MPDZ promotes DLL4-induced Notch signaling during angiogenesis
Figures
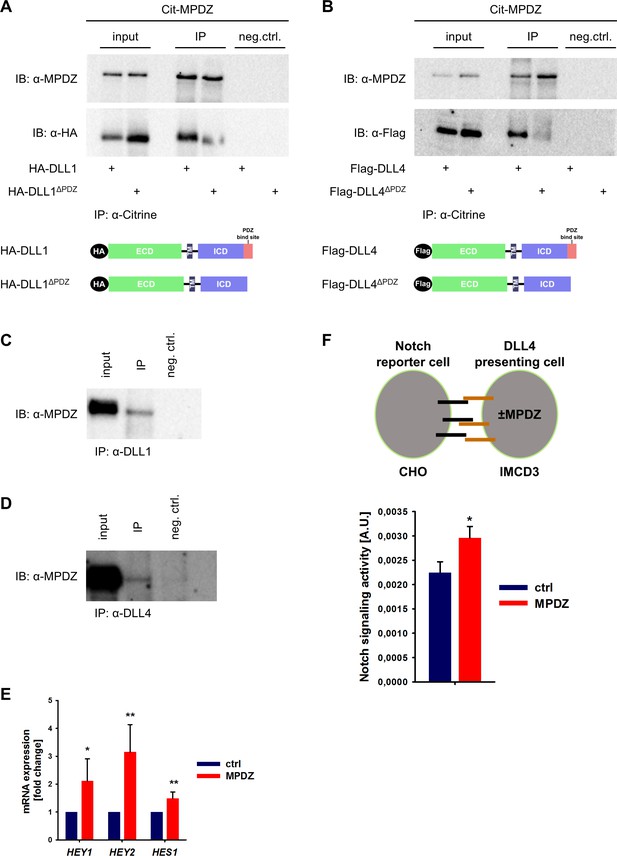
MPDZ interacts with DLL1 and DLL4.
(A, B) HEK293T cells were transfected with Citrine-MPDZ together with HA-tagged DLL1, HA-tagged DLL1ΔPDZ (lacking the PDZ-binding site), Flag-tagged DLL4 or Flag-tagged DLL4ΔPDZ. Antibodies against Citrine were used to immunoprecipitate Citrine-MPDZ. HA and FLAG-tagged proteins as well as MPDZ were detected by immunoblot (IB). Scheme shows structures of the constructs used for co-immunoprecipitation. Input, 10% of the immunoprecipitate. Cit-MPDZ, Citrine-MPDZ; IP, immunoprecipitation; neg.ctrl., negative control. (C, D) MPDZ was co-expressed with either DLL1 or DLL4 in primary endothelial cells (HUVEC). DLL1 and DLL4 were pulled down by using specific antibodies. DLL1, DLL4 and MPDZ were detected by immunoblot (IB). Input, 5% of the immunoprecipitate. IP, immunoprecipitation; neg.ctrl., negative control. (E) HUVEC were either transduced with adenovirus expressing GFP (ctrl) or MPDZ (MPDZ). Expression level of Notch target genes HEY1, HEY2 and HES1 were analyzed by qPCR 48 hr after transduction. Data are presented as mean ±SD. n = 4; *, p<0.05; **, p<0.01 unpaired Student’s t-test. (F) Scheme of the co-culture Notch reporter assay. IMCD3 cells expressing the Notch ligand DLL4 were co-cultured with CHO-N1-CIT cells carrying a Notch luciferase reporter construct. The IMCD3 sender cells were modified by expression of MPDZ or an empty vector control. After 48 hours, cells were lysed and the light emission of the luciferin and the Renilla luciferase activities were measured. Signaling activity is calculated by normalizing the luciferase signal with the Renilla signal. Data are presented as mean ± SEM. n = 5; *, p<0.05 unpaired Student’s t-test.
-
Figure 1—source data 1
Source data of qantitative PCR analysis related to Figure 1E.
- https://doi.org/10.7554/eLife.32860.005
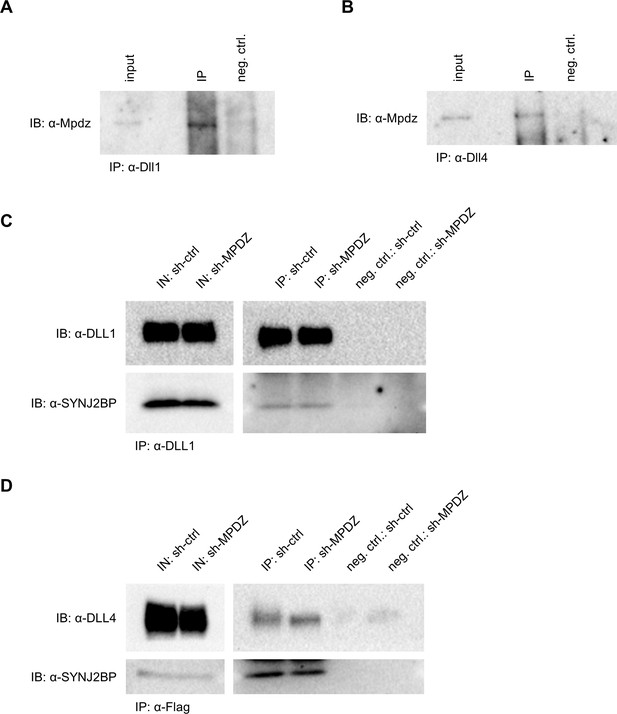
MPDZ interacts with DLL1 and DLL4.
(A, B) Dll1 and Dll4 were immunoprecipitated by using specific antibodies from lysates of mouse kidneys. Mpdz was detected by immunoblot (IB). Input, 10% of the immunoprecipitate. IP, immunoprecipitation; neg.ctrl., negative control. (C, D) HEK293T control cells (293T sh-ctrl) as well as MPDZ-silenced HEK293T cells (293T sh-MPDZ) were transfected with SYNJ2BP and HA-tagged DLL1 or Flag-tagged DLL4. For immunoprecipitation either a DLL1 or a Flag-tag antibody was used and DLL1, DLL4 and SYNJ2BP were detected by Western Blotting. Input, 10% of immunoprecipitate; IB, Immunoblot; IP, Immunoprecipitation; neg.ctrl., negative control.
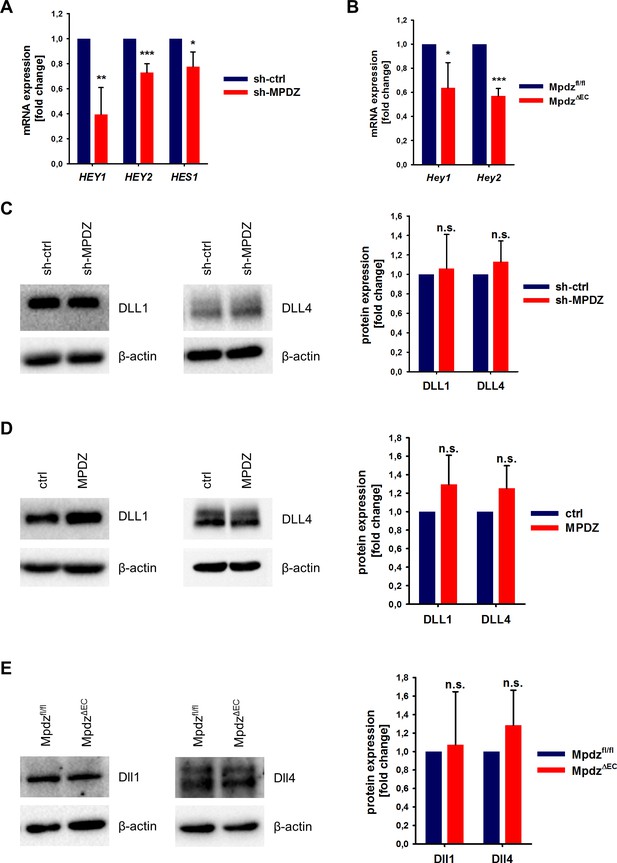
MPDZ promotes Notch signaling activity.
(A) HUVECs were either transduced with lentivirus expressing GFP (sh-ctrl) or with lentivirus expressing shRNA against MPDZ (sh-MPDZ). Expression level of Notch target genes HEY1, HEY2 and HES1 were analyzed by qPCR 48 hr after transduction. Data are presented as mean ±SD. n ≥ 3; *, p<0.05; **, p<0.01; ***, p<0.001 unpaired Student’s t-test. (B) Cardiac endothelial cells were isolated from Mpdzfl/fl and MpdzΔEC mice by magnetic beads bound with CD31 antibodies. Expression levels of Notch target genes Hey1 and Hey2 were analyzed by qPCR. Data are presented as mean ±SD. n = 3; *, p<0.05; ***, p<0.001 unpaired Student’s t-test. (C) HUVECs were either transduced with lentivirus expressing GFP (sh-ctrl) or with lentivirus expressing shRNA against MPDZ (sh-MPDZ). Expression levels of DLL1 and DLL4 were analyzed by immunoblotting 48 hr after transduction. β-actin served as loading control. Data are presented as mean ±SD. n ≥ 3; n.s., not significant. (D) HUVECs were either transduced with adenovirus expressing GFP (ctrl) or with adenovirus expressing MPDZ. Expression levels of DLL1 and DLL4 were analyzed by immunoblotting 48 hr after transduction. β-actin served as loading control. Data are presented as mean ±SD. n ≥ 3; n.s., not significant. (E) Lung endothelial cells were isolated from Mpdzfl/fl and MpdzΔEC mice by CD31 magnetic beads. Protein amounts of Dll1 and Dll4 were analyzed by immunoblotting. β-actin served as loading control. Data are presented as mean ±SD. n = 3; n.s., not significant.
-
Figure 2—source data 1
Source data of qantitative PCR analysis related to Figure 2A and B.
- https://doi.org/10.7554/eLife.32860.007
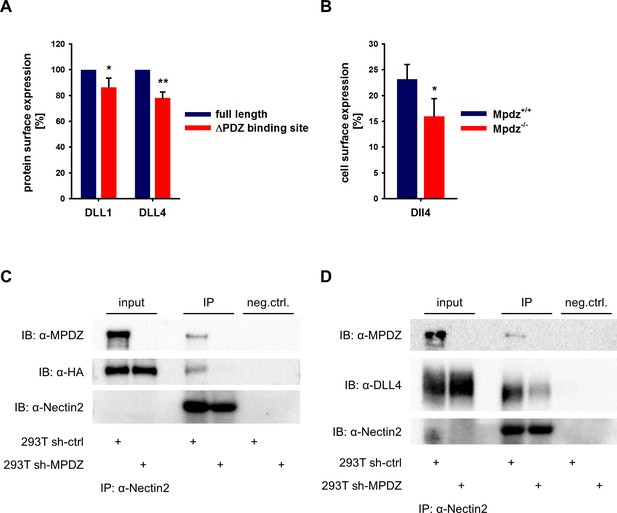
MPDZ recruits DLL1 and DLL4 to Nectin-2.
(A) DLL1 and DLL4 full length and such lacking the PDZ-binding site (ΔPDZ) constructs containing a mCherry tag were expressed in HUVEC. Cells were stained with antibodies against DLL1 or DLL4. DLL1 and DLL4 surface expression of mCherry positive cells was analyzed by flow cytometry. n = 3; *, p<0.05; **, p<0.01 unpaired Student’s t-test. (B) Endothelial cells were isolated from Mpdz+/+ embryos and Mpdz-/- littermates at embryonic day E11.5. Cells were purified by CD31 magnetic dynabeads and stained with anti-CD34 and anti-Dll4 antibodies for flow cytometric analysis. n = 4; *, p<0.05; unpaired Student’s t-test. (C, D) HEK293T control cells (293T sh-ctrl) as well as MPDZ-silenced HEK293T cells (293T sh-MPDZ) were transfected with Nectin-2 and HA-tagged DLL1 or Flag-tagged DLL4. For immunoprecipitation a Nectin-2 antibody was used and HA-tagged DLL1, Flag-tagged DLL4 and MPDZ were detected by western Blotting. Input, 10% of immunoprecipitate. IB, Immunoblot; IP, Immunoprecipitation; neg.ctrl., negative control.
-
Figure 3—source data 1
Source data of FACS analysis related to Figure 3A and B.
- https://doi.org/10.7554/eLife.32860.010
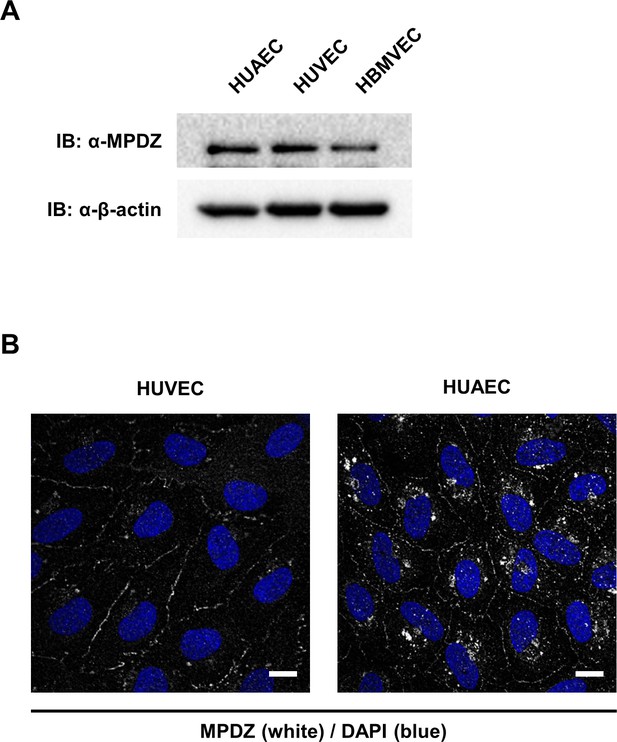
MPDZ recruits DLL1 and DLL4 to Nectin-2.
(A) Human umbilical vein endothelial cells (HUVEC), human umbilical artery endothelial cells (HUAEC), human brain microvessel endothelial cells (HBMVEC) were lysed, proteins isolated and expression level of MPDZ was analyzed by immunoblotting. β-actin served as loading control. (B) Methanol-fixed HUVEC and HUAEC were stained for MPDZ and counterstained with DAPI. Images were acquired with the confocal microscope LSM 700. Scale bar: 10 µm. Staining of MPDZ in HBMVEC did not give specific signals.
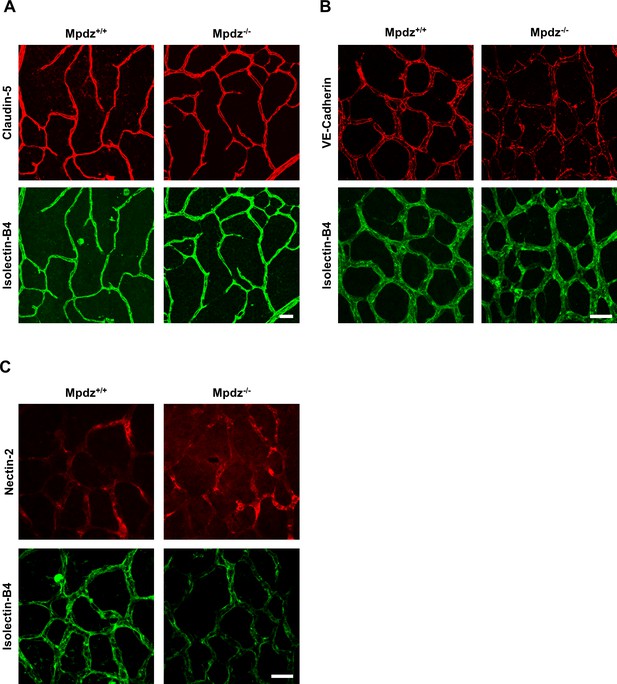
Mpdz does not affect cell cell junction assembly.
(A) Retinae isolated from 14 days old Mpdz-/- pups and control littermates (Mpdz+/+) were stained with Isolectin-B4 and antibodies against Claudin-5. Images were acquired with the confocal microscope LSM 700. Scale bar: 25 µm. (B, C) Retinae were isolated 9 days old Mpdz-/- pups and control littermates. Staining for endothelial cells with Isolectin-B4 and VE-Cadherin or Nectin-2. Images were acquired with the confocal microscope LSM 700. Scale bar: 25 µm.
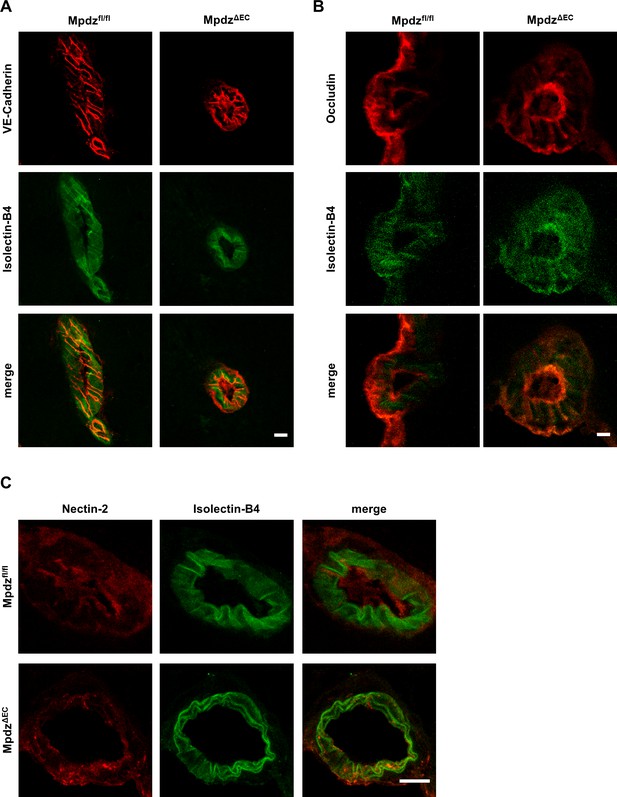
Mpdz does not affect cell junction assembly.
(A, B, C) Brains were resected from 8 week old MpdzΔEC mice and control littermates (Mpdzfl/fl). Sections were stained with Isolectin-B4 and antibodies against VE-Cadherin (A), Occludin (B) and Nectin-2 (C). Images were acquired with the confocal microscope LSM 700. Scale bar: 10 µm.
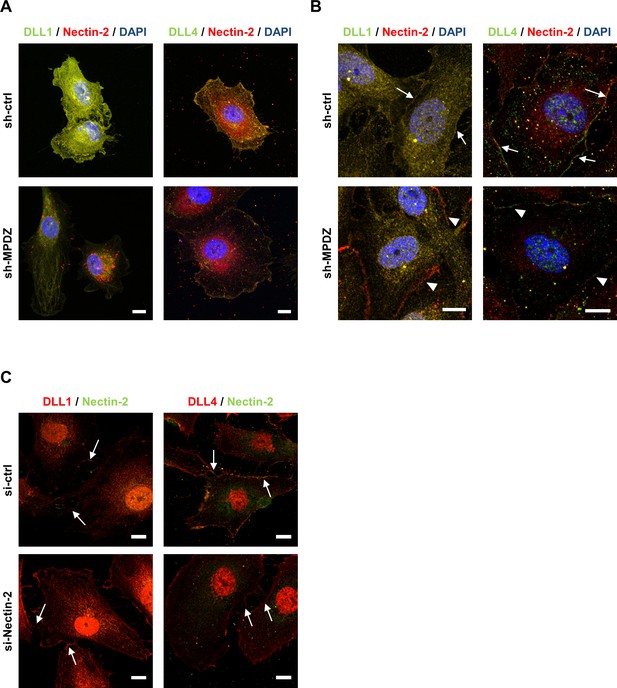
Mpdz does not affect cell cell junction assembly.
(A, B) HUVECs were either transduced with lentivirus expressing GFP (sh-ctrl) or with lentivirus expressing shRNA against MPDZ (sh-MPDZ). Cells were cultured under sparse conditions (A) or confluent conditions (B). After PFA fixation cells were stained for DLL1 and Nectin-2 or DLL4 and Nectin-2 and counterstained with DAPI. Images were acquired with the confocal microscope LSM 700. Arrow indicates co-localization of DLL1/4 with Nectin-2 at the cell membrane. Arrow head indicates diminished co-localization at the cell membrane. Scale bar: 10 µm. (C) HUVECs were either transfected with control siRNA (si-ctrl) or with siRNA against Nectin-2 (si-Nectin-2). After PFA fixation cells were stained for DLL1 and Nectin-2 or DLL4 and Nectin-2. Images were acquired with the confocal microscope LSM 700. Arrow indicates localization of DLL1/4 at the cell membrane.Scale bar: 10 µm.
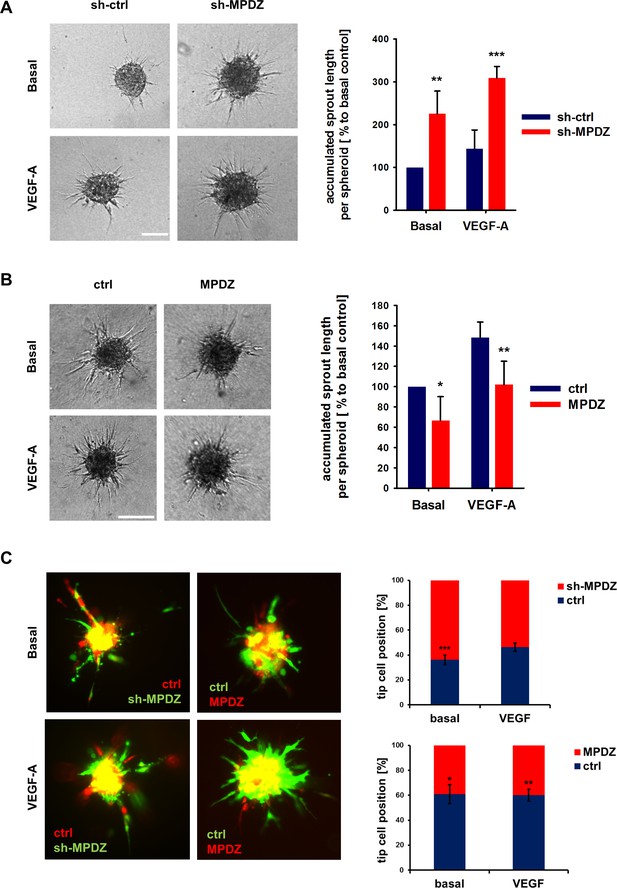
MPDZ inhibits sprouting angiogenesis in vitro.
(A) HUVEC were transduced with lentivirus-expressing shRNA against MPDZ (sh-MPDZ) or expressing GFP (sh-ctrl). Sprouting angiogenesis of collagen-embedded spheroids was analyzed 72 hr after transduction. Spheroids were cultured under basal conditions or stimulated with VEGF-A (25 ng/ml). Quantification shows length of all sprouts of each spheroid. n = 4 experiments with 10 spheroids per condition. **, p<0.01; ***, p<0.001 one-way ANOVA (Holm-Sidak method). (B) HUVEC were transduced with control (ctrl) or MPDZ-expressing adenovirus. Sprouting angiogenesis of collagen-embedded spheroids was analyzed 72 hr after transduction. Spheroids were cultured under basal conditions or stimulated with VEGF-A (25 ng/ml). Quantification shows length of all sprouts of each spheroid. n = 5 experiments with 10 spheroids per condition. *, p<0.05; **, p<0.01; One Way ANOVA (Holm-Sidak method). (C) Mixed spheroids of HUVEC transduced with lentivirus expressing shRNA against MPDZ (sh-MPDZ) or expressing mCherry (ctrl) or spheroids of HUVEC transduced with control (ctrl) or MPDZ-expressing (MPDZ) adenovirus were embedded in a collagen matrix and analyzed 72 hr after transduction. Cells at the most distal end were considered as tip cells. Tip cell numbers were analyzed under basal conditions or after stimulation with VEGF-A (25 ng/ml). n = 3 experiments with 10 spheroids per condition. *, p<0.05; **, p<0.01; ***, p<0.001 unpaired Student’s t-test.
-
Figure 5—source data 1
Source data of the sprouting assay and the tip-stalk-cell competition assay related to Figure 5 A, B, C.
- https://doi.org/10.7554/eLife.32860.015
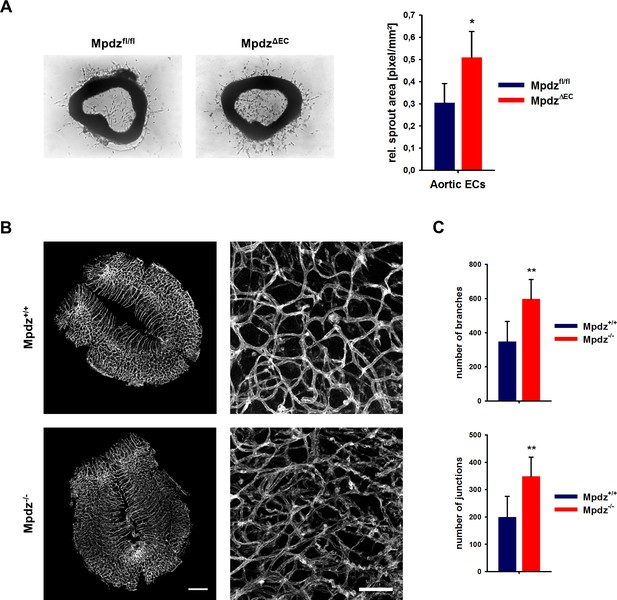
Loss of Mpdz leads to increased vessel branching in the embryonic mouse hindbrain.
(A) Aortae were isolated from Mpdzfl/fl and MpdzΔEC mice. Aortic rings were embedded in Matrigel and EC outgrowth was analyzed 24 hr after embedding. n = 4 mice per genotype; *, p<0.05 unpaired Student’s t-test. (B) Embryos at developmental stage E12.5 were used for IsolectinB4-FITC staining (endothelial cells). Left panel shows whole hindbrains of Mpdz+/+ and Mpdz-/- embryos. Right panel shows zoom-ins. Left panel: scale bar, 500 µm; Right panel: scale bar, 100 µm. (C) Quantification of vessel branches and junctions per area. n ≥ 6 mice per genotype; **, p<0.01 unpaired Student’s t-test.
-
Figure 6—source data 1
Source data of the aortic ring assay and the blood vessel analysis of embryonic hindbrains related to Figure 6 A and C.
- https://doi.org/10.7554/eLife.32860.017
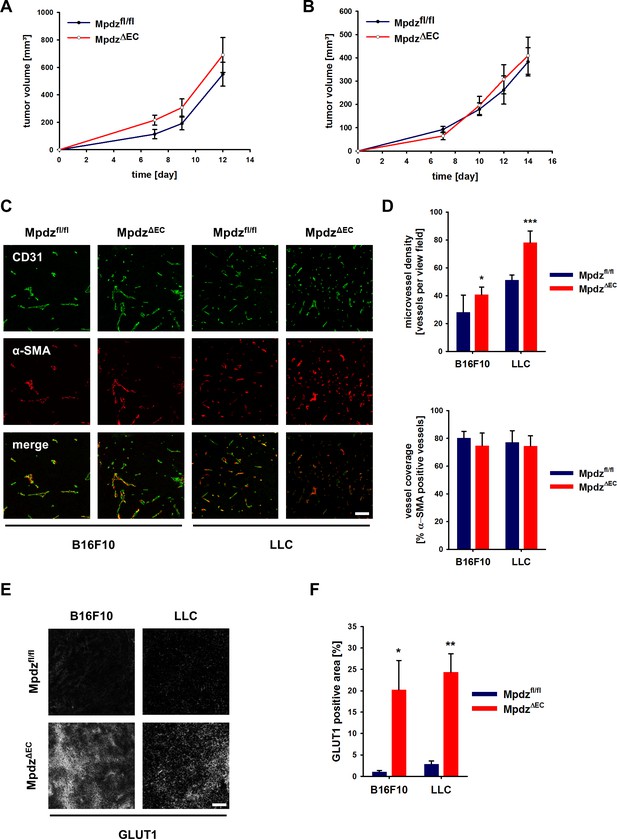
Excessive tumor angiogenesis upon endothelial-specific inactivation of Mpdz.
Tumor growth curve of B16F10 (A) and LLC (B) tumors subcutaneously implanted into Mpdzfl/fl and MpdzΔEC mice. n ≥ 5; Results are shown as mean ±SEM. (C) Representative images of B16F10 and LLC tumors grown in Mpdzfl/fl and MpdzΔEC mice, stained against CD31 (endothelial cells) and α-SMA (smooth muscle cells). Scale bar: 100 µm (D) Quantification of the vessel staining. Microvessel density was determined by counting the CD31-positive vessels per area. For the analysis of vessel coverage, the percentage of α-SMA-positive vessels was determined. n ≥ 5; results are shown as mean ±SD; *, p<0.05; ***, p<0.001; unpaired Student’s t-test. (E) Representative images of B16F10 and LLC tumors stained for Glut1 (hypoxia marker) grown in Mpdzfl/fl and MpdzΔEC mice. Scale bar: 100 µm (F) Quantification of the Glut1-positive area. n ≥ 4; results are shown as mean ±SEM; *, p<0.05; **, p<0.01; unpaired Student’s t-test. Figure legends – figure supplements.
-
Figure 7—source data 1
Source data of the microvessel density analysis and the Glut1 expression analysis related to Figure 7D and F.
- https://doi.org/10.7554/eLife.32860.020
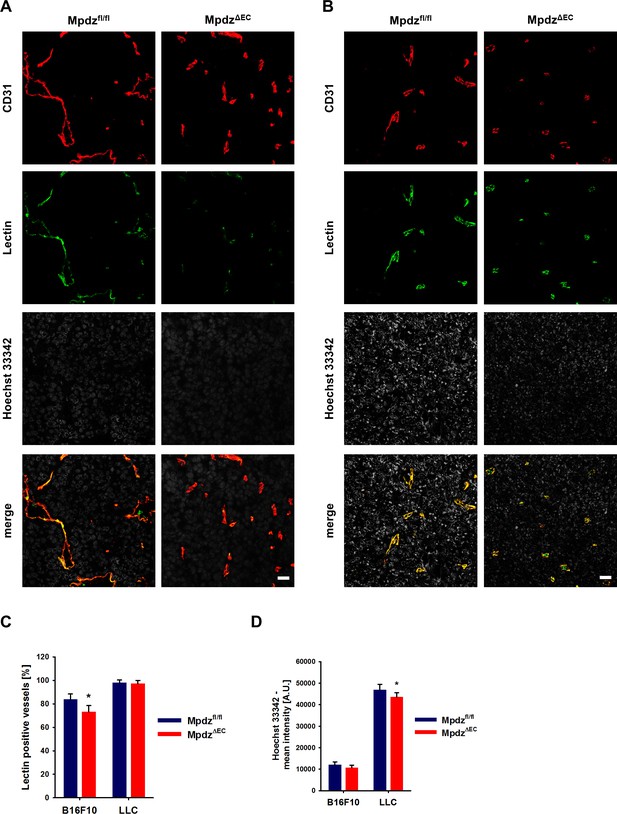
Excessive tumor angiogenesis upon endothelial-specific inactivation of Mpdz.
(A, B) Representative images of B16F10 (A) or LLC (B) tumors subcutaneously implanted into Mpdzfl/fl and MpdzΔEC mice. Sections show CD31-immunolabeled and fluorescein-lectin-labeled tumor vessels and Hoechst-33342-positive tumor areas. Lycopersicon Esculentum lectin and Hoechst 33342 was tail vein injected 5 min prior to sacrifice. Scale bar: 25 µm (C) Quantification of the lectin-positive vessels. n ≥ 3; results are shown as mean ±SD; *, p<0.05; unpaired Student’s t-test. (D) Quantification of the mean intensity of Hoechst 33342 per area. n ≥ 3; results are shown as mean ±SD; *, p<0.05; unpaired Student’s t-test.
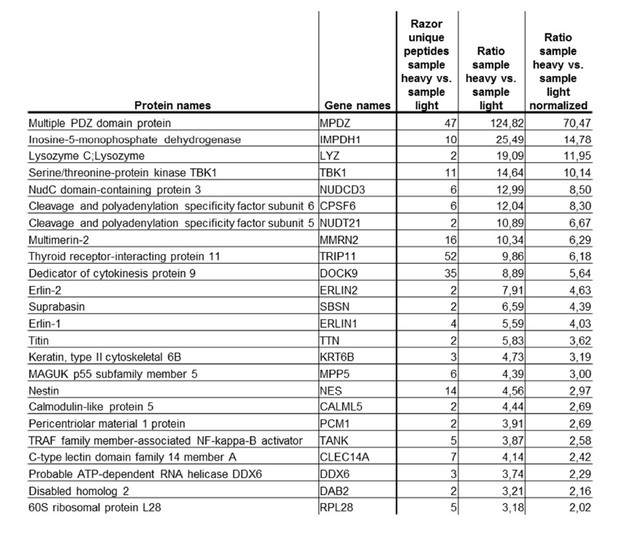
Tables
| Reagent type (species) or resource | Designation | Source or reference | Identifiers | Additional information |
|---|---|---|---|---|
| Strain, strain background (Mus musculus) | Mpdz-/- | DOI: 10.15252/emmm.201606430 | ||
| Strain, strain background (Mus musculus) | MpdzΔEC | DOI: 10.15252/emmm.201606430 | ||
| Genetic reagent (Homo sapiens) | MPDZ shRNA | Biocat | V2LHS_3656, 16945 16946 | |
| Genetic reagent (Homo sapiens) | Nectin-2 siRNA | Origene | SR321541 | |
| Antibody | MPDZ | Sigma-Aldrich | HPA020255 | Western Blot (1:500)/ICC (1:50) on methanol fixed cells |
| Antibody | Mpdz | Invitrogen | 42–2700 | Western Blot (1:500) |
| Antibody | HA | Cell Signaling | #3724 | Western Blot (1:1000) |
| Antibody | Flag | Sigma-Aldrich | F3165 | Western Blot (1:1000) |
| Antibody | DLL1 | abcam | ab85346 | Western Blot (1:1000)/ICC (1:100) on PFA fixed cells |
| Antibody | Dll1 | R and D Systems | AF3970 | Western Blot (1:500) |
| Antibody | DLL4 | Cell Signaling | #2589 | Western Blot (1:1000) |
| Antibody | DLL4 | Sigma-Aldrich | WH0054567M4 | ICC (1:100) on PFA fixed cells |
| Antibody | Dll4 | R and D Systems | AF1389 | Western Blot (1:500) |
| Antibody | SYNJ2BP | abcam | ab69431 | Western Blot (1:250) |
| Antibody | GFP | abcam | ab290 | Western Blot (1:2500) |
| Antibody | Nectin-2 | abcam | ab135246 | ICC (1:50) on PFA fixed cells |
| Antibody | Nectin-2 | Santa Cruz Biotechnology | sc-32804 | Western Blot (1:500)/ICC (1:50) on PFA fixed cells |
| Antibody | Nectin-2 | abcam | ab16912 | ICC (1:100) on fresh frozen sections |
| Recombinant DNA reagent | MPDZ | BioCat | clone BC140793 | |
| Recombinant DNA reagent | Citrine-MPDZ | this paper | Citrine tag fused to the N-terminus of MPDZ (BioCat, clone BC140793) | |
| Recombinant DNA reagent | DLL1 | OpenBiosystems | OHS4559-99847851 | |
| Recombinant DNA reagent | HA-DLL1 | this paper | Gateway cloning: Dll1 (OHS4559-99847851) into pDest26-HA | |
| Recombinant DNA reagent | DLL1-mCherry | this paper | mCherry inserted between extracellular and transmembrane domain of DLL1. Gateway vector: pLenti6.2 | |
| Recombinant DNA reagent | HA-DLL1ΔPDZ | this paper | Stop codon inserted before PDZ-binding site by site-directed mutagenesis. Gateway vector: pDest26-HA | |
| Recombinant DNA reagent | DLL1-mCherryΔPDZ | this paper | Stop codon inserted before PDZ-binding site by site-directed mutagenesis. Gateway vector: pLenti6.2 | |
| Recombinant DNA reagent | DLL4 | PMID:17045587 | cDNA cloned into pEntr3C | |
| Recombinant DNA reagent | Flag-DLL4 | this paper | cDNA cloned into pCS2p-FLAG | |
| Recombinant DNA reagent | DLL4-mCherry | this paper | mCherry inserted between extracellular and transmembrane domain of DLL4. Gateway vector: pLenti6.2 | |
| Recombinant DNA reagent | Flag-DLL4ΔPDZ | this paper | Stop codon inserted before PDZ-binding site by site-directed mutagenesis. Vector: pCS2p-FLAG | |
| Recombinant DNA reagent | DLL4-mCherryΔPDZ | this paper | Stop codon inserted before PDZ-binding site by site-directed mutagenesis. Gateway vector: pLenti6.2 | |
| Recombinant DNA reagent | Nectin-2 | DKFZ Genomics and Proteomics Core Facility | BC003091 (cDNA). Gateway vector: pLenti6.2 |
Additional files
-
Transparent reporting form
- https://doi.org/10.7554/eLife.32860.021





