The neuropeptide F/nitric oxide pathway is essential for shaping locomotor plasticity underlying locust phase transition
Figures
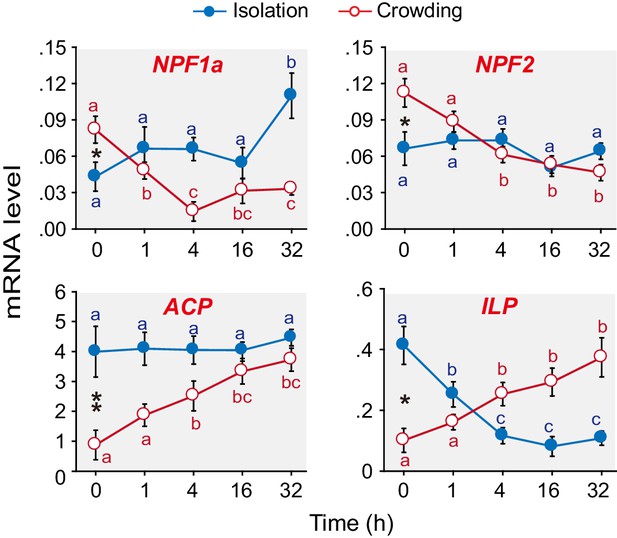
Levels of transcripts encoding the neuropeptides NPF1a, NPF2, ACP and ILP change during the G/S phase transition in the migratory locust.
qPCR was performed to determine the transcript levels of 15 neuropeptide-encoding genes in locust brains in the time course of the isolation of gregarious (G-phase) locusts or the crowding of solitarious (S-phase) locusts. Four neuropeptide genes displayed clear expression changes during isolation or crowding or both (in the case of NPF1a and ILP). Raw data measuring the mRNA levels of the four neuropeptide genes are shown in Figure 1—source data 1. For the transcript levels of the other 11 neuropeptide genes, see Figure 1—figure supplement 1. The data are presented as mean ± s.e.m. Significant differences at different times are denoted by letters (n = 4 samples per timepoint, 8 animals/sample, one-way ANOVA, p<0.05). *indicates a significant difference between typical G-phase (0 hr after isolation) and typical S-phase (0 hr after crowding) locust brains (Student’s t-test, *p<0.05, **p<0.01).
-
Figure 1—source data 1
mRNA levels of the four neuropeptide-encoding genes during isolation and crowding processes.
- https://doi.org/10.7554/eLife.22526.004
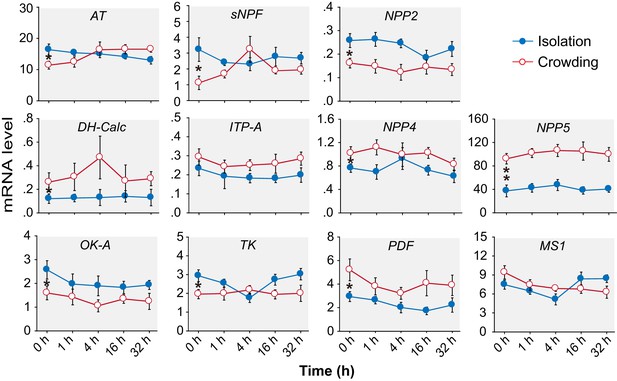
The transcript levels of 11 neuropeptides do not change during the G/S phase transition in the migratory locust.
Isolation is shown in blue; crowding is shown in red. The data are presented as mean ± s.e.m., *indicates a significant difference between typical G-phase (0 hr after isolation) and typical S-phase (0 hr after crowding) locust brains (n = 4 samples, 8 locusts/sample, one-way ANOVA for multi-group comparisons, Student’s t-test for two-group comparison, *p<0.05, **p<0.01).
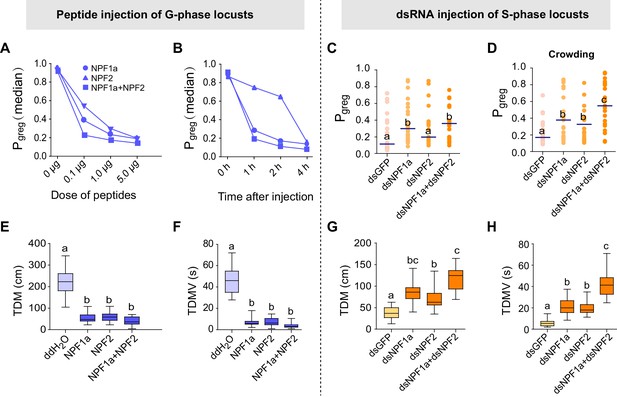
Perturbations of NPF1a or NPF2 peptide levels or of their transcript levels leads to changes in locomotor activity related to the G/S phase transition.
Locust behaviors are measured by the term Pgreg, which is a combined assessment of movement and inter-insect attraction (indicated as attraction index, see Figure 2—figure supplement 2). Pgreg = 0 represents a fully S-phase behavioral state; Pgreg = 1 represents a fully G-phase behavioral state. (A) and (B) Dose- and time-dependent changes in the median Pgreg of G-phase locusts after injection of NPF1a and NPF2 peptides, separately and together. For detailed Pgreg distributions and statistics, see Figure 2—figure supplement 2 (n ≥ 18 locusts, Mann–Whitney U test, p<0.05). (C) Pgreg in S-phase locusts 48 hr after transcript knockdown of NPF1a, or NPF2, or both (n ≥ 20 locusts, Mann–Whitney U test, p=0.020, 0.064 and 0.017, respectively). Lines indicate median Pgreg. Significant differences are denoted by letters. (D) Pgreg in crowded S-phase locusts after transcript knockdown of NPF1a, or NPF2, or both (n ≥ 20 locusts, Mann–Whitney U test, p=0.024, 0.039 and 0.037, respectively). Locusts were forced into a crowd 32 hr after dsRNA injection, and their behaviors were measured after 16 hr of crowding (that is 48 hr after dsRNA injection). (E) and (F) Total distance moved (TDM) and total duration of movement (TDMV) 4 hr after injection of NPF1a or NPF2 or both peptides in G-phase locusts (5 μg/individual). The data are presented as mean ± s.e.m. Significant differences are denoted by letters (n ≥ 18 locusts, one-way ANOVA, p<0.05). (G) and (H) Total distance moved (TDM) and total duration of movement (TDMV) 48 hr after transcript knockdown of NPF1a or NPF2 or both genes in S-phase locusts (n ≥ 20 locusts).
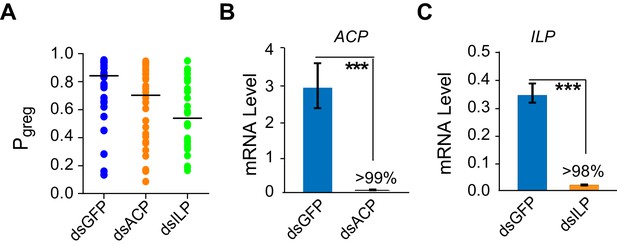
Transcript knockdown of ACP or ILP does not significantly affect behavioral phase state in G-phase locusts.
(A) Behavioral tests (measured as Pgreg) after transcript knockdown of ACP or ILP in G-phase locusts (n ≥ 27 locusts, Mann–Whitney U test, p=0.053 and 0.042 for dsACP and dsILP injection, respectively). (B) and (C) Efficiency and specificity of ACP and ILP transcript knockdown in G-phase locusts. RNAi effects were examined by qPCR 48 hr after dsRNA injection. The data are presented as mean ± s.e.m. (n = 4 samples, 6–8 locusts/sample, Student’s t-test, ***p<0.001).
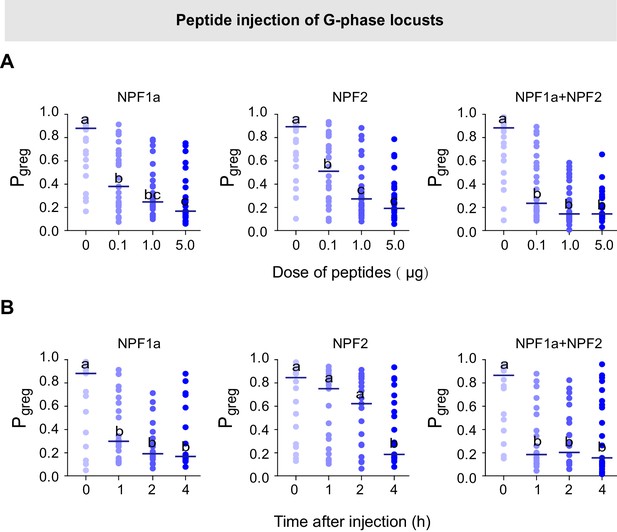
Injection of NPF1a or NPF2 peptide into G-phase locusts induces S-phase-like behaviors in a dose- and time-dependent manner.
(A) Dose-dependent effects on the Pgreg values of G-phase locusts 4 hr after injection of NPF1a and NPF2 peptides, separately and together. Significant differences between treatments are denoted by letters (n ≥ 20 locusts, Mann–Whitney U test, p=0.002, 0.006 and 0.000 for 0.1, 1 and 5 μg NPF1a peptide injections; p=0.031, 0.008 and 0.001 for 0.1, 1 and 5 μg NPF2 peptide injections; p=0.004, 0.000 and 0.000 for 0.1, 1 and 5 μg NPF1a and NPF2 co-injections, respectively). (B) Time-dependent effects on the Pgreg values of G-phase locusts after injection of NPF1a and NPF2 peptides, separately and together. Significant differences are denoted by letters (n ≥ 18 locusts, Mann–Whitney U test, p=0.002, 0.006 and 0.000 for 1, 2 and 4 hr NPF1a peptide injections; p=0.031, 0.008 and 0.001 for 1, 2 and 4 hr NPF2 peptide injections; p=0.004, 0.000 and 0.000 for 1, 2 and 4 hr NPF1a and NPF2 co-injections, respectively).
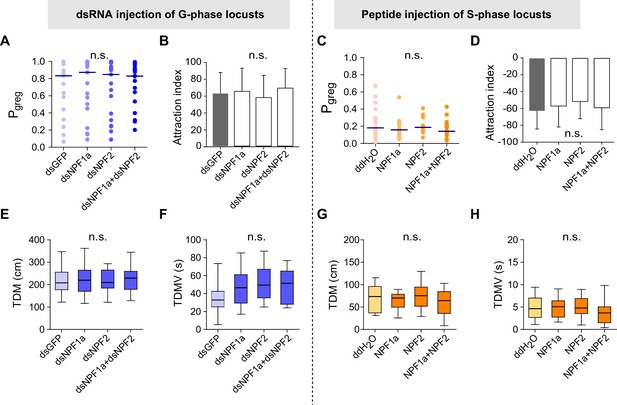
Transcript knockdown of NPF1a or NPF2 in G-phase locusts and peptide injection of NPF1a or NPF2 in S-phase locusts do not affect phase-related behaviors.
Attraction index (AI) represents the extent by which the tested animals are attracted by the stimulus group (AI = total duration in stimulus area − total duration in the opposite of stimulus area). (A) Pgreg of G-phase locusts 48 hr after transcript knockdown of NPF1a and NPF2, separately and together (n ≥ 18 locusts, Mann–Whitney U test, p=0.426, 0.584 and 0.636, respectively). Lines indicate median Pgreg. n.s. indicates no significant difference. (B) Attraction index of G-phase locusts 48 hr after transcript knockdown of NPF1a and NPF2, separately and together (n ≥ 18 locusts, one-way ANOVA). (C) Pgreg of S-phase locusts 4 hr after injection of NPF1a or NPF2 or both peptides (n ≥ 18 locusts, Mann–Whitney U test, p=0.381, 0.939 and 0.475, respectively). (D) Attraction index of S-phase locusts 4 hr after injection of NPF1a or NPF2 or both peptides (n ≥ 18 locusts, one-way ANOVA). (E) and (F) Total distance moved (TDM) and total duration of movement (TDMV) 48 hr after transcript knockdown of NPF1a or NPF2 or both genes in G-phase locusts (n ≥ 18 locusts, one-way ANOVA). (G) and (H) Total distance moved (TDM) and total duration of movement (TDMV) 4 hr after injection of NPF1a or NPF2 or both peptides in S-phase locusts. (n ≥ 18 locusts, one-way ANOVA).
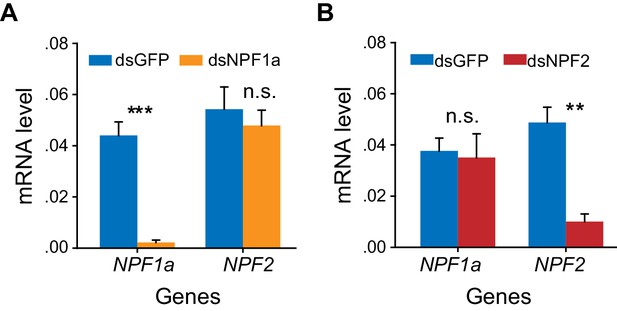
Efficiency and specificity of NPF1a and NPF2 transcript knockdown.
The S-phase locusts were injected with dsNPF1a or dsNPF2 or dsGFP. RNAi effects were examined by qPCR 48 hr after injection. The data are presented as mean ± s.e.m. (n = 4 samples, 6–8 locusts/sample, Student’s t-test, **p<0.01; ***p<0.001). n.s. indicates no difference in a paired comparison.
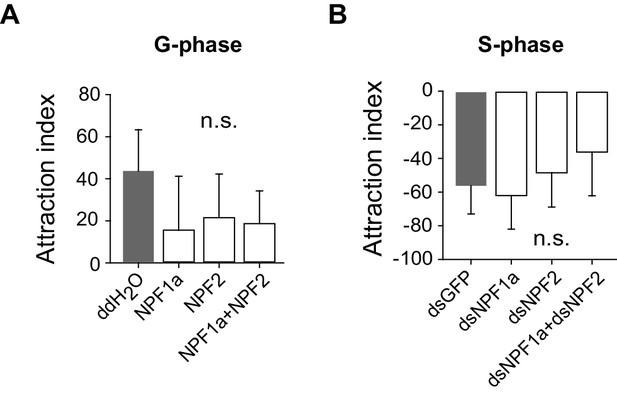
Perturbation of NPF1a or NPF2 peptide, or of their transcript levels, do not change attraction index related to the G/S phase transition.
(A) Effects on attraction index of G-phase locusts 4 hr after injection of NPF1a or NPF2 or both peptides. The data are presented as mean ± s.e.m. (n ≥ 20 locusts, Student’s t-test). n.s. means not significant. (B) Effects on attraction index of S-phase locusts 48 hr after transcript knockdown of NPF1a or NPF2 or both genes (n ≥ 20 locusts, Student’s t-test).
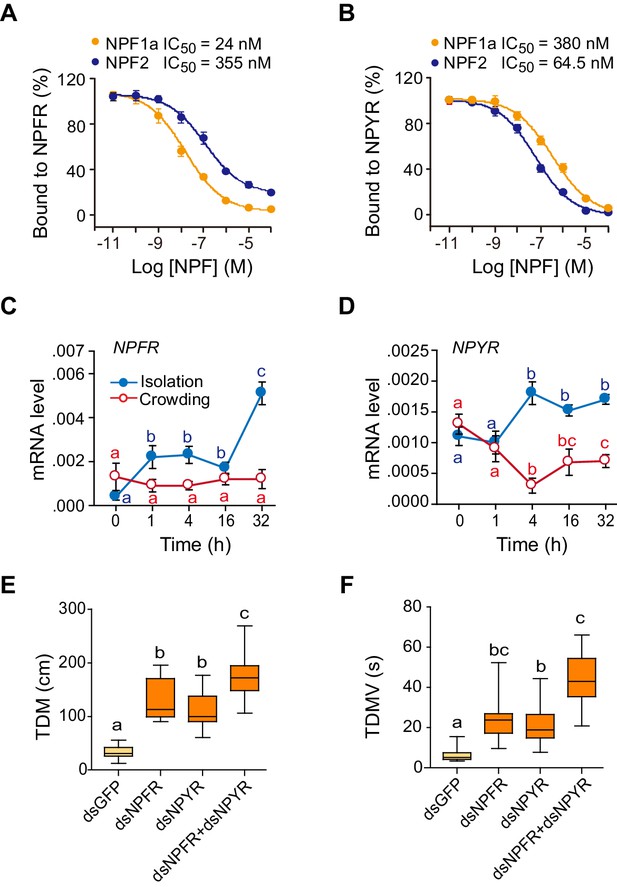
Receptors for NPF1a and NPF2 are involved in transmitting the effects of these neuropeptides on locomotor activity.
(A) Competitive inhibition of TAMRA-NPF1a binding to HEK 293 T cells transfected with pcDNA3.1-NPFR vector (n = 6). (B) Competitive inhibition of TAMRA-NPF2 binding to HEK 293 T cells transfected with pcDNA3.1-NPYR vector (n = 6). (C) and (D) Time course patterns of NPFR and NPYR transcript levels during the G/S locust phase transition (isolation, shown in blue; crowding, shown in red). The data are presented as mean ± s.e.m (n = 4 samples per timepoint, 8 locusts/sample, one-way ANOVA, p<0.05). Detailed expression levels of the two NPF receptors are shown in Figure 3—source data 1. (E) and (F) Total distance moved (TDM) and total duration of movement (TDMV) 48 hr after transcript knockdown of NPFR or NPYR or both genes in S-phase locusts. Significant differences are denoted by letters (n ≥ 19 locusts, one-way ANOVA, p<0.05).
-
Figure 3—source data 1
Transcript levels of NPFR and NPYR during the G/S locust phase transition.
- https://doi.org/10.7554/eLife.22526.013

Phylogenetic relationship of NPF or NPY precursors and their receptors in different species.
(A) Phylogenetic relationship of NPF and NPY precursors. The NPF precursors were obtained from Acyrthosiphon pisum, Aedes aegypti, Anopheles gambiae, Apis mellifera, Aphis gossypii, Bombyx mori, Culex pipiens, Drosophila melanogaster, Danaus plexippus, Nilaparvata lugens, Nasonia vitripennis, Schistocerca schistostatin, and Locusta migratoria; mammalian NPY precursors were obtained from Ovis aries, Mus musculus, and Homo sapiens. Most insect NPF peptides share an identical phenylalanine (F) instead of tyrosine (Y) at their C-terminus. The migratory locust genome contains two NPF1 precursors (NPF1a and NPF1b) and an NPF2 precursor. NPF1b, which encodes an 86 aa peptide, is hardly detected in the locust brain because of its extremely low expression level (Hou et al., 2015). (B) Phylogenetic relationship between insect NPF/Y receptors and human NPY recepetors. The receptors are obtained from A. pisum, A. aegypti, A. florea, B. mori, Culex quinquefasciatus, D. melanogaster, T. castaneum and L. migratoria, together with mammalian NPY receptors from H. sapiens. The locust NPFR is close to 'type two' mammalian NPYR (Homo Y2).
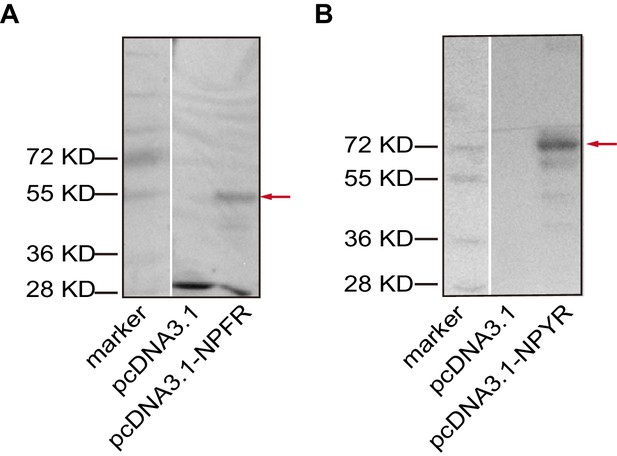
Overexpressions of (A) NPFR and (B) NPYR in HEK 293 T cells validated by western blot.
DNA fragments encoding either NPFR (492 aa) or NPYR (641 aa), followed by a Flag-tag, were inserted to a pcDNA3.1-expressing vector. Red arrow indicates target protein. The total protein of cells that were transiently transfected with pcDNA3.1-NPFR or pcDNA3.1-NPYR was used for western blot analysis. The HEK 293 T cells transfected with pcDNA3.1 were used as a control. Mouse monoclonal antibody against Flag (CoWin, 1:5000) was used to validate the expression of the two receptors.
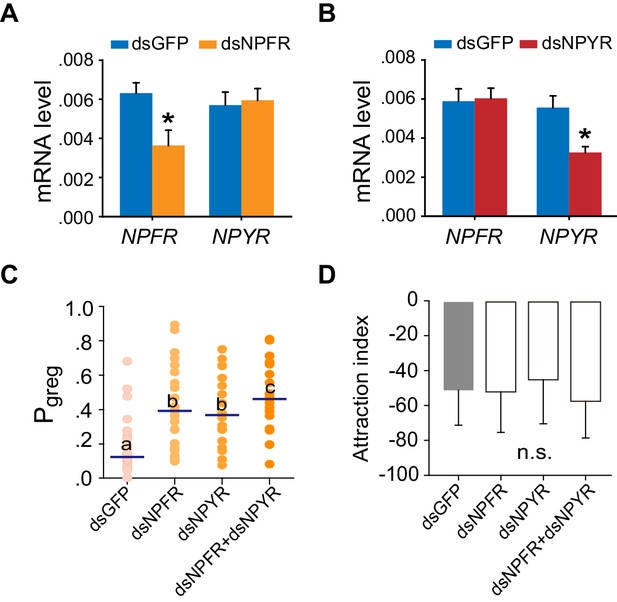
Transcript knockdown of NPFR or NPYR in S-phase locusts induces G-phase-like behaviors without affecting attraction index.
(A) and (B) Efficiency and specificity of NPFR and NPYR transcript knockdown examined by qPCR. dsNPFR or dsNPYR or dsGFP was microinjected into the locust brains. The effects of RNAi were analyzed 48 hr after dsRNA injection. The data are presented as mean ± s.e.m. (n = 4 samples, 6–8 locusts/sample, Student’s t-test, *p<0.05). (C) Effects on Pgreg of S-phase locusts 48 hr after transcript knockdown of NPFR or NPYR or both genes. Lines indicate median Pgreg. Significant differences are denoted by letters (n ≥ 19 locusts, Mann–Whitney U test, p=0.0005, 0.0015 and 0.0002 for dsNPFR, dsNPYR, or dsNPFR and dsNPYR injection, respectively). (D) Attraction index of S-phase locusts 48 hr after transcript knockdown of NPFR or NPYR or both genes. The data are presented as mean ± s.e.m. n.s. indicates not significant (n ≥ 19 locusts, Student’s t-test).
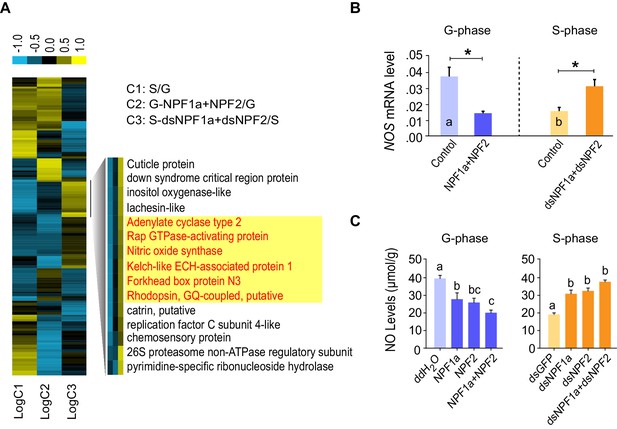
Cluster analysis of RNA-Seq data leads to the identification of nitric oxide synthase (NOS) as a downstream component of the NPF1a and NPF2 pathway.
(A) Cluster analysis of differentially expressed genes in the transcriptome. Several important genes (highlighted in yellow) involved in signaling in other organisms display expression patterns that correlate with behavioral change after the manipulation of NPF1a and NPF2 peptides or transcript levels. Logarithmic fold alteration of treatment versus control is shown in the heat map. Yellow and blue colors indicate up- and downregulation, respectively (n = 3 samples per treatment, 10 animals/sample). For detailed gene-expression data, please see Figure 4—source data 1. (B) Transcript levels of NOS in the brains after co-injection of NPF1a and NPF2 peptides in G-phase locusts or transcript knockdown of both NPF1a and NPF2 in S-phase locusts (n = 5 samples, 8 locusts/sample, Student’s t-test, *p<0.05, different letters labeled in columns indicate a significant difference). (C) NO levels after injection of NPF1a and NPF2 peptides, separately and together, in G-phase locusts, or after transcript knockdown of NPF1a and NPF2, separately and together, in S-phase locusts. The data are presented as mean ± s.e.m. Significant differences are denoted by letters (n = 4 samples, 16 locusts/sample, one-way ANOVA, p<0.05).
-
Figure 4—source data 1
The effects of NPF1a and NPF2 on the expression of annotated genes in the brains of fourth-instar locusts.
- https://doi.org/10.7554/eLife.22526.018
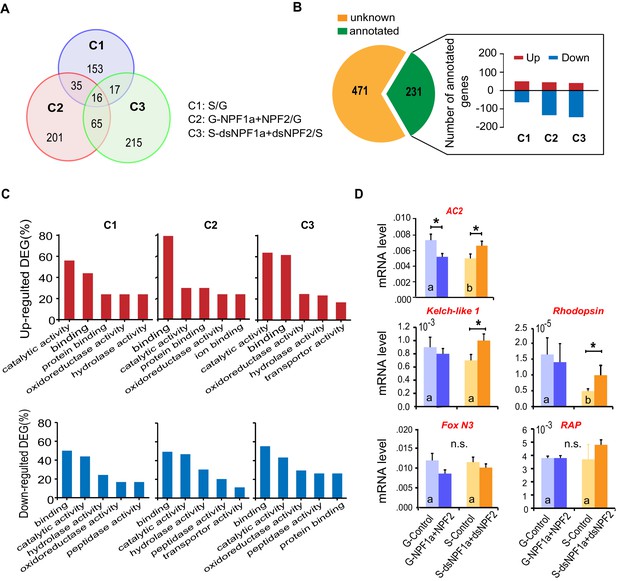
Transcriptomic profiles influenced by NPF1a and NPF2 in locust brains revealed by RNA-seq.
(A) Venn diagram of differentially expressed genes (DEGs) in the transcriptome. (B) The numbers of up- and downregulated genes in three comparisons: C1 (G vs. S); C2 (G-NPF1a+NPF2 vs. G); C3 (S-dsNPF1a +dsNPF2 vs. S). (C) Molecular function analysis of DEGs in the transcriptome. Genes encoding catalytic and binding activities are obviously enriched in each treatment. (D) Verification of changes in the transcription of candidate genes based on the manipulation of NPF1a and NPF2 peptides or their transcript levels in the locust brains. Data are presented as mean ± s.e.m. (n = 5 samples, 8 locusts/sample, Student’s t-test, *p<0.05, different letters labeled in columns indicate a significant difference).
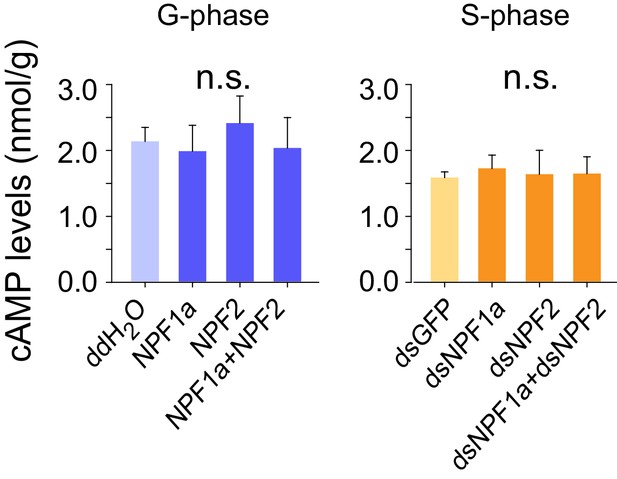
cAMP levels after artificial manipulation of NPF1a or NPF2 peptide or their transcript levels.
The data are presented as mean ± s.e.m. n.s. indicates no significance (n = 4 samples, 12–16 locusts/sample, one-way ANOVA).
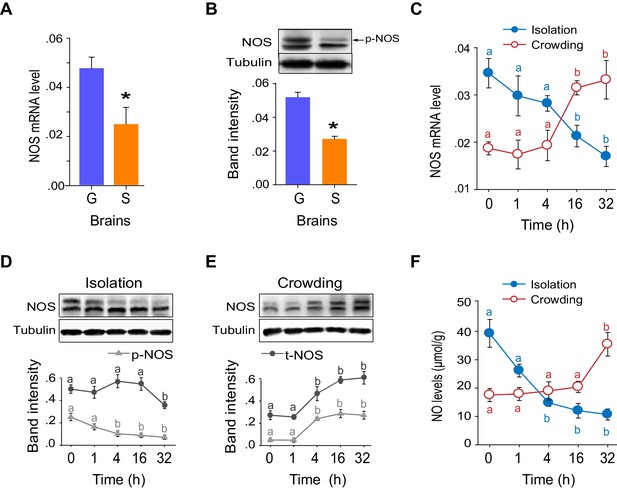
NOS transcript levels and phosphorylation states and NO levels differ in G-phase and S-phase locust brains.
(A) NOS mRNA levels in the brains of G-phase and S-phase locusts (n = 4 samples, 8 locusts/sample, Student’s t-test, *p<0.05). (B) NOS protein levels in the brains of G-phase and S-phase locusts. The upper band detected by anti-uNOS indicates phosphorylated NOS (p-NOS, see Figure 5—figure supplement 1) (n = 3 samples, 12 locusts/sample, Student’s t-test, *p<0.05). (C) Time course of NOS mRNA levels during the G/S phase transition (n = 4 samples/timepoint, 8 locusts/sample, one-way ANOVA, p<0.05, isolation shown in blue; crowding shown in red). (D) and (E) Time course of NOS protein levels during the G/S phase transition (n = 3 samples, 10–12 locusts/sample, phosphorylated NOS data are represented by triangles; total NOS data are represented by dots). The protein level is referenced to β-tubulin. (F) Time course of NO levels during the G/S phase transition. All data are presented as mean ± s.e.m. Significant differences are denoted by letters (n = 4 samples, 16 locusts/sample, one-way ANOVA, p<0.05). Raw data showing the changes in NOS mRNA level, NOS protein level and NO level are shown in Figure 5—source data 1.
-
Figure 5—source data 1
Time-course changes in NOS mRNA level, NOS protein level and NO level during the G/S phase transition.
- https://doi.org/10.7554/eLife.22526.022
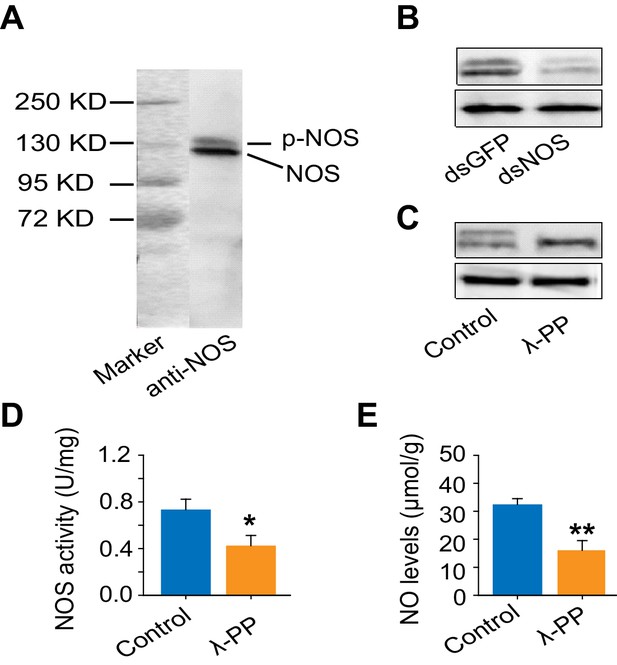
Reducing NOS expression and reducing NOS phosphorylation levels decrease NOS activity and NO level.
(A) Western blot confirms the specificity of anti-uNOS, and two bands are detected. (B) The specificity of anti-uNOS was validated by transcript knockdown and Western blot analyses. The locust NOS is probably modified at the post-translational level. (C) NOS phosphorylation was confirmed using lambda phosphatase (λPP, an enzyme that can remove protein phosphorylation). In Western blots, the upper band is removed, whereas the intensity of the lower band is increased by λPP treatment. (D) and (E) NOS activity and NO levels after the removal of NOS phosphorylation by λPP. The data are presented as mean ± s.e.m. (n = 4 samples, 12–16 locusts/sample, Student’s t-test, *p<0.05; **p<0.01).
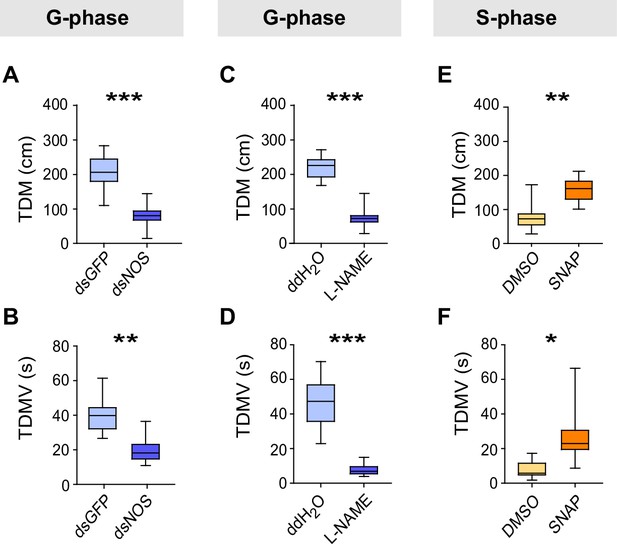
Perturbations of NO levels by transcript knockdown and drug injection dramatically change G-phase and S-phase locust behaviors.
(A) and (B) Total distance moved (TDM) and total duration of movement (TDMV) of G-phase locusts 48 hr after knockdown of the NOS transcript. All data are presented as mean ± s.e.m. (n ≥ 23 locusts, Student’s t-test, *p<0.05, **p<0.01, ***p<0.001). (C) and (D) Total distance moved (TDM) and total duration of movement (TDMV) of G-phase locusts 2 hr after injection of NOS inhibitor (L-NAME). (E) and (F) Total distance moved (TDM) and total duration of movement (TDMV) of S-phase locusts 2 hr after injection of NO donor (SNAP).
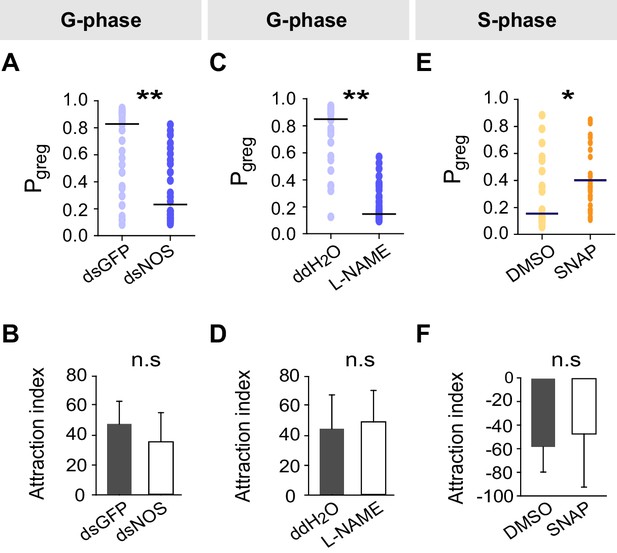
Effects on Pgreg and attraction index after NOS transcript knockdown and drug treatments in G-phase and S-phase locusts.
(A) and (B) Effects on Pgreg and attraction index 48 hr after transcript knockdown of NOS in G-phase locusts (n ≥ 23 locusts, Mann–Whitney U test for Pgreg, p=0.005; lines indicate median Pgreg; Student’s t-test for attraction index analysis, the data are presented as mean ± s.e.m.). n.s. means no significant difference. (C) and (D) Effects on Pgreg (n ≥ 23 locusts, p=0.001) and attraction index 2 hr after injection of NOS inhibitor (L-NAME) in G-phase locusts. (E) and (F) Effects on Pgreg (n ≥ 23 locusts, p=0.036) and attraction index 2 hr after injection of NO donor (SNAP) in S-phase locusts.
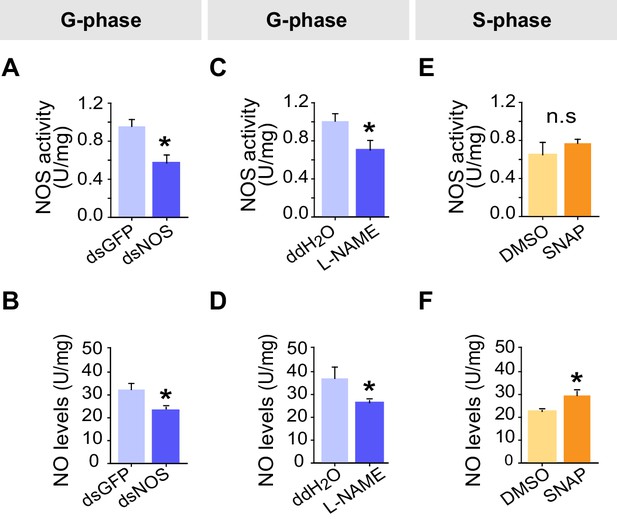
Effects on NOS activity and NO levels after NOS transcript knockdown and drug treatments in G-phase and S-phase locusts.
(A) and (B) Effects on NOS activity and NO levels 48 hr after transcript knockdown of NOS into G-phase locusts. The data are presented as mean ± s.e.m. (n = 4 samples, 12–16 locusts/sample, Student’s t-test, *p<0.05). (C) and (D) Effects on NOS activity and NO levels 2 hr after injection of NOS inhibitor (L-NAME) into G-phase locusts (n = 4 samples, Student’s t-test, *p<0.05). (E) and (F) Effects on NOS activity and NO levels 2 hr after injection of NO donor (SNAP) into S-phase locusts (n = 4 samples, Student’s t-test, *p<0.05). n.s. means no significant difference.
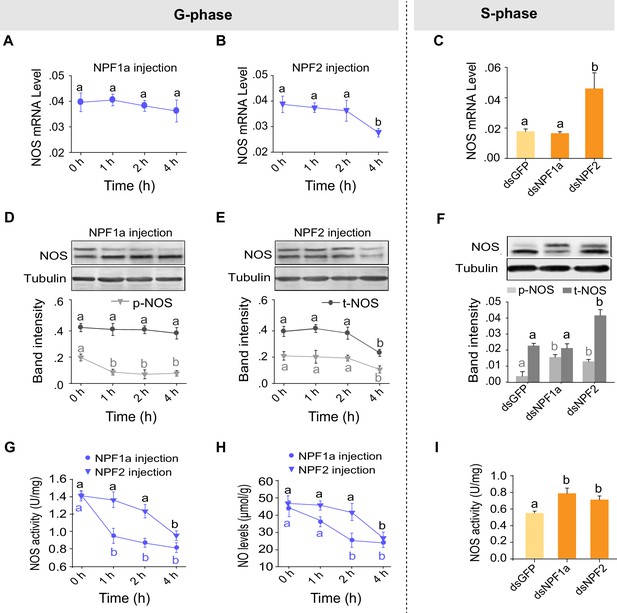
Manipulations of NPF1a and NPF2 levels alter NOS activity and phosphorylation states in the brains of G-phase and S-phase locusts.
(A) and (B) NOS mRNA levels after injection of NPF1a or NPF2 peptide into G-phase locusts. The data are presented as mean ± s.e.m. Significant differences are denoted by letters (n = 4 samples, 8 locusts/sample, one-way ANOVA, p<0.05). (C) NOS mRNA levels 48 hr after transcript knockdown of NPF1a or NPF2 in S-phase locusts (n = 4 samples, one-way ANOVA, p<0.05). (D) and (E) NOS protein levels after injection of NPF1a or NPF2 peptide into G-phase locusts (n = 3 samples, 10–12 locusts/sample, one-way ANOVA, p<0.05). (F) NOS protein levels 48 hr after transcript knockdown of NPF1a or NPF2 in S-phase locusts (n = 3 samples, one-way ANOVA, p<0.05). (G) NOS activity after injection of NPF1a or NPF2 peptide into G-phase locusts (n = 4 samples, 12–16 locusts/sample, one-way ANOVA, p<0.05). (H) NO levels after injection of NPF1a or NPF2 peptide into G-phase locusts (n = 4 samples, 12–16 locusts/sample, one-way ANOVA, p<0.05). (I) NOS activity 48 hr after transcript knockdown of NPF1a or NPF2 in S-phase locusts (n = 4 samples, one-way ANOVA, p<0.05).
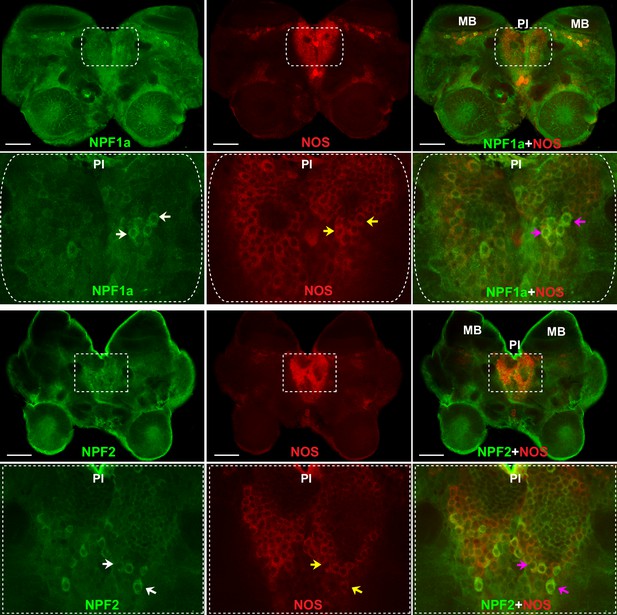
NOS and the two neuropeptides NPF1a and NPF2 co-localize in the pars intercerebralis of the locust brain.
NPF1a and NOS also co-localize in the neurons of pars lateralis anterior to the calyces of mushroom in each hemisphere in the locust brain (see Figure 8—figure supplement 1). White arrows indicateNPF1a or NPF2 staining, yellow arrows show NOS staining, pink arrows indicate merged signal of NOS and NPF1a or NOS and NPF2. Scale bars represent 100 μm.
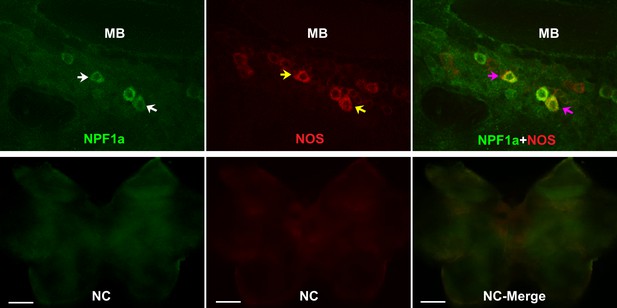
NOS and the two neuropeptides NPF1a and NPF2 co-localize in the pars lateralis around the mushroom bodies in each hemisphere of locust brain.
NC indicates negative control. White arrows indicate NPF1a staining, yellow arrows show NOS staining, pink arrows indicate merged signal of NPF1a and NOS. Scale bars represent 100 μm.
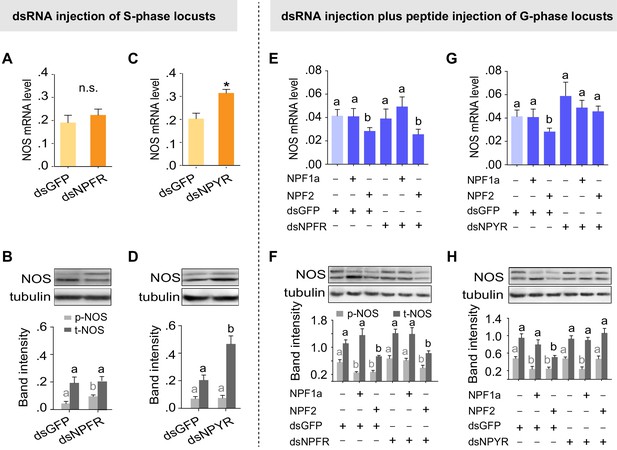
Two receptors mediate distinct effects of NPF1a and NPF2 neuropeptides on NOS phosphorylation and on NOS transcript levels, respectively.
(A) and (B) NOS mRNA levels (n = 5 samples, 6–8 locusts/sample) and NOS protein levels (n = 3 samples, 10–12 locusts/sample) 48 hr after transcript knockdown of NPFR in S-phase locusts. The data are presented as mean ± s.e.m. Significant differences are denoted by letters. n.s. means not significant (Student’s t-test, *p<0.05). (C) and (D) NOS mRNA levels (n = 5 samples) and NOS protein levels (n = 3 samples) 48 hr after transcript knockdown of NPYR in S-phase locusts. (E) and (F) NOS mRNA levels (n = 4 samples) and NOS protein levels (n = 3 samples) 4 hr after injection of NPF1a or NPF2 peptide in G-phase locusts pre-injected with dsNPFR. (G) and (H) NOS mRNA levels (n = 4 samples) and NOS protein levels (n = 3 samples) 4 hr after injection of NPF1a or NPF2 peptide in G-phase locusts pre-injected with dsNPYR. Detailed data describing NOS expression after injection of NPF1a or NPF2 peptide in G-phase locusts pre-injected with dsNPFR or dsNPYR are shown in Figure 9—source data 1.
-
Figure 9—source data 1
NOS mRNA levels and NOS protein levels after injection of NPF1a or NPF2 peptide into G-phase locusts pre-injected with dsNPFR or dsNPYR.
- https://doi.org/10.7554/eLife.22526.031
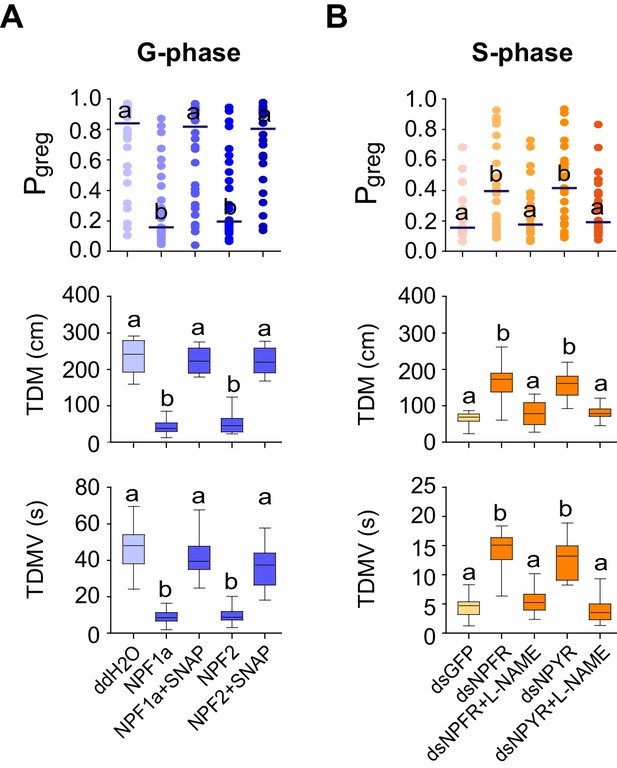
NPF1a, NPF2 and compounds that affect NO levels mediate effects on the locomotor behavior of G-phase and S-phase locusts.
(A) Behavioral test after administration of NO donor (SNAP) to G-phase locusts pre-injected with NPF1a or NPF2 peptide. Significant differences are denoted by letters. For Pgreg analysis, lines indicate median value (n ≥ 24 locusts; Mann–Whitney U test, p=0.0003 and 0.0001 for Pgreg NPF1a&SNAPvs. Pgreg NPF1a and Pgreg NPF2&SNAPvs. Pgreg NPF2, respectively). For TDM and TDMV analysis, the data are presented as mean ± s.e.m. (n ≥ 24 locusts, Student’s t-test, p<0.05). (B) Behavioral test after administration of NOS inhibitor (L-NAME) in S-phase locusts pre-injected with dsNPFR or dsNPYR (n ≥ 16 locusts, Mann–Whitney U test, p=0.022 and 0.042 for Pgreg dsNPFR&L-NAMEvs. Pgreg dsNPFR and Pgreg dsNPYR&L-NAMEvs. Pgreg dsNPYR, respectively). For TDM and TDMV analysis, the data are presented as mean ± s.e.m. (n ≥ 16 locusts, Student’s t-test, p<0.05).
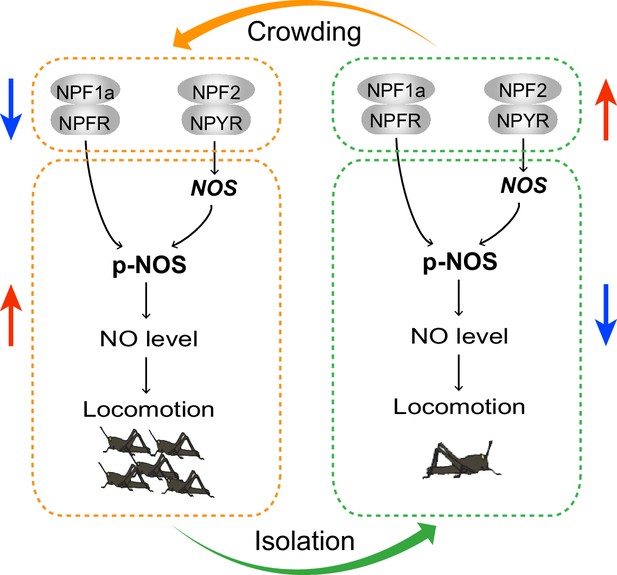
A model for the effects of neuropeptides NPF1a and NPF2 on locomotor activity related to phase transition of the migratory locust through NO signaling.
During the G/S phase transition, changes in the expression of two NPFs and their receptors act in concert to regulate the NO level, thus shaping phase-related locomotor plasticity. During crowding, the levels of two NPF systems decrease and promote NO level, resulting in higher locomotor activity. During isolation, the levels of two NPF systems increase and reduce NO level, thus leading to lower locomotor activity. Arrows indicate increased or decreased levels or activity.
Additional files
-
Supplementary file 1
Protein sequences of two NPF receptors, NPFR and NPYR, in the migratory locust.
- https://doi.org/10.7554/eLife.22526.034
-
Supplementary file 2
Primers used in qPCR and RNAi experiments.
- https://doi.org/10.7554/eLife.22526.035





