Molecular basis of outer kinetochore assembly on CENP-T
Figures
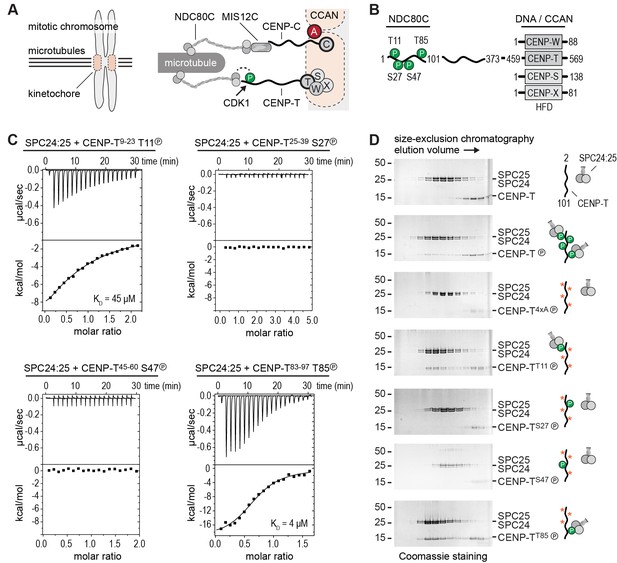
Phosphorylation of CENP-T2-101 at T11 or T85 is sufficient for the binding of SPC24:SPC25.
(A) Schematic representation of CENP-C and CENP-T recruiting MIS12C:NDC80C and NDC80C. (B) Besides the histone-fold domain (HFD) at the carboxy terminus, CENP-T mainly consists of regions of compositional bias, rich in polar residues, and likely to be intrinsically disordered. The boundaries of CENP-T constructs used in this study and domains involved in the binding of NDC80C and DNA/CCAN are indicated. In vitro phosphorylated residues in CENP-T1-101 are marked by a P (see also Table 1). (C) The binding between SPC24:SPC25 and CENP-T phosphopeptides was determined by isothermal titration calorimetry. The y-axis indicates kcal/mole of injectant. Dissociation constants (Kd) between SPC24:SPC25 and phosphopeptides containing T11 and T85 were determined to be 45 μM and 4 μM respectively. (D) SDS-PAGE analysis of various CENP-T2-101 mutants that were incubated with SPC24:SPC25 and separated by analytical size-exclusion chromatography. Red asterisks indicate mutated phosphorylation sites. See Figure 1—figure supplement 1 for the entire dataset.
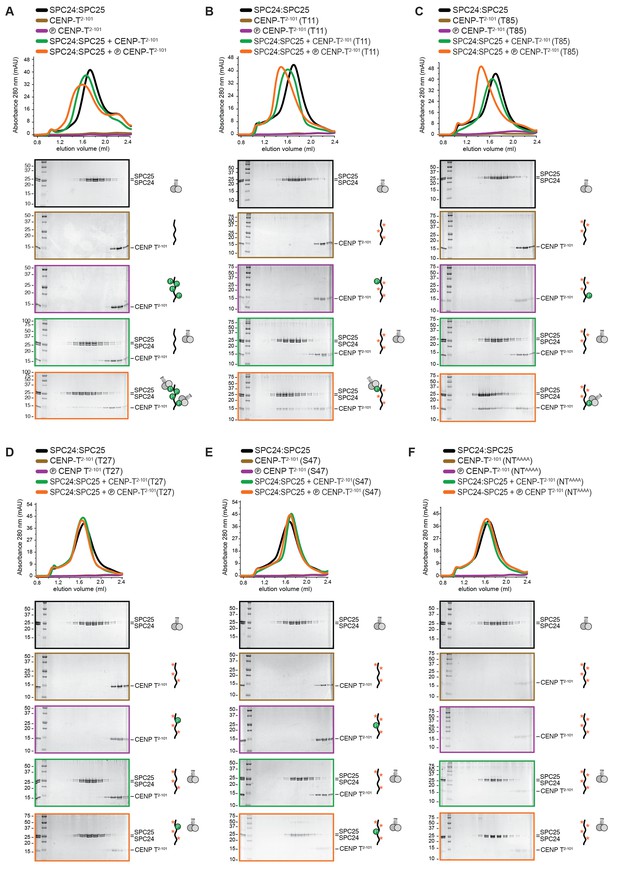
Phosphorylation of CENP-T2-101 at T11 or T85 is sufficient for the binding of SPC24:SPC25.
SDS-PAGE analysis of various CENP-T2-101 mutants that were incubated with SPC24:SPC25 and separated by analytical size-exclusion chromatography (Superdex 200 5/150). Six gels boxed in orange are also displayed in Figure 1D.
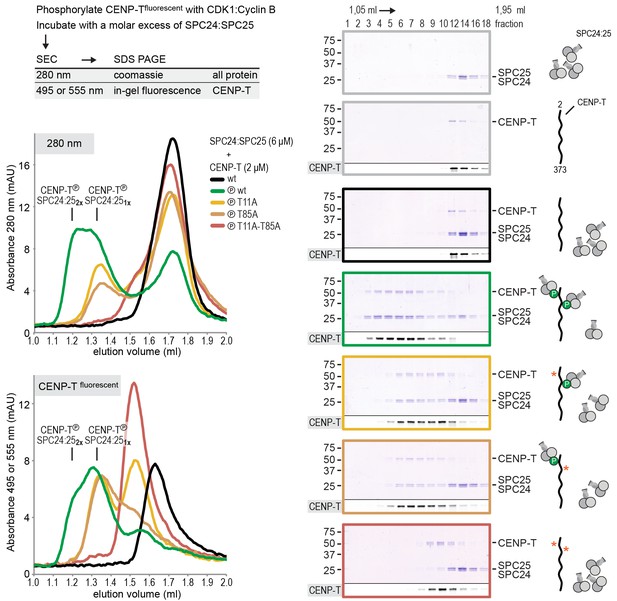
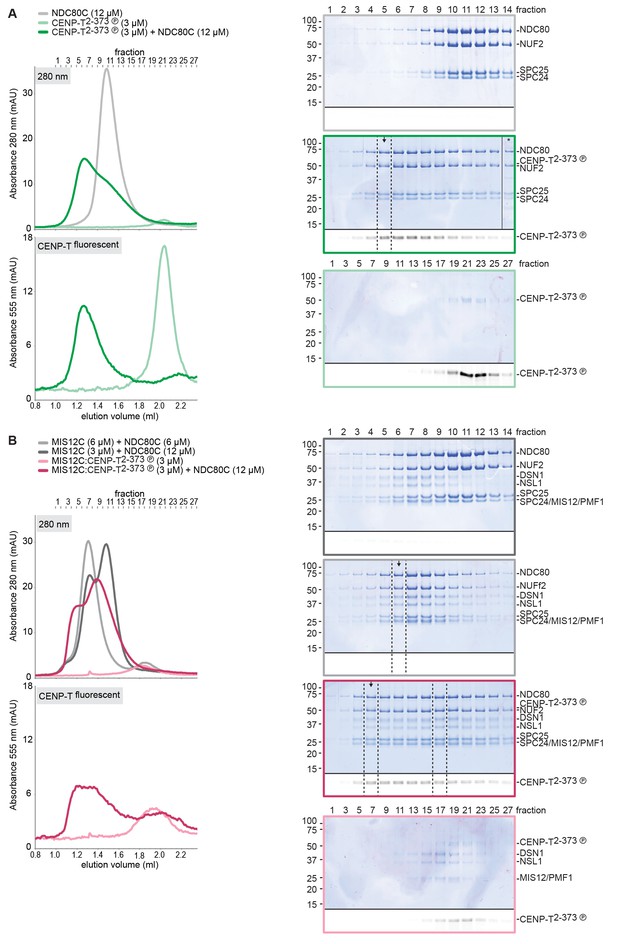
CENP-T phosphorylated at T11 and T85 binds two copies of SPC24:SPC25.
Analytical size-exclusion chromatography (Superdex 200 5/150 increase) and SDS-PAGE show that CENP-T2-373 phosphorylated by CDK1:Cyclin B at positions T11 and T85 can bind two copies of SPC24:SPC25. CENP-T was fluorescently labeled with FAM (wt) or TMR (mutants) and monitored specifically during chromatography and after SDS-PAGE. Red asterisks indicate mutated phosphorylation sites.
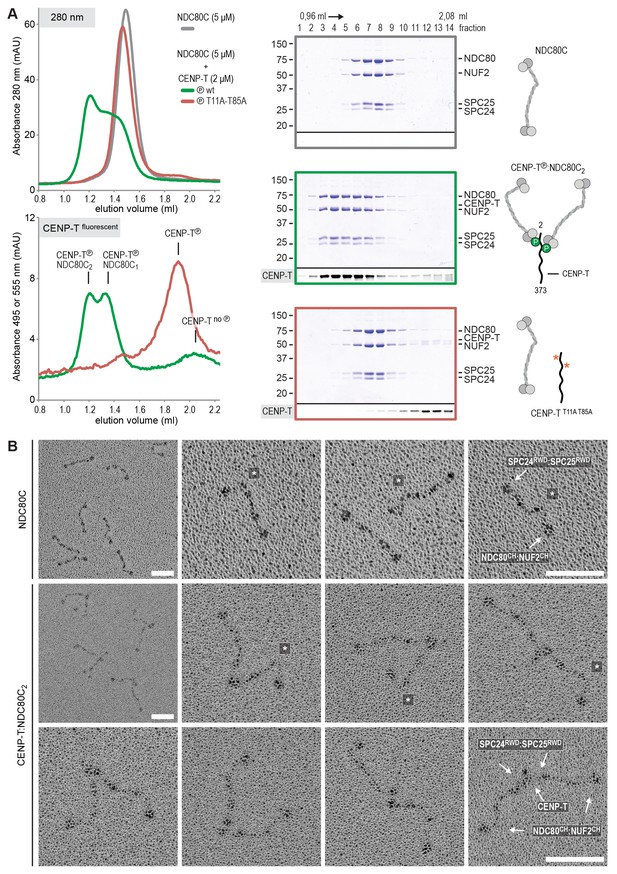
Phosphorylated CENP-T recruits two full-length NDC80 complexes.
(A) Analytical size-exclusion chromatography (Superose 6 5/150) and SDS-PAGE show that CENP-T2-373 phosphorylated by CDK1:Cyclin B at positions T11 and T85 can bind two full-length NDC80 complexes. The mixture of CENP-T and NDC80C contained NDC80C, CENP-T:NDC80C, as well as CENP-T:NDC80C2 species (green trace). The specific monitoring of fluorescently labeled CENP-T was used to distinguish these species. Red asterisks indicate mutated phosphorylation sites. Analysis of the single T11A and T85A CENP-T mutants and additional controls are included in Figure 3—figure supplement 1. (B) NDC80C (top row) and CENP-T:NDC80C2. (middle and bottom rows) were visualized by electron microscopy after glycerol spraying and low-angle platinum shadowing. Asterisks mark the kink in the NDC80:NUF2 coiled coil region. The first micrographs show a representative field of view at a lower magnification. All scale bars represent 50 nm. More micrographs of NDC80C and CENP-T:NDC80C2 as well as sample preparation information are included in Figure 3—figure supplements 2–4.
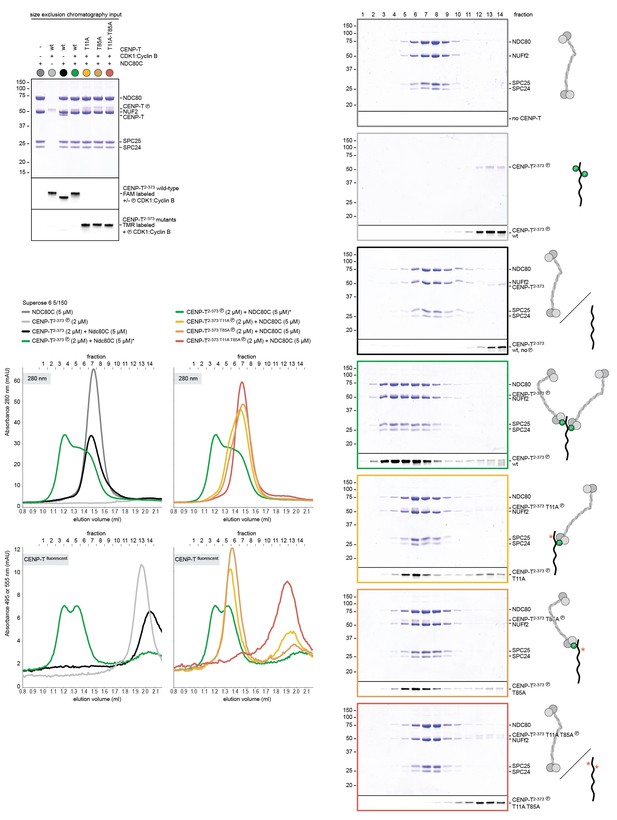
Phosphorylated CENP-T recruits two full-length NDC80 complexes.
(A) A fraction of the samples was analyzed by SDS-PAGE prior to size-exclusion chromatography. The phosphorylation-induced shift of CENP-T is clearly visible. (B) Analytical size-exclusion chromatography (Superose 6 5/150) and SDS-PAGE showing that CENP-T phosphorylated by CDK1 at positions T11 and T85 can bind two full-length NDC80 complexes. Data for NDC80C alone (grey), NDC80C mixed with phosphorylated wild-type (green) or T11A-T85A (red) CENP-T are displayed in Figure 3.
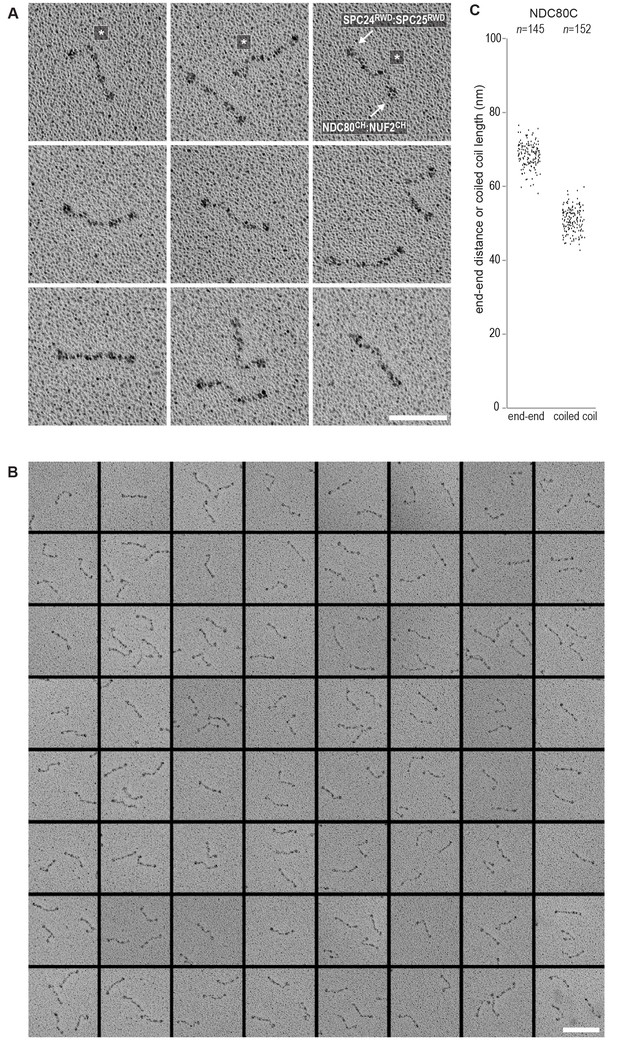
NDC80C gallery and measurements.
(A) Purified NDC80 complexes were separated by size-exclusion chromatography and visualized after glycerol spraying followed by low-angle metal shadowing. The three micrographs in the top row are shown in Figure 3. (B) Gallery showing 64 selected areas with one or multiple NDC80Cs. Scale bars represent 50 nm. (C) End-end and coiled coil distances were measured for the indicated number of shadowed NDC80Cs. The displayed values are not corrected for an estimated low-angle shadowing contribution of 3 nm at either end of NDC80C.
Sample preparation for low-angle metal shadowing and EM.
NDC80C was mixed with phosphorylated CENP-T (green traces) or with an isolated CENP-T:MIS12C complex (red traces) and analyzed by analytical size-exclusion chromatography (Superose 6 5/150) and SDS-PAGE. Different gels from the same material were used for Coomassie staining and the stain-free identification of fluorescent CENP-T. Samples that were used for low-angle metal shadowing are indicated by the black arrow. Results are shown in Figure 3B and Figure 6B as well as in Figure 3—figure supplement 4 and Figure 6—figure supplements 3–4.
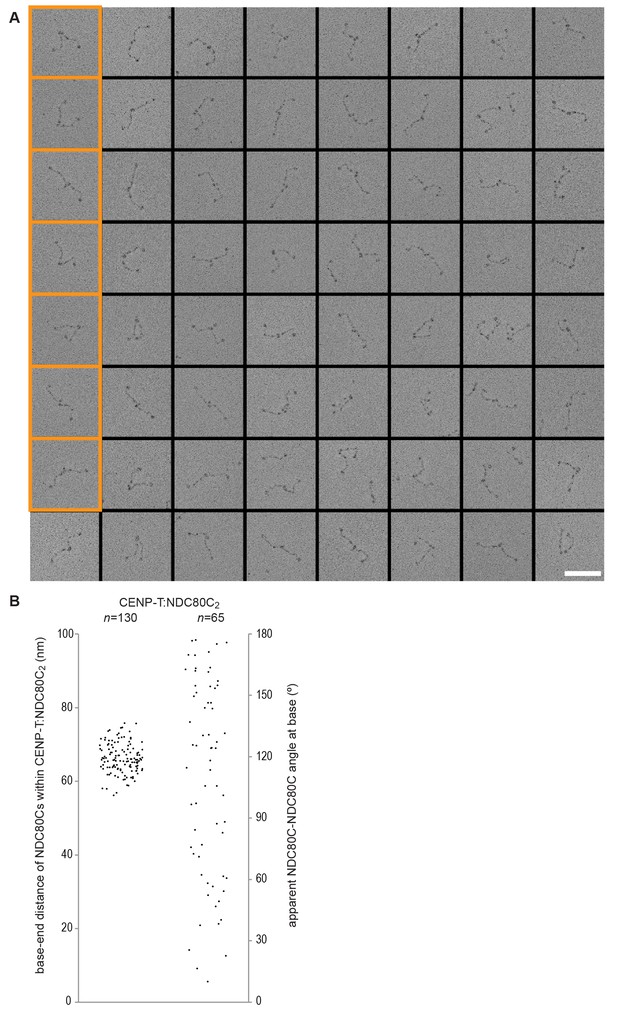
Gallery and measurements of CENP-T:NDC80C2.
(A) CENP-T:NDC80C2 complexes were visualized after glycerol spraying followed by low-angle metal shadowing. The orange-boxed micrographs are shown in Figure 3B. Scale bar 50 nm. (B) The apparent relative orientation of the two NDC80Cs in CENP-T:NDC80C2 complexes was measured.
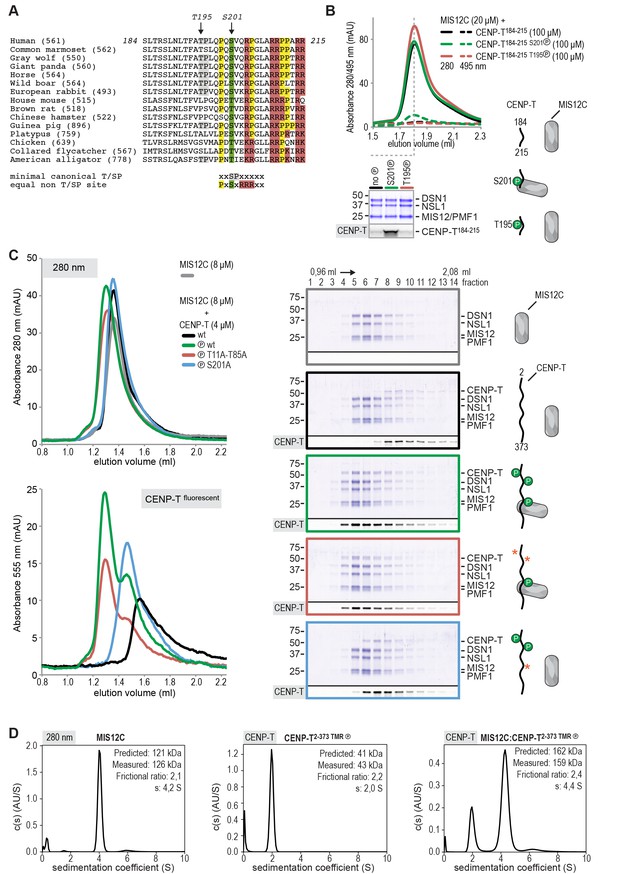
CENP-T phosphorylated by CDK1 at position S201 binds the MIS12 complex.
(A) Multiple sequence alignment of the CENP-T184-215 region. The CENP-T length in the various species, as well as the T195 and S201 phosphosites are indicated. The indicated non-canonical CDK1 recognition site, to which S201 and neighboring residues conform perfectly, and the shown CDK1 minimal canonical T/SP site were found to be equally good CDK1 substrates (Suzuki et al., 2015b). (B) Analytical size-exclusion chromatography (Superose 6 5/150 increase) shows that CENP-T184-215 binds to MIS12C when phosphorylated at position S201. Dashed traces indicate absorption at 495 nm and represent peptide that co-elutes with MIS12C. Free peptides have a retention volume of 2.6 ml. Peak fractions were analyzed by Coomassie staining and in-gel fluorescence after SDS-PAGE. (C) Analytical size-exclusion chromatography (Superdex 200 5/150 increase) and SDS-PAGE show that CENP-T2-373 phosphorylated by CDK1:Cyclin B at S201 binds to MIS12C. Red asterisks indicate mutated phosphorylation sites. Additional controls are included in Figure 4—figure supplement 2. (D) Sedimentation velocity analytical ultracentrifugation of MIS12C, CENP-T, and MIS12C:CENP-T show that MIS12C and CENP-T bind in a 1:1 complex. MIS12C was monitored by its absorbance at 280 nm whereas CENP-T and the CENP-T:MIS12C mixture were followed using the absorbance of the fluorescently labeled CENP-TTMR at 555 nm.
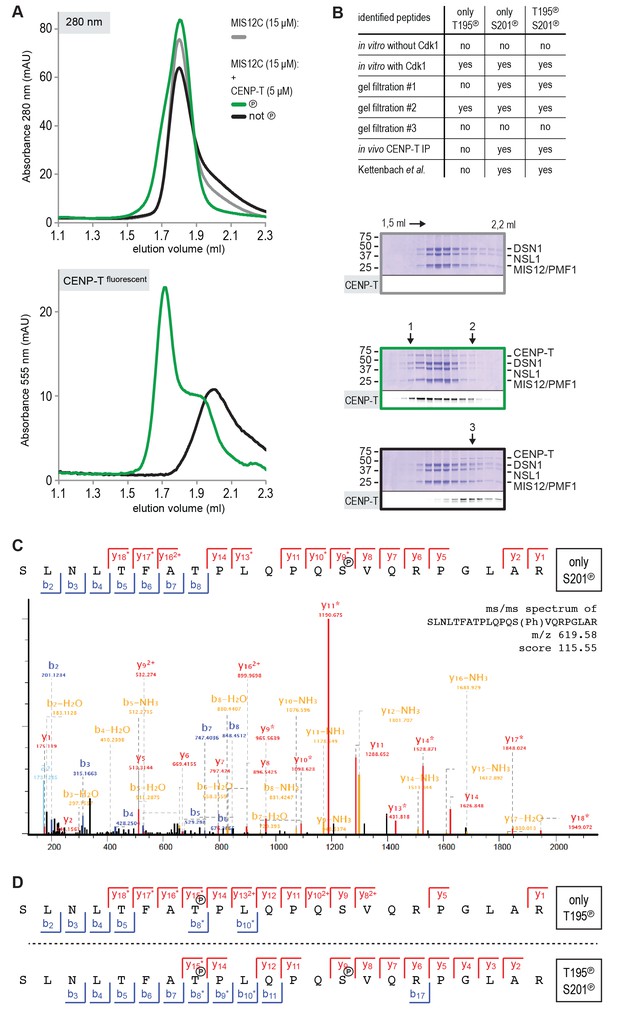
CENP-T phosphorylation by CDK1:Cyclin B at positions T195 and S201.
(A) CENP-T:MIS12C complexes were prepared by size-exclusion chromatography (Superose 6 5/150 increase). The phosphorylation status of CENP-T in the fractions indicated by 1, 2, and three were analyzed by mass spectrometry. (B) Specification of the identified single T195 or S201 phosphopeptides and the double phosphopeptide. Endogenous CENP-T was purified using a specific antibody. (C– E) Representative ms/ms spectra of SLNLTFATPLQPQSVQRPGLAR peptides phosphorylated at positions S201 and/or T195 from in vitro phosphorylated CENP-T (panels C and D) and from in vivo immunoprecipitated CENP-T (panel E).
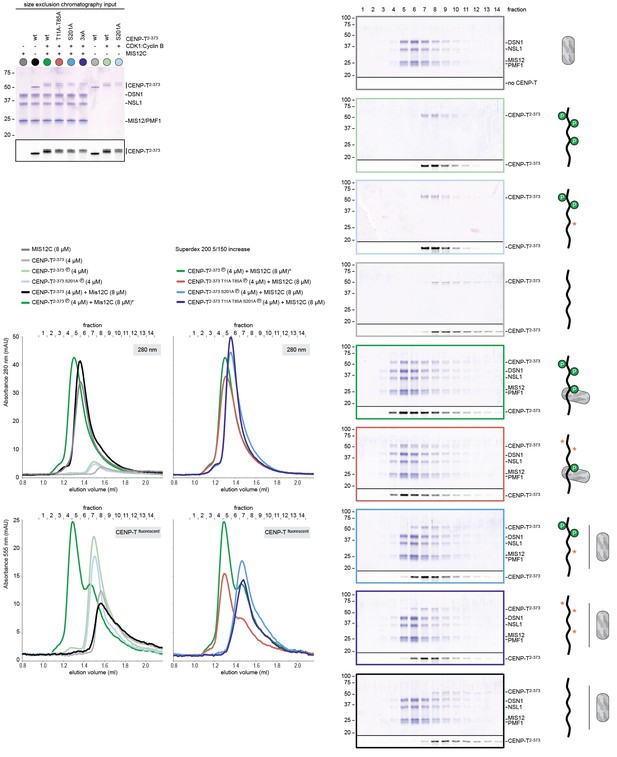
CENP-T phosphorylated by CDK1:Cyclin B at position S201 binds the MIS12 complex.
(A) A fraction of the mixed samples was analyzed by SDS-PAGE prior to size-exclusion chromatography. The phosphorylation-induced shift of CENP-T is clearly visible. (B) Analytical size-exclusion chromatography (Superdex 200 5/150 increase) and SDS-PAGE show that CENP-T phosphorylated by CDK1 at position S201 binds MIS12C. Data for MIS12C alone (grey), MIS12C mixed with a unphophorylated CENP-T (black), and MIS12C mixed with phosphorylated wild type (green), T11A-T85A (red), or S201A (blue) CENP-T are displayed in Figure 4C.
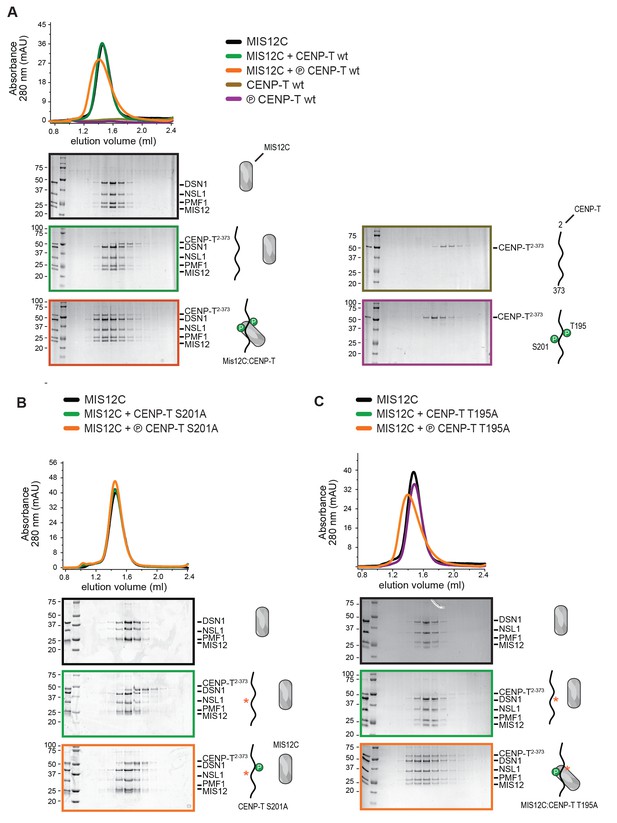
CENP-T mutated at position T195 binds MIS12C.
(A–C) Complex formation between MIS12C and phosphorylated or not-phosphorylated forms of CENP-T was analyzed by size-exclusion chromatography (Superdex 200 5/150) and SDS-PAGE. The shift of the orange compared to the green traces indicates binding between MIS12C and phosphorylated CENP-Twt (panel A) or phosphorylated CENP-TT195A (panel C). Phosphorylated CENP-TS201A does not bind to MIS12C (panel B).
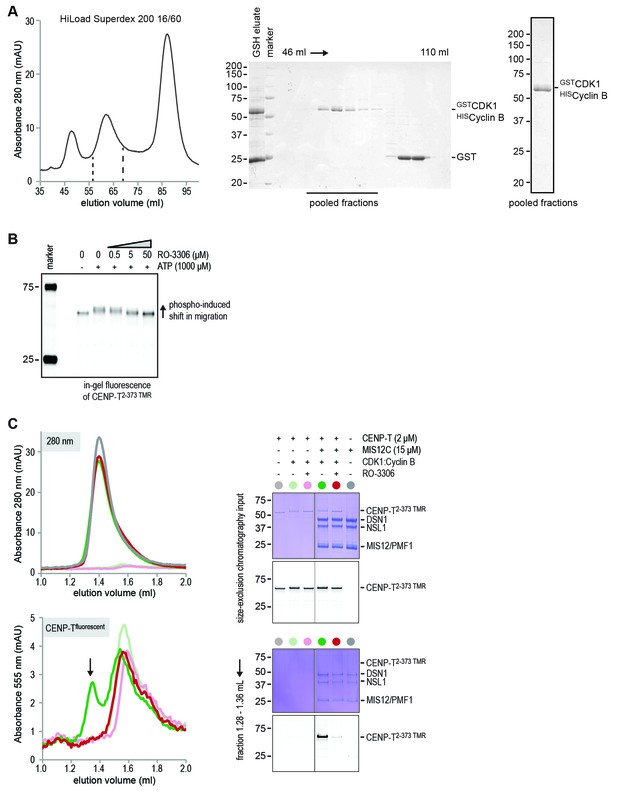
Purification of CDK1:Cyclin B and inhibition of CENP-T phosphorylation by RO-3306.
(A) Glutathione-based affinity purification and size-exclusion chromatography were used to purify the CDK1:Cyclin B complex. Selected fractions are indicated and the final concentrated product is shown. (B) CENP-T2-373 TMR (1 μM) was incubated with CDK1:Cyclin B (100 nM) in the absence or presence of ATP (1 mM) and in the absence or presence of increasing amounts of RO3306 (0, 0.5, 5, and 50 μM). In-gel fluorescence of CENP-T2-373 TMR was used to monitor the CDK1-induced shift in migration and its inhibition by the CDK1 inhibitor RO-3306. (C) CENP-T2-373 TMR (10 μM) was incubated with CDK1:Cyclin B (100 nM) in the presence of ATP (1 mM) and in the absence or presence of RO3306 (25 μM). CENP-T was thereafter incubated with a molar excess of MIS12C and analyzed by size-exclusion chromatography (Superdex 200 5/150 increase). RO-3306 inhibited the integration of CENP-T in a complex with MIS12C (retention volume 1.28–1.36 ml).
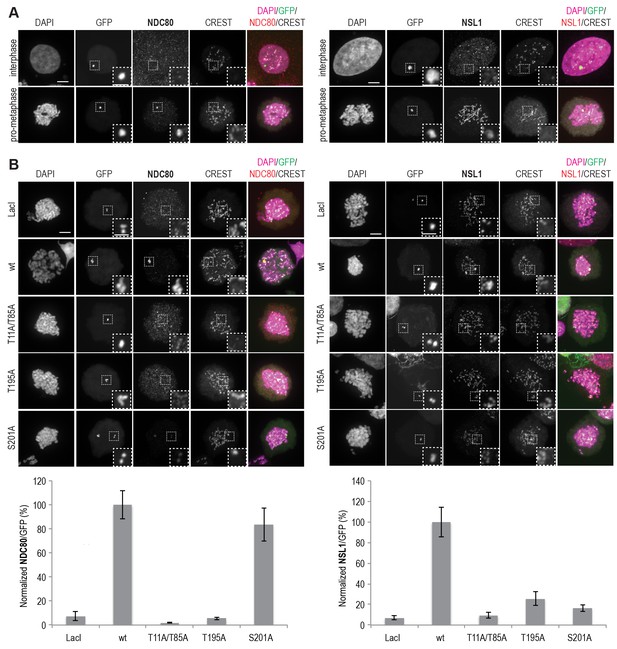
MIS12C and NDC80C are recruited to ectopically localized CENP-T in U2OS cells.
(A) U2OS cells that contain a lacO array recruit transiently expressed LacI-EGFP-CENP-T2-373 fusion constructs throughout the cell cycle. NDC80C (antibody against NDC80) and MIS12C (antibody against NSL1) are recruited to ectopic CENP-T loci in prometaphase, but not in interphase. Endogenous kinetochores are marked by CREST, DAPI was used to stain the DNA, and the EGFP signal indicates LacI-EGFP or LacI-EGFP-CENP-T constructs and marks the position of the lacO array. Scale bars represent 5 µm for the overview images and 2 µm for the magnified insets. (B) Representative prometaphase cells expressing LacI-EGFP or LacI-EGFP-CENP-T (wild-type or mutant) fusion constructs. (C) The mean NDC80C/EGFP and MIS12C/EGFP signal intensity ratios were determined from three independent experiments and normalized to the ratio for the wild-type construct. A total of 12–27 cells were analyzed per LacI-EGFP-CENP-T fusion construct and error bars indicate standard error of mean.
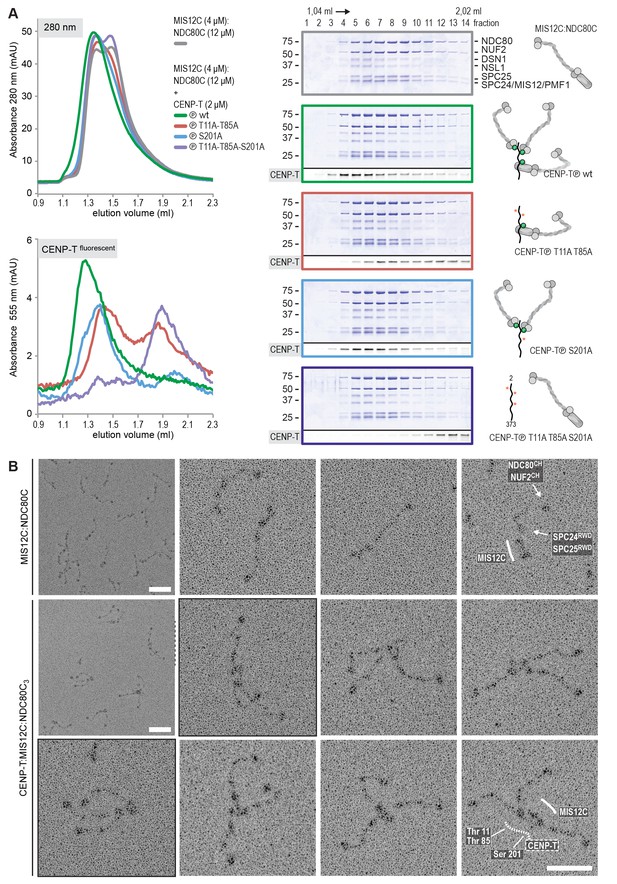
Reconstitution and visualization of CENP-T:MIS12C bound to three full-length NDC80 complexes.
(A) Analytical size-exclusion chromatography (Superose 6 5/150 increase) and SDS-PAGE show that CENP-T2-373 phosphorylated by CDK1:Cyclin B can bind MIS12C and three full-length NDC80 complexes. The triple mutation of phospho-sites T11, T85, and S201 is sufficient to prevent CENP-T from interacting with NDC80C and MIS12C:NDC80C (purple). Red asterisks indicate mutated phosphorylation sites. Fluorescently labeled CENP-T was monitored specifically during chromatography and after SDS-PAGE. The analysis of the complete set of CENP-T mutants is included as Figure 6—figure supplement 1. (B) MIS12C:NDC80C (top row) and CENP-T:MIS12C:NDC80C3 (middle and bottom rows) were visualized by electron microscopy after glycerol spraying and low-angle platinum shadowing. MIS12C forms a rod-like extension in the MIS12C:NDC80C complexes and can in some cases also be distinguished as a module in CENP-T:MIS12C:NDC80C3 assemblies. In those cases, CENP-T T11 and T85 are positioned at the base of the other two NDC80Cs (as annotated in the bottom right micrograph). The first micrographs show a representative field of view at a lower magnification. Scale bars represent 50 nm. Sample preparation is described in Figure 3—figure supplement 3. More micrographs of MIS12C:NDC80C and CENP-T:MIS12C:NDC80C3 are included in Figure 6—figure supplements 3–4.
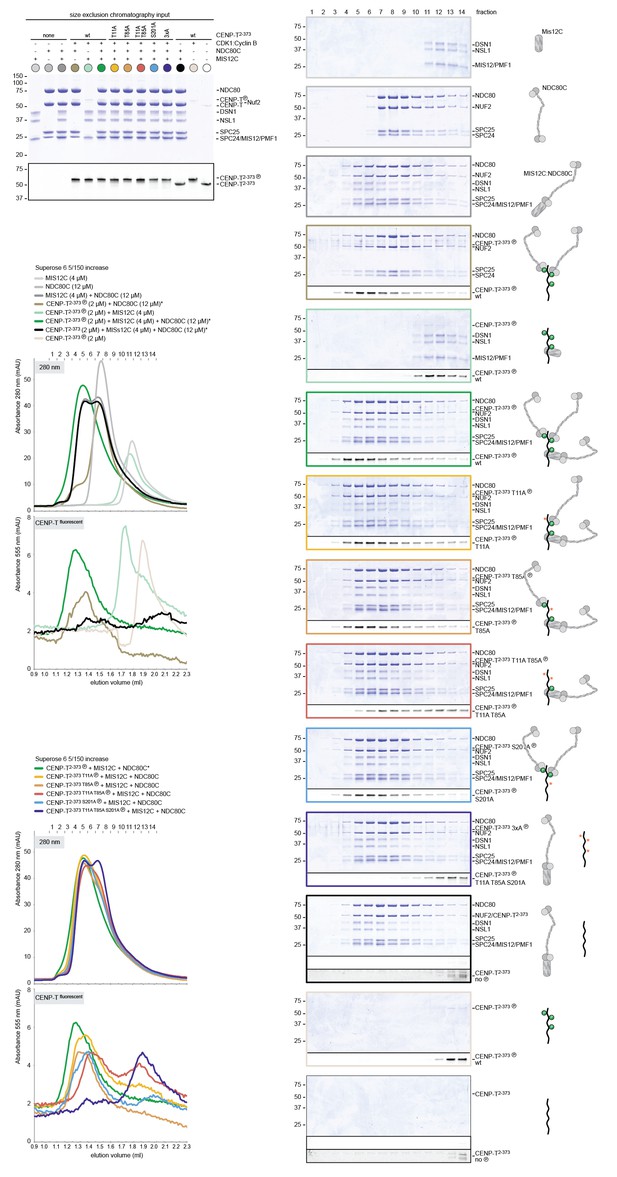
Supercomplex formation depends on the phosphorylation of CENP-T T11, T85, and S201.
A fraction of the mixed samples was analyzed by SDS-PAGE prior to size-exclusion chromatography. The phosphorylation-induced shift of CENP-T is clearly visible, also for the triple CENP-T mutant. Analytical size-exclusion chromatography (Superose 6 5/150 increase) and SDS-PAGE show that phosphorylation by CDK1:Cyclin B of CENP-T at positions T11, T85, and S201 is required for the formation of CENP-T:MIS12C:NDC80C3 assemblies. Data for MIS12C:NDC80C alone (grey) and various phosphorylated CENP-T mutants (green, red, blue, purple) are part of Figure 6.
CENP-T phosphorylated at position S201 can recruit the KMNZ network.
A fraction of the mixed samples was analyzed by SDS-PAGE prior to size-exclusion chromatography. The phosphorylation-induced shift of CENP-T is clearly visible. Analytical size-exclusion chromatography (Superose 6 5/150 increase) and SDS-PAGE show that phosphorylation by CDK1:Cyclin B enables the formation of CENP-TT11A T85A:KNL1:MIS12C:NDC80C:ZWINT assemblies.

Gallery and measurements of MIS12C:NDC80C.
(A) MIS12C:NDC80C complexes were visualized after glycerol spraying followed by low-angle metal shadowing. The three micrographs in the top row are also shown in Figure 6B. (B) Gallery showing 64 MIS12C:NDC80C complexes. Scale bars represent 50 nm. (C) End-end and coiled coil distances were measured for the indicated number of shadowed MIS12C:NDC80C complexes. The values shown here are not corrected for an estimated low-angle shadowing contribution of 3 nm at either end.

Galleries of CENP-T:MIS12C:NDC80C.
CENP-T:MIS12C:NDC80C complexes were visualized after glycerol spraying followed by low-angle metal shadowing. The orange-boxed CENP-T:MIS12C:NDC80C3 micrographs are shown in Figure 6B. The number of shown CENP-T:MIS12C:NDC80C3 (panel A; 64), CENP-T:MIS12C:NDC80C2 (panel B; 32), and CENP-Tx:MIS12Cx:NDC80C>3 (panel C; 16) micrographs is roughly indicative of the relative abundance of the various assemblies. Scale bars 50 nm.
CENP-T and CENP-C are competitive binders of the MIS12 complex.
(A) The binding of phosphorylated CENP-T to MIS12CNano, a version of MIS12C designed for structural analysis (Petrovic et al., 2016), is abolished by the mutation of Tyr8, Phe12, and Phe13 to alanine in the MIS12 subunit. (B) Fluorescent polarization was used to determine the displacement of CENP-C1-21 from MIS12CNano upon the addition of either CENP-C1-71 or, at higher concentrations, CENP-T195-215. (C) Analytical size-exclusion chromatography (Superdex 200 5/150) shows that phosphorylated CENP-T is displaced from MIS12C when incubated with CENP-C, but not when incubated with a version of CENP-C that cannot bind to MIS12C. Fluorescently labeled CENP-T was monitored specifically during chromatography. Red asterisks indicate K10A and Y13A mutations in CENP-C. See Figure 7—figure supplement 2 for the binding of CENP-C and CENP-CK10A Y13A to MIS12C.
Phosphorylated CENP-T192-215 binds to MIS12C.
A binding affinity of approximately 30 μM between MIS12CNano and CENP-T192-215 phosphorylated at position S201 was determined by fluorescence polarization. Non-phosphorylated CENP-T192-215 as well as a mutated version of MIS12C (see Figure 7A) do not form a complex.
CENP-C with K10A and Y13A mutations does not bind to MIS12C.
As reported earlier (Screpanti et al., 2011), analytical size-exclusion chromatography (Superdex 200 5/150) shows that CENP-C2-545, but not CENP-C2-545 K10A Y13A, forms a complex with MIS12C.
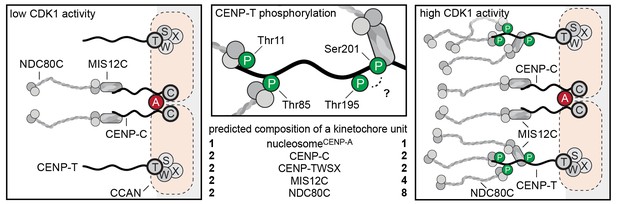
Molecular basis of stoichiometric assembly of microtubule binders in the outer kinetochore.
Schematic representation of the results presented here and their implications. CDK activity results in the recruitment of NDC80C and MIS12C:NDC80C to CENP-T. The predicted stoichiometry of a single kinetochore unit is indicated. In the middle panel, we summarize the role of T11, T85, and S201 in recruitment of NDC80C and MIS12C, and suggest that T195 may contribute to phosphorylation of S201 in vivo.
Tables
Phospho-sites on CENP-T2-373 in vitro phosphorylated by CDK1:Cyclin B. Identified phospho-peptides with a localization probability for the phospho-site >80% and an Andromeda search engine score >100 are shown. All peptides have a posterior error probability (PEP) below 1 × 10−25. The last column indicates if sites were identified as single (S) and/or double (D) phospohorylated peptides.
| Position | Peptide with pS or pT | Score | P-sites |
|---|---|---|---|
| 8 | MADHNPDpSDSpTPRTLLR | 144 | S and D |
| 10 | MADHNPDSDpSTPRTLLR | 209 | S |
| 11 | MADHNPDpSDpSpTPRTLLRR | 209 | S and D |
| 27 | VLDTADPRpTPR | 135 | S |
| 45 | RALLEpTApSPRK | 112 | D |
| 47 | RALLEpTApSPRK | 112 | S and D |
| 78 | SAHIQApSGHLEEQpTPR | 101 | S and D |
| 85 | SAHIQApSGHLEEQpTPR | 167 | S and D |
| 184 | LSVFQQGVDQGLSLSQEPQGNADASpSLTR | 129 | S |
| 195 | SLNLTFApTPLQPQpSVQRPGLAR | 152 | S and D |
| 201 | SLNLTFApTPLQPQpSVQRPGLAR | 186 | S and D |





