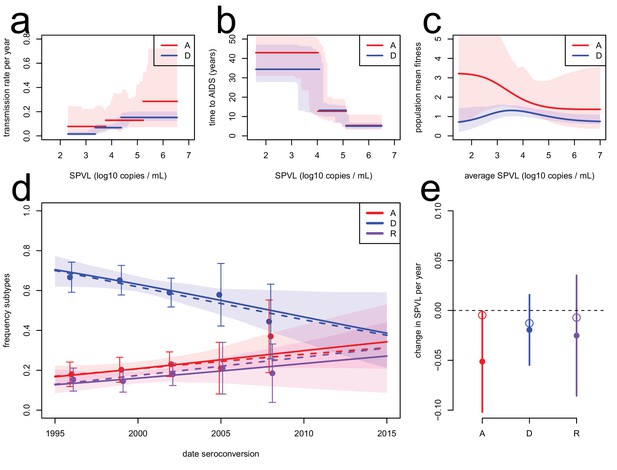
Subtype-specific evolutionary predictions.
Maximum likelihood functions for transmission (a) and time to AIDS (b) as a function of SPVL, stratified by subtype, for heritability h2=0.36 and biased mutation α = −0.093 (scenario 2). Shaded areas are bootstrap confidence intervals. (c) Predicted fitness function for subtype A (red) and subtype D (blue). (d) Subtype dynamics in the Rakai cohort as inferred by fitting a multinomial linear model with a 'date seroconversion' effect (solid lines, and confidence intervals as a shaded area; points show the actual frequency in the data, binned in five time categories, with confidence intervals), together with subtype dynamics predicted by the ODE model stratified by subtype (dashed lines). Recombination occurs upon co-infection and generates 'R' subtypes (purple). (e) Rates of evolution of SPVL per year within subtype, in the data (points, with 95% confidence intervals) and in the ODE simulation stratified by subtype (open circles).
Tables
Epidemiological and demographic characteristics of the HIV-1 incident cases in the Rakai cohort, used for the analysis of time trends in SPVL and for the analysis of time to AIDS. *Multiple subtypes (possibly dual infection) ** Recombinants, primarily A/D.
| Gender | N | Mean SPVL, [0.025; 0.975] quantiles |
|---|---|---|
| F | 362 | 4.3 [2.3; 5.85] |
| M | 285 | 4.54 [2.3; 6.03] |
| Date of infection | ||
| 1995–1999 | 269 | 4.47 [2.3; 6.01] |
| 2000–2004 | 297 | 4.46 [2.3; 5.83] |
| 2005–2009 | 54 | 3.98 [2.3; 5.77] |
| ≥2010 | 27 | 3.97 [2.2; 5.33] |
| HIV-1 subtype | ||
| A | 96 | 4.34 [2.78; 5.61] |
| C | 6 | 3.92 [3.42; 4.71] |
| D | 292 | 4.56 [2.62; 5.92] |
| M* | 14 | 3.99 [2.48; 5.35] |
| R** | 74 | 4.38 [2.33; 5.84] |
| Unknown | 165 | 4.22 [2.3; 6.03] |
| Age at infection | ||
| 15–19 | 61 | 4.17 [2.3; 5.51] |
| 20–29 | 327 | 4.43 [2.3; 5.97] |
| 30–39 | 182 | 4.45 [2.3; 5.88] |
| 40–49 | 67 | 4.43 [2.28; 6.09] |
| ≥50 | 10 | 4.05 [2.3; 5.94] |
Epidemiological and demographic characteristics of the infected individual in serodiscordant couples in the Rakai cohort, used for the analysis of time trends in SPVL and for the analysis of time to AIDS. ** Including recombinants, primarily A/D.
| Gender | N | Mean SPVL, [0.025; 0.975] quantiles |
|---|---|---|
| F | 324 | 3.99 [2.3; 5.61] |
| M | 493 | 4.23 [2.3; 5.85] |
| Date of infection | ||
| Unknown | 595 | 4.1 [2.3; 5.64] |
| 1995–1999 | 93 | 4.13 [2.3; 5.53] |
| 2000–2004 | 96 | 4.41 [2.3; 5.98] |
| 2005–2009 | 30 | 4.08 [2.3; 5.62] |
| ≥2010 | 3 | 3.19 [2.36; 3.77] |
| HIV-1 subtype | ||
| A | 54 | 4.11 [2.42; 5.72] |
| D | 430 | 4.27 [2.4; 5.77] |
| Other/Unknown** | 333 | 3.97 [2.3; 5.67] |
Summary of adjusted effects and p-values obtained by type II analysis of deviance for the logistic regression of 'detectable versus undetectable SPVL' over epidemiological covariates. n = 647. ‘p < 0.1, *p<0.05, ** p<0.01, ***p< 0.001.
| Factor | Effect size | p-value |
|---|---|---|
| Intercept | 0.217 | 0.002 ** |
| John Hopkins | Reference | - |
| RHSP | −0.029 | 0.354 |
| Walter Reed | −0.041 | 0.117 |
| Abbott | Reference | - |
| Roche 1.5 | −0.158 | 0.001 ** |
| Not RCCS visit | Reference | - |
| RCCS visit | 0.01 | 0.747 |
| Female | Reference | - |
| Male | −0.021 | 0.261 |
| Age | 0.001 | 0.378 |
| Date seroconversion | −0.005 | 0.149 |
| Subtype A | Reference | - |
| Subtype C | −0.012 | 0.899 |
| Subtype D | −0.011 | 0.693 |
| Recombinant | 0.002 | 0.948 |
| Dual infections | 0.051 | 0.434 |
Additional files
-
Supplementary file 1
Individual viral load trajectories within patients for 603 incident cases with a SPVL value (when undetectable viral load were removed from the SPVL calculation).
Points are viral load values, shown as solid bullets when used for the SPVL calculation, and open circles otherwise. The vertical red line is the mid-point between last negative test and first positive test. The vertical light green line is the date ART started. The vertical dark green line is the date of first self-reported ART. The horizontal black line is the SPVL value. This data relates to Figure 2.
- https://doi.org/10.7554/eLife.20492.018
-
Reporting standard 1
Summary table of statistical tests.
- https://doi.org/10.7554/eLife.20492.019





