Translation initiation by the hepatitis C virus IRES requires eIF1A and ribosomal complex remodeling
Figures
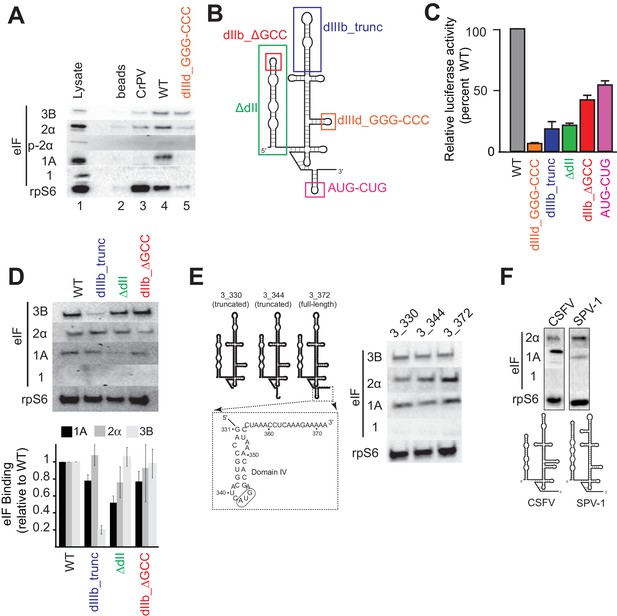
Analysis of HCV IRES preinitiation complex composition, formed in lysate.
(A) Results of pull-down experiments. HCV IRES was 5’ biotinylated and immobilized on ’ streptavidin agarose beads. Factors and subunits that bound to the IRES after incubation in lysate were detected by western blot analysis. Lane 1: Input lysate. Lane 2: streptavidin beads only. Lane 3: CrPV IGR IRES RNA control. Lane 4: WT HCV IRES RNA. Lane 5: HCV IRES mutant dIIId_GGG-CCC. (B) Secondary structure cartoon of the HCV IRES RNA, with the location of mutations indicated. (C) Translation initiation activity of the HCV IRES, WT and mutants, with RNA transfections of Huh 7.5 cells. Error bars represent averages ±SEM of ≥3 independent experiments. (D) Pull-downs as in panel (A) with HCV IRES mutants shown in panel (B). Blots were quantitated by densitometry, and the intensity of each eIF band was normalized to the rpS6 band. Graph shows the average of three independent experiments with the amount of each eIF bound to WT set to 1. Error bars represent one standard error from the mean. (E) Pull-downs as in panel (A) with truncated HCV IRESs. Cartoon diagrams of these three truncations are shown, as is the portion of the IRES in which these truncations were made. Visual examination shows a similar qualitative pattern. (F) Pull-downs as in panel (A) with the IRESs from CSFV and SPV-1, along with corresponding secondary structure cartoons.
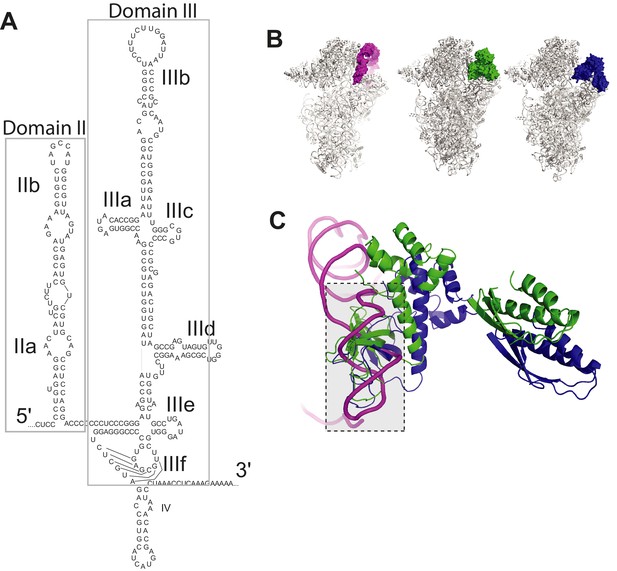
HCV IRES structure and clash between HCV IRES domain II and eIF2.
(A) Secondary structure of the HCV IRES, with domains labeled. (B) Left: Cryo-EM reconstructions of human 40S subunit in complex with the HCV IRES (magenta, PDB: 5A2Q) Middle: yeast PIC in the open conformation (PDB: 3JAQ) with all factors but eIF2 (green) removed. Right: yeast PIC in closed conformation (PDB: 3JAP) and eIF2 (blue). (C) Steric clash between HCV IRES dII and eIF2.
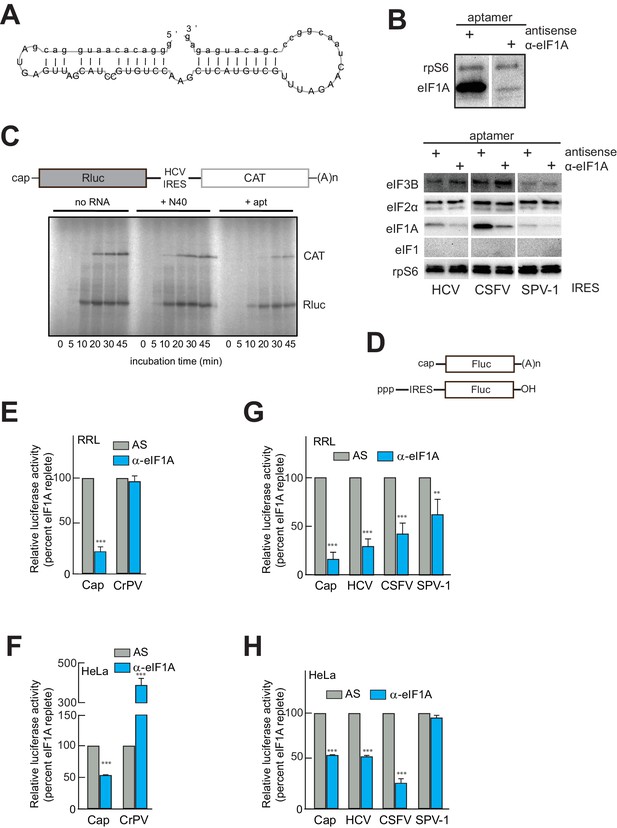
An RNA aptamer targeting eIF1A for inactivation negatively affects both cap- and IRES-driven translation in lysate.
(A) Sequence and predicted secondary structure of the α-eIF1A aptamer used in this study. (B) Top: Results of pull-down experiment with the HCV IRES (similar to Figure 1), showing substantial depletion of eIF1A with aptamer treatment compared to negative control antisense (AS) aptamer RNA. Bottom: Pull-down experiments with several IRES RNAs, with blotting to detect eIFs that should not be removed or depleted by the aptamer. (C) 35S-Met labeling of translation products in RRL using a dual-reporter mRNA template. The aptamer treatment reduces protein levels over time from IRES-driven (CAT) and cap-driven (Rluc) messages. The effect of apatamer (+ apt) addition is shown compared to the addition of no RNA or a randomized 40 nucleotide RNA (+ N40). (D) Diagram of monocistronic reporters used in the experiments of panels (E–H). (E) Results of translation assays in RRL using reporter RNAs. The level of each RNA in untreated lysate is set at 100% and the effect of aptamer treatment is reported as a percentage of that for each RNA. Capped (Cap) RNA is the positive control and the CrPV IRES is the negative control for a requirement for eIF1A. (F) Identical to panel (E) but assayed in HeLa cell extract. (G) Translation assays in RRL using reporter RNAs with the HCV, CSFV, and SPV-1 IRESs. Cap is included as a control. (H) Identical to panel (G) but assayed in HeLa cell extract. In panels (E–H), error bars represent averages ±SEM of ≥3 independent experiments. Statistical significance shown by: *p<0.05, **p<0.01, ***p<0.001.
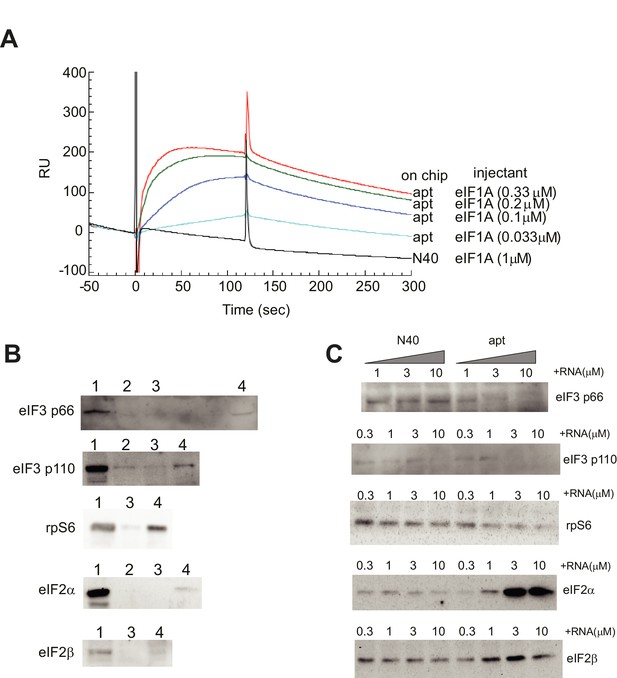
Validation of α-eIF1A aptamer.
(A) Binding profiles of the eIF1A aptamer using surface plasmon resonance. The aptamer RNA (apt) or the ‘N40’ random sequence RNA was immobilized on a sensor chip for Biacore2000 analysis. eIF1A solution was prepared and was injected at time 0 for 120 s. From the fitting of these sensorgrams by the instrument software BIAevaluation, the dissociation constant (Kd) of the aptamer-eIF1A interaction was estimated as 5.2 nM. (B) Pulldown assay of translation factors bound to eIF1A. The following samples were loaded on each lane: lane 1, 1 μl of HeLa cell extract; lane 2, coprecipitated sample by Ni-NTA agarose; lane 3, coprecipitated sample by Ni-NTA agarose with His-CAT; and lane 4, coprecipitated sample by Ni-NTA agarose with 6XHis-eIF1A. Separated and transferred proteins are detected by Western blotting using antibodies against eIF3 p66, eIF3 p110, rpS6, eIF2α and eIF2β (from upper to lower). These proteins bound with eIF1A specifically. (C) Pull-down assay from HeLa cell extracts using Ni-NTA agarose and 6XHis-eIF1A, with 0.3, 1, 3 or 10 μM N40 random RNA (N40) or 0.3, 1, 3 or 10 μM aptamer (labeled). Two components of eIF3, p66 and p110, plus rpS6, eIF2α and eIF2β bound to eIF1A. When aptamer was added to this pull-down reaction, the precipitation of eIF3 components and rpS6 was reduced, suggesting that the aptamer blocked the interaction of eIF1A with eIF3 and rpS6.
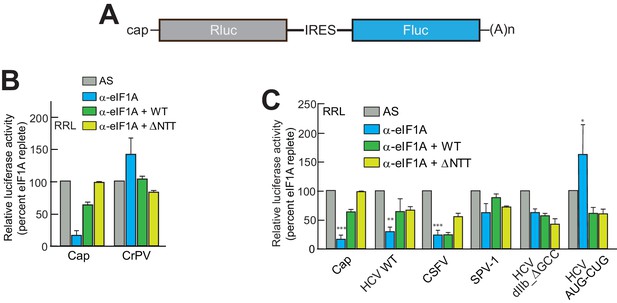
eIF1A depletion inhibits translation from the HCV IRES.
(A) Cartoon showing design of bicistronic dual-luciferase reporter constructs used in the experiments of panels (B–C). (B) Effect of depletion of eIF1A using the aptamer followed by add-back of WT full-length eIF1A (WT) or an N-terminally truncated mutant (ΔNTT) on translation from the reporter shown in panel (A). Experiments were done in RRL. This panel shows the effect on the Rluc (Cap) and a control CrPV IRES. (C) Same as panel (B) but with WT HCV, CSFV, and SPV-1 IRESs, plus two HCV IRES mutants shown in Figure 1B. For panels (B and C), activity in lysate treated with AS control aptamer was set at 100%. Error bars represent averages ±SEM of ≥3 independent experiments. Statistical significance shown by: *p<0.05, **p<0.01, ***p<0.001.
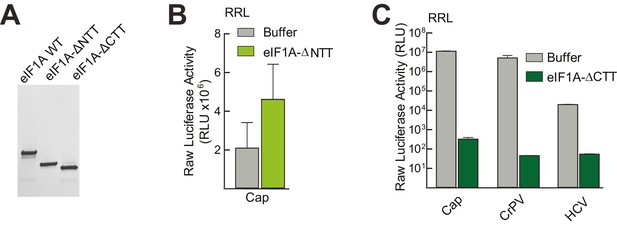
Purity of WT and mutant eIF1A and effects in untreated lysate.
(A) Recombinantly expressed and purified WT, N-terminally truncated (ΔNTT), and C-terminally truncated (ΔCTT) human eIF1A shown on a Coomassie-stained SDS-PAGE gel. (B) Translation assays in RRL using a cap-driven message (Figure 3A) with or without the addition of ΔNTT, without aptamer treatment. (C) Translation assays in RRL showing potent translation inhibition by adding ΔCTT. Monocistronic reporters with cap-, CrPV IRES-, or HCV IRES-driven translation are shown. Translation level is shown as raw light units from Fluc. Error bars represent averages ±SEM of ≥3 independent experiments.
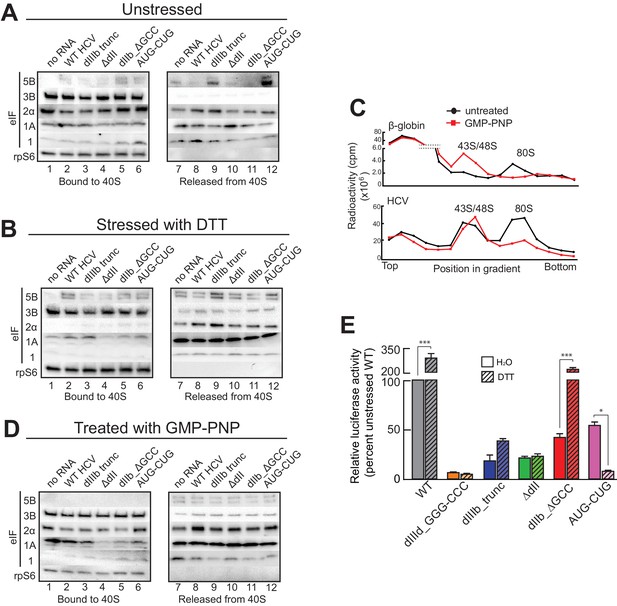
HCV IRES binding alters natively purified PICs into complexes suited for IRES-driven translation.
(A, B, and D) Results of the experimental protocol shown in Figure 4—figure supplement 1. Natively purified 40S subunit-containing complexes were challenged with WT HCV IRES, then the composition of the complex (bound to 40S) and the factors released (released from 40S) were analyzed using western blot analysis. Panel (A): PICs from unstressed 293F cells. Panel (B): PICs from 293F cells stressed with dithiothreitol (DTT). Panel (D): PICs from unstressed cells incubated with non-hydrolyzable GTP analog GMP-PNP. (C) 80S ribosome formation on radiolabeled β-globin leader or HCV IRES in treated 293F lysate either untreated or treated with GMP-PNP. Reactions were fractionated by ultracentrifugation through a sucrose gradient. Total radioactivity per fraction is plotted. (E) Translation from bicistronic reporter mRNA (Figure 3A) containing WT or mutant HCV IRES, used to transfect Huh 7.5 cells that were left untreated (H2O control) or pretreated with DTT. Translation of Fluc is plotted relative to translation of WT HCV IRES reporter in unstressed cells (set to 100%). Error bars represent averages ±SEM of ≥3 independent experiments. Statistical significance shown by: *p<0.05, **p<0.01, ***p<0.001.
-
Figure 4—source data 1
Quantified blot data supporting Figure 4A–D.
Tables containing quantified band intensities from blots resulting from multiple biological replicates (n = 3). Blots were quantitated by densitometry, and the intensity of each eIF band was normalized to the rpS6 band. Graph shows the average of three independent experiments with the amount of each eIF bound to 40S subunit in the presence of WT HCV IRES set to 1. Errors represent one standard error from the mean. ND = not detectable.
- https://doi.org/10.7554/eLife.21198.009
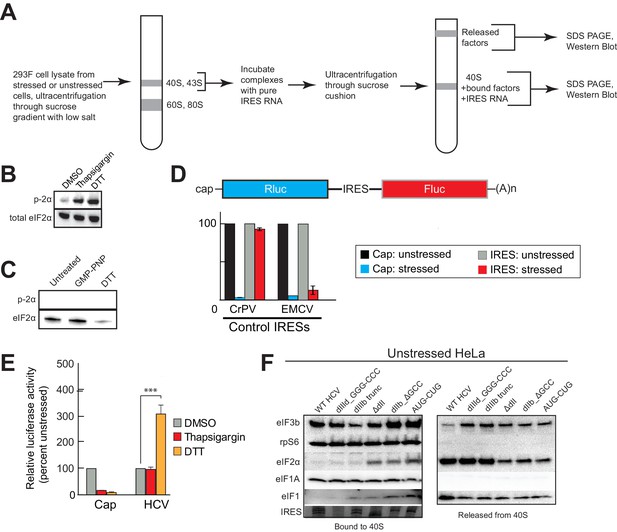
Experimental protocol for analysis of HCV IRES remodeling of natively purified PICs.
(A) Diagram of the PIC preparation strategy and experiment to analyze bound and released factors. (B) Western blot showing effective phosphorylation of eIF2α (p-2α) with both thapsigargin and DTT treatments. (C) Western blot analysis showing the presence of eIF2 in the three PIC preparations presented in Figure 4A,B and D. Inactive, phosphorylated eIF2α did not co-purify with these complexes. (D) Diagram of bicistronic reporter construct and translation activity in Huh 7.5 cells from two control IRESs: The eIF2-independent cricket paralysis virus (CrPV) intergenic region (IGR) IRES, and the eIF2-dependent encephalomyocarditis virus (EMCV) IRES. Treatment of the cells with thapsigargin induced eIF2α phosphorylation and reduced EMCV IRES-dependent translation, while the CrPV IGR IRES retained activity. For each construct, unstressed cap (black) and unstressed IRES (grey) activities are normalized to 100%. Error bars represent averages ±SEM of ≥3 independent experiments. (E) Translation initiation activity of the HCV IRES (compared to cap-driven translation) in Huh 7.5 cells, comparing stress induced by thapsigargin and DTT. Error bars represent averages ±SEM of ≥3 independent experiments. Statistical significance shown by: *p<0.05, **p<0.01, ***p<0.001. (F) Natively purified PICs from unstressed HeLa cells, analyzed for the effect of HCV IRES binding, as in Figure 4A.
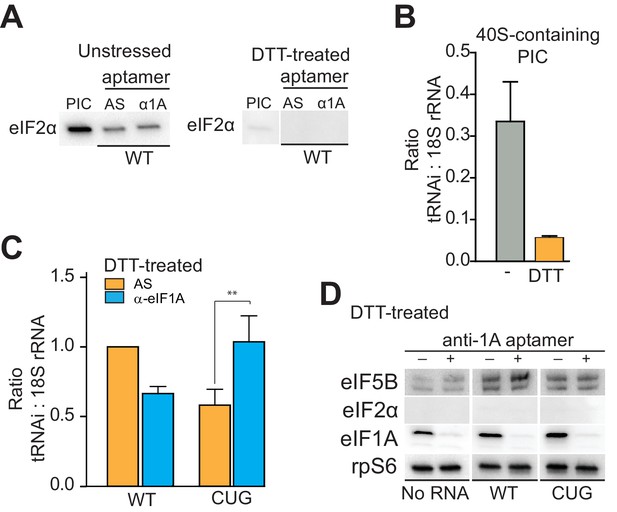
eIF1A stabilizes tRNAiMet binding and inspects the start codon on HCV IRES-bound PICs.
(A) Western analysis of eIF2 bound to natively purified PICs treated with α-eIF1A aptamer. α-eIF1A aptamer treatment has no effect on eIF2 binding in unstressed or DTT-stressed PICs bound with WT HCV IRES. Input PIC shown for comparison. (B) Quantified data from Northern blots measuring the amount of tRNAi bound to PICs from unstressed (grey) or DTT-stressed (orange) cells. PICs were not treated with aptamer or IRES. tRNAi signal presented as a ratio to internal control 18S rRNA signal. Experimental protocol shown in Figure 5—figure supplement 1. (C) Identical to panel (B), but measuring tRNAi binding to PICs treated with AS (orange) or α-eIF1A (cyan) aptamers and bound to WT HCV IRES or the AUG-CUG mutant (CUG). Ratio is expressed as tRNAi compared to 18S rRNA signal, and the signal from untreated, HCV IRES-bound PICs set to 1.0. Error bars represent averages ±SEM of ≥3 independent experiments. Statistical significance shown by: *p<0.05, **p<0.01, ***p<0.001. (D) Analysis of factors bound to PICs from DTT-stressed cells, depleted of eIF1A. Western blots to various factors are shown. These are the same samples analyzed in panel (C).
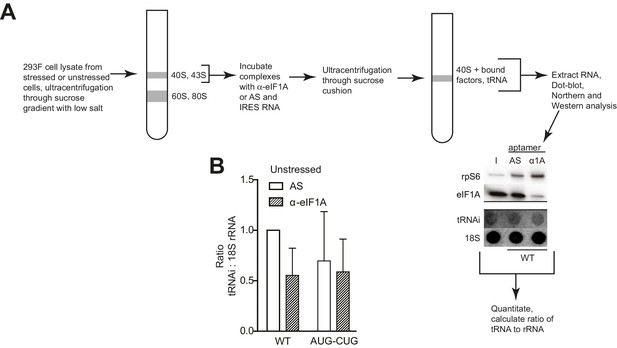
PIC preparation strategy for northern blotting quantification.
(A) Diagram of the PIC preparation strategy and experiment to analyze the amount of tRNAi bound to natively purified PICs. Example of Western and Northern blots used for analysis shown. (B) Quantification of tRNAi bound to PICs from unstressed cells (eIF2-bound PICs). In the presence of eIF2, eIF1A depletion had negligible effects on tRNAiMet from WT and start mutant (AUG-CUG) HCV IRESs. Error bars represent averages ±SEM of ≥3 independent experiments.
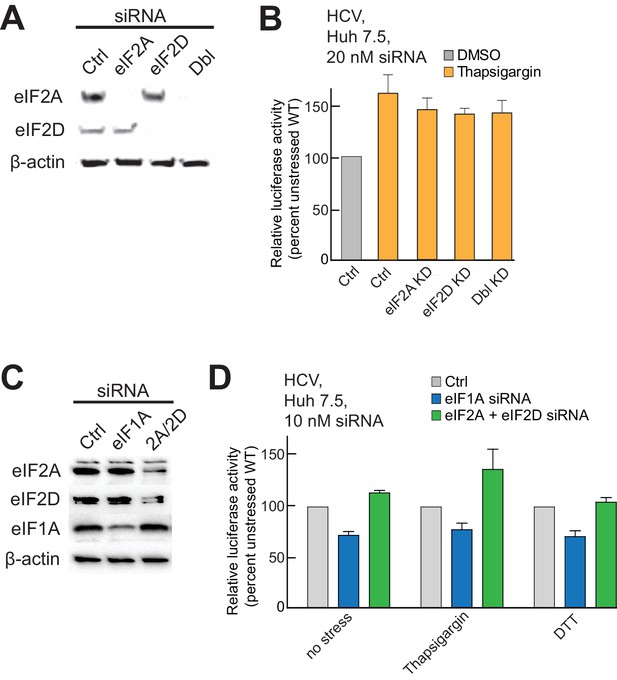
Presence of alternate eIFs 2A and 2D in PICs, and effect of knockdowns.
(A) Analysis of siRNA-induced knockdown of eIF2A and eIF2D and double knockdown in Huh 7.5 cells with β-actin as a loading control. [siRNA] = 20 mM. (B) Activity of WT HCV IRES in eIF2A- and eIF2D-depleted cells, treated with thapsigargin. ‘Ctrl’ is from cells treated with non-targeting siRNA. Error bars represent averages ±SEM of ≥3 independent experiments. (C) Analysis of siRNA-induced double knockdown of eIF2A and eIF2D in Huh 7.5 cells. eIF1A knockdown is included as a control. [siRNA] = 10 mM, as higher concentrations resulted in knockdown of eIF1A that caused cell death. (D) Activity of WT HCV IRES in doubly eIF2A- and eIF2D-depleted cells and in cells depleted of eIF1A. Cells were treated with either thapsigargin or DTT. ‘Ctrl’ is from cells treated with non-targeting siRNA. Error bars represent averages ±SEM of ≥3 independent experiments. Lowering eIF1A concentrations results in decreased HCV IRES activity, but depletion of eIF2A+D has no effect.
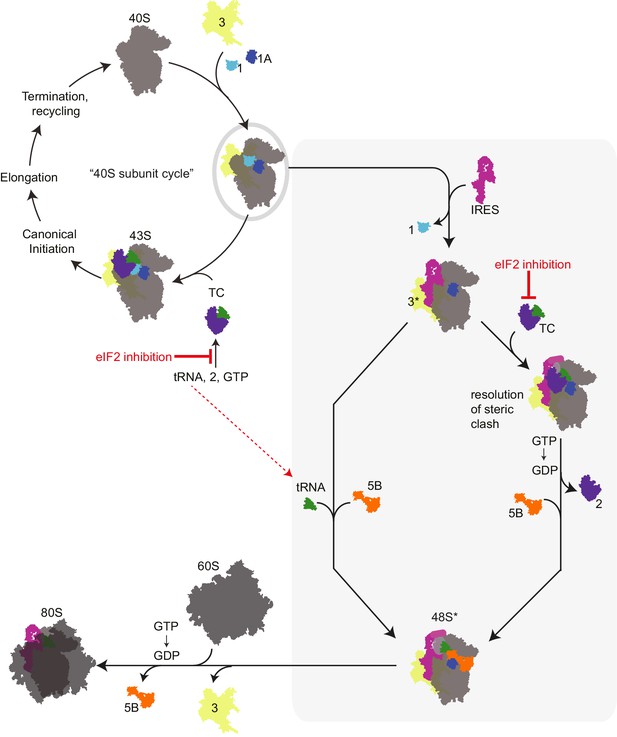
Revised mechanism for HCV IRES-driven initiation.
This model is described in the main text. Briefly, we propose that the HCV IRES (and probably other type 3 IRESs) recruits a pre-assembled ‘TC-deficient’ PIC containing eIF1, 1A, and 3 (grey oval), which is an intermediate in the normal 40S recycling process. IRES binding to this PIC partially remodels it to include displacing eIF3 (yellow, ‘3*’) and either passively or actively releasing eIF1. This remodeled PIC is used under both eIF2 active and inactive conditions (grey shaded box). When active eIF2 is abundant, delivery of tRNA through the TC presumably requires movement of IRES domain II to an alternate position (Yamamoto et al., 2014) to resolve a steric clash (Figure 1—figure supplement 1B and C). If eIF2 is inhibited, this would have the effect of increasing the amount of available ‘TC-deficient’ PICs and perhaps free tRNAi. While we cannot prove that an ‘alternate’ factor is not used to deliver tRNAi under these conditions, we assert that available evidence supports a mode of tRNAi recruitment using eIF1A and eIF5B..





