Concerted IL-25R and IL-4Rα signaling drive innate type 2 effector immunity for optimal helminth expulsion
Figures
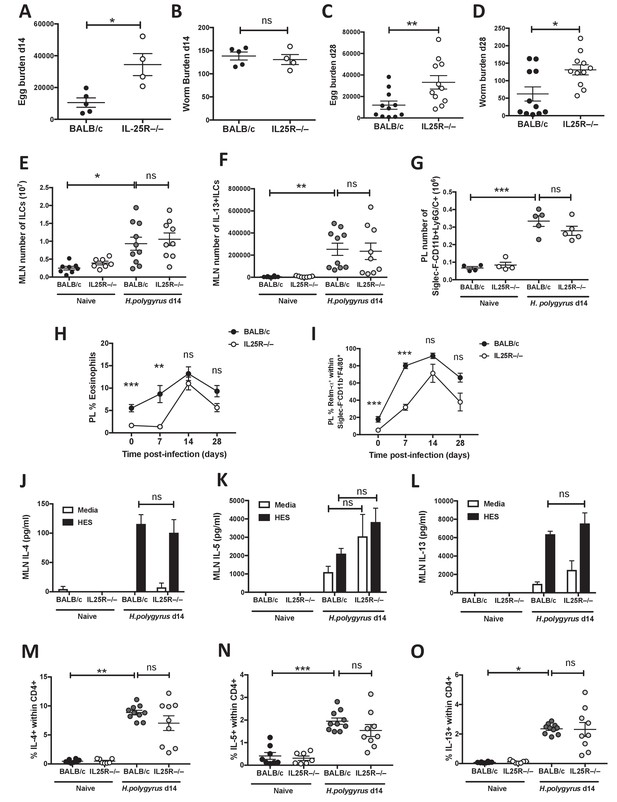
IL-25R signaling is required for expulsion of H.
polygyrus from BALB/c mice. H. polygyrus-infected BALB/c or Il17rb-deficient (IL25R–/–) BALB/c mice were analyzed at day 14 post-infection for fecal egg counts (A) and intestinal adult worm burden (B) or day 28 post-infection for fecal egg counts (C) and intestinal adult worm burden (D). MLN cells underwent intracellular cytokine staining (ICCS) to compare the number of Lin-ICOS+innate lymphoid cells (ILCs) (E) and IL-13+ ILC2s (F) by flow cytometry in the different naïve and day 14 infected genotypes. Peritoneal lavage cells (PL) were stained with Siglec-F, CD11b and Ly6G/C to compare the number of Siglec-F-CD11b+Ly6G/C+ monocytes (G) in the different naïve and day 14 infected genotypes. Percentages of eosinophils (H) and RELMα+ alternatively-activated macrophages (Ι) were also determined over a 4 week infection timecourse. ELISA of supernatants from MLN cells incubated with media or 1 μg HES for 72 hr was performed to compare IL-4 (J), IL-5 (K) and IL-13 (L) production in the different naïve and day 14 infected genotypes. ICCS of MLN allowed a comparison of the proportion of IL-4+, IL-5+ and IL-13+ CD4+ T cells by flow cytometry in the different naïve and day 14 infected genotypes (M–O). Results shown are one representative of three experiments with n ≥ 4 mice/group (A–D), pooled data from two experiments with n ≥ 4 mice/group (C–F, M–O) or one representative of two experiments with n ≥ 4 mice/group (G–L). Data were analysed by unpaired t test or one way ANOVA, where *=p≤0.05,**=p≤0.01,***=p≤0.001 and ns = not significant. Error bars represent Standard Error of the Mean.
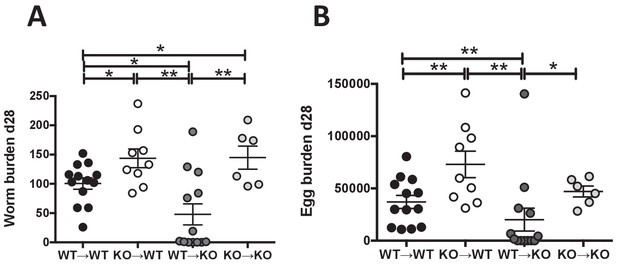
IL-25R signaling is required within the hematopoietic compartment for efficient expulsion.
Bone marrow chimeras generated from BALB/c (WT) or Il17rb-deficient (KO) donor and BALB/c (WT) or Il17rb-deficient (KO) recipient mice were infected with H. polygyrus and intestinal adult worm burden (A) and fecal egg burden (B) performed at day 28 post-infection. Results shown are pooled data from two experiments performed with n ≥ 3 mice/group, and data from all individual mice are presented. Data were analysed by unpaired t test, where *=p≤0.05,**=p≤0.01,***=p≤0.001 and ns = not significant. Error bars represent Standard Error of the Mean.
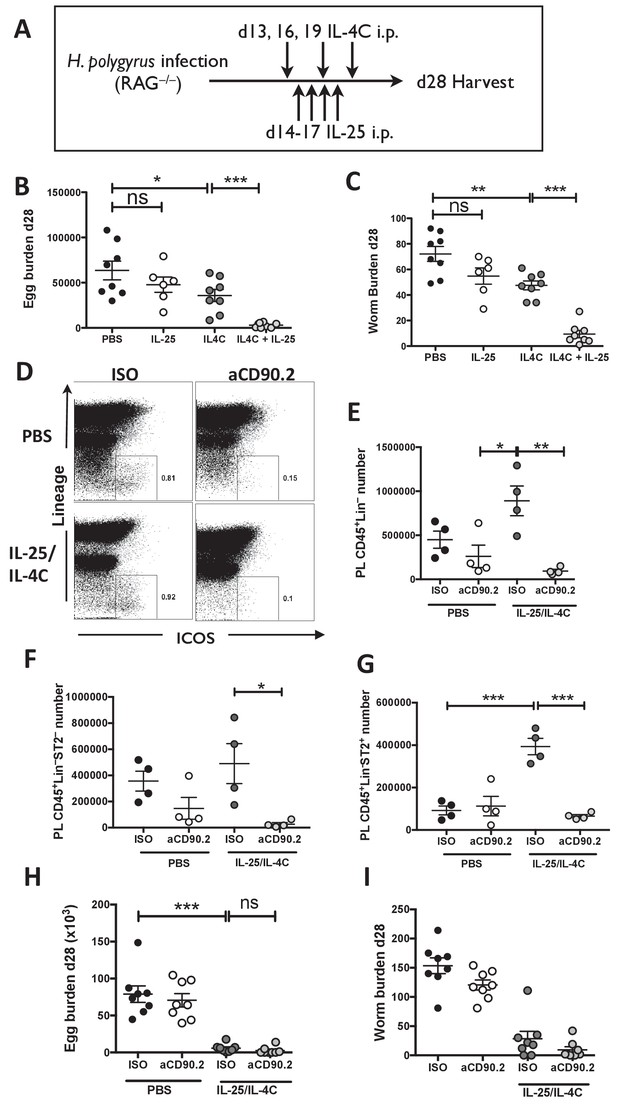
IL-25R signaling synergises with IL-4Rα within the innate immune compartment to facilitate efficient worm expulsion.
H. polygyrus-infected Rag1-/- mice (RAG-/-) were given 0.4 µg recombinant IL-25 i.p. days 14–17 (late) post-infection with or without a complex of 5 μg rIL-4 and 25 μg anti-IL-4 (IL-4C) on days 13, 16 and 19 post-infection, according the schedule shown in (A). Mice were analyzed at 28 days post-infection for fecal egg burden (B) and intestinal adult worm burden (C). H. polygyrus-infected Rag1-/- mice were given IL-25 and IL-4C according to the same schedule, as well as 200 µg of anti-CD90.2/Th1.2 antibody or rat IgG2b control (days 12, 15, 18 and 21). The peritoneal lavage was analyzed at 28 days post-infection for CD45+lin- (CD3, CD5, CD8α, CD11c, CD19, DX5, F4/80, GR-1, TCRβ, CD11b), ICOS and ST2 staining by flow cytometry as shown (D) and the number of CD45+lin– (E), CD45+lin–ST2– (F) and CD45+lin–ST2+ (G) ILCs was determined. Mice were analysed at 28 days post-infection for fecal egg burden (H) and intestinal adult worm burden (I). Results shown are one representative of two experiments with n = 4 mice/group (D–G), or pooled data from two experiments with n ≥ 3 mice/group (B,C,H,I). Data were analysed by unpaired t test, where *=p≤0.05,**=p≤0.01,***=p≤0.001 and ns = not significant. Error bars represent Standard Error of the Mean.
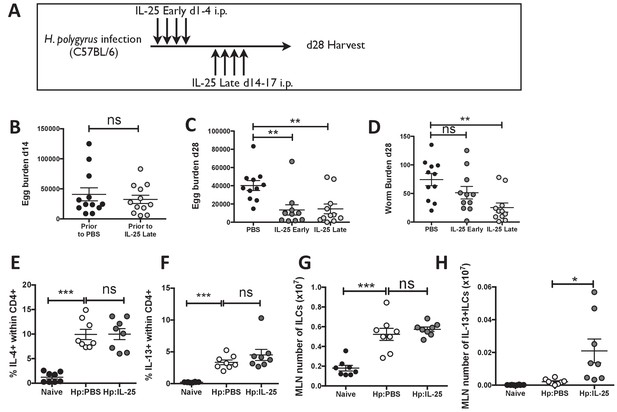
IL-25 induces protective immune responses in the late stage of infection.
H. polygyrus-infected C57BL/6 mice were given 0.4 μg recombinant IL-25 i.p. at day 1–4 (early) or day 14–17 (late) post-infection according to the schedule shown in (A). Before administration of IL-25, intestinal egg burden was analysed in two groups at day 14 post-infection (B). Mice were then analysed at day 28 post-infection for fecal egg counts (C) and intestinal adult worm burden (D) following administration of IL-25 early or late. H. polygyrus-infected C57BL/6 mice were given 0.4 μg recombinant IL-25 i.p. at day 14–17 (late) post-infection. Day 18 post-infection, MLN cells underwent ICCS to compare the proportion of IL-4+CD4+ (E) and IL-13+CD4+ (F) T cells, as well as the number of ILCs (G) and Lin-IL-13 + ILC2 s (H) by flow cytometry. Results shown are data pooled from three experiments with n ≥ 3 mice/group (B–D), or are representative of two experiments with n = 4 mice/group (E,F) or pooled from two experiments with n = 4 mice/group (G, H). Data were analysed by unpaired t test, where *=p≤0.05,**=p≤0.01,***=p≤0.001 and ns = not significant. Error bars represent Standard Error of the Mean.
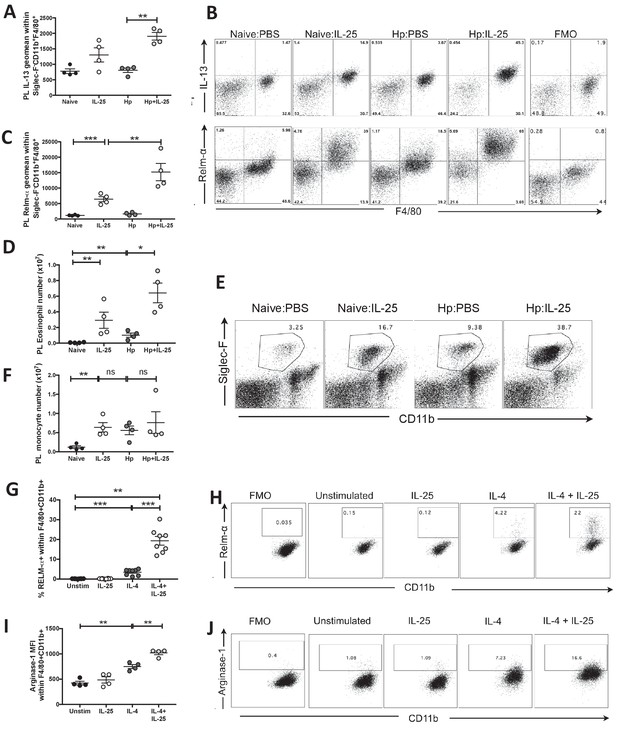
IL-25 drives alternative activation and IL-13 expression of macrophages.
H. polygyrus-infected C57BL/6 mice were given 0.4 μg recombinant IL-25 i.p. at day 14–17 (late) post-infection. At day 18 post-infection, PL cells were taken and stimulated with 10 μg Brefeldin A to determine the expression intensity (geomean) of IL-13 (A) or were unstimulated to determine the expression intensity (geomean) of RELM-α (B, C) within Siglec-F–CD11b+F4/80+ monocytes by flow cytometry, as shown by the example flow cytometry plots in B and compared to fluorescence minus one (FMO) samples stained from the Hp:IL-25 group. Total Siglec-F+CD11b+ eosinophil (D) numbers were calculated from populations shown in the example flow cytometry plots (E), numbers of Siglec-F–CD11b+ (F) monocytes. Bone marrow-derived macrophages from C57BL/6 mice were generated in vitro and stimulated with 10 ng/ml IL-4, 200 ng/ml IL-25 or a combination of both for 16 hr before analysis of the percentage of RELM-α expression (G, H) and the mean fluorescence intensity (geometric mean) of Arginase-1 expression (I, J) within CD11b+F4/80+ cells by flow cytometry. Results shown are one representative of 2 experiments with n = 4 mice/group (A–F, I, J) or pooled from two experiments with n = 4 replicates/group (G, H). Data were analysed by unpaired t test, where *=p≤0.05,**=p≤0.01,***=p≤0.001 and ns = not significant. Error bars represent Standard Error of the Mean.
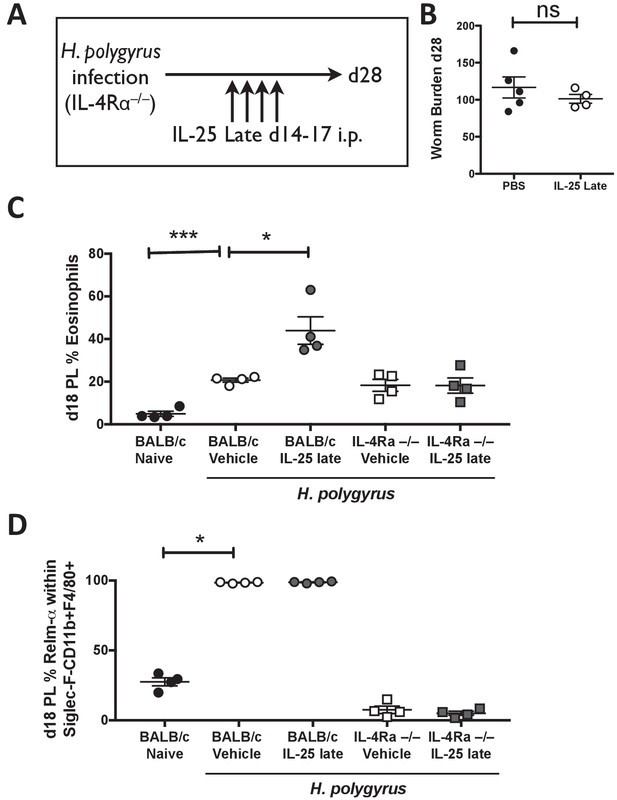
IL-4Rα expression is required for activation of the immune system by IL-25.
H. polygyrus-infected Il4ra-/- mice (IL-4R-/-) were given 0.4 μg recombinant IL-25 i.p. days 14–17 (late) post-infection (A). Mice were analyzed at day 28 post-infection for intestinal adult worm burden (B). H. polygyrus-infected BALB/c and Il4ra-/- mice (IL-4R-/-) were given 0.4 μg recombinant IL-25 i.p. days 14–17 (late) post-infection. Day 18 post-infection, PL cells were taken to determine the percentage of eosinophils (C) and percentage of RELM-α expression within Siglec-F-CD11b+F4/80+ macrophages by flow cytometry (D). Results shown are one representative of 2 experiments with n ≥ 4 mice/group (A–D). Data were analysed by unpaired t test, where *=p≤0.05,**=p≤0.01,***=p≤0.001 and ns = not significant. Error bars represent Standard Error of the Mean.
Tables
| Reagent type (species) or resource | Designation | Source or reference | Identifiers |
|---|---|---|---|
| Mus musculus, BALB/c | Il17rb-/- (IL-25R-/-) | Neill et al. (2010) Nuocytes represent a new innate effector leukocyte that mediates type-2 immunity. Nature 464:1367–1370 | |
| Mus musculus, C57BL/6 | Rag1-/- | Mombaerts,P., Iacomini,J., Johnson, R.S., Herrup, K., Tonegawa,S., and Papaioannou, V. E. 1992. Cell. Vol 68: 869–877. PMID: 1547488 | |
| Mus musculus, C57BL/6 | Il4ra-/- | Noben-Trauth N, Shultz LD, Brombacher F, Urban JF, Jr., Gu H, Paul WE. 1997. An interleukin 4 (IL-4)-independent pathway for CD4 + T cell IL-4 production is revealed in IL-4 receptor-deficient mice. Proc Natl Acad Sci USA 94:10838–10843. | |
| Parasite | Heligmosomoides polygyrus bakeri | Johnston et al., 2015. Cultivation of Heligmosomoides polygyrus: an immunomodulatory nematode parasite and its secreted products. Journal of Visualized Experiments 98:e52412. | |
| Antibody | Anti-IL-4 | BioXCell | Clone 11B11 |
| Antibody | Anti-IL-13 | BioLegend | JES10-5A2 |
| Antibody | Anti-CD90.2 | BioXCell | 30H12 |
| Antibody | Anti-SiglecF | BD Pharmingen | E50-2440 |
| Antibody | Anti-CD11b | BioLegend | M1/70 |
| Antibody | Anti-F4/80 | BioLegend | BM8 |
| Antibody | Anti-Ly6G | BioLegend | 1A8 |
| Antibody | Anti-Ly6C | BioLegend | AL-21 |
| Antibody | Anti-RELMα | R and D Systems | 22603 |
| Antibody | Anti-Arginase-1 (Polyclonal) | R and D Systems | IC5868P |
| Antibody | Anti-Lineage | BioLegend | 17A2,RB6-8C5,RA3-6B2,Ter-119,M1/70 |
| Peptide, recombinant protein | IL-4 | Peprotech | 214–14 |
| Peptide, recombinant protein | IL-25 | BioLegend | 587302 |
| Commercial assay or kit | Foxp3 Staining Kit | eBioScience | 88–8118 |
| Other | Control IgG2b immunoglobulin | BioXcell | LTF-2 |
| Other | Anti-CD90.2 Microbeads | Miltenyi | 130-049-101 |
Additional files
-
Transparent reporting form
- https://doi.org/10.7554/eLife.38269.008





