Evolutionary changes in transcription factor coding sequence quantitatively alter sensory organ development and function
Figures
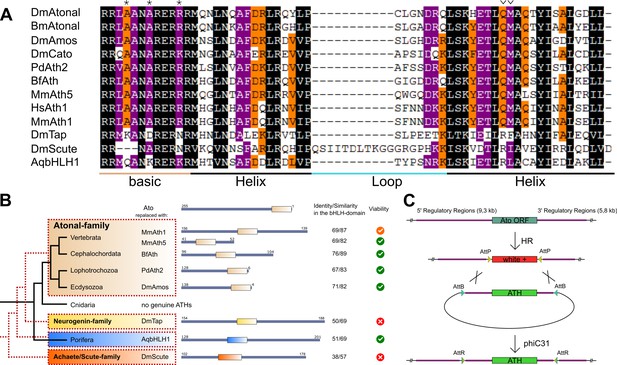
ATHs represent a functionally distinct proneural group.
(A) Alignment of bHLH domains of the ATHs used in this study. The bHLH domains of the three proneural TF families are characterized by the presence of family-specific AA (marked with * and ∨). Asterisks: AA distinguishing ATHs from members of the Neurogenin family (Quan et al., 2004). (B) Phylogenetic position of the selected homologues and evolutionary relationship of the control proteins. The black tree shows the phylogeny of the organisms. The red dashed tree shows the relationships between protein families. The diagrams show the primary structures of the proteins; the bold box is the bHLH domain, numbers indicate the length of the protein outside the bHLH domain. Green tick: homozygous viable stocks , orange tick: semi-viable, red cross: absence of unbalanced flies. Dm: Drosophila melanogaster; Mm: Mus musculus; Bf: Branchiostoma floridae; Pd: Platynereis dumerilii; Aq: Amphimedon queenslandica. (C) Schema of the improved IMAGO approach (Choi et al., 2009). See also Figure 1—figure supplement 1.
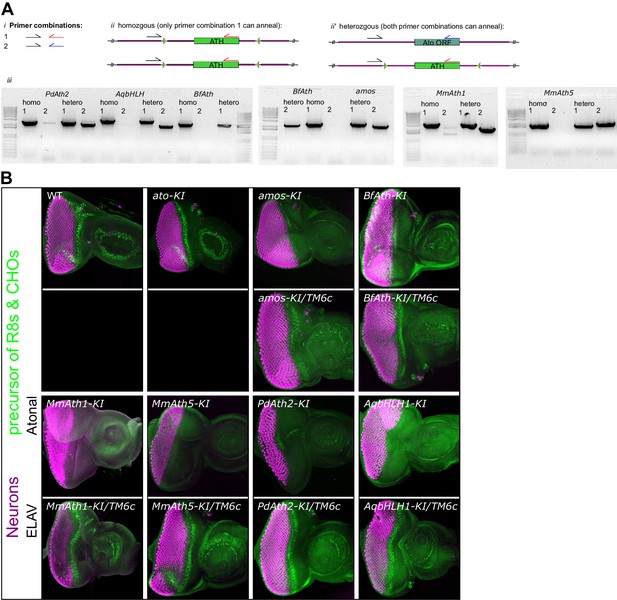
Upon replacement in ato endogenous locus, all ATHs and AqbHLH1 rescue the lethality caused by loss of ato in the fruit fly.
(A) PCR reactions (iii) with the primer combinations (i) that result in a product only if the ATH (ii) or ato (ii’) is present in the template. Balanced and unbalanced flies were processed in parallel. (B) Anti-ELAV and anti-Ato immunostainings on late L3 eye-antennal discs of the indicated genotypes. Balanced and unbalanced flies were processed in parallel. Note that the magnification is not constant across all samples.
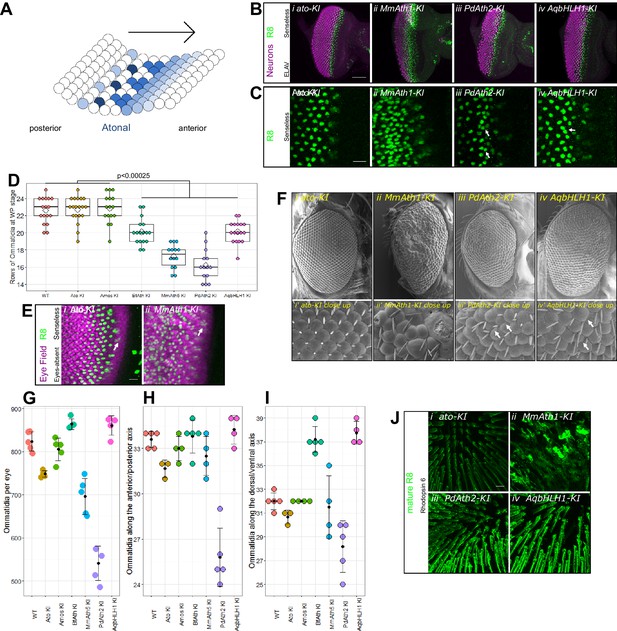
ATHs specify retinal precursors at different rates.
(A) Ato specifies the R8 photoreceptors in a sequential manner (from posterior to anterior) in an indentation in the imaginal disc called the morphogenetic furrow (MF), leaving behind regularly spaced R8 precursors. (B) Anti-ELAV and anti-Senseless immunostainings on late L3 eye-antennal discs: (i) ato-KI, (ii) MmAth1-KI, (iii) PdAth2-KI, (iv) AqbHLH1-KI (scale bar 50 μm). (C) Anti-Senseless immunostainings at the level of the MF: (i) ato-KI, (ii) MmAth1-KI, (iii) PdAth2-KI, (iv) AqbHLH1-KI (scale bar 10 μm). (D) Number of rows of ommatidia consisting of at least six photoreceptors at white pupae stage; boxes indicate interquartile ranges, lines medians, diamonds means and whiskers data ranges. Summary statistics can be found in Supplementary file 6A and all p-values (t-test) for all pairwise comparisons in Supplementary file 6B. (E) Anti-Eyes-absent and anti-Senseless immunostainings on P0 eye-antennal discs: (i) ato-KI, (ii) MmAth1-KI (scale bar 50 μm). (F) Scanning electron microscopy images of compound eyes (i–iv) and close ups (i’–iv’) of (i+i’) ato-KI, (ii+ii’) MmAth1-KI, (iii+iii’) PdAth2-KI, (iv+iv’) AqbHLH1-KI (scale bar 50 μm); arrows indicate smaller or larger ommatidia. (G) Ommatidia number of female compound eyes; black dots indicate means, error bars SEM. (H+I) Ommatidia numbers along the anterior/posterior axis (H) and (I) dorsal ventral axis of adult female compound eyes; black dots indicate means, error bars SEM. (J) Anti-hodopsin 6 immunostainings of adult KI fly retinas (i) ato-KI, (ii) MmAth1-KI, (iii) PdAth2-KI, (iv) AqbHLH1-KI (scale bar 50 μm); All confocal images (B, C, E, J) are maximum intensity projections. See also Figure 2—figure supplements 1 and 2.
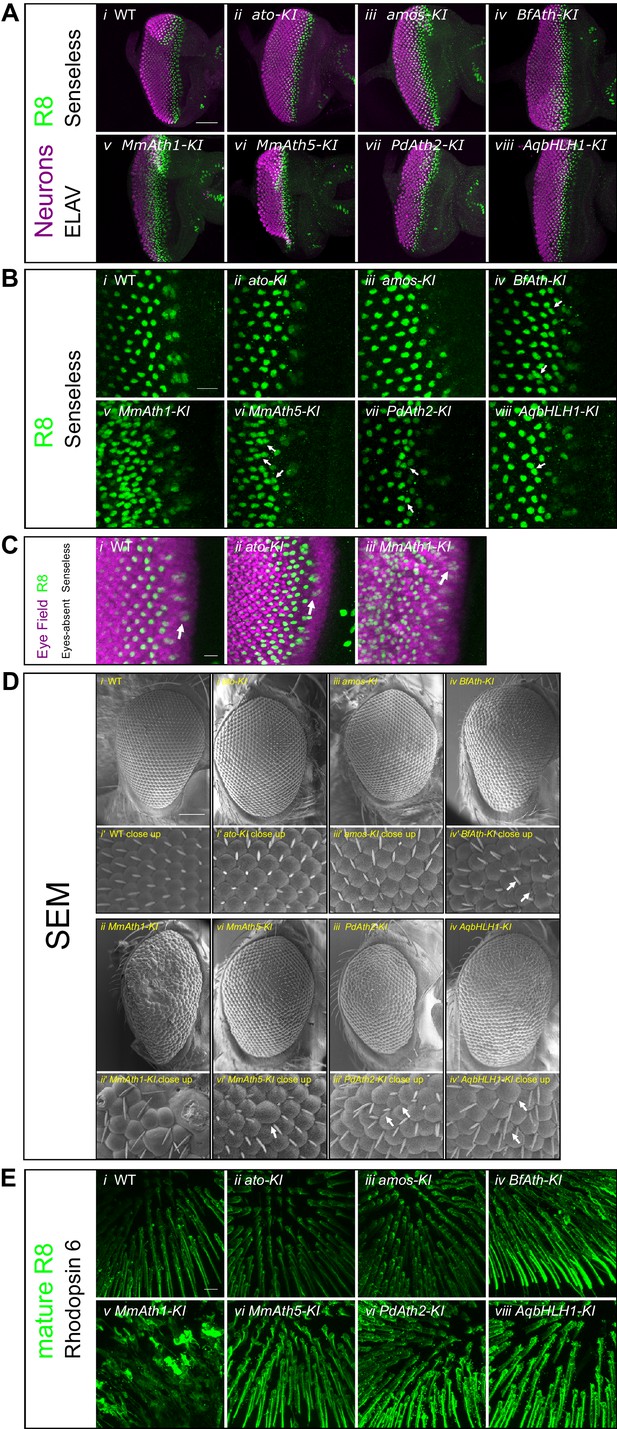
ATHs specify retinal precursors at different rates.
(A) Anti-ELAV and anti-Senseless immunostainings on late L3 eye-antennal discs: (i) WT, (ii) ato-KI, (iii) amos-KI, (iv) BfAth-KI (v) MmAth1-KI, (vi) MmAth5-KI, (vii) PdAth2-KI, (viii) AqbHLH1-KI (scale bar 50 μm). (B) Anti-Senseless immunostainings at the level of the MF: (i) WT, (ii) ato-KI, (iii) amos-KI, (iv) BfAth-KI (v) MmAth1-KI, (vi) MmAth5-KI, (vii) PdAth2-KI, (viii) AqbHLH1-KI (scale bar 10 μm). (C) Anti-Eyes-absent and anti-Senseless immunostainings on P0 eye-antennal discs: (i) WT , (ii) ato-KI, (iii) MmAth1-KI (scale bar 50 μm). (D) Scanning electron microscopy images of compound eyes (i-viii) and close ups (i’-viii’) of (i+i’) WT, (ii+ii’) ato-KI, (iii+iii’) amos-KI, (iv+iv’) BfAth-KI, (v+v’) MmAth1-KI, (vi+vi’) MmAth5-KI, (vii+vii’) PdAth2-KI, and (viii+viii’) AqbHLH1-KI (scale bar 50 μm); arrows indicate smaller or larger ommatidia. (E) Anti-rhodopsin 6 immunostainings of adult fly retinas of (i) WT, (ii) ato-KI, (iii) amos-KI, (iv) BfAth-KI (v) MmAth1-KI, (vi) MmAth5-KI, (vii) PdAth2-KI, (viii) AqbHLH1-KI (scale bar 50 μm). All confocal images (A, B, C, E) are maximum intensity projections.
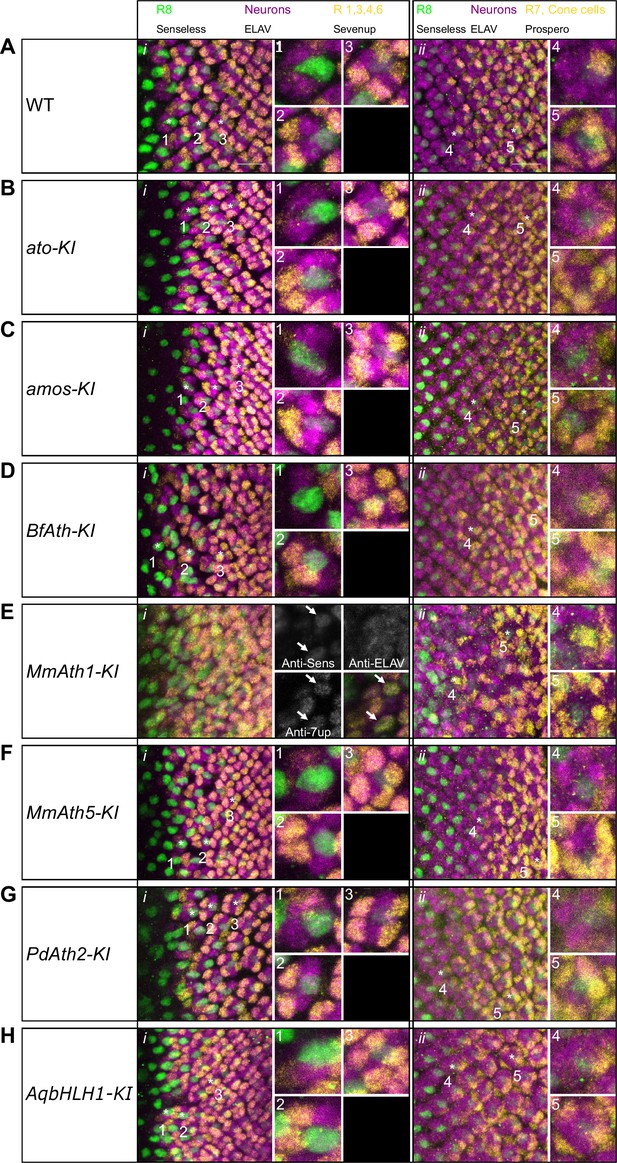
Correct ommatidia differentiation takes place in all KI-lines except MmAth1-KI.
Late L3 eye-antennal disc immunostainings against Senseless, ELAV and Sevenup (i) and Senseless, ELAV and Prospero (ii): (A) WT, (B) ato-KI, (C) amos-KI, (D) BfAth-KI (E) MmAth1-KI, (F) MmAth5-KI, (G) PdAth2-KI, (H) AqbHLH1-KI (all images are maximum intensity projections; scale bar 10 μm). Numbers highlight key differentiation events: 1: recruitment of R2/5; 2: recruitment of R3/4; 3: recruitment of R1/6; 4: recruitment of R7; 5: recruitment of cone cells.
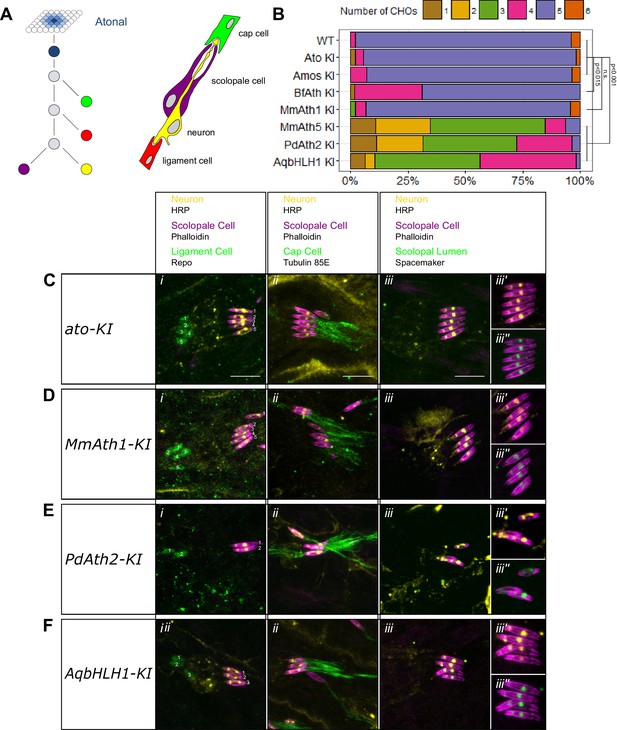
ATHs show quantitative variation in chordotonal organ specification.
(A) Ato specifies the ChO precursor cell that gives rise to the four ChO cells via a fixed division pattern. (B) Frequency plot of the number of ChOs present in the embryonic lateral ChO cluster (14–17 hr after egg laying). Summary statistics can be found in Supplementary file 6C and all p-values (Fisher exact test) for all pairwise comparisons in Supplementary file 6D. (C–F) Cell content of embryonic lateral ChOs formed by the different ATHs. Neurons (yellow) are labeled using anti-HRP; scolopale cells (magenta) with Phalloidin labeling. In addition, ligament cells are revealed using anti-repo (i), cap cells using anti-tubulin 85E (ii) and the scolopale lumen using anti-spacemaker (iii), and immunoreactivity. (iii’ + iii’’): close ups of phalloidin labeling together with HRP (iii’) or spacemaker (iii’’) immunostainings. All images are maximum intensity projections. (C) ato-KI, (D) MmAth1-KI, (E) PdAth2-KI, (F) AqbHLH1-KI (scale bar 10 μm). See also Figure 3—figure supplement 1.
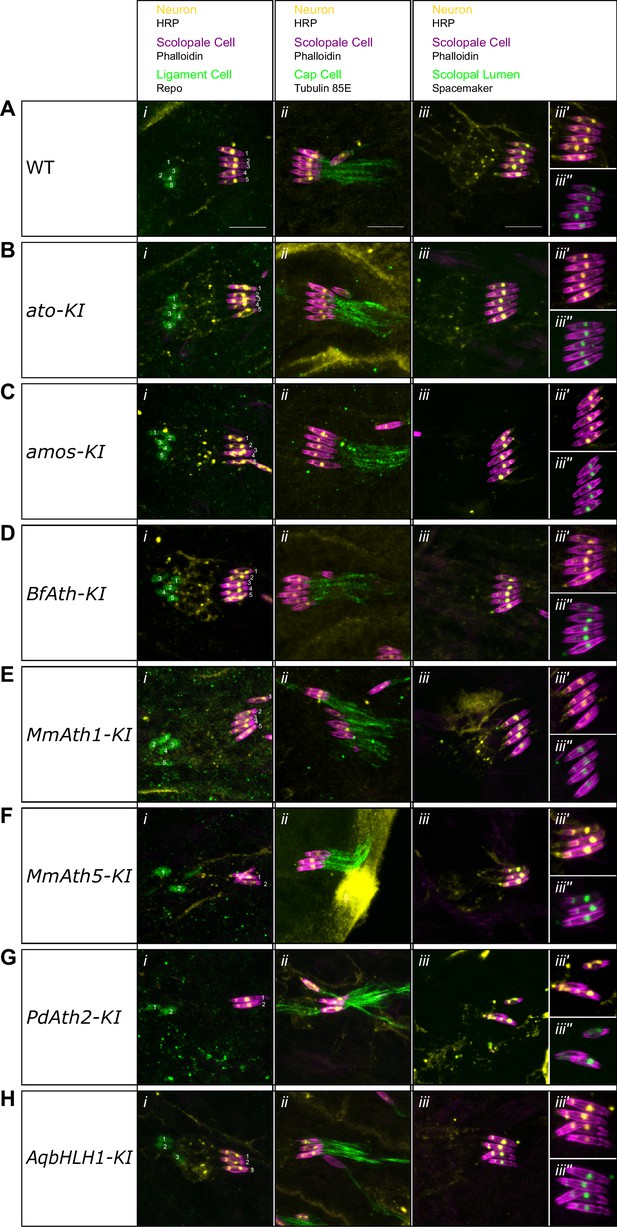
ATHs and AqbHLH1 - derived chordotonal organs present correct cell content.
(A-H) Maximum intensity projections of Phalloidin, HRP and Repo (i), Phalloidin, HRP and Tubulin 85E (ii), and Phalloidin, HRP and Spacemaker (iii) immunostainings of late embryo lateral chordotonal organ cluster of (A) WT, (B) ato-KI, (C) amos-KI, (D) BfAth-KI (E) MmAth1-KI, (F) MmAth5-KI, (G) PdAth2-KI, (H) AqbHLH1-KI (scale bar 10 μm). (iii’) are Phalloidin and HRP and (iii’’) are Phalloidin and Spacemaker close up magnifications of (iii).
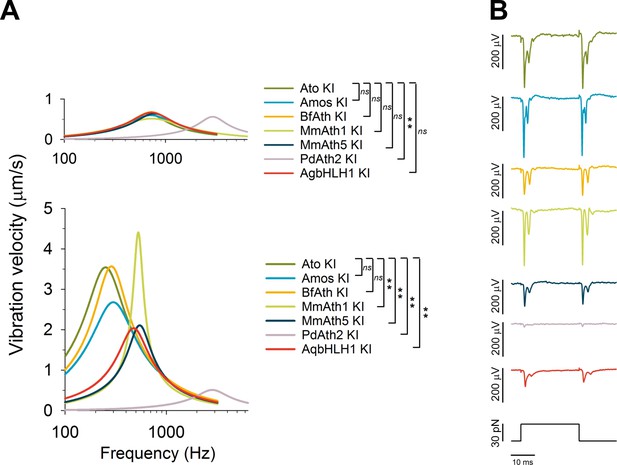
ChOs of Johnston’s organ (JO) specified by ATHs and AqbHLH1 are functional and different functional aspects are restored in a quasi-modular way.
(A) All viable KI-lines, at least partly, restored the rotational joint of the flies’ antennal sound receivers (top: metabolically inactive – passive - receivers of CO2-sedated, O2-deprived flies; bottom: metabolically active receivers of awake, O2-supplied flies). The free mechanical fluctuations of the unstimulated receivers reveal the specific antennal best frequencies for all ATHs (color legend to the right indicates statistically significant differences compared with the ato-KI). Whereas the passive receivers (top) are virtually identical in all ATH-KI rescues (with the exception of PdAth2-KI), the active receivers (bottom) display characteristic differences in frequency tuning and auditory amplification. Summary statistics and p-values (t-test or Mann-Whitney Rank Sum) can be found in Supplementary file 6Q and R. (B) In response to a medium-range force step (30 pN corresponding to ~ half-maximal activation of ato-KI controls), all antennal nerves of viable KI-lines produce a symmetric compound action potential (CAP) response to both the onset and the offset of the antennal deflection, as reported for wild-type controls. The amplitudes of the CAP responses, however, vary among the different KI-lines, with amos-KI and MmAth1-KI rescues reaching ato-KI control levels and the remaining ATHs producing significantly smaller CAPs (BfAth1 > MmAth5 > AqBHLH1 > PdAth2). See also Figure 4—figure supplement 1.
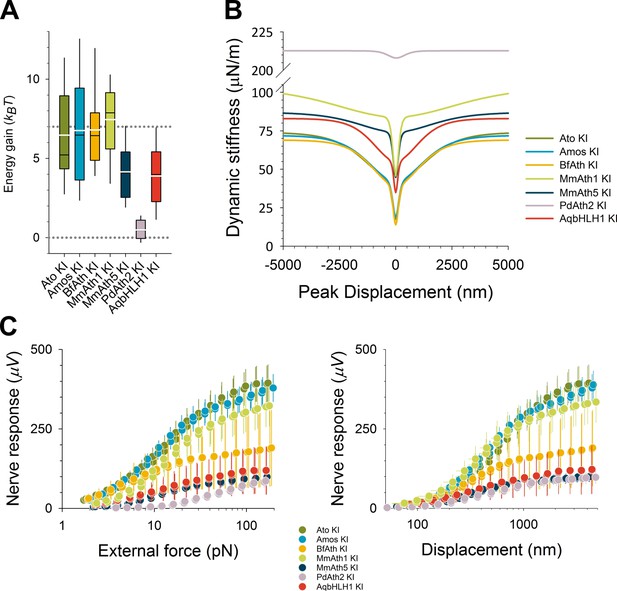
Analysis of dynamic stiffness of the antenna and the nerve responses over a wide range of stimuli revealed a quasi-modular JO function in the KI-lines.
(A) All viable KI-lines had, at least partly, restored antennal energy gain, a hallmark of transducer-based auditory amplification. Whereas the energy gain reaches (ato-KI) control levels in amos-, BfAth- and MmAth1-KI rescues, energy injections are reduced to ~50% of control levels in MmAth5-KI and AqbHLH1-KI rescues. In PdAth2-KI rescues, energy gains are almost entirely lost (lower dotted line indicates zero power gain, upper dotted line indicates wild-type, Canton-S control levels). (B) All viable KI-lines, at least partly, restored the mechanical signatures of mechanotransduction (gating compliances). Average fits are shown of a gating spring model to the receivers’ dynamic stiffness following force-step actuation for all tested ATHs. With the exception of the PdAth2-KI rescue, which was sufficiently described by a one transducer-type-model (test: corrected Akaike Information Criterion, AIC), all ATHs restored mechanical signatures of two transduction-related mechanical nonlinearities, which have been previously suggested to reflect two different types of mechanotransducers. The various ATHs, however, display characteristic differences in the extents of, and the proportions in between, these two nonlinear compliances, with most variation affecting the shallower, outer nonlinearity, which has previously been linked to a less sensitive population of transducers. Note that only Amos and BfAth restore the dynamic stiffness around the resting position (i.e. at zero displacement) to ato-KI control levels. Summary statistics and p-values (t-test or Mann-Whitney Rank Sum) can be found in Supplementary file 6S. (C) Comparing the mechanically evoked responses over the entire working range (left: against size of force step, right: against antennal peak displacement) shows that for small stimulus forces (< ~8 pN, left panel) only Amos and BfAth restore CAP amplitudes to ato-KI control levels. All other ATHs and AqbHLH1 shift the response curves to the right hand side of the controls, indicating a partial loss of force sensitivity. For large stimuli, close to response saturation, in turn, only Amos appears to restore full CAP responses, suggesting that only Amos fully restores all subgroups of Johnston organ neurons. Plotting CAP nerve responses against antennal displacements (right panel), leaves curves closer to the controls, indicating that parts of the response deficits seen between ATHs- and ato-KI controls result from a reduced force sensitivity (most likely caused by a partial loss of nonlinear compliance) and not from changes in displacement sensitivity. If the necessary force to deflect the antenna to a certain displacement is provided, then mechanotransducer channels will open. Note that in this plot the MmAth1-KI rescue largely superimposes with the atoKI control curve. But also here, only Amos restores the full (saturation) response amplitudes.
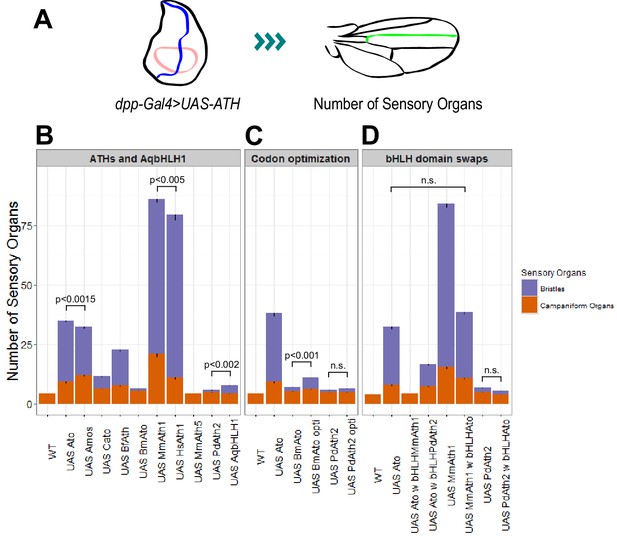
ATHs ectopic expression induces formation of distinct numbers of ectopic sensory organs.
(A) Schematics of the wing disc assay; ectopic sensory organs formed upon ectopic expression of ATHs and AqbHLH1 driven by dpp-Gal4 were counted along the mid-vein until and including the anterior cross-vein. (B–D) Numbers of ectopic sensory organs; data are presented as mean with SEM. Statistics: Wilcoxon rank sum test on the number of bristles except for MmAth1 andHsAth1 on number of campaniform organs. Summary statistics can be found in Supplementary file 6E,F,I,J,M and N and all p-values (Wilcoxon rank sum test) for all pairwise comparisons in Supplementary file 6G,H,K,L,O and P. See also Figure 5—figure supplement 1.
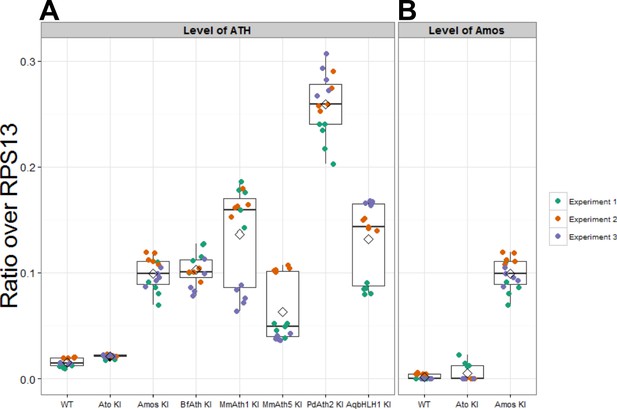
Expression of ATHs and AqbHLH1 is upregulated compared with ato.
(A) Expression levels of ato and KI transgenes in eye-antennal discs (plotted as ratio over RPS13). (B) Expression levels of amos in the eye-antennal imaginal discs (plotted as ratio over RPS13). Note that the values for the amos-KI in (A) and (B) are identical as the experiments were performed in parallel. Boxes indicate interquartile ranges, lines medians, diamonds means and whiskers data ranges.
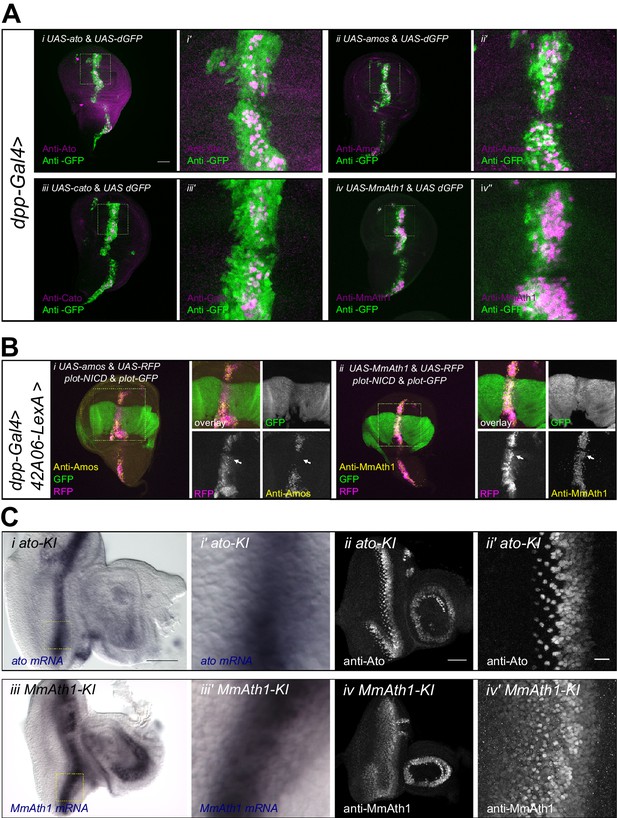
ATH protein steady state dynamics correlate with proneural potency and are regulated by Notch signaling.
(A) Wing disc ectopic expression and immunostainings of (i+i’) ato and dGFP, (ii+ii’) amos and dGFP, (iii+iii’) cato and dGFP, (iv+iv’) MmAth1 and dGFP. (i+ii) overviews (scale bar 50 μm), (i’+ii’) close ups. (B) (i) Anti-Amos; (ii) anti-MmAth1 immunostainings and endogenous fluorescence of RFP and GFP in L3 wing discs. ATHs and RFP are ectopically expressed along the dorso-ventral axis (dpp-GAL4) while 42A06-LexA drives the expression of GFP and NICD. (C) Comparison of the expression levels of ato and MmAth1 mRNAs and proteins; (i+i’) ato in situ hybridization in an ato-KI eye-antennal disc; (i) overview (scale bar 50 μm) and (i’) close up indicated by square in (i). (ii+ii’) Anti-Ato immunostaining in an ato-KI eye-antennal disc; (ii) overview (scale bar 50 μm); (iii) close up at the MF (scale bar 10 μm). (iii+iii’) MmAth1 in situ hybridization in a MmAth1-KI eye-antennal disc; (iii) overview and (iii’) close up indicated by square in (ii). (iv+iv’) Anti-MmAth1 immunostaining in a MmAth1-KI eye-antennal disc.; (ii) overview; (iii) close up at the MF. Confocal images in A, B and C (ii+ii’; iv, iv’) are maximum intensity projections. See also Figure 6—figure supplement 1.
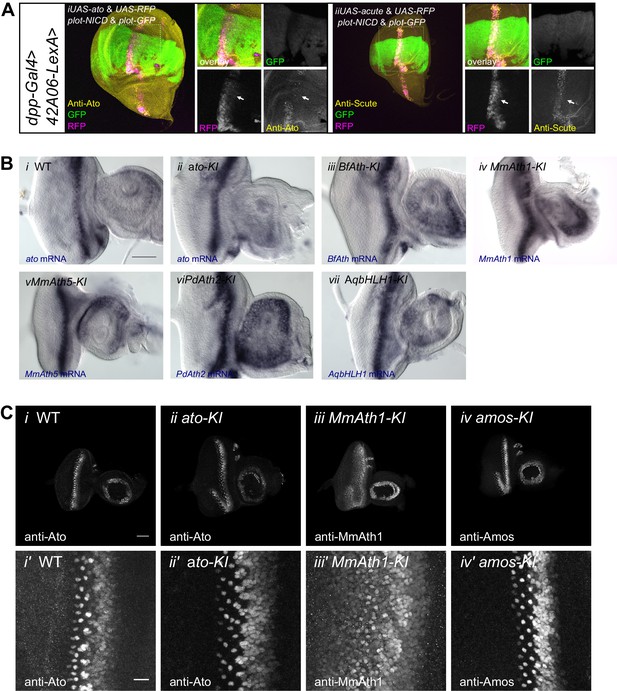
ATH mRNA and protein expression.
(A) (i) Anti-Ato; (ii) anti-Scute immunostainings and endogenous fluorescence of RFP and GFP in L3 wing discs. ato or scute and RFP are ectopically expressed along the dorso-ventral axis (dpp-GAL4) while 42A06-LexA drives the expression of GFP and NICD. (B) In situ hybridization of eye-antennal imaginal discs of WT and KI-lines with anti-sense probes against ato or the transgene (i) WT, (ii) ato-KI, (iii) BfAth-KI, (iv) MmAth1-KI, (v) MmAth5-KI, vi) PdAth2-KI, vii) AqbHLH1-KI (scale bar 50 μm). (C) Immunostainings of eye-antennal imaginal discs of WT and KI-lines with anti-Ato or corresponding anti-ATH antibodies; overview (i–iv), close up (i’–iv’) of (i+i’) WT, (ii+ii’) ato-KI, (iii+iii’) MmAth1-KI, iv+iv’) amos-KI (scale bar 50 μm overview and 10 μm close up). All confocal imges (A and C) are maximum intensity projections.
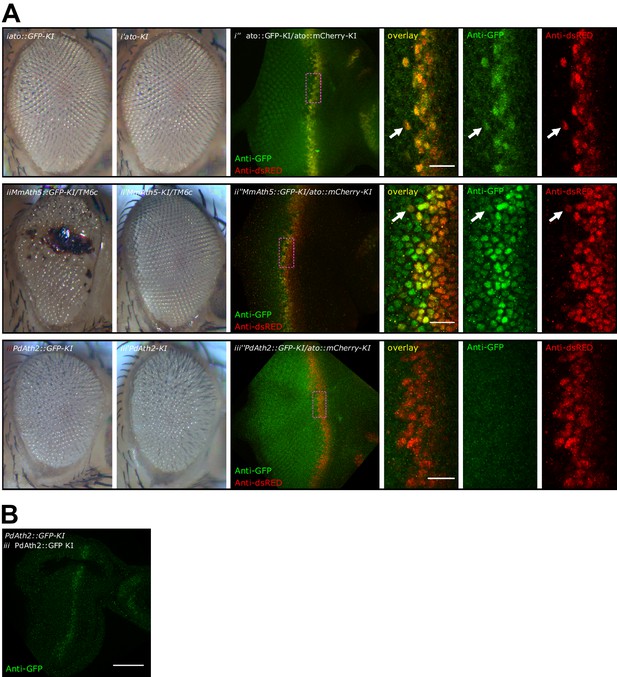
C-terminal tags differently affect ATH function.
(A) Compound eyes of female adults: (i) Ato::GFP-KI, (i’) Ato-KI, (ii) Math5::GFP-KI/TM6c,( ii’) Math5-KI/TM6c, (iii) PdAth2::GFP-KI, (iii’) PdAth2-KI flies. Anti-GFP and anti-dsRed immunolabelings of L3 eye-antennal discs co-expressing a GFP-tagged ATH and cherry-tagged Ato: (i’’) Ato::GFP-KI/Ato::mCherry-KI, (ii’’) MmAth5::GFP-KI/Ato::mCherry-KI, (iii’’) PdAth2::GFP-KI/Ato::mCherry-KI; overviews correspond to maximum intensity projections, close up images are single confocal planes (scale bar 10 μm). (B) Single confocal plane of anti-GFP immunostaining of PdAth2::GFP-KI homozygous L3 eye-antennal discs (scale bar 50 μm).
Tables
Comparison of proneural potency across tissue.
| Tissue | Order |
|---|---|
| R8 | MmAth1 ≈ Ato ≈ Amos > BfAth ≈ AqbHLH1 > MmAth5 > PdAth2 |
| ChO | MmAth1 ≈ Ato ≈ Amos > BfAth >> AqbHLH1 > PdAth2 ≈ MmAth5 |
| Ect. Ex. | MmAth1 >> Ato > Amos > BfAth >> AqbHLH1 > PdAth2 > MmAth5 |
Additional files
-
Supplementary file 1
Organisms for tBLASTn search.
- https://doi.org/10.7554/eLife.26402.017
-
Supplementary file 2
Sources of DNA.
- https://doi.org/10.7554/eLife.26402.018
-
Supplementary file 3
Antibodies used in this study.
- https://doi.org/10.7554/eLife.26402.019
-
Supplementary file 4
Codon adaptation index (CAI) of the genes used in this study.
- https://doi.org/10.7554/eLife.26402.020
-
Supplementary file 5
Primer pairs used for qPCR.
- https://doi.org/10.7554/eLife.26402.021
-
Supplementary file 6
(A) Summary statistics of Figure 2D; number of observations (N), mean, median, standard deviation, 95% confidence interval and p-value of Shapiro Wilk test. (B) p-values of Figure 2D; p-values (t-test) and adjusted p-values (by Holm method). (C) Summary statistics of Figure 3B; number of observations (N), mean, median, standard deviation, 95% confidence interval and p-value of Shapiro Wilk test. (D) p-values of Figure 3B; p-values (Fisher exact test) and adjusted p-values (by Holm method). (E) Summary statistics of Figure 5B for bristles; number of observations (N), mean, median, standard deviation, 95% confidence interval and p-value of Shapiro Wilk test. (F) Summary statistics of Figure 5B for campaniform organs; number of observations (N), mean, median, standard deviation, 95% confidence interval and p-value of Shapiro Wilk test. (G) p-values of Figure 5B for bristles; p-values (Wilcoxon Rank Sum and Signed Rank Tests) and adjusted p-values (by Holm method). (H) p-values of Figure 5B for campaniform organs; p-values (Wilcoxon Rank Sum and Signed Rank Tests) and adjusted p-values (by Holm method) (I) Summary statistics of Figure 5C for bristles; number of observations (N), mean, median, standard deviation, 95% confidence interval and p-value of Shapiro Wilk test. (J) Summary statistics of Figure 5C for campaniform organs; number of observations (N), mean, median, standard deviation, 95% confidence interval and p-value of Shapiro Wilk test. (K) p-values of Figure 5C for bristles; p-values (Wilcoxon Rank Sum and Signed Rank Tests) and adjusted p-values (by Holm method). (L) p-values of Figure 5C for campaniform organs; p-values (Wilcoxon Rank Sum and Signed Rank Tests) and adjusted p-values (by Holm method). (M) Summary statistics of Figure 5D for bristles; number of observations (N), mean, median, standard deviation, 95% confidence interval and p-value of Shapiro Wilk test. (N) Summary statistics of Figure 5D for campaniform organs; number of observations (N), mean, median, standard deviation, 95% confidence interval and p-value of Shapiro Wilk test. (O) p-values of Figure 5D for bristles; p-values (Wilcoxon Rank Sum and Signed Rank Tests) and adjusted p-values (by Holm method). (P) p-values of Figure 5D for campaniform organs; p-values (Wilcoxon Rank Sum and Signed Rank Tests) and adjusted p-values (by Holm method). (Q) Summary statistics for passive antennal mechanics (best frequency, f0; tuning sharpness, Q; apparent mass) across ATH rescues (relating to Figure 4A); number of observations (N), mean, median, standard deviation, standard error, 95% confidence interval and p-values for pair-wise comparison with Ato KI control (t-test for normally distributed data and Mann-Whitney Rank Sum test (MWRS) for non-normally distributed data. Significances are highlighted in color (<0.05 and<0.001). (R) Summary statistics for active antennal mechanics (best frequency, f0; tuning sharpness, Q; energy gain) across ATH rescues (relating to Figure 4A); number of observations (N), mean, median, standard deviation, standard error, 95% confidence interval and p-values for pair-wise comparison with Ato KI control (t-test for normally distributed data and Mann-Whitney Rank Sum test (MWRS) for non-normally distributed data. Significances are highlighted in color (<0.05 and<0.001). (S) Summary statistics for gating compliance analysis (relating to Figure 4—figure supplement 1B) across ATH rescues (number of sensitive ion channels, Ns; number of insensitive ion channels, Ni; single channel gating force of sensitive ion channels, zs; single channel gating force of insensitive ion channels, zi; asymptotic stiffness, Kinf; steady state stiffness, Ksteady; total gating spring stiffness, KGS); number of observations (N), mean, median, standard deviation, standard error, 95% confidence interval and p-values for pair-wise comparison with Ato KI control (t-test for normally distributed data and Mann-Whitney Rank Sum test (MWRS) for non-normally distributed data. Significances are highlighted in color (<0.05 and<0.001). Note that parameter values for the PdAth2-KI were dispensed from statistical comparison to the control condition, as the transduction system in the antennae of PdAth2-KI flies did not comply with the two transducer population model (from Effertz et al., 2012), but rather conformed to a one transducer population model. It was thus not immediately evident how to compare the single transducer populations of PdAth2-KI flies to the two transducer (sensitive and insensitive, respectively) populations of control flies.
- https://doi.org/10.7554/eLife.26402.022
-
Supplementary file 7
Sequences codon optimized genes.
- https://doi.org/10.7554/eLife.26402.023





