CCR5 is a suppressor for cortical plasticity and hippocampal learning and memory
Figures
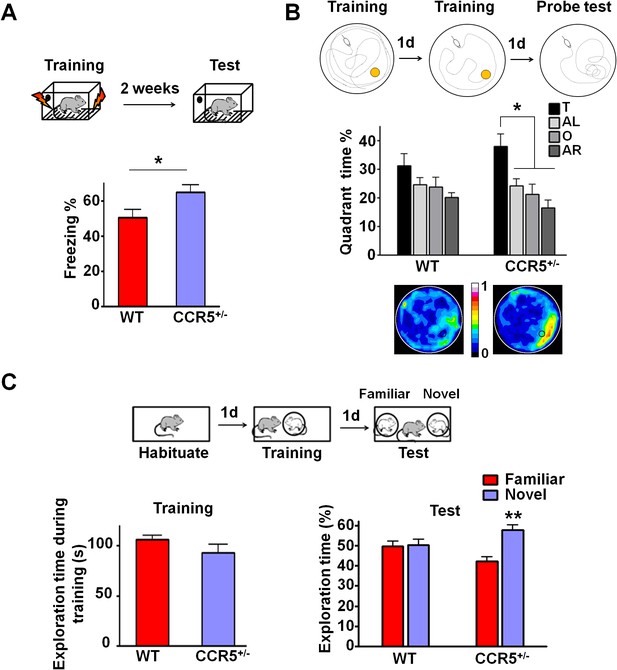
Ccr5+/- mice show enhanced memory in multiple memory tasks.
(A) In a fear conditioning test given two weeks after training, Ccr5+/- mice showed enhanced contextual memory (WT n = 11, Ccr5+/- n = 11; *p<0.05, Student’s t-test). (B) In a water maze probe test given after two days of training, Ccr5+/- mice spent significantly more time in the target quadrant than in the other three quadrants of the water maze. In contrast, WT mice did not search selectively for the platform (WT n = 14, Ccr5+/- n = 16; *p<0.05, Two-way ANOVA with repeated measure). Heat maps below the bar graphs show the combined traces of the mice from each group during the probe test. (C) There was no difference between WT mice and Ccr5+/- mice in social training. In the social recognition test given 24 hr after training (7 min, a training time chosen to undertrain WT mice), Ccr5+/- mice, but not WT mice, spent more time exploring the novel OVX mouse (WT n = 14, Ccr5+/- n = 19; **p<0.01, one sample t-test compared to 50%). Error bars indicate SEM.
-
Figure 1—source data 1
Full names and MGI accession # of the 148 mutant strains in the reverse genetic memory screen (*Jax stock#: 370200; **Jax stock#: 370202).
- https://doi.org/10.7554/eLife.20985.003
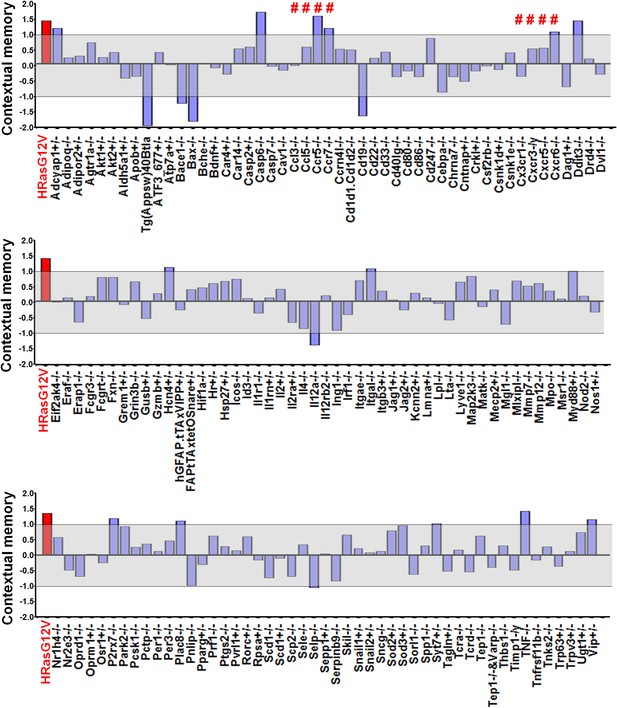
Identification of Ccr5 knockout mice in a reverse genetic memory screen.
Combined mean Z scores for freezing and suppression ratios of 148 mutant strains screened for remote contextual memory phenotypes. For each mutant strain screening experiment, 8–10 mice (Wild-type group: n = 8–10, Mutant group: n = 8–10, half male and half female) were used. Mice were trained with three tone-shock pairs and their contextual memory was tested two weeks after training. The gray shaded area denotes values that are within ±1.0 standard deviation from controls. Z scores of 8 chemokine/chemokine receptor mutant strains were marked by #. Z scores for HRasG12V mice, a strain already known to have enhanced memory, were shown in red for comparison.
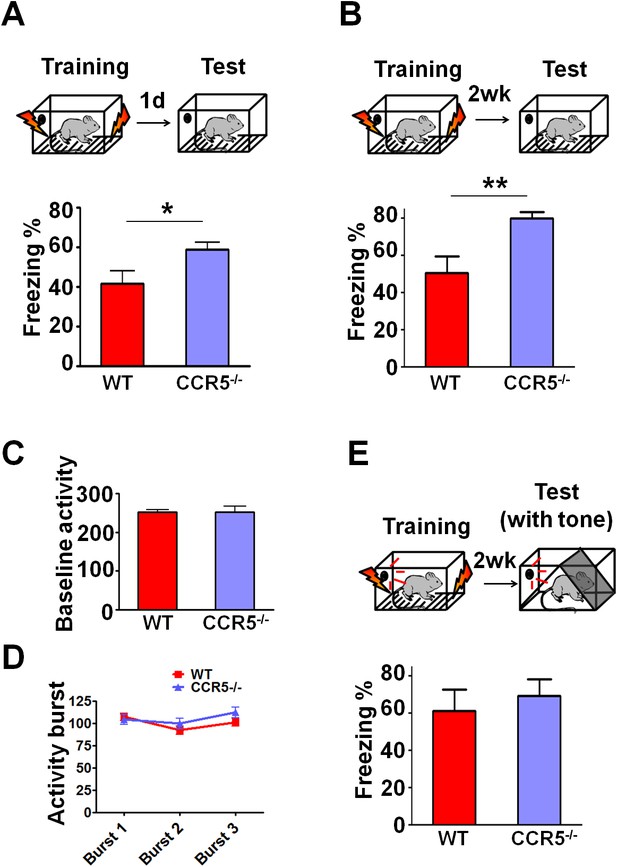
Ccr5-/- mice show enhanced contextual memory.
(A) When tested 24 hr after fear conditioning training, Ccr5-/- mice showed enhanced contextual memory compared to WT mice (WT n = 8, Ccr5-/- n = 8; *p<0.05, Student’s t-test). (B) When tested at two weeks after fear conditioning training, Ccr5-/- mice showed enhanced contextual memory compared to WT mice (WT n = 8, Ccr5-/- n = 8; **p<0.01, student’s t-test). (C) There were no differences between WT mice and Ccr5-/- mice in baseline activity (WT n = 16, Ccr5-/- n = 16). (D) There were no differences in the activity bursts triggered by the three shocks given during training between WT and Ccr5-/- mice (WT n = 16, Ccr5-/- n = 16). (E) When tested in a novel context 2-weeks after training, there were no differences in cued memory (freezing levels during the tone) between WT mice and Ccr5-/- mice (WT n = 8, Ccr5-/- n = 8). Error bars indicate SEM.
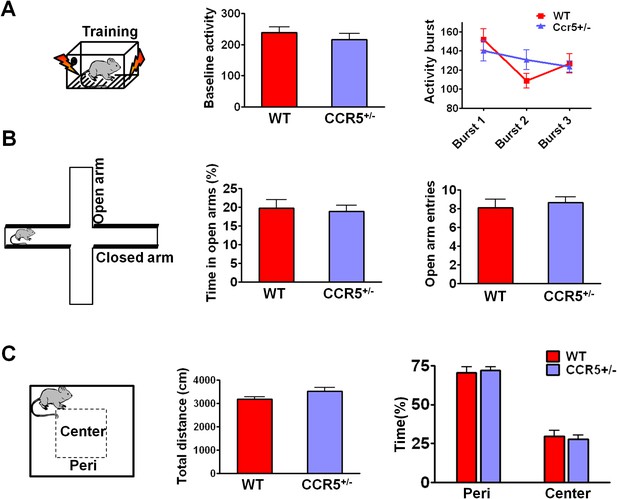
Behavioral tasks to test the baseline activity and activity burst during fear conditioning, and anxiety and locomotive levels in WT and Ccr5+/- mice.
(A) During fear conditioning training, there were no differences between WT mice and Ccr5+/- mice in baseline activity. There were no differences in the activity bursts triggered by the three shocks given during training between WT and Ccr5+/- mice either (WT n = 11, Ccr5+/- n = 11). (B) In the elevated plus maze test, WT and Ccr5+/- mice spent similar time in the open arms, and there was no difference in the number of open arm entries between WT and Ccr5+/- mice (WT n = 11, Ccr5+/- n = 20). (C) In the open field test, Ccr5+/- and WT mice showed similar travel distance when exposed to a novel open field, and there was no difference in the percent time WT and Ccr5+/- mice spent in the peri- or center- zone of the open field (WT n = 11, Ccr5+/- n = 21). Error bars indicate SEM.
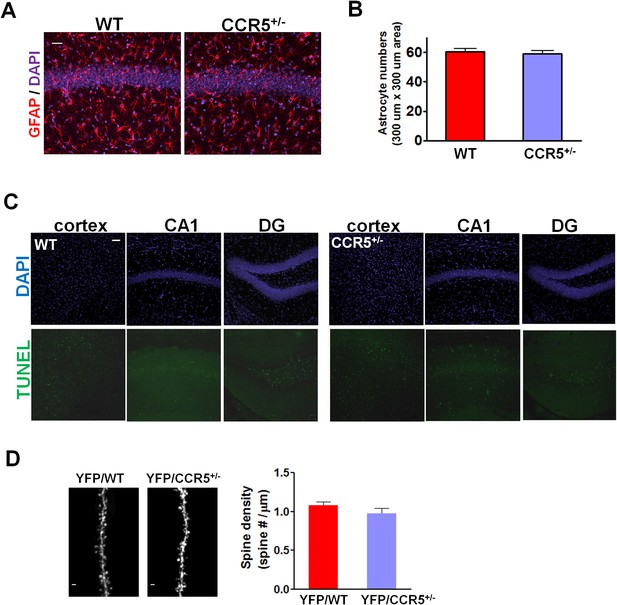
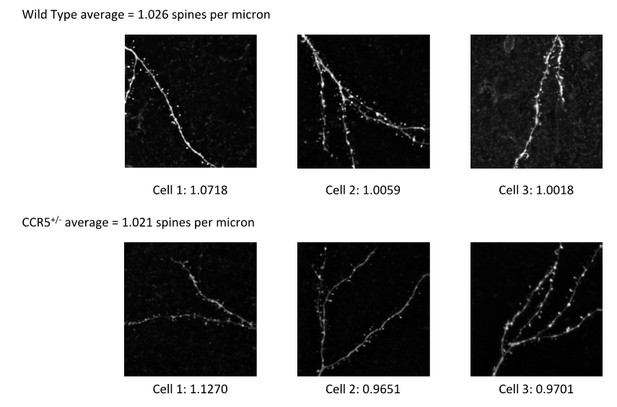
WT and Ccr5+/- mice show similar GFAP and TUNEL immunostaining and similar spine density.
(A) Representative images of brain slices stained with GFAP (astrocytes) and DAPI (nucleus) antibodies. Scale bar, 20 μm. (B) WT and Ccr5+/- mice had similar number of astrocytes in the CA1 subregion (WT n = 4; Ccr5+/- n = 4). (C) TUNEL staining showed very few apoptotic cells both in WT and Ccr5+/- mice in the retrosplenial cortex, CA1, and DG. Scale bar, 50 μm. (D) YFP/WT and YFP/Ccr5+/- mice have similar spine density in the hippocampal CA1 subregion (n = 3 mice for YFP/WT or YFP/Ccr5+/- group; for each mouse, over 200 spines from 10 different dendritic segments were counted. Scale bar, 1 μm). Error bars indicate SEM.
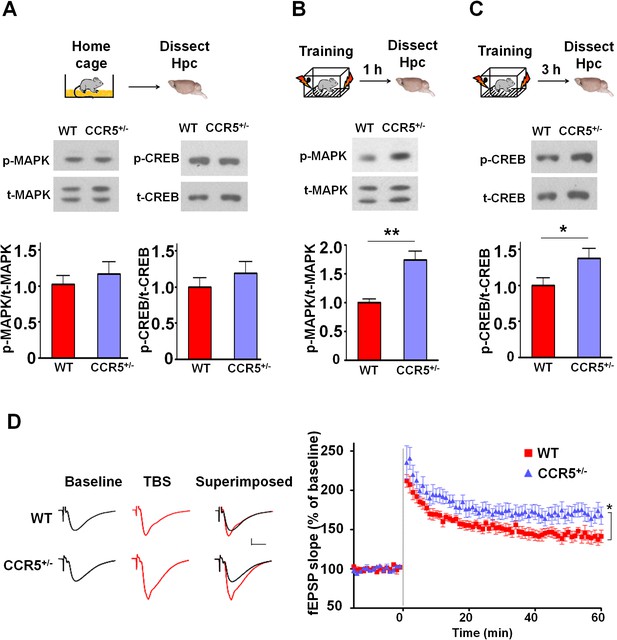
Ccr5+/- mice show post-training increases in MAPK and CREB signaling and enhanced hippocampal LTP.
(A) Hippocampus collected from home cage WT and Ccr5+/- mice had similar levels of phosphorylated p44/42 MAPK and CREB (MAPK: WT n = 4, Ccr5+/- n = 4; CREB: WT n = 6, Ccr5+/- n = 7). (B) Ccr5+/- mice showed enhanced levels of phosphorylated p44/42 MAPK (normalized with total MAPK) in hippocampal samples collected one hour after fear conditioning training (WT n = 6, Ccr5+/- n = 6; **p<0.01, Student’s t-test). (C) Ccr5+/- mice showed enhanced levels of phosphorylated CREB (normalized with total CREB) in hippocampal samples collected 3 hr after fear conditioning training (WT n = 7, Ccr5+/- n = 7; *p<0.05, Student’s t-test). (D) CA1 fEPSPs were recorded in hippocampal slices before (baseline) and after 5 TBS (theta bursts stimulation, each burst consists of four stimuli at 100 Hz, 200 ms inter-burst interval). Ccr5+/- slices show a significant LTP enhancement in the fEPSP measured during the last 10 min of recordings following the tetanus (n = 7 mice for both WT and Ccr5+/-; *p<0.05, Student’s t-test). Error bars indicate SEM.
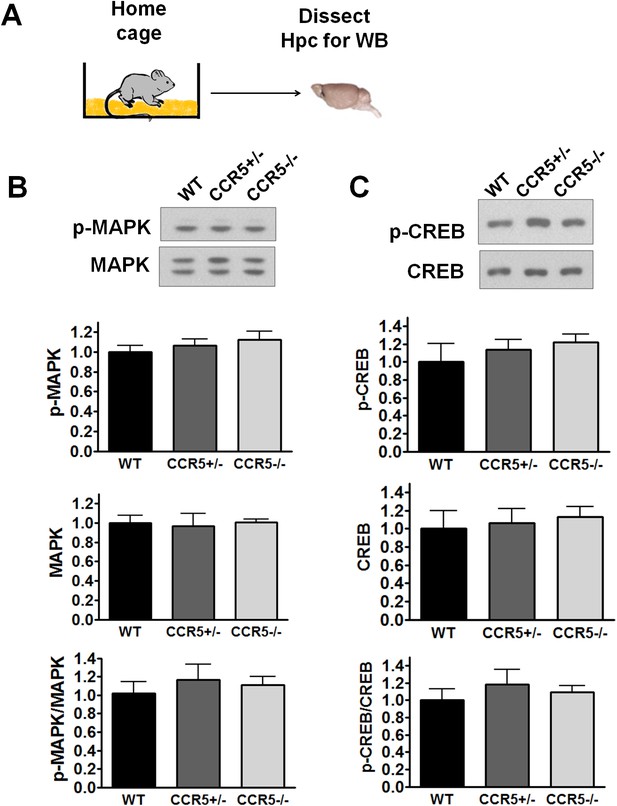
Hippocampal MAPK/CREB signaling of WT, Ccr5+/- and Ccr5-/- mice.
(A) Schematic of experiments. Hippocampal samples were collected from home cage mice. (B) Phospho- and total p44/42 MAPK levels (WT n = 4, Ccr5+/- n = 4, Ccr5-/- n = 4). (C) Phospho- and total CREB levels (WT n = 6, Ccr5+/- n = 7, Ccr5-/- n = 6). Error bars indicate SEM.
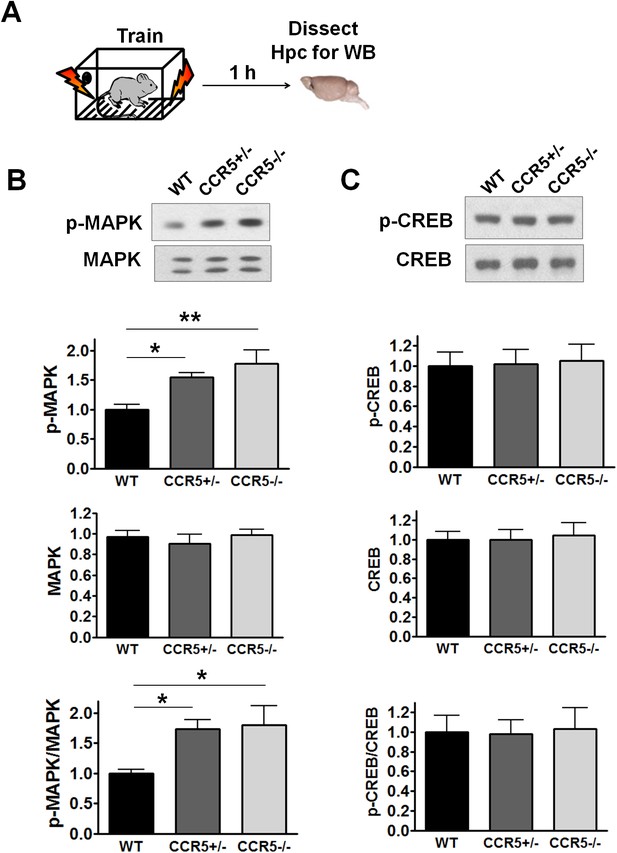
Hippocampal MAPK/CREB signaling of WT, Ccr5+/- and Ccr5-/- mice after fear conditioning.
(A) Schematic of experiments. Hippocampal samples were collected from mice at 1 hr after fear conditioning. (B) Phospho- and total p44/42 MAPK levels (WT n = 6, Ccr5+/- n = 6, Ccr5-/- n = 5, *p<0.05, **p<0.01, One-way ANOVA). (C) Phospho- and total CREB levels (WT n = 8, Ccr5+/- n = 8, Ccr5-/- n = 5). Error bars indicate SEM.
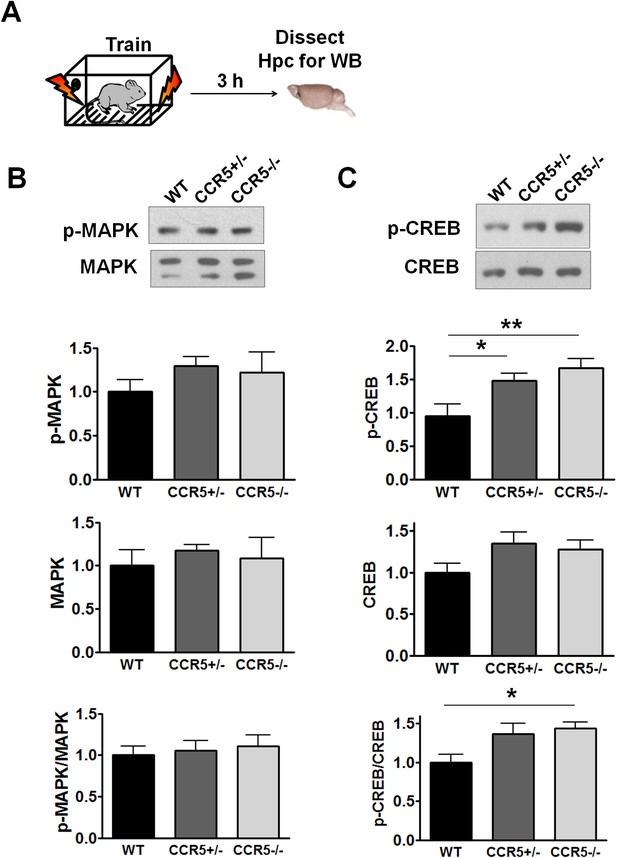
Hippocampal MAPK/CREB signaling of WT, Ccr5+/- and Ccr5-/- mice after fear conditioning.
(A) Schematic of experiments. Hippocampal samples were collected from mice at 3 hr after fear conditioning. (B) Phospho- and total p44/42 MAPK levels (WT n = 5, Ccr5+/- n = 5, Ccr5-/- n = 4). (C) Phospho- and total CREB levels (WT n = 7, Ccr5+/- n = 7, Ccr5-/- n = 7, *p<0.05, **p<0.01, One-way ANOVA). Error bars indicate SEM.
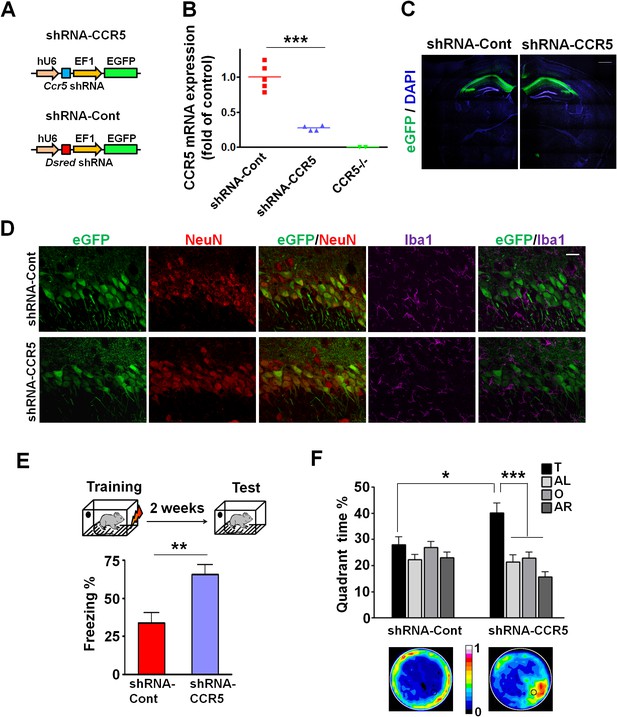
Ccr5 knockdown in the adult hippocampus results in enhanced memory.
(A) Schematics of the shRNA-CCR5 and shRNA-dsRed (shRNA-Cont) plasmids. (B) AAV containing shRNA-CCR5 or shRNA-Cont was injected into the hippocampus CA1/CA2 region, and dorsal CA1/CA2 Ccr5 mRNA was measured one-month after virus injection. Compared to shRNA-Cont, shRNA-CCR5 triggered a significant reduction in Ccr5 mRNA expression (***p<0.001, student’s t-test). Also, Ccr5-/- mice showed no Ccr5 mRNA expression in hippocampus. (C) AAV containing shRNA-CCR5 or shRNA-Cont was injected into the hippocampus. One month after virus injection, brain slices were stained with DAPI (nuclear labeling) and GFP (virus infection). Scale bar, 500 μm. (D) AAV containing shRNA-CCR5 or shRNA-Cont was injected into the hippocampus. One month after virus injection, brain slices were stained with GFP (virus infection), NeuN (neurons), and Iba1 (microglia). GFP was exclusively expressed in neurons. Scale bar, 20 μm. (E) AAV containing shRNA-CCR5 or shRNA-Cont was injected into the hippocampus, and mice were subjected to behavioral testing one month after virus injection. In the fear conditioning test, shRNA-CCR5 mice showed enhanced contextual memory when compared to those injected with shRNA-Cont virus (shRNA-cont n = 7, shRNA-CCR5 n = 10; **p<0.01, Student’s t-test). (F) In the probe test given after two days of water maze training, only shRNA-CCR5 mice but not shRNA-Cont mice spent significantly more time in the target quadrant than the other three quadrants (shRNA-cont n = 22; shRNA-CCR5 n = 20; *p<0.05, Student’s t-test; ***p<0.001, Two-way ANOVA with repeated measure). Heat maps below the bar graphs show the combined traces of the mice from each group during the probe test. shRNA-Cont mice showed a pattern of wall-hugging swim after two days training, but they learned the water maze task with extended training (Figure 3—figure supplement 1D). Error bars indicate SEM.
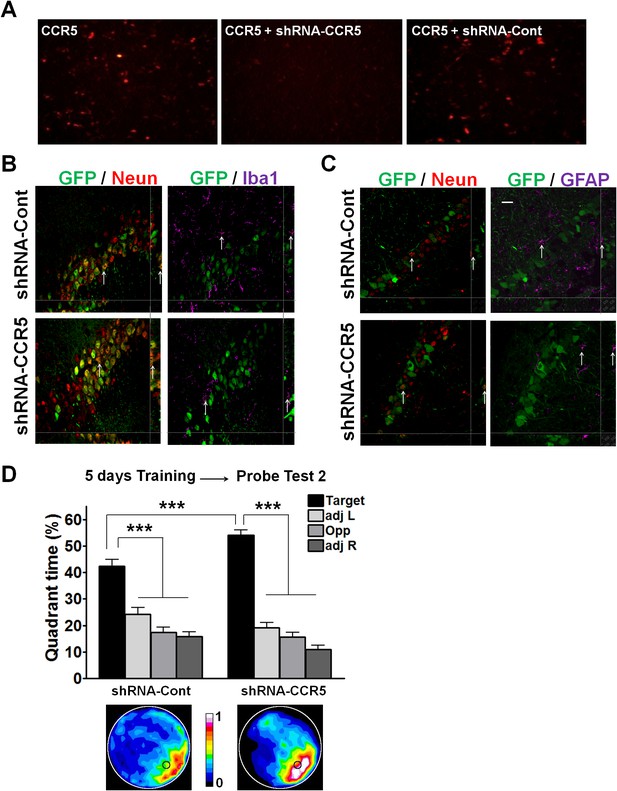
shRNA-CCR5 knockdown efficiency in HEK 293 cells, AAV infection specificity, and mice performance in the water maze probe test 2.
(A) HEK 293 cells were co-transfected with CCR5-tdTomato plus either shRNA-CCR5 or shRNA-Cont (dsRed) plasmids, and cells were imaged 1-day after plasmid transfection. Only shRNA-CCR5, but not shRNA-Cont, reduced CCR5-tdTomato expression in HEK 293 cells. (B) Brain slices were stained with GFP (viral infection), NeuN (neurons), and Iba1 (microglia) antibodies. (C) Brain slices were stained with GFP (viral infection), NeuN (neurons), and GFAP (astrocyte) antibodies. GFP is exclusively expressed in neurons, but not in microglia or astrocytes. Scale bar, 20 μm. (D) After 5 days of training, both shRNA-Cont mice and shRNA-CCR5 mice spent significantly more time in the target quadrant than in the other three quadrants. Heat maps show the combined traces of the mice from each group during the probe test (shRNA-cont, n = 22; shRNA-CCR5, n = 20; Between target quadrant and the other three quadrants, ***p<0.001, Two-way ANOVA with repeated measure; Between shRNA-Cont target quadrant and shRNA-CCR5 target quadrant, ***p<0.001, Student’s t-test). Error bars indicate SEM.
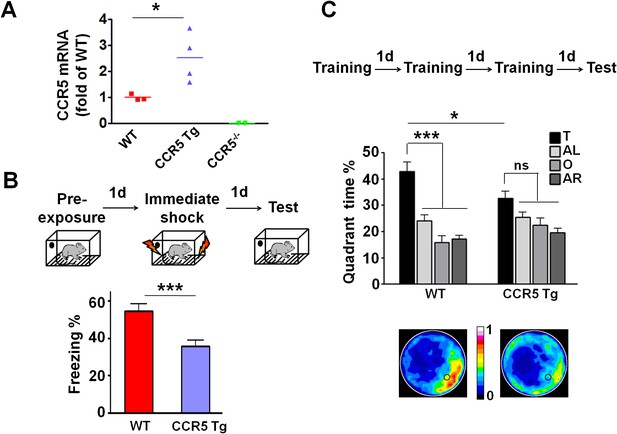
CCR5 overexpression leads to learning and memory deficits.
(A) Compared to WT mice, CCR5-overexpressing transgenic (Tg) mice showed enhanced Ccr5 mRNA expression in the hippocampus. (B) In a context pre-exposure fear-conditioning paradigm, CCR5 Tg mice showed contextual memory deficits compared to WT mice (WT n = 13, CCR5 Tg n = 15; ***p<0.001, Student’s t-test). (C) In the probe test given after 3 days of training in water maze, only WT mice but not CCR5 Tg mice, spent significantly more time in the target quadrant than the other three quadrants; CCR5 Tg mice also had lower searching times in the target quadrant than WT mice, indicating a learning and memory deficit (WT n = 18, CCR5 Tg n = 18; *p<0.05, Student’s t-test; ***p<0.001, Bonferroni post-tests, Two-way ANOVA with repeated measure). Error bars indicate SEM.
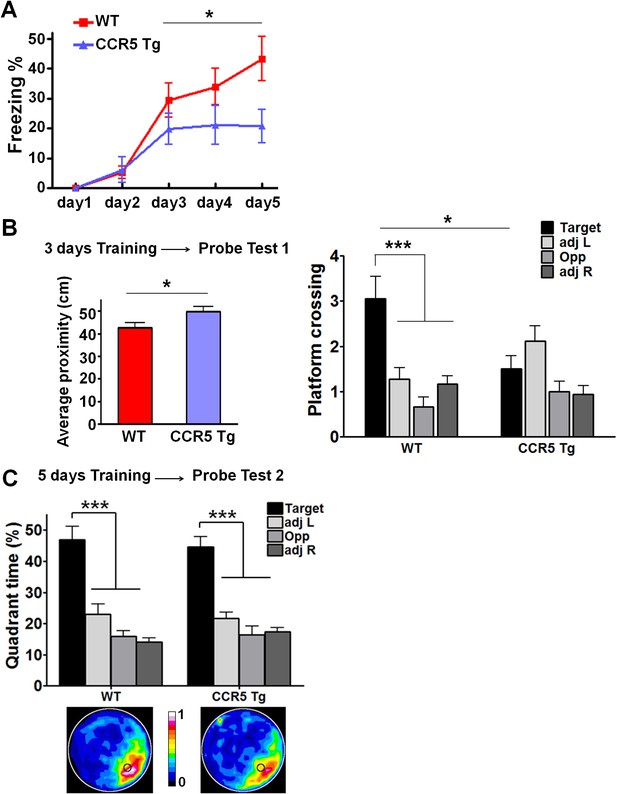
CCR5 overexpression leads to learning and memory deficits.
(A) CCR5 transgenic (Tg) mice showed a memory deficit with multi-day fear conditioning. WT and CCR5 Tg mice were subjected to daily weak training (25 s baseline, followed by a 0.4 mA (1 s) shock, followed by a 5 s post-shock, and then the mice were removed from the training chamber) for 5 days. The contextual memory was measured by the freezing% during the 25 s baseline exposure. Compared to WT mice, CCR5 Tg mice showed a contextual memory deficit between day 3 and day 5 (WT n = 18, CCR5 Tg n = 17, *p<0.05, overall (genotype × training days) interaction: F(2,66) = 1.15; Main effect of genotype: F(1,66) = 4.48, Two-way ANOVA with repeated measure). (B) In the probe test given after 3 days of training in water maze, CCR5 Tg mice showed significantly higher average proximity values (i.e., searched further away from the training platform site) than WT mice (WT n = 18, CCR5 Tg n = 18, *p<0.05, Student’s t-test). In the probe test, WT mice crossed the platform zone located in the target quadrant more than in the other three quadrants; In contrast, CCR5 Tg mice did not (WT n = 18, CCR5 Tg n = 18, *p<0.05, Student’s t-test; ***p<0.001, Bonferroni posttests, Two-way ANOVA with repeated measure). (C) In the water maze task, in probe test 2 after 5 days of training, both WT mice and CCR5 Tg mice spent more time in the target quadrant than the other three quadrants. Heat maps below the bar graphs show the combined traces of the mice from each group during the probe test (WT n = 18, CCR5 Tg n = 18, ***p<0.001, Two-way ANOVA with repeated measure). Error bars indicate SEM.
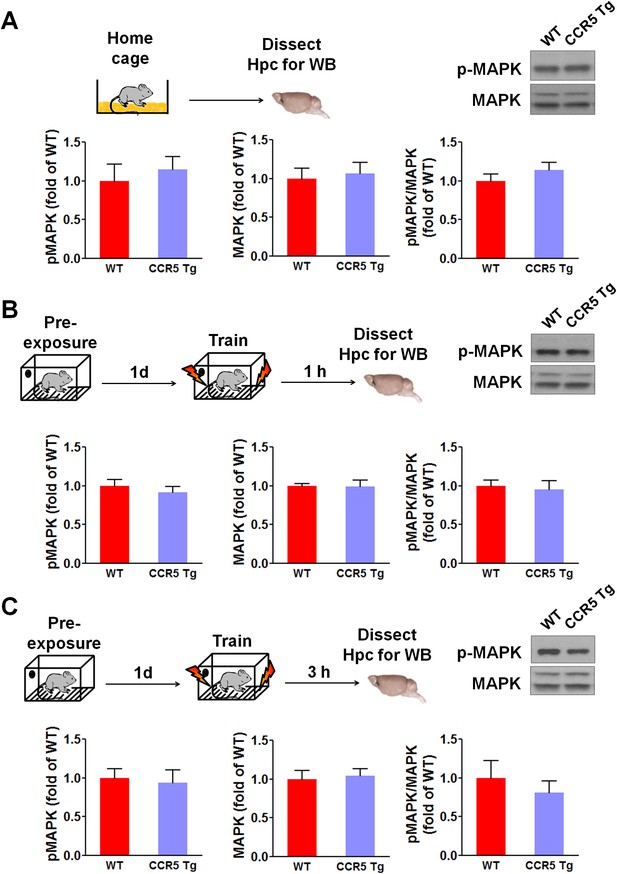
Hippocampal p44/42 MAPK signaling of WT and CCR5 transgenic (Tg) mice.
(A) Phospho- and total p44/42 MAPK levels of hippocampal samples collected from home cage mice (n = 6 for each group). (B) Phospho- and total p44/42 MAPK levels of hippocampal samples collected from mice at 1 hr after fear conditioning (n = 6 for each group). (C) PhosphoPhospho- and total p44/42 MAPK levels of hippocampal samples collected from mice at 3 hr after fear conditioning (n = 6 for each group). Error bars indicate SEM.
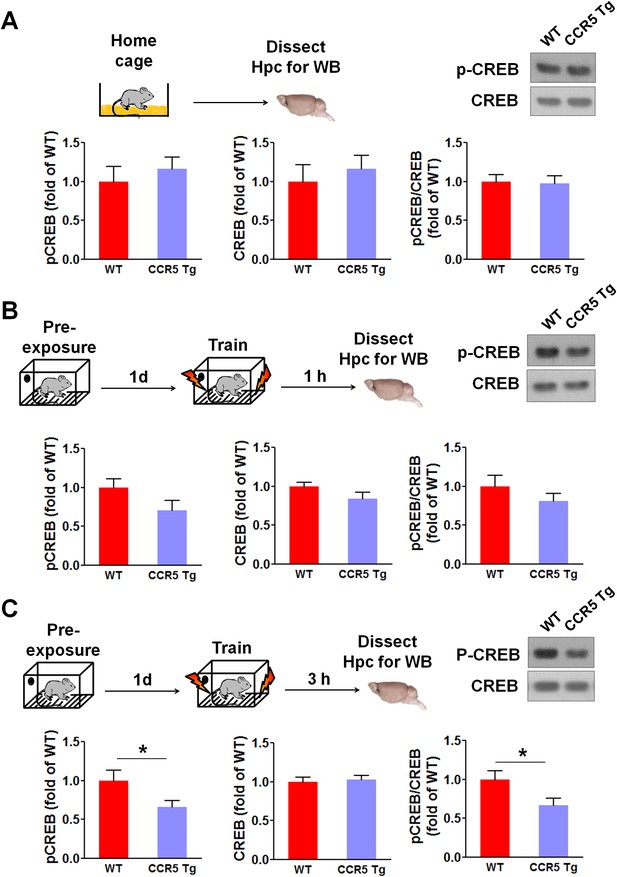
Hippocampal CREB signaling of WT and CCR5 transgenic (Tg) mice.
(A) Phospho- and total CREB levels of hippocampal samples collected from home cage mice (n = 6 for each group). (B) Phospho- and total CREB levels of hippocampal samples collected from mice at 1 hr after fear conditioning (n = 6 for each group). (C) PhosphoPhospho- and total CREB levels of hippocampal samples collected from mice at 3 hr after fear conditioning (n = 7 for each group; *p<0.05, Student’s t-test). Error bars indicate SEM.
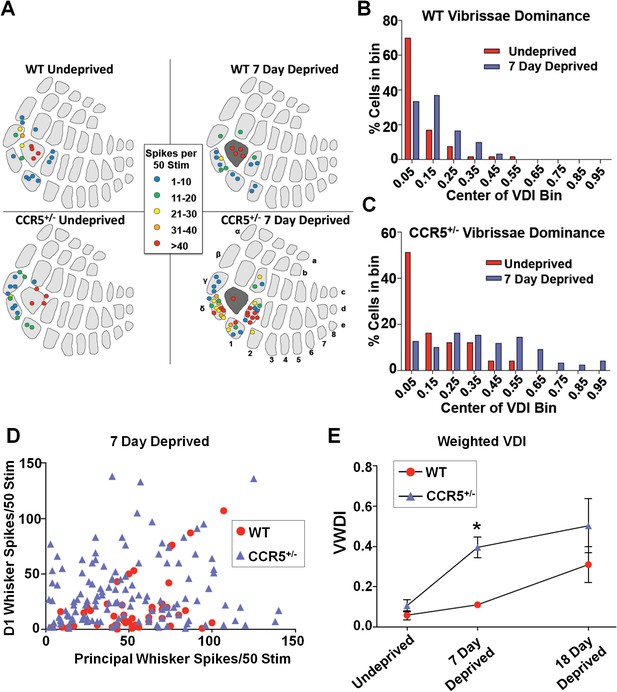
Ccr5+/- mice display accelerated experience dependent plasticity.
(A) Recording locations plotted on a standard barrel field map. The barrel shaded dark grey indicates the principal barrel for the spared whisker. The color of each circle represents the average response to D1 whisker stimulation (spikes per 50 stimulations) for cells in L2/3. Deprived Ccr5+/- mice showed greater proportion of penetrations responding strongly to D1 stimulation (WT: control vs deprived, p>0.99;Ccr5+/-: control vs deprived, p=0.0019, binomial test). (B) Vibrissae dominance histograms. Vibrissae dominance is calculated as D1/(PW + D1) and sorted into 10 bins (see Materials and methods). In WT mice, there is very little shift in the dominance histogram after 7 days of deprivation (n = 14). (C) The vibrissae dominance histogram shows a substantial shift right toward D1 dominance in the D1-spared Ccr5+/- mice (blue) compared with undeprived Ccr5+/- mice (red) (n = 24; p<0.01, Student’s t-test). (D) The value of the response to D1 stimulation is plotted against the same L2/3 cell’s response to principal whisker (PW) stimulation for mice subject to 7 days deprivation. A large number of Ccr5+/- (but not WT) cells lie above the unity line. (E) The average weighted vibrissae dominance index (WVDI) is plotted against deprivation period for WT and Ccr5+/- mice. Naïve WT and Ccr5+/- mice do not exhibit differences in their vibrissae dominance, however after 7 days deprivation there is an increase in WVDI in Ccr5+/- mice but not in WT mice (7-day WT vs Ccr5+/- mice: p=0.0030, ANOVA, Bonferroni post-tests). Error bars indicate SEM.
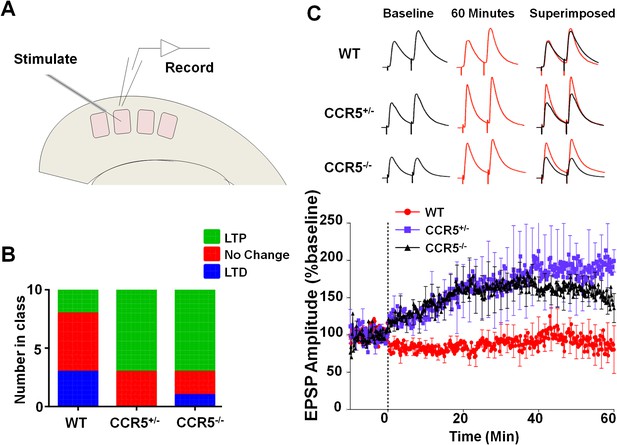
Ccr5 knockout mice exhibit a higher probability of LTP than WT Mice.
(A) Schematic of LTP in vitro recordings. Whole-cell patch clamp recordings were made from pyramidal cells in L2/3 of the barrel cortex. A stimulating electrode was placed in the center of the barrel immediately below the recording site and the columnar projection from L4 to L2/3 was stimulated. (B) When LTP was induced with a spike-timing dependent protocol with a low probability of potentiation in WT mice, Ccr5+/- and Ccr5-/- mice showed a higher incidence of LTP (70% LTP in Ccr5+/- and Ccr5-/- mice vs 20% in WT, p<0.001, χ-squared test). (C) Mean amplitude of LTP. Failures and successes are averaged together to give an overall average of all recordings. Ccr5+/- and Ccr5-/- cells had larger LTP than WT cells which on average did not potentiate (n = 10 per group).
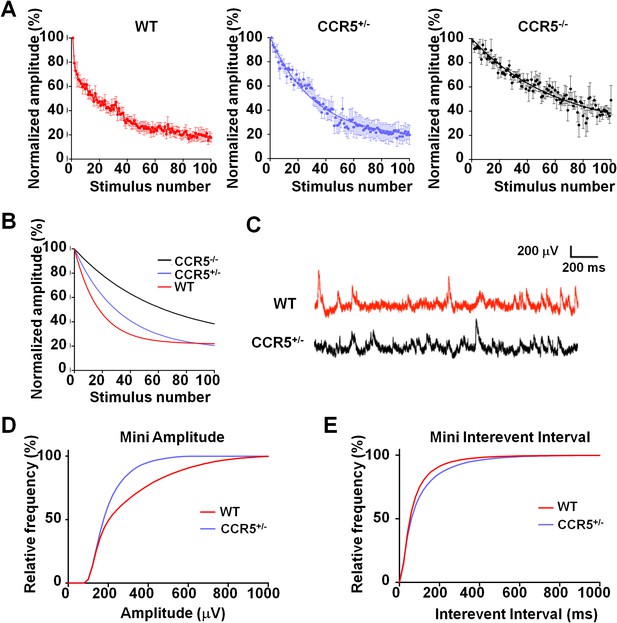
Ccr5 knockout mice exhibit lower release probability, and smaller, less frequent mEPSPs than WT mice.
(A) Cells from Ccr5+/-and Ccr5-/- mice exhibited lower release probability than cells from WT mice. Traces represent normalized amplitude of NMDA-receptor mediated evoked EPSPs in the presence of the use-dependent antagonist MK-801. A faster decrease in the peak EPSP amplitude is indicative of a higher release probability. (B) Single exponential curves were fitted for the data presented in panel A, and Ccr5+/- and Ccr5-/- mice demonstrated lower Pr than WT mice (n = 10 per group; p<0.01, Kruskal-Wallis test). (C) Example miniature EPSPs recordings from WT and Ccr5+/- mice. (D) Cells from Ccr5+/- mice displayed smaller mEPSPs than WT (WT n = 10, Ccr5+/- n = 10; p<0.001, K-S test). (E) Cells from Ccr5+/- mice displayed less frequent mEPSPs than WT and therefore greater inter-event intervals (WT n = 10, Ccr5+/- n = 10;p<0.001, K-S test).
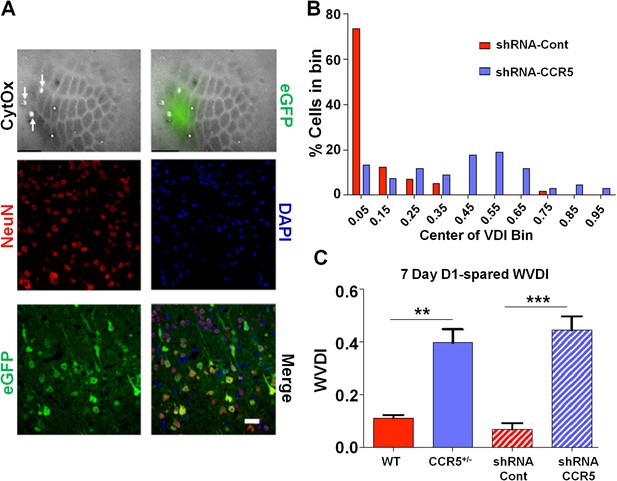
Viral knockdown of Ccr5 in barrel cortex enhances experience-dependent plasticity.
(A) Specificity and spread of viral infection. Interleaved slices processed for cytochrome oxidase (CytOx) confirmed viral location (GFP) over the recorded area of the barrel cortex. Arrows show lesions marking recording penetrations. Confocal images of the neuronal marker (NeuN) and viral expression (GFP) suggest that the shRNA is expressed exclusively in neurons. Scale bar = 20 µm. (B) Vibrissae dominance histogram after 7 days D1-spared single-whisker experience. Compared to control group, knockdown of Ccr5 leads to a shifted histogram (shRNA-cont n = 6; shRNA-CCR5 n = 6; p<0.001, Student’s t-test). (C) Weighted VDI values comparing Ccr5+/- mice with viral knockdowns. Viral knockdown has a similar effect to that of constitutive mutants after 7 days deprivation. Error bars indicate SEM.
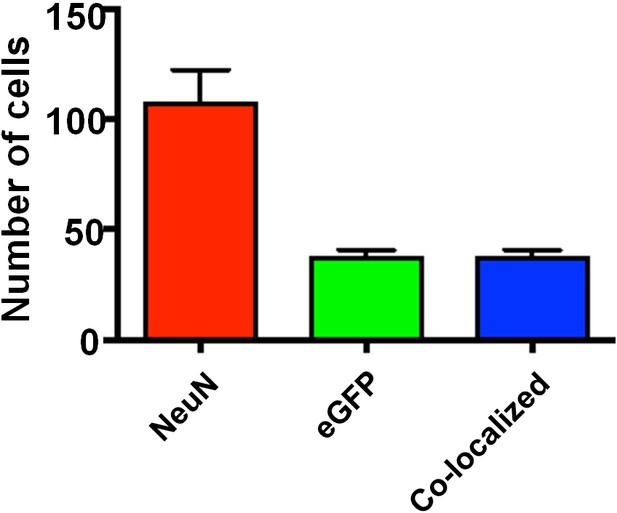
Specificity of viral infection in the barrel cortex.
Quantification of infected cells and those positive for NeuN. The result suggests Ccr5 viral knockdown is highly restricted to neurons. Using threshold detection we found an average of 107 ± 15.3 cells per region of interest labeled with the neuronal marker NeuN and 37.20 ± 3.54 cells labeled with virally-delivered GFP (Imaris software, Bitplane). 100% of cells labeled with GFP were co-localized with NeuN staining, indicating an exclusive infection of neurons by the virus. Error bars indicate SEM.
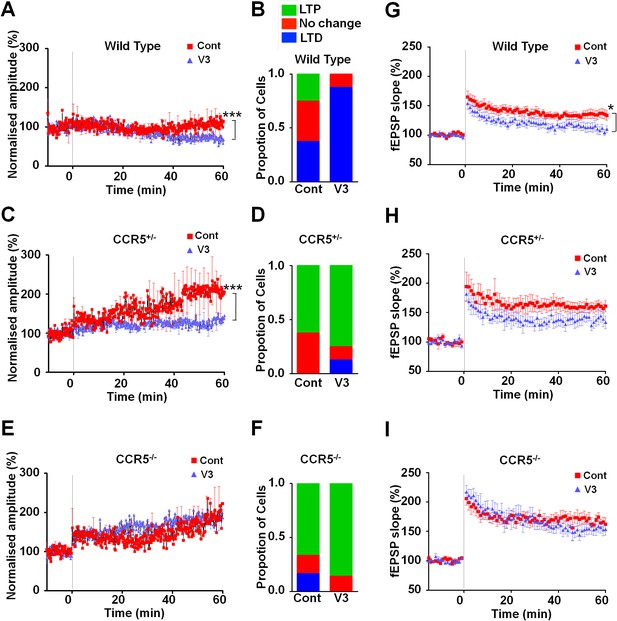
Ccr5 knockout prevents gp120 V3 peptide induced long-term potentiation deficits both in hippocampus and in barrel cortex.
(A) Whole-cell patch clamp recordings were made from pyramidal cells in L2/3 of the barrel cortex of WT mice. When LTP was induced with a spike-timing dependent protocol, in control conditions the trace showed no significant potentiation, while V3 peptide treatment caused significant depression (n = 8 for both Control and V3; ***p<0.001, Student’s t-test). (B) WT control cells showed equal proportions of LTP, LTD and no change; In contrast, 7 out of 8 V3-treated cells showed LTD. (C) Cells from Ccr5+/- mice showed strong LTP under control conditions, while V3-treated cells show significantly reduced LTP (n = 8 for both Control and V3; ***p<0.001, Student’s t-test). (D) Although LTP magnitude was reduced in V3-treated cells, the proportion of cells undergoing potentiation in slices from Ccr5+/- mice was similar to control. (E, F) Both control cells and V3-treated cells from Ccr5-/- mice showed strong potentiation, with no significant difference in amplitude and in the probability of LTP (Control n = 6, V3 n = 7; p=0.25). (G) Hippocampal CA1 fEPSPs were recorded in hippocampal slices before (baseline) and after 5 TBS (theta bursts stimulation). V3 peptide resulted in LTP deficits in the fEPSP measured during the last 10 min of recordings in hippocampal slices from WT mice (n = 8 for both Control and V3; *p<0.05, Student’s t-test). (H, I) V3 peptide treatment had no significant effect on Ccr5+/- (control n = 8, V3 n = 7; p=0.105, Student’s t-test) and Ccr5-/- mice (control n = 7, V3 n = 7, p=0.340, Student’s t-test). Error bars indicate SEM.
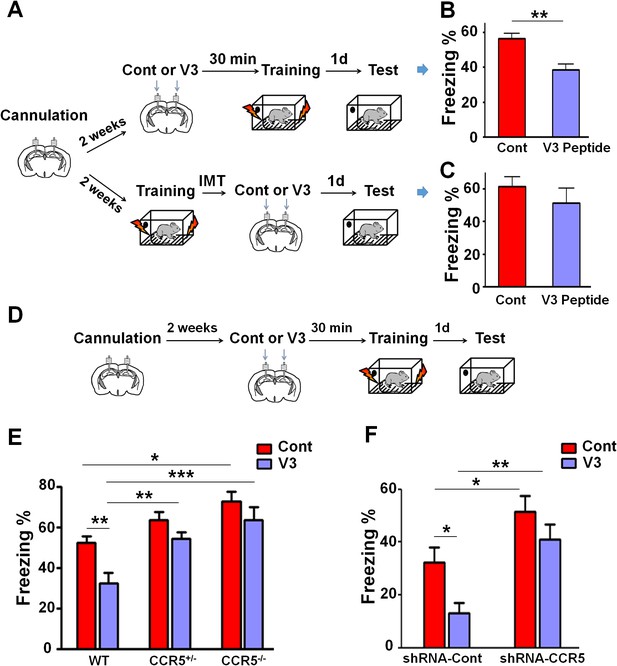
Both Ccr5 knockout and knockdown protect against gp120 V3 peptide induced memory deficits.
(A) Schematic of V3 peptide infusion and fear conditioning for the experiments shown in B and C. (B) 30 min after V3 peptide infusion into hippocampus, C57BL/6N mice were trained with fear conditioning. V3 peptide caused contextual memory deficits (Cont n = 6, V3 n = 6; **p<0.01, Student’s t-test). (C) When V3 peptide was infused into hippocampus immediately after fear conditioning training, no difference was observed between the control group and the V3 peptide group (Cont n = 10, V3 n = 10). (D) Schematic for experiments shown in E and F. (E) 30 min after V3 peptide infusion into hippocampus, mice were trained with fear conditioning. V3 peptide caused contextual memory deficits in WT mice, but Ccr5 knockout protected against V3-induced memory deficits (WT/Cont n = 10, WT/V3 peptide n = 11, Ccr5+/-/Cont n = 8, Ccr5+/-/V3 peptide n = 11, Ccr5-/-/Cont n = 8, Ccr5-/-/V3 peptide n = 8; *p<0.05, **p<0.01, ***p<0.001, Two-way ANOVA). (F) V3 peptide caused contextual fear conditioning memory deficits in mice injected with shRNA-Cont AAV, but hippocampal Ccr5 knockdown protected against V3-induced memory deficits (shRNA-Cont/Cont n = 10, shRNA-Cont/V3 n = 9, shRNA-CCR5/Cont n = 10, shRNA-CCR5/V3 n = 9; *p<0.05, **p<0.01, Two-way ANOVA). Error bars indicate SEM.
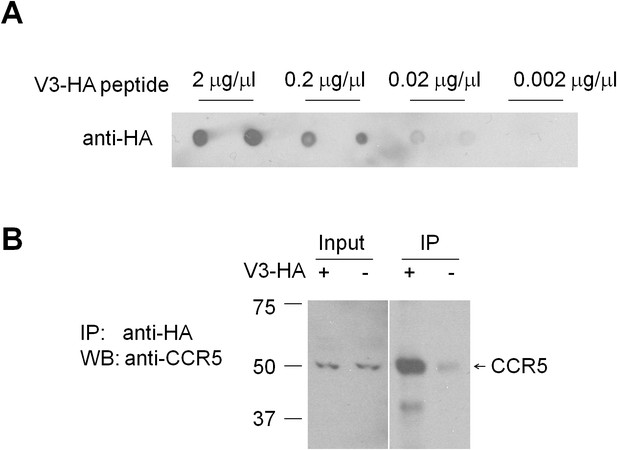
V3-HA peptide binds to hippocampal CCR5.
(A) Dot blots showed that different concentrations of V3-HA peptide could be detected by HA antibody. A final concentration of V3-HA peptide (0.02 μg/μl) was used for immunoprecipitation. (B) Immunoprecipitation with anti-HA-agarose showed that V3-HA peptide could co-precipitate CCR5 in the hippocampal lysate.
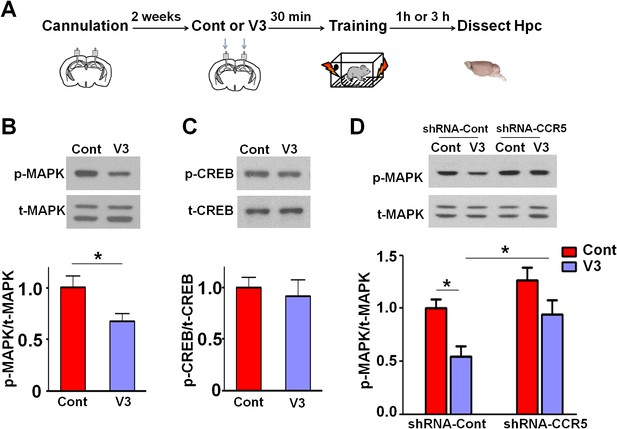
Ccr5 knockdown ameliorates hippocampal p44/42 MAPK signaling deficits caused by V3 peptide treatment.
(A) The dorsal hippocampal CA1 subregion was extracted 1 hr after training. V3 peptide reduced p44/42 pMAPK levels at 1 hr after fear conditioning (Cont n = 6, V3 n = 7; *p<0.05, Student’s t-test). (B) V3 peptide had no effect on pCREB when dorsal hippocampal CA1 subregion was extracted 3 hr after fear conditioning (Cont n = 5, V3 n = 6). (C) V3 peptide reduced p44/42 pMAPK in the dorsal CA1 of mice injected with shRNA-Cont virus at 1 hr after fear conditioning, but Ccr5 knockdown ameliorated the decrease in p44/42 pMAPK levels observed after learning (n = 8 for each group; *p<0.05, Two-way ANOVA). Error bars indicate SEM.
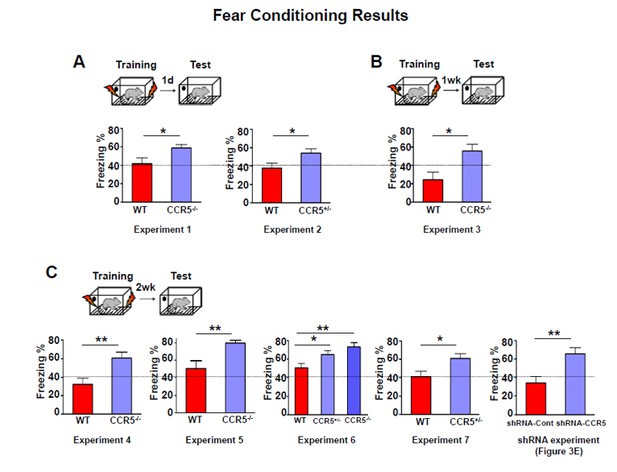
Fear conditioning results of WT and Ccr5+/-, Ccr5-/- mice or mice with Ccr5 knockdown.
(A) Mice were tested 1 day after fear conditioning training. (B) Mice were tested 1 week after fear conditioning training. (C) Mice were tested 2 weeks after fear conditioning training. A dash line marking the freezing at 40% level was shown in the figures to help compare mouse freezing across different experiments.





