The β-alanine transporter BalaT is required for visual neurotransmission in Drosophila
Figures
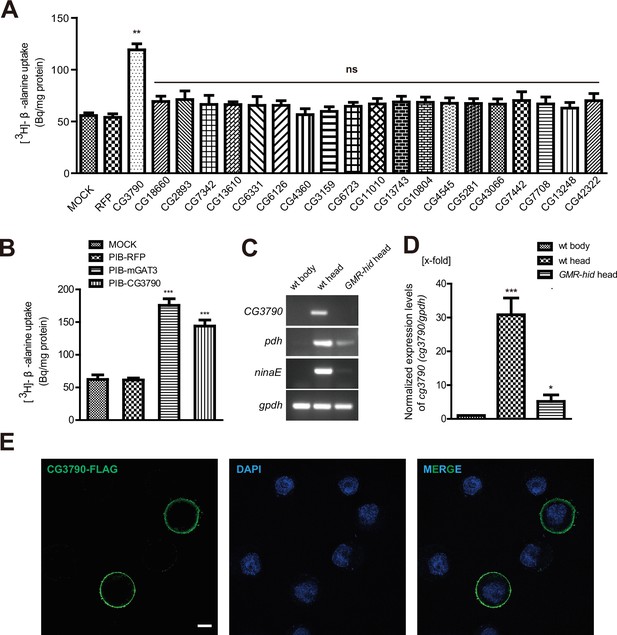
CG3790 is a retina-enriched β-alanine transporter.
(A) Screening for a β-alanine transporter. HEK293T cells transiently expressing one of ~20 head-enriched potential transporters were exposed to [3H]-β-alanine, which was added to the ECF buffer (final concentration 3.7 × 104 Bq). RFP (red fluorescent protein) was expressed as a negative control. (B) CG3790 transported β-alanine into S2 cells. Mouse GAT3 and RFP were used as positive and negative control, respectively. The results given are the mean values ± S.D. of four experiments. (C–D) CG3790 was expressed at high levels in the compound eye. (C) Relative RNA transcript levels (RT-PCR experiments) show that CG3790 expression is enriched in wild-type (w1118) heads compared with wild-type bodies or GMR-hid heads. (D) Relative CG3790 transcript levels from wild-type (wt) bodies, wt heads, and GMR-hid heads (gpdh served as an internal control). RNA levels were normalized to levels in wt bodies, which were set to 1. Error bars indicate standard deviations (SDs) from three replicate experiments. Significant differences between candidates and control were determined using unpaired t-tests (**p<0.01; ns, not significant). (E) S2 cells were transiently transfected with 3xFlag-tagged CG3790, and then labeled with Flag antibody (green) and DAPI (blue). Scale bar, 2 μm.
-
Figure 1—source data 1
Relates to Figure 1A and B.
[3H]-β-alanine uptake data for screening for β-alanine transporter in HEK 293 T cells and S2 cells (.xlsx file).
- https://doi.org/10.7554/eLife.29146.004
-
Figure 1—source data 2
Relates to Figure 1D.
Relative CG3790 transcript levels from wild-type (wt) bodies, wt heads, and GMR-hid heads. RNA levels were normalized to levels in wt bodies, which were set to 1 (.xlsx file).
- https://doi.org/10.7554/eLife.29146.005

CG3790 is a SLC22 family protein.
Alignment of the Drosophila CG3790 amino acid sequence with mouse OCT3 (mOCT3) and human OCT3 (hOCT3). Identical residues, found in at least two proteins, are enclosed in green boxes. CG3790 is 29% identical to mOCT3% and 30% identical to hOCT3. The transmembrane domains are indicated by solid lines above the sequences. The running tally of amino acids is indicated to the right.
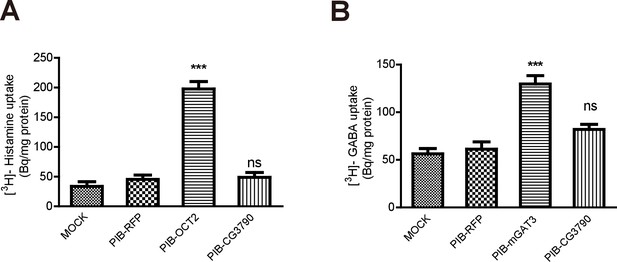
CG3790 is unable to transport histamine and GABA.
(A) CG3790 did not transport GABA into S2 cells. Mouse GAT3 was used as a positive control, and RFP was used as a negative control. (B) CG3790 did not exhibit histamine transporting activity in S2 cells. Human OCT2 and RFP were used as positive and negative control, respectively. [3H]-GABA or [3H]-histamine was added to the ECF buffer (final concentration 3.7 × 104 Bq). The results given are the mean values ± S.D. of three experiments. Significant differences were determined using unpaired t-tests (***p<0.001; ns, not significant).
-
Figure 1—figure supplement 2—source data 1
Relates to Figure 1—figure supplement 2A and B.
[3H]-histamine uptake data in S2 cells transfected mock and RFP negative control or mGAT3. [3H]-GABA uptake data in S2 cells transfected mock and RFP negative control or hOCT2 (.xlsx file).
- https://doi.org/10.7554/eLife.29146.008
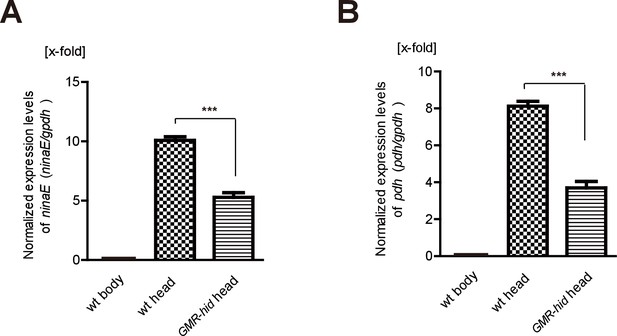
GMR-hid abolished expression of ninaE and pdh.
(A–B) Wild-type heads contain more transcripts for (A) ninaE and (B) pdh than wild-type bodies and GMR-hid heads (gpdh served as an internal control, and mRNA levels were normalized to wild-type body). Data is processed by logarithmic 2 and averaged from three replicate experiments. Error bars indicate SDs. Significant differences between candidates and control were determined using unpaired t-tests (***p<0.001).
-
Figure 1—figure supplement 3—source data 1
Relates to Figure 1—figure supplement 3A and B.
Relative ninaE and pdh transcript levels from wild-type (wt) bodies, wt heads, and GMR-hid heads. RNA levels were normalized to levels in wt bodies. Data is processed by logarithmic 2 (.xlsx file).
- https://doi.org/10.7554/eLife.29146.010
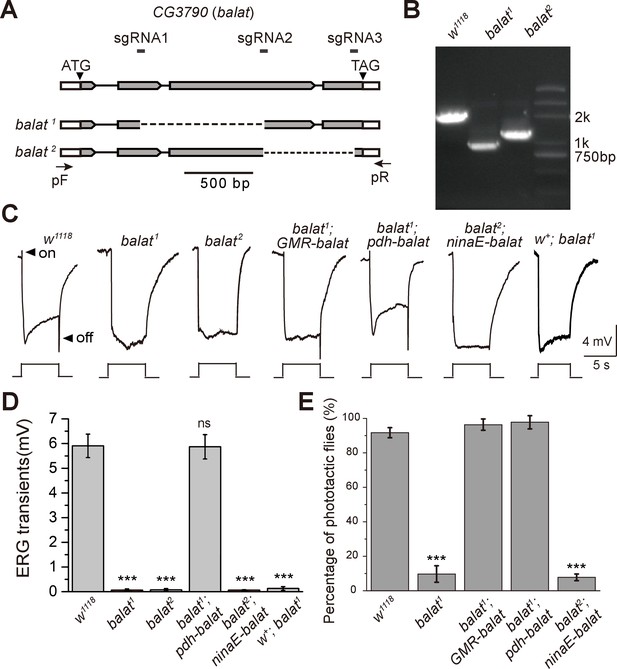
Mutations in balat disrupt photoreceptor cell synaptic transmission and visual behavior.
(A) Schematic for balat knock-out by sgRNA targeting. The organization of the balat locus and the expected structures of balta1 and balat2 alleles are shown. Boxes represent exons with the coding region between ATG and TAG. The sgRNA1 and sgRNA2 primer pair was used to generate the balat1 allele; the sgRNA2 and sgRNA3 primer pair was used to generate the balat2 allele. Arrows indicate the primers used for genomic PCR. (B) PCR products obtained from balat1 and balat2 mutants show successful gene deletions. (C) ERG recordings from: wild-type (w1118), balat1, balat2, balat1;GMR-balaT, balat1;pdh-balaT, balat2;ninaE-balaT and w+; balat1 flies. Young flies (<3 days after eclosion) were dark adapted for 1 min and subsequently exposed to a 5 s pulse of orange light. ON and OFF transients are indicated by arrows. All flies contained the w1118 mutation except for the w+; balat1 flies. (D) Quantitative analysis of the amplitudes of ERG OFF transients shown in C. (E) Phototaxis behaviors of wt, balat1 balat1;GMR-balaT, balat1;pdh-balaT and balat2;ninaE-balaT flies. Significant differences between mutant and wild-type flies were determined using unpaired t-tests (***p<0.001; ns, not significant).
-
Figure 2—source data 1
Relates to Figure 2D and E.
Quantitative analysis of the amplitudes of ERG OFF transients about wild-type (w1118), balat1, balat2, balat1;GMR-balaT, balat1;pdh-balaT, balat2;ninaE-balaT and w+; balat1 flies. Quantitative of phototaxis behaviors of wt, balat1 balat1;GMR-balaT, balat1;pdh-balaT and balat2;ninaE-balaT flies (.xlsx file).
- https://doi.org/10.7554/eLife.29146.013
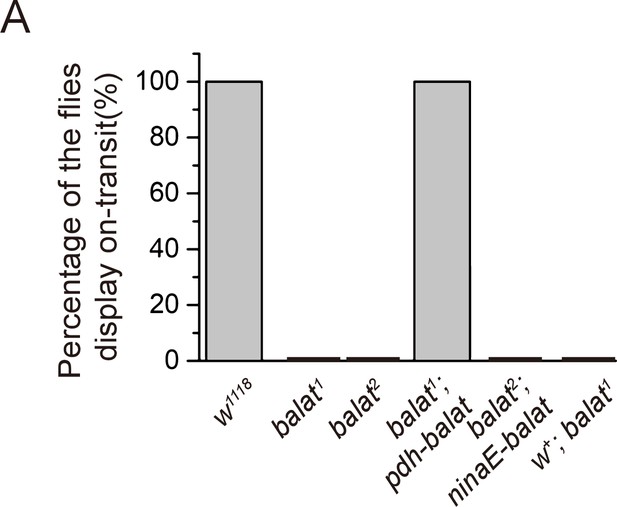
Quantification of the ERG ON transients.
Percentage of flies of indicated genotypes that have ON transients were quantified. The ERG profiles from at least 7 flies of each genotype were scored.
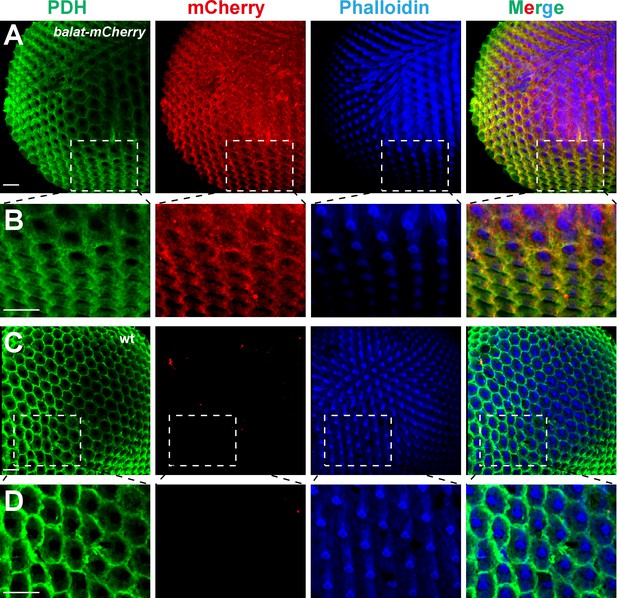
BalaT localizes to pigment cells.
(A–B) Compound eyes from balat-mcherry flies that express mCherry (red) driven by the endogenous balat promoter were labeled for PDH (green), mCherry (red), and phalloidin (blue). High-magnification images are shown in (B). (C–D) mCherry signals were not detected in wild-type (wt) retinas. Scale bars represent 20 µm.
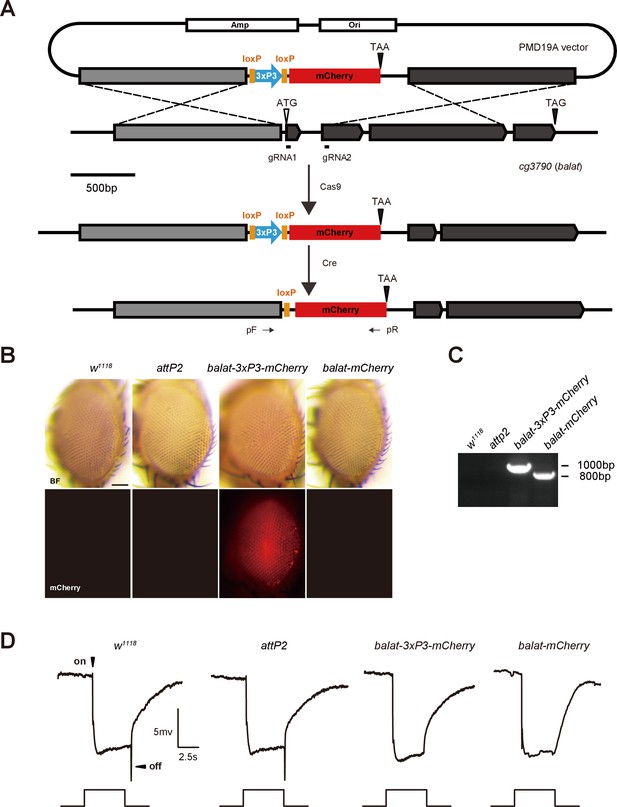
Generation of balat-mcherry flies.
(A) Schematics for generating balat-mcherry flies. The mcherry fluorescent protein, driven by an eye-specific 3XP3 promoter, was inserted into the balat genomic locus using CRISPR/Cas9-mediated homologous recombination. The 3XP3 promoter region was subsequently removed by Cre recombinase. PCR primers (arrows, pF and pR) were used to verify the balat-mcherry knock-in flies. (B) Images show flies with the 3XP3-mcherry reporter inserted into the balat locus (balat-3XP3-mcherry), and the balat-mcherry knock-in flies after Cre/LoxP-mediated recombination. Scale bar represents 150 µm. (C) Genomic PCR products from wild-type (w1118), attP2, balat-3XP3-mcherry, and balat-mcherry flies show successful gene targeting. (D) ERG recordings from wild-type (w1118), attP2, balat-3XP3-mcherry, and balat-mcherry flies. The mcherry insertion caused the loss of ‘on’ and ‘off’ transients.
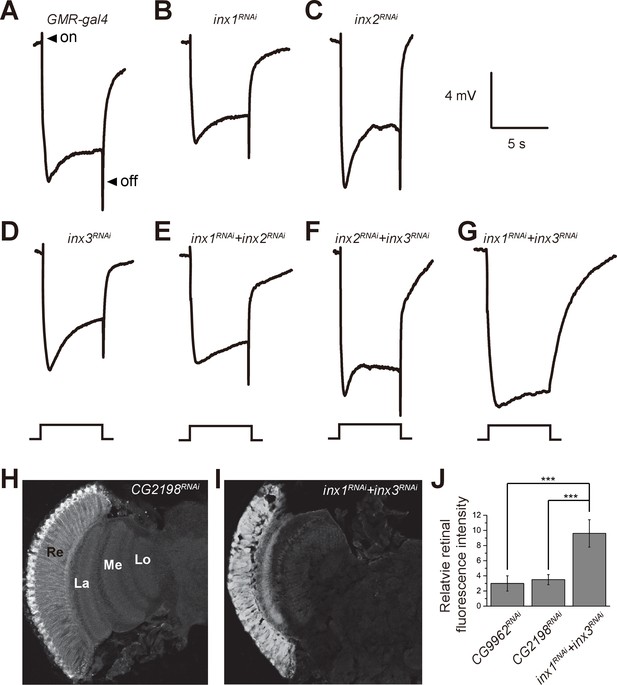
Specific knockdown of Inx1 and Inx3 in compound eyes blocks visual neurotransduction.
ERG recordings of (A) control (GMR-gal4), (B) inx1RNAi (GMR-gal4/ UAS-inx1RNAi), (C) inx2RNAi (GMR-gal4/UAS-inx2RNAi), (D) inx3RNAi (GMR-gal4/ UAS-inx3RNAi), (E) inx1RNAi + inx2RNAi (GMR-gal4/UAS-inx1RNAi UAS-inx1RNAi), (F) inx2RNAi + inx3RNAi (GMR-gal4/UAS-inx2RNAi UAS-inx3RNAi), and (G) inx1RNAi + inx3RNAi (GMR-gal4/UAS-inx1RNAi UAS-inx3RNAi) are shown. Arrows indicate ON and OFF transients in (A). Flies (~1 day after eclosion) were dark adapted for 1 min and subsequently exposed to a 5 s pulse of orange light. (H–I) β-alanine was immunolabeled in horizontal sections of heads from (H) control: CG2198RNAi (GMR-gal4/UAS- CG2198RNAi) and (I) inx1RNAi + inx3RNAi (GMR-gal4/UAS-inx1RNAi UAS-inx3RNAi) flies. Re, retina; La, lamina; Me, medulla; Lo, lobula. (J) Fluorescence intensity ratios of β-alanine signals between retina and lobula. Quantifications of all genotypes are averages of six replicate experiments. Significant differences between inx1RNAi + inx3RNAi and controls (CG2198RNAi and CG9962RNAi) were determined using unpaired t-tests (***p<0.001; ns: not significant).
-
Figure 4—source data 1
Relates to Figure 4J.
Quantitative of fluorescence intensity ratios of β-alanine signals between retina and lobula of inx1RNAi + inx3RNAi and controls (CG2198RNAi and CG9962RNAi) (.xlsx file).
- https://doi.org/10.7554/eLife.29146.018
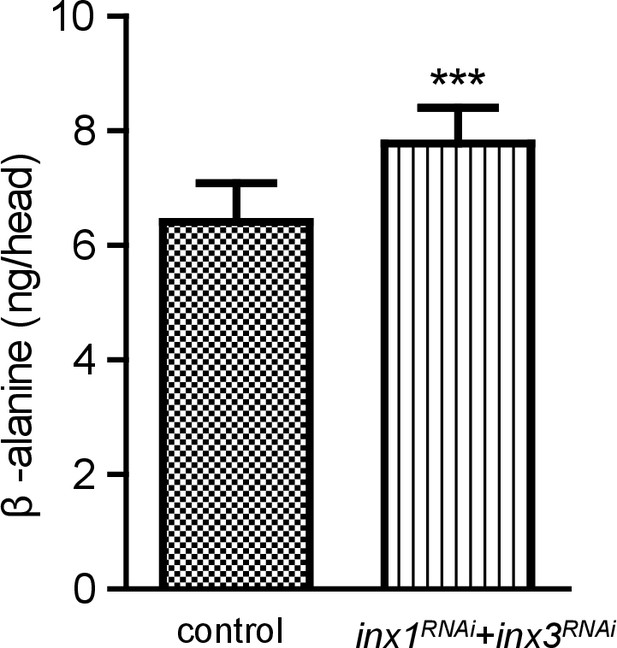
Accumulation of β-alanine in heads of Inx1 and Inx3 double RNAi flies.
Levels of β-alanine in heads of control and inx1RNAi + inx3RNAi (GMR-gal4/UAS-inx1RNAi UAS-inx3RNAi) flies. Error bars indicate SDs, and significant differences between mutant and wt flies were determined using unpaired t-tests (***p<0.001).
-
Figure 4—figure supplement 1—source data 1
Relates to Figure 4—figure supplement 1.
Quantitative of levels of β-alanine in heads of control and inx1RNAi + inx3RNAi (GMR-gal4/UAS-inx1RNAi UAS-inx3RNAi) flies (.xlsx file).
- https://doi.org/10.7554/eLife.29146.020
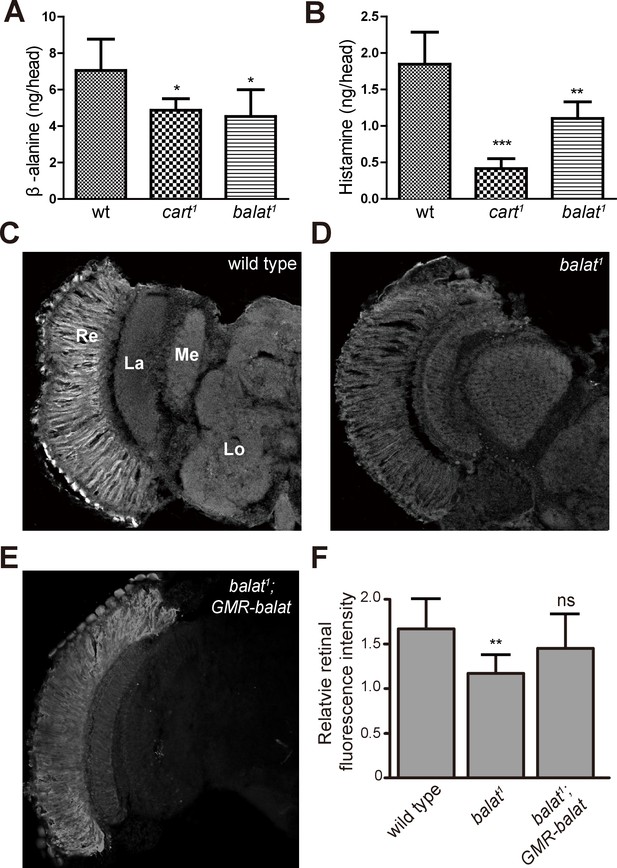
Loss of BalaT affects the level and distribution of β-alanine in vivo.
(A–B) Levels of (A) β-alanine and (B) histamine in heads of wild-type (wt, w1118), cart1, and balat1 flies are shown. Error bars indicate SDs, and significant differences between mutant and wt flies were determined using unpaired t-tests (*p<0.05; **p<0.01). (C–E) β-alanine was immunolabeled in horizontal sections of heads from (C) wild-type (w1118), (D) balat1, and (E) balat1;GMR-balat flies. Re, retina; La, lamina; Me, medulla; Lo, lobula. (F) Fluorescence intensity ratios of β-alanine labeling between retina and lobula. Significant differences between mutant and wild-type flies were determined using unpaired t-tests (**p<0.01; ns: not significant).
-
Figure 5—source data 1
Relates to Figure 5A and B.
Quantitative of levels of β-alanine and histamine in heads of wild-type, cart1, and balat1 flies (.xlsx file).
- https://doi.org/10.7554/eLife.29146.022
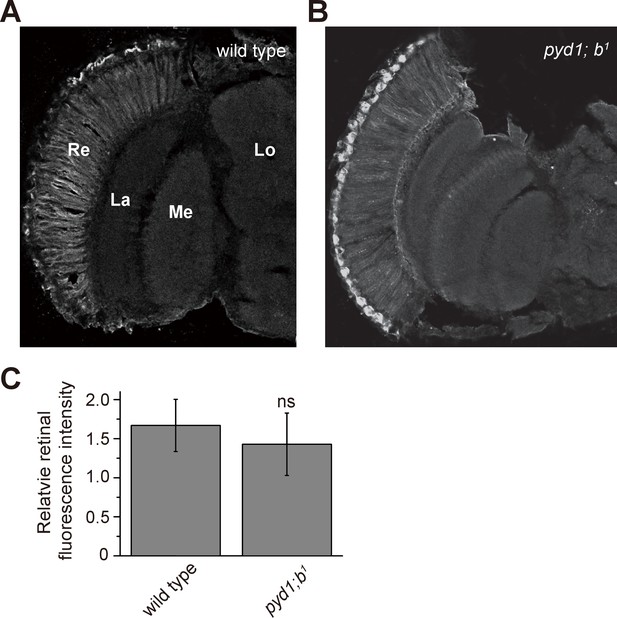
Distribution of β-alanine was unaffected in pyd1 and balck double mutant flies.
β-alanine was immunolabeled in horizontal sections of heads from (A) wild type (w1118) and (B) pyd1;b1 flies. Re, retina; La, lamina; Me, medulla; Lo, lobula. (C) Fluorescence intensity ratios of β-alanine signals between retina and lobula. Quantifications of both genotypes are averages of six replicate experiments. Significant difference between wild type and pyd1;b1 was determined using unpaired t-tests (ns: not significant).
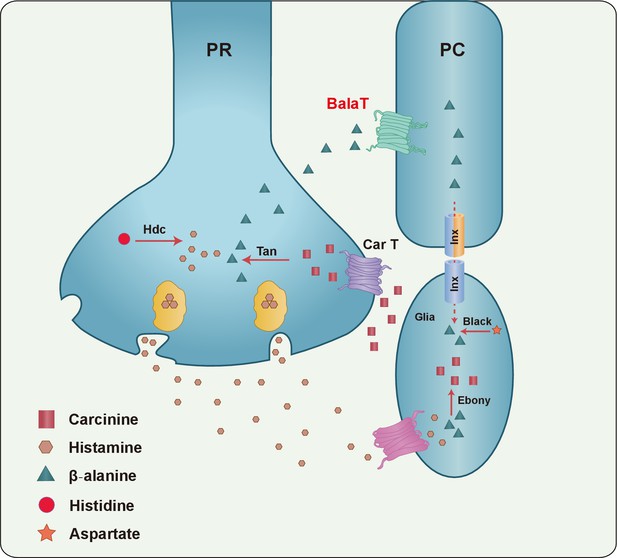
A model of the histamine recycling pathway.
Histamine is initially synthesized by histidine decarboxylase (Hdc) in photoreceptor cells (PR). Upon light stimulation, PRs release histamine into the synaptic cleft. Released histamine is quickly taken up by an unknown histamine transporter into epithelial glial cells that express Ebony. In these glia, histamine is conjugated to β-alanine, which inactivates histamine and generates carcinine. Carcinine is released into the synaptic cleft and subsequently internalized, via CarT, by the PRs. After carcinine is hydrolyzed to histamine and β-alanine by Tan hydrolase in the PR, histamine is re-packaged into synaptic vesicles, whereas β-alanine is delivered to and subsequently internalized, via the BalaT transporter, by retinal pigment cells (PCs). PCs can store β-alanine or deliver β-alanine to the laminar glia cells through a gap junction network involving Inx1 and Inx3, which are expressed in PCs. In glial cells, β-alanine is conjugated to histamine, and the cycle repeats. Moreover, β-alanine can be synthesized by Black, which is an aspartate decarboxylase that is expressed together with Ebony in optic lobe glia.
Tables
Description of 20 head-enriched genes.
| CG number | Name | Description |
|---|---|---|
| CG3790 | Organic anion transmembrane transporter activity | |
| CG18660 | Nckx30C | Sodium/potassium/calcium exchanger |
| CG2893 | zydeco | Potassium-dependent sodium/calcium exchanger |
| CG7342 | Organic anion transmembrane transporter activity | |
| CG13610 | Orct2 | Organic cation transporter |
| CG6331 | Orct | Organic anion transmembrane transporter activity |
| CG6126 | Organic anion transmembrane transporter activity | |
| CG4360 | Nucleic acid binding | |
| CG3159 | Eaat2 | L-aspartate transmembrane transporter activity |
| CG6723 | Sodium/solute symporter | |
| CG11010 | Ent3 | Nucleoside transmembrane transporter activity |
| CG13743 | Amino acid transporter | |
| CG10804 | Neurotransmitter transporter activity | |
| CG4545 | SerT | Monoamine transmembrane transporter activity |
| CG5281 | EamA domain | |
| CG43066 | Neurotransmitter transporter activity | |
| CG7442 | SLC22A | Choline transmembrane transporter activity |
| CG7708 | Sodium/solute symporter | |
| CG13248 | Amino acid/polyamine transporter I | |
| CG42322 | Solute carrier family 35 member F3/F4 |





