DNA-mediated association of two histone-bound complexes of yeast Chromatin Assembly Factor-1 (CAF-1) drives tetrasome assembly in the wake of DNA replication
Figures
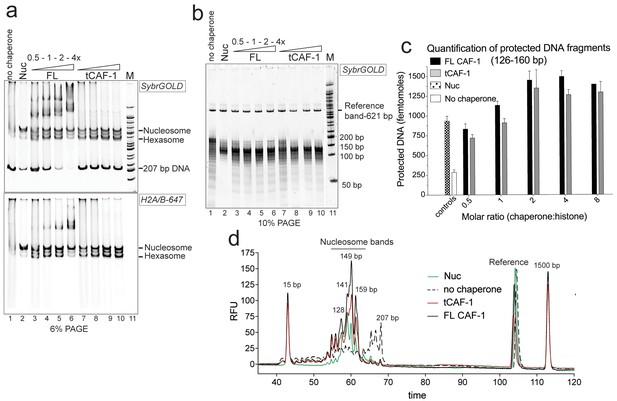
tCAF-1 is competent for nucleosome assembly in absence of other factors.
(a) Products from the nucleosome assembly assay on 207 bp DNA, with FL CAF-1 or tCAF-1. DNA is 200 nM, (H3–H4)2 200 nM and H2A-H2B-AttoN647 400 nM; 100-200-400-800 nM CAF-1 is titrated. Assemblies were analyzed by native PAGE. (b) Products of MNase digestion performed on samples shown in panel a, after DNA purification. The 207 bp band in lane 1 results from nonproductive association of histones with DNA that renders it MNase-resistant. The disappearance of the 207 bp protection as CAF-1 is titrated demonstrates the ability of CAF-1 to relieve non-specific histone-DNA complexes, as observed for other chaperones. (c) Quantification of protected nucleosomal DNA (126–160 bp) obtained from the samples shown in panel a and b. The reference DNA is used to normalize amounts in each lane. Mean ± SD is shown for at least three repeats. The data used for this panel is included in Figure 1—source data 1. (d) Bioanalyzer electropherograms of protected DNA fragments from nucleosome assembly with 400 nM FL CAF-1 and tCAF-1 shown in b, with controls from salt-assembled nucleosomes (Nuc) and a no-chaperone sample.
-
Figure 1—source data 1
FL and tCAF-1 assemble nucleosomes in vitro.
- https://doi.org/10.7554/eLife.22799.003
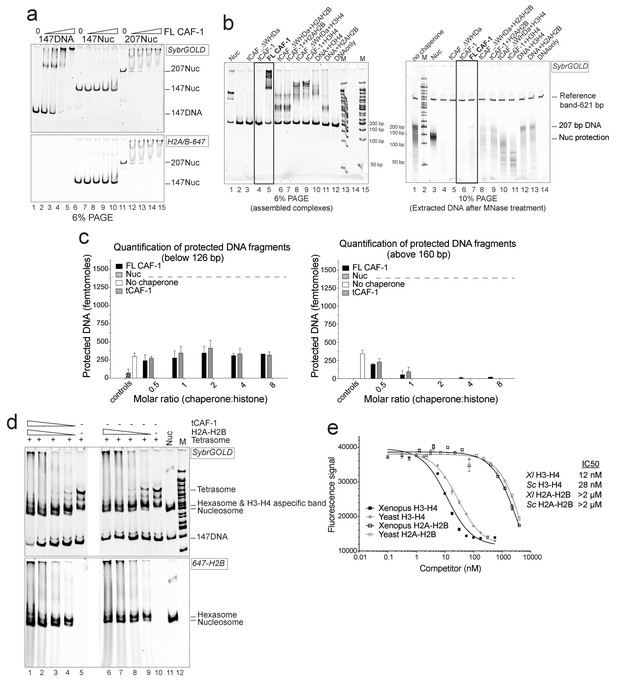
Control experiments for Figure 1.
(a) FL CAF-1 binds DNA that is not assembled into nucleosomes. DNA and salt-assembled nucleosomes (Nuc) are 50 nM; CAF-1 was titrated from 50-100-200-400 nM. (b) DNA protection at 126–160 bp from the NAQ assay is specific for nucleosomes and hexasomes. Left panel: 6% PAGE of NAQ control reactions on 207 bp of DNA. Right panel: products of the MNase digestion performed on the same samples, after DNA purification. The black box highlights the control for the FL CAF-1•DNA and tCAF-1•DNA complexes, which show no significant protection after MNase treatment. These controls (lanes 5–6 on the left gel and 6–7 on the right gel) demonstrate that DNA protection from MNase treatment is a result of nucleosome formation. The other controls confirm that deposition of either H3-H4 (lanes 10–12 on the left gel) or H2A-H2B (lanes 8–9 and 13 on the left gel) do not lead to significant DNA protection pattern. (c) In addition to nucleosomes, no significant products are made by CAF-1 in the NAQ assay. NAQ assay quantification of protected bands below 126 bp (left panel) and above 160 bp (right panel), from the data shown in Figure 1c. The dashed line indicates the amount of nucleosome bands (i.e. 120–160 bp protection) assembled by CAF-1. The data shown in this panel is included in Figure 1—source data 1. Error bars show the SD from at least three replicates. (d) H2A-H2B do not require histone chaperones to be incorporated into pre-assembled tetrasomes to form hexasome and nucleosomes. tCAF-1 has no additional effect on H2A-H2B association with the tetrasome. (e) CAF-1 does not distinguish between yeast and Xenopus laevis histones, but has significantly lower affinity for H2A-H2B compared to H3-H4. Competition assays were performed with 10 nM of FL CAF-1•DMH3-H4 complex as a probe, where H4 was labeled with Alexa488 on T71C. Unlabeled Xenopus laevis or yeast histones were titrated to follow competition of the labeled H3-H4 dimer. Fluorescence quenching was monitored. Error bars show the S.E.M. from three independent measurements.
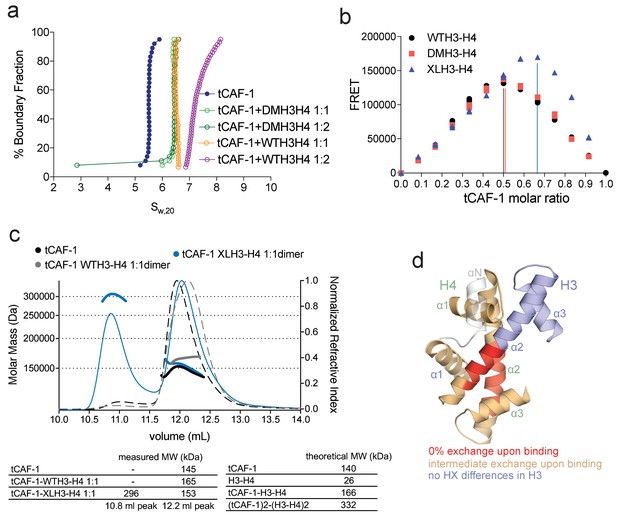
CAF-1 has a single binding site for a H3-H4 dimer.
(a) van Holde-Weischet analysis of SV-AUC runs performed with tCAF-1 and WTH3-H4, titrated in a 1:1 or 1:2 molar ratio of CAF-1 to H3-H4 dimer (orange and purple). A constitutively dimeric form of H3-H4, DMH3-H4 (green) was analyzed likewise. (b) FRET-based Job plot assay with tCAF-1 and H3-H4 shows a single binding event with WT or DMH3-H4 dimers, while with XL(H3-H4)2 a stoichiometry of two CAF-1 complexes per tetramer is observed (total protein concentration was kept at 150 nM, where WT H3-H4 is dimeric). Two independent measurements are depicted; the data points are mostly overlapping. (c) SEC-MALS experiment of tCAF-1 alone or in complex with WT or XL(H3-H4)2. The sample containing XL(H3-H4)2 shows an additional peak at ~300 kDa. The protein elution traces (refractive index, RI) refer to the right y axis, the calculated molar masses refer to the left y axis. Validation that tCAF-1 binds WTH3-H4 on SEC is shown in Figure 2—figure supplement 1g. (d) Protection of regions on the H3-H4 dimer (from PDB: 1AOI) upon interaction with tCAF-1, based on HX-MS experiments (Figure 2—figure supplement 2). Red areas: regions with complete protection on both WT and DMH3 upon interaction with tCAF-1 (near 0% uptake upon binding); orange regions: sites of intermediate HX protection upon interaction with tCAF-1. Blue indicate s regions with no significant protection in H3, upon binding to tCAF-1. H3 αN is displayed with transparency because it is likely in a different conformation in the free H3-H4 dimer; no peptide coverage was observed for this region.
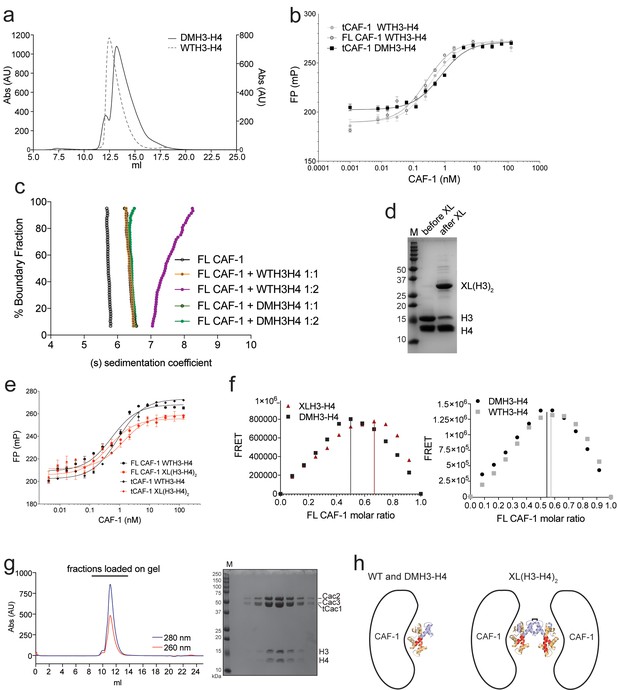
Control experiments for Figure 2.
(a) DMH3-H4 is primarily a dimer, even under conditions that favor the tetrameric state of WTH3-H4. Gel filtration of WTH3-H4 and DMH3-H4 in refolding buffer (2M NaCl). (b) tCAF-1 binds equally well to WT or DMH3-H4, determined by fluorescence polarization (Kd ~0.5–0.7 nM). Error bars show the S.E.M. from three independent measurements. (c) van Holde-Weischet analysis of SV-AUC experiments performed with FL CAF-1 and H3-H4 (wild type and DMH3-H4), combined in a 1:1 or 1:2 molar ratio of CAF-1 to H3-H4 dimer. An increased S-value upon addition of a second equivalent of H3-H4 dimer (but not with a second DM-H3H4) indicates that a (H3–H4)2 tetramer may form on CAF-1. (d) SDS PAGE of histones before and after the chemical crosslinking to form XL(H3-H4)2. (e) tCAF-1 binds equally well to WT or XL(H3-H4)2, as determined by fluorescence polarization (Kd ~0.5–0.9 nM). Error bars show the S.E.M. from three independent measurements. (f) FRET-based Job plot experiment, using FL CAF-1 and either WT or DMH3-H4, shows a single binding event for both histone complexes (protein concentration was kept at 150 nM, where WT histones are dimeric). With XL(H3-H4)2, we observed a stoichiometry of two CAF-1 complexes per tetramer. Error bars show the SEM of two independent titrations. (g) Size-exclusion chromatography (S200 10/300 GL) and SDS PAGE of tCAF-1•WTH3-H4 complex. The buffer contained 30 mM Tris pH 7.5, 100 mM NaCl, 1 mM EDTA, 1 mM TCEP. (h) Schematics to show the binding stoichiometry of CAF-1 to different H3-H4 isoforms.
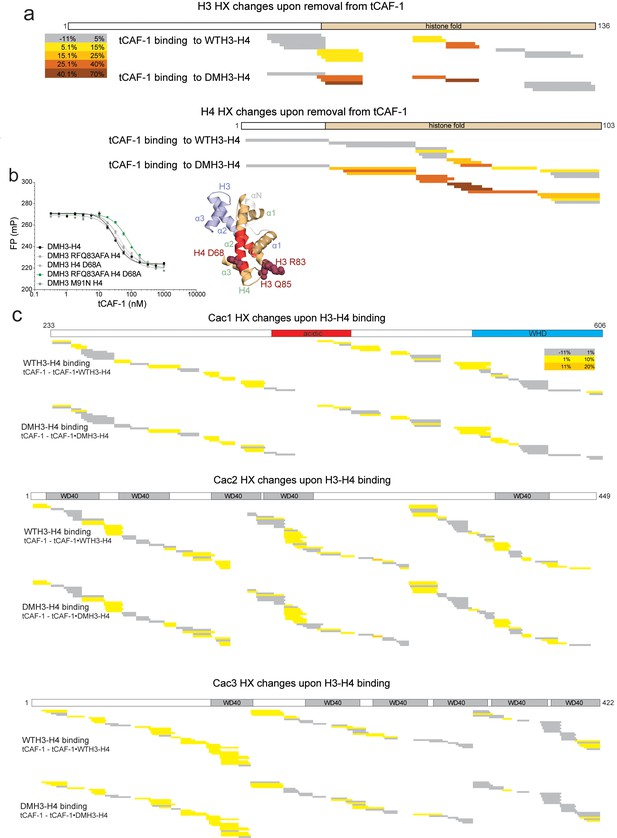
HX-MS data on CAF-1•H3-H4 complexes.
(a) HX-MS heatmap of the differences in deuterium uptake per H3 (top) or H4 (bottom) peptide when binding to tCAF-1 (60 min). The difference was calculated as percent uptake in free H3-H4 (unbound form) minus the percent uptake in tCAF-1•H3-H4 (bound form). Data related to WTH3-H4 histones are shown in the upper row; DMH3-H4 is shown below. The differences in uptake observed between WT and DMH3-H4 originate primarily from the fact that DMH3-H4 histones start less stably folded than the WTH3-H4 (DeNizio et al., 2014). CAF-1-bound DM or WTH3-H4 display highly similar deuterium uptake, although few peptides show differences, suggesting the possibility for minor dynamic changes on the histones upon CAF-1 binding (Supplementary file 1). (b) Competition assay to measure the binding properties of H3-H4 mutants designed to interfere with the CAF-1 interaction. The probe was 10 nM tCAF-1•DMH3-H4 complex. The biggest difference was observed with DM H3 RFQ83AFA – H4 D68A (mutant residues mapped in dark red on the H3-H4 dimer structure on the right), which reduced the affinity 5-fold. Error bars show the SEM from three experiments. (c) Heatmap showing the differences in deuterium uptake at 60 min for Cac1, Cac2 and Cac3 peptides analyzed in HX-MS experiments upon addition of either WT (top) or DM (bottom) H3-H4. The difference was calculated as percent uptake in tCAF-1 (unbound form) minus percent uptake in tCAF-1•H3-H4 (bound form). Supplementary file 1 contains the data used to generate the HX-MS heatmaps. The deuterium uptake was not significantly different at shorter time points.
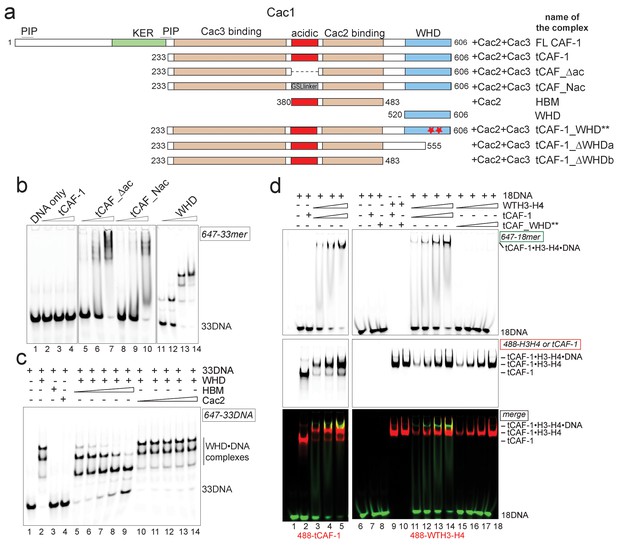
H3-H4 binding activates DNA binding by the CAF-1 complex.
(a) Schematic of the constructs and complexes used in this and subsequent figures. Cac1 domains that are combined with full length Cac2 and/or Cac3 as indicated, are depicted. PIP stands for PCNA binding peptide; tCAF_WHD** contains mutations at K564E and K568E. tCAF_∆ac contains a deletion of residues 397–431 in Cac1, while in tCAF_Nac aa 397–431 were replaced with a Gly-Ser-Leu linker. KER identifies a Lys-Arg-Glu rich region. The Cac2 and Cac3 binding regions were mapped using HX-MS. Gel filtration profiles and SDS PAGE of these complexes are shown in Figure 3—figure supplement 1a. (b) The acidic domain of Cac1 inhibits DNA binding by tCAF-1. 100 nM 33 bp DNA (33DNA) was mixed with tCAF-1, isolated WHD, or the tCAF-1 complexes mutated in the acidic region (depicted in a). CAF-1 complexes are titrated as 1–0.5-0.25–0.125 µM. (c) The acidic domain competes WHD away from DNA. The WHD was kept constant at 1 µM and DNA at 100 nM. Cac2 and HBM were titrated as 4-2-1-0.5–0.25 µM. (d) Histone binding releases the DNA binding activity of tCAF-1. EMSA performed with 100 nM 18 bp DNA (18DNA). tCAF-1 is titrated 0.37–0.75-1.5–3 µM, and was added either alone (in control lanes) or as a 1:1 complex with WTH3-H4 (H3-H4 dimer concentration) to the DNA. Alexa488-labeled tCAF-1 was used in lanes 1–5, while Alexa488-labeled H3-H4 were used in lanes 6–18).
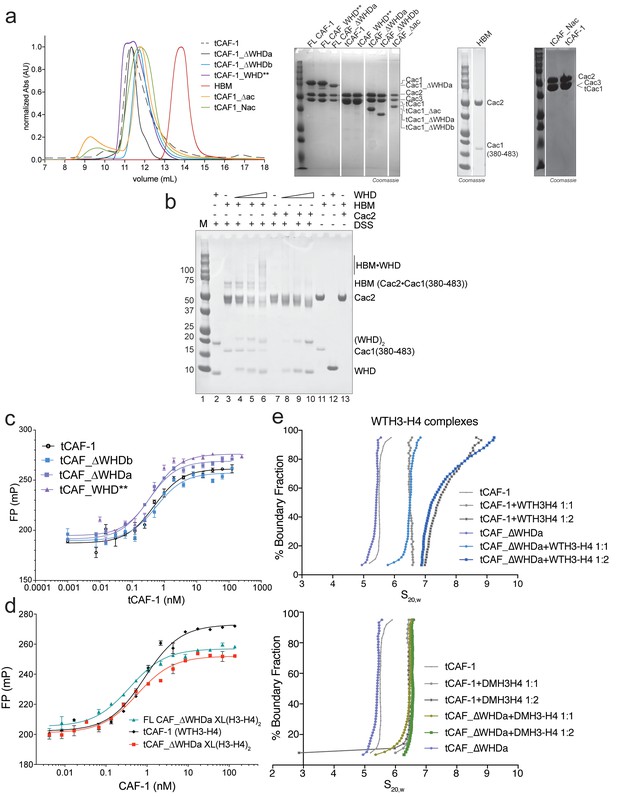
Control experiments for Figure 3.
(a) Gel filtration profiles (S200 10/300 GL column) for the complexes described in Figure 3a. SDS PAGE analysis of the purified samples. The HBM sample is also shown on SDS PAGE in panel b) lane 11. All the complexes do not show any aggregation and elute from gel-filtration at the expected volume for their size, indicating that the stoichiometry of the subunits is not altered. (b) In solution cross-linking experiments with 5 µM of either HBM [Cac1(380-483)•Cac2] or Cac2 alone and variable amounts of WHD (0-5-10-20 µM). DSS was added at 2 mM and incubated for 30 min, before quenching and analysis by SDS PAGE. (c–d) WHD mutation or deletion does not alter the affinity for H3-H4. Affinity measurements (FP) of the tCAF-1 complexes mutated or deleted in the WHD, to WT (c) or XL(H3-H4)2 (d) show no changes in Kd (Kd between 0.5–0.9 nM). Error bars depicts the SEM from three measurements. (e) WHD deletion does not affect the stoichiometry of the CAF-1 interaction with histones. van Holde-Weischet analysis of SV-AUC experiments performed with tCAF_∆WHDa and either WT (top panel) or DMH3-H4 (bottom panel) titrated as 1:1 or 1:2 ratio of CAF-1 to H3-H4 dimer. The curves also shown in Figure 2a obtained with the tCAF-1 complex are shown in gray as reference.
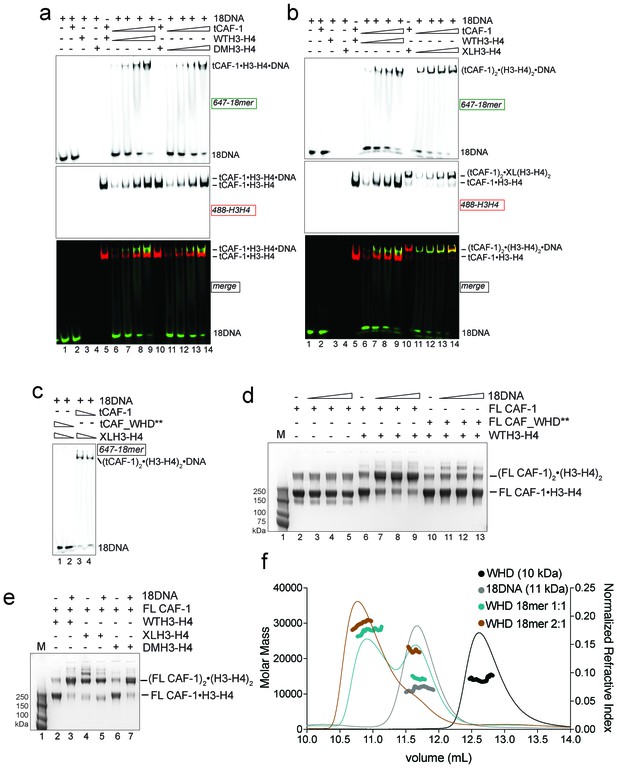
Two CAF-1•H3-H4 complexes associate on a short DNA fragment.
(a–b) A ternary tCAF-1•histone•18DNA complex can be formed with DMH3-H4(a) or with XL(H3-H4)2 (b). EMSA was performed with 100 nM 18 bp DNA (18DNA). tCAF-1 is titrated 0.37–0.75-1.5–3 µM, and is added at a 1:1 ratio to either DMH3-H4 (a) or XL(H3-H4)2 (b). H3-H4 was calculated as a dimer, even with XL(H3-H4)2 to the DNA. WTH3-H4 is shown as a control in both panels. (c) Both H3-H4 moieties in the XL(H3-H4)2 are shielded from DNA binding in complex with tCAF-1. EMSA performed with 100 nM 18 bp DNA (18DNA). tCAF-1 or tCAF_WHD** in complex with XL(H3-H4)2 [1 CAF-1 complex per 1 H3-H4 dimer] is at 1 and 3 µM. (d) In solution cross-linking experiments with 2 µM of either FL CAF-1 alone, FL CAF-1•H3-H4 complex, or FL CAF_WHD**•H3-H4 complex, and variable amounts of DNA (0-1-2-4 µM). DSS was added at 1 mM and incubated for 30 min, before quenching and analysis by SDS PAGE. Full gel image shown in Figure 4—figure supplement 1a. (e) In solution cross-linking experiments with DSS as in panel (d), but here FL CAF-1 was premixed with either WT, DMH3-H4 or XLH3-H4. DNA is at 4 µM. Full gel image shown in Figure 4—figure supplement 1c. (f) SEC-MALS experiment of the isolated WHD alone or in complex with 18DNA. In buffer containing 150 mM NaCl, the WHD is monomeric in absence of DNA, but on 18DNA it favors binding in a 2:1 stoichiometry (WHD to 18DNA). The protein/DNA elution traces (refractive index, RI) refer to the right y axis, the calculated molar masses refer to the left y axis.
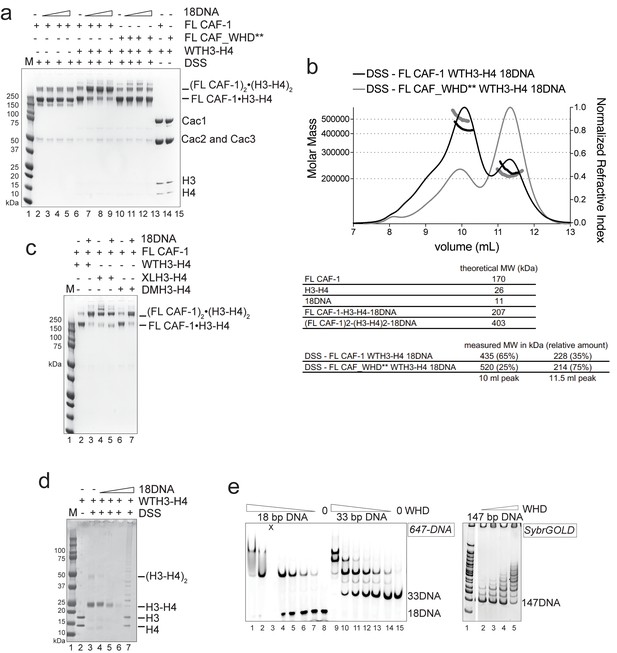
Control experiments for Figure 4.
(a) In solution cross-linking experiments with 2 µM of either FL CAF-1 alone, FL CAF-1•H3-H4 complex, or FL CAF_WHD**•H3-H4 complex, and variable amounts of DNA (0-1-2-4 µM). DSS was added at 1 mM and incubated for 30 min, before quenching and analysis by SDS PAGE. Top part of this gel is shown in main Figure 4d. (b) SEC-MALS experiment of FL CAF-1 or FL CAF_WHD** in complex with WTH3-H4 after DSS crosslinking treatment, as shown in Figure 4d. With an intact WHD, the main species in solution appear to have a molar mass consistent with two CAF-1•H3-H4 moieties. The protein elution traces (refractive index, RI) refer to the right y axis, the calculated molar masses refer to the left y axis. Note that DNA is not chemically cross-linked by DSS. (c) In solution cross-linking experiments with DSS as in panel a, but here FL CAF-1 was premixed with either WT, DMH3-H4 or XLH3-H4. DNA is at 4 µM. Top part of this gel is shown in main Figure 4e. (d) In solution cross-linking experiments using WTH3-H4 at 2 µM and 18DNA titration at 0.5-1-2-4 µM. (e) DNA binding assay of the isolated WHD (titrated 8-4-2-1-0.5–0.25 µM) to 18 or 33 bp DNA (1 µM) (left panel). X denotes a skipped lane. Right panel depicts WHD binding to 147 bp DNA (100 nM), the WHD was titrated (1–0.5-0.25–0.125 µM).
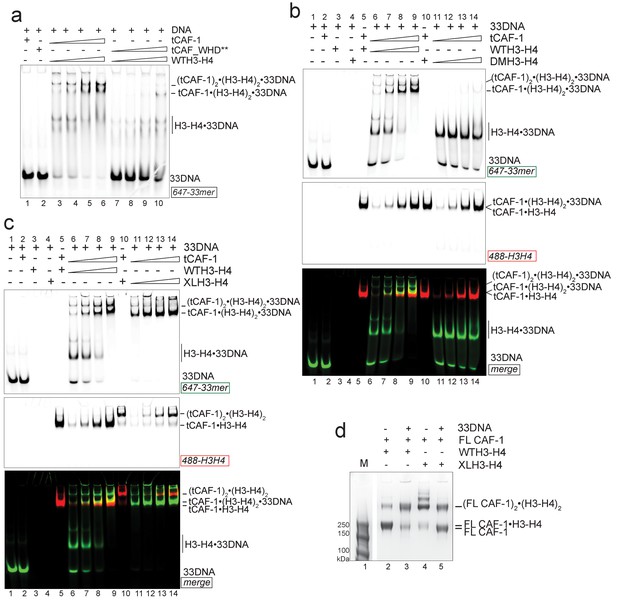
DNA of sufficient length sequesters (H3–H4)2 from CAF-1.
(a) EMSA performed with 100 nM 33mer DNA. tCAF-1 or tCAF-1_WHD** were titrated 0.37–0.75-1.5–3 µM, and were added with WTH3-H4 to the DNA. (b) EMSA performed with 100 nM 33 bp DNA (33DNA). tCAF-1 was titrated 0.37–0.75-1.5–3 µM, and was added with WT or DMH3-H4 (calculated as a H3-H4 dimer) to the DNA. (c) EMSA performed with 100 nM 33 bp DNA (33DNA). tCAF-1 was titrated 0.37–0.75-1.5–3 µM, and was added with WT or XL(H3-H4)2 (calculated as a H3-H4 dimer) to the DNA. (d) In solution cross-linking experiments with 2 µM of FL CAF-1 premixed with either WT, DMH3-H4 or XL(H3-H4)2, in presence of 4 µM of DNA. DSS was added at 1 mM and incubated for 30 min, before quenching and running of SDS PAGE. Full gel image is shown in Figure 5—figure supplement 1c.
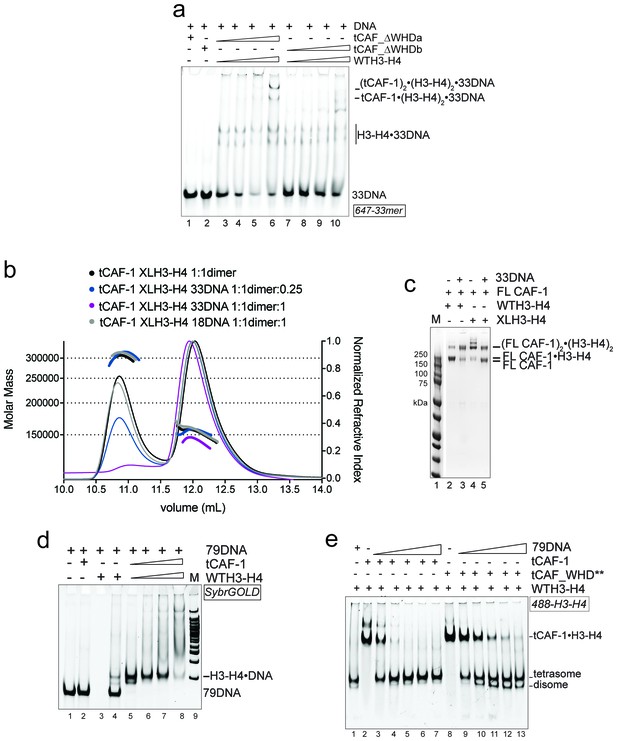
Control experiments for Figure 5.
(a) EMSA performed with 100 nM 33mer DNA. tCAF-1_∆WHDa or tCAF-1__∆WHDb were titrated 0.37–0.75-1.5–3 µM, and were added with WTH3-H4 to the DNA. (b) SEC-MALS experiment titrating 33DNA into a tCAF-1 in complex with XL(H3-H4)2 (mixed as 1 CAF-1 per histone dimer). The sample with 18DNA is used as a control. The protein/DNA elution traces (refractive index, RI) refer to the right y axis, the calculated molar masses refer to the left y axis. (c) In solution cross-linking experiments with 2 µM of FL CAF-1 premixed with either WT, DMH3-H4 or XLH3-H4, in presence of 4 µM of DNA. DSS was added at 1 mM and incubated for 30 min, before quenching and running of SDS PAGE. Top part of the gel is shown in main Figure 5d. (d) EMSA performed with 100 nM 79 bp DNA (79DNA). tCAF-1•WTH3-H4 is titrated 0.37–0.75-1.5–3 µM, to the DNA. (e) EMSA performed with 200 nM tCAF-1•WTH3-H4 or tCAF_WHD**•WTH3-H4, 79 bp DNA (79DNA) is titrated at 25-50-100-200-400 nM.
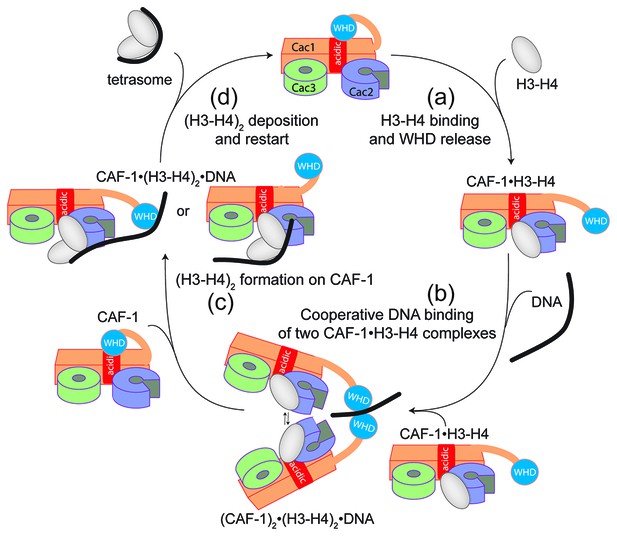
Model of the molecular mechanism of CAF-1 mediated tetrasome assembly.
(a) The nucleosome assembly mechanism of CAF-1 is activated by H3-H4 binding, which releases the WHD domain from an intramolecular interaction with the acidic region on Cac1. (b) DNA binding promotes the association of two CAF-1•H3-H4 complexes to join the histones into a (H3–H4)2 tetramer (c). In the presence of DNA of sufficient length, the (H3–H4)2 histones are directly sequestered from CAF-1. (d) (H3–H4)2 are transferred to the DNA to form the tetrasome, and the WHD re-binds to the now free acidic region, resulting in its dissociation from DNA.
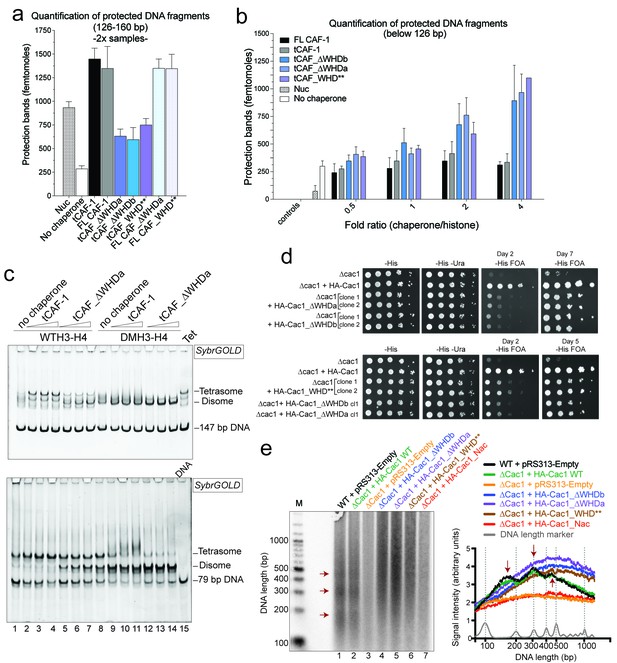
DNA binding by the Cac1 WHD is required for nucleosome assembly in vitro and in vivo.
(a) Quantification of the nucleosome bands (126–160 bp) from the NAQ assays. Mean ± SD is shown from two or more independent measurements. For simplicity, we only show results from experiments with a 2-fold chaperone to histone ratio. The trends are identical for the 0.5-1-4 fold chaperone to histone ratio. The data use for this panel is included in Figure 7—source data 1. (b) Quantification of bands below 126 bp (sub-nucleosomal species) from the NAQ assays. The error bars indicate SD from at least three repeats. The data use for this panel is included in Figure 7—source data 2. (c) Tetrasome assembly assay on 147 bp (top panel) or 79 bp DNA (bottom panel) with tCAF-1 and the tCAF_∆WHDa mutant, comparing WT and DMH3-H4 deposition. Assembly for the other WHD mutants is shown in Figure 7—figure supplement 1a. (d) Epigenetic silencing of a telomere-proximal URA3 reporter performed with yeast strains expressing the Cac1 mutation or deletion of the WHD. Samples were spotted at 0-101-102-103-104 dilutions from a OD600 = 1 stock. (e) Okazaki fragment ends isolated from WT yeast strains, WHD mutant strains, and the acidic region mutant strain (Cac1_Nac, residues 397–431 were replaced with a Gly-Ser-Leu linker), as indicated, were radiolabeled and separated on a denaturing agarose gel. The right-hand panel depicts a normalized trace of signal intensity for each lane; DNA ladder is shown in light gray. Red arrows highlight the nucleosome-dependent length of the fragments in the positive control lanes (lane 1 and 2).
-
Figure 7—source data 1
Mutation of the Cac1 WHD inhibits nucleosome assembly in tCAF-1.
- https://doi.org/10.7554/eLife.22799.016
-
Figure 7—source data 2
Mutation of the Cac1 WHD in tCAF-1 promotes the formation of sub-nucleosomal species.
- https://doi.org/10.7554/eLife.22799.017
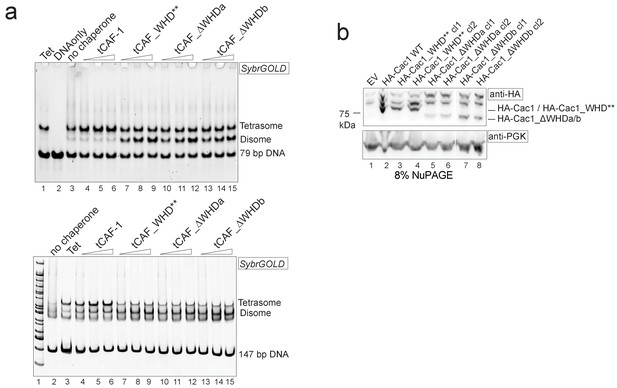
Control experiments to Figure 7.
(a) Cac1 WHD mutation or deletion result in the deposition of a H3-H4 dimer onto DNA. Tetrasome assembly assay with tCAF-1 WHD mutants on 79 (top) or 147 bp DNA (bottom). (b) Cac1 mutants used in Figure 7d are expressed, as shown by immunoblot using anti-HA antibodies.
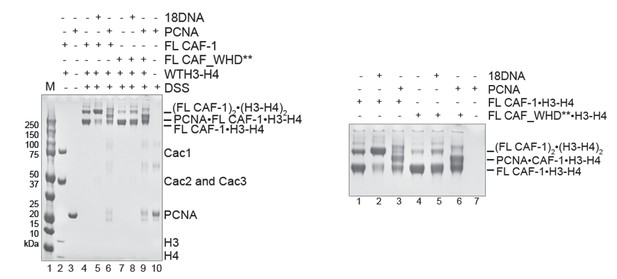
In solution cross-linking studies of the complexes formed by FL CAF-1/H3-H4 in presence of 1 18 bp DNA (18DNA) or PCNA.
CAF-1/H3-H4 was kept at 2 µM, 18DNA and PCNA were added at 4 and 8 µM, respectively. The whole gel is shown on the left, the close-up is shown on the right.
Additional files
-
Supplementary file 1
HX-MS data and uptake plots for tCAF-1 alone and in complex with WT or DMH3-H4.
- https://doi.org/10.7554/eLife.22799.019





