Negative regulation of conserved RSL class I bHLH transcription factors evolved independently among land plants
Figures
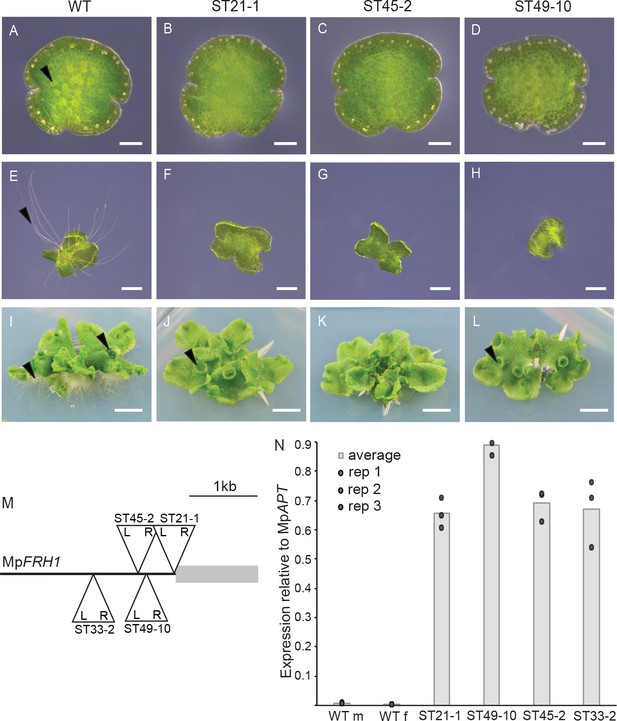
T-DNA insertion within the MpFRH1 promoter results in elevated steady state levels of MpFRH1 mRNA and defective rhizoid precursor cell differentiation.
(A–L) Phenotype of wild type M. polymorpha and the few rhizoid mutants ST21-1, ST45-2 and ST49-10. (A–D) One day old gemma (scale bar 100 μm), (E–H) four day old gemma (scale bar 1 mm), (I–L) 28 day old gemma (scale bar 5 mm) of wild type (A, E, I) and the few rhizoid mutants ST21-1 (N, F, J), ST49-10 (C, G, K) and ST45-2 (D, H, J). The arrowheads indicate rhizoid precursor cells (in A-D) rhizoids (in E-L) and gemma cups (in I-L). (M) Location and orientation of the T-DNA insertion sites within the MpFRH1 locus. L and R stand for T-DNA left and right border, respectively. (N) qRT-PCR analysis of steady state MpFRH1 mRNA levels in 15 day old gemmae of wild type and the few rhizoid mutants ST21-1, ST45-2 and ST49-10. The MpFRH1 transcript level was normalised against MpAPT1.
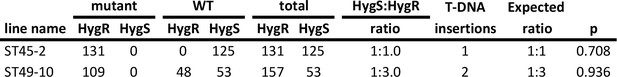
The few rhizoids phenotype of mutants ST45-2 and ST49-10 co-segregates with the hygromycin resistance marker on the T-DNA.
Frequencies of hygromycin resistant and hygromycin sensitive plants showing mutant and wild type phenotypes in F1 generation from a cross between ST45-2 and ST49-10 with wild type M. polymorpha. Probability (p) of obtaining the observed numbers of hygromycin sensitive and hygromycin resistant F1 plants with the estimated T-DNA copy number was determined using the Chi-square test.
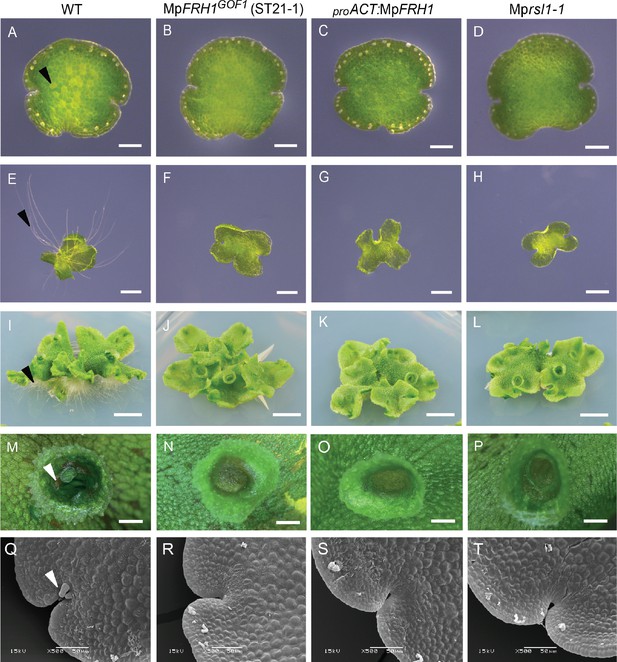
The phenotype of the MpFRH1 gain-of-function mutants and plants transformed with proACT:MpFRH1 is similar to Mprsl1 loss-of-function mutant phenotype.
(A–T) Phenotype of wild type M. polymorpha, T-DNA insertion line MpFRH1GOF1/ST21-1, plant transformed with proACT:MpFRH1 and Mprsl1-1 loss-of-function mutant. One day old gemma (A-D, scale bar 100 μm), four day old gemma (E-H, scale bar 1 mm), 28 day old gemma (I-L, scale bar 5 mm), gemma cup of mature plant (M-P, scale bar 600 μm) and meristematic region of one day old gemma (Q-T, scale bar 50 μm) of wild type (A, E, I, M, Q), MpFRH1GOF1/ST21-1, (B, F, J, N, R), proACT:MpFRH1 (C, G, K, O, S) and Mprsl1-1 (D, H, L, P, T). The arrowheads indicate rhizoid precursor cells (in A-D), rhizoids (in E-L), gemmae (in M-P) and mucilage papillae (in Q-T).
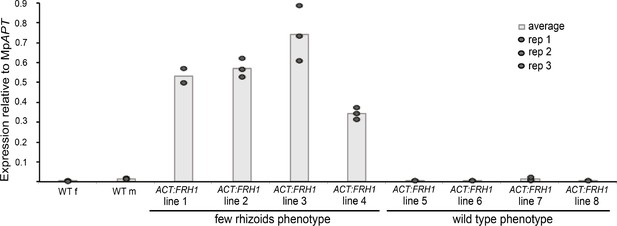
Over-expression of the full-length MpFRH1 transcript behind the strong constitutive rice ACTIN promoter in the wild type background is sufficient to reproduce the few rhizoids phenotype.
qRT-PCR analysis of steady state MpFRH1 transcript levels in 15 day old gemmae of wild type and eight lines transformed with proACT:MpFRH1. MpFRH1 transcript levels were normalised against MpAPT1..
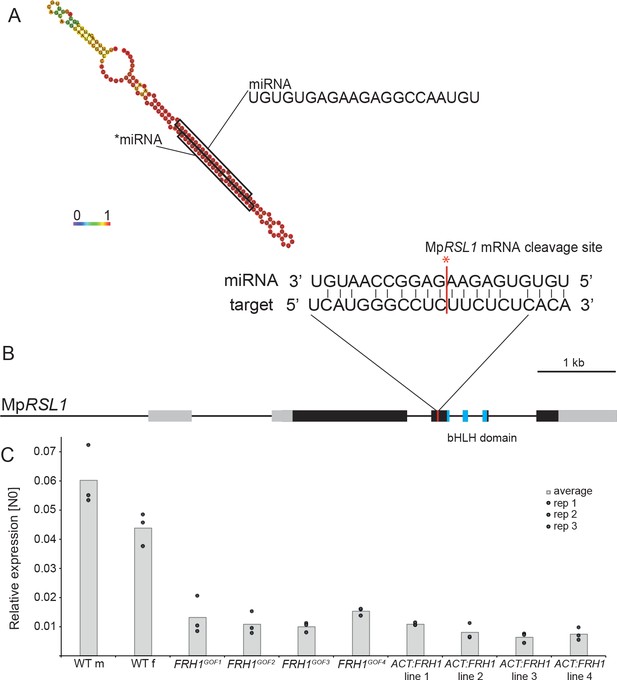
The MpFRH1 locus produces a miRNA that targets the MpRSL1 transcript.
(A) RNA folding prediction of the 150 bp sequence sufficient to reproduce the few rhizoids phenotype when over-expressed in the wild type carried out using RNAfold (Gruber et al., 2008). The colours represent base-pairing probabilities. The small RNA sequences corresponding to MpFRH1 miRNA (mpo-MIR11861) and the complementary *miRNA are indicated. (B) MpRSL1 gene model, the MpFRH1 miRNA target site is indicated in red. (C) MpFRH1 negatively regulates MpRSL1 transcript level. qRT-PCR quantification of steady state MpRSL1 transcript levels in 15 day old gemmae of wild type, the four MpFRH1GOF mutant lines and four proACT:FRH1 lines with a strong few rhizoid phenotype. MpRSL1 transcript levels were normalised against MpAPT1 and MpCUL3..
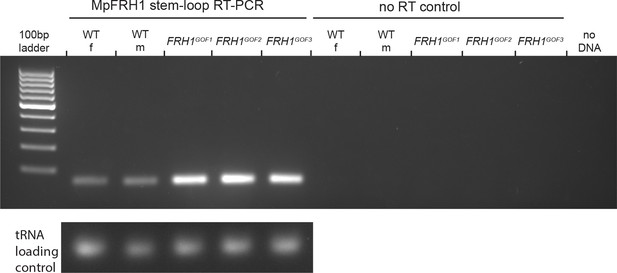
Stem-loop PCR detection of the MpFRH1 miRNA.
RNA extracted from 15 day old wild type and MpFRH1GOF1-3 gemmae was used as a template for the stem-loop PCR amplification. Amplification products after 28 PCR cycles were visualised on a EtBr stained 2% agarose gel. The tRNA band was used as a loading control.

Predicted targets of MpFRH1 miRNA.
Lower scores indicate a more likely target based on sequence similarity between the miRNA and the target transcript. Transcript number corresponds to the M. polymorpha gametophyte transcriptome (Honkanen et al., 2016). Mapoly gene ID correspond to the M. polymorpha genome sequence published in Bowman et al. (2017).
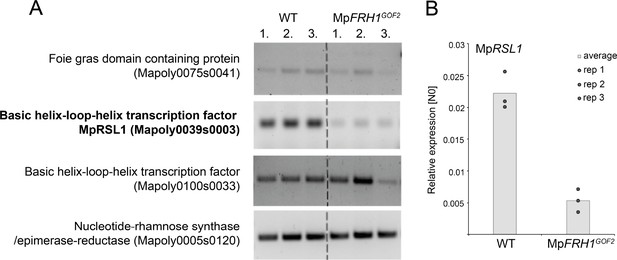
Transcripts of one of the four predicted MpFRH1 miRNA targets are less abundant in MpFRH1GOF2 compared to wild type.
(A) RT-PCR quantification of transcript levels of the four predicted FRH1 miRNA target mRNAs in WT and MpFRH1GOF2 plants. (B) qRT-PCR quantification of transcript levels of MpRSL1. MpRSL1 transcript levels were normalised against MpAPT1 and MpCUL3.
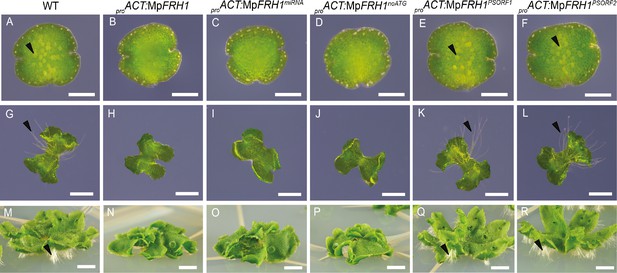
MpFRH1 functions as miRNA.
One day old gemma (A-F, scale bar 200 μm), four day old gemma (F-L, scale bar 1 mm) and 28 day old gemma (M-R, scale bar 5 mm) of wild type (A, G, M), proACT:MpFRH1 (B, H, N), proACT:MpFRH1miRNA (C, I, O), proACT:MpFRH1noATG (D, J, P), proACT:MpFRH1PSORF1 (E, K, Q) and proACT:MpFRH1PSORF2 (F, L, R). The arrowheads indicate rhizoid precursor cells (in A-F) and rhizoids (in G-R).
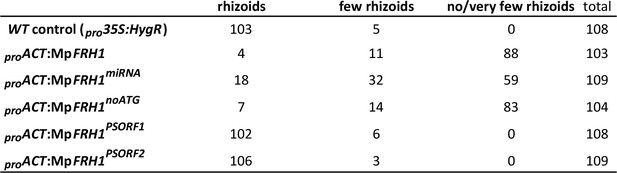
Frequencies of different phenotypes observed in sporelings transformed with partial or modified versions of the MpFRH1 transcript sequence driven by the strong constitutive rice actin promoter.
Phenotypes of the transformant sporelings were scored four weeks after the initial transformation.
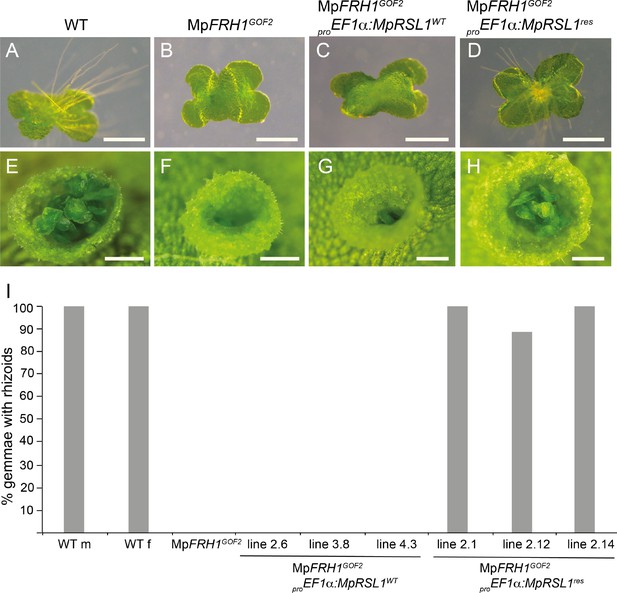
MpFRH1 miRNA-resistant form of MpRSL1 restores rhizoid and gemma development in the MpFRH1GOF2 mutant background.
Three day old gemmae (A–D) of WT (A), MpRSL1GOF2 (B), MpFRH1GOF2; proEF1a:MpRSL1WT (C) and MpFRH1GOF2; proEF1a:MpRSL1res (D), scale bar 500 μm. Gemma cup of mature WT (E), MpRSL1GOF2 (F), MpFRH1GOF2; proEF1a:MpRSL1WT (G) and and MpFRH1GOF2; proEF1a:MpRSL1res (H), scale bar 500 μm. (I) Percentage of three day old gemmae forming rhizoids, n = 18 for each line.
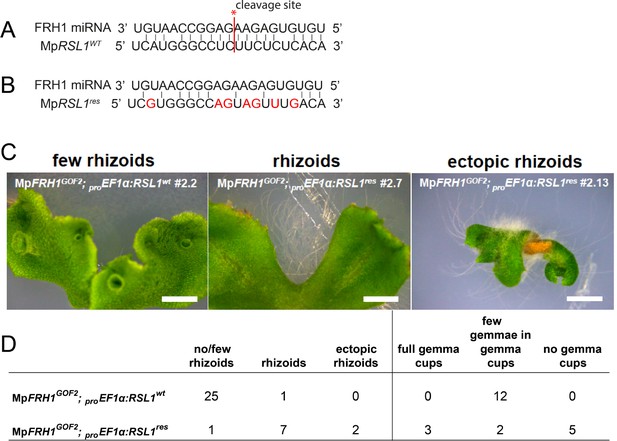
MpFRH1 miRNA-resistant version of MpRSL1 (MpRSL1res) suppresses the MpFRH1GOF2 few rhizoids and few gemmae phenotype.
(A) Sequence of the MpFRH1 miRNA and its target site on the wild type MpRSL1 mRNA. The cleavage site is indicated in red. (B) Point mutations introduced into the MpFRH1 miRNA target site on MpRSL1 mRNA to create MpFRH1 miRNA resistant version of MpRSL1 (MpRSL1res). Mutated bases are indicated in red. (C) Examples phenotypes observed in MpFRH1GOF2 plants transformed with proEF1a:MpRSL1WT or proEF1a:MpRSL1res. Mature transformant plants, Scale bar 2 mm. (D) Frequencies of each phenotype observed in MpFRH1GOF2 plants transformed with either proEF1a:MpRSL1WT or proEF1a:MpRSL1res.
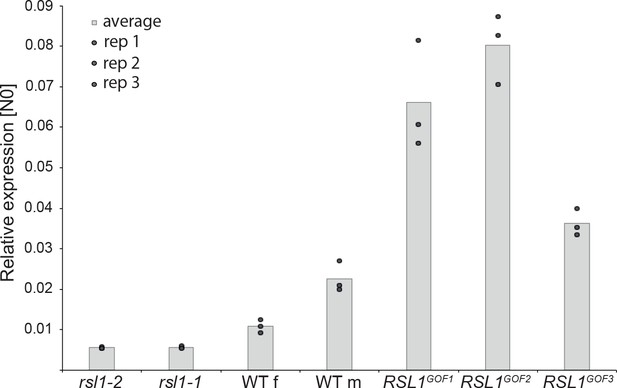
MpRSL1 positively regulates MpFRH1 transcript level.
qRT-PCR quantification of steady state MpFRH1 transcript levels in 15 day old gemmae of wild type M. polymorpha, Mprsl1 loss-of-function mutants Mprsl1-1 and Mprsl1-2, and MpRSL1 gain-of-function mutants MpRSL1GOF1, MpRSL1GOF2 and MpRSL1GOF3. MpFRH1 transcript levels were normalised against MpAPT1 and MpCUL3.
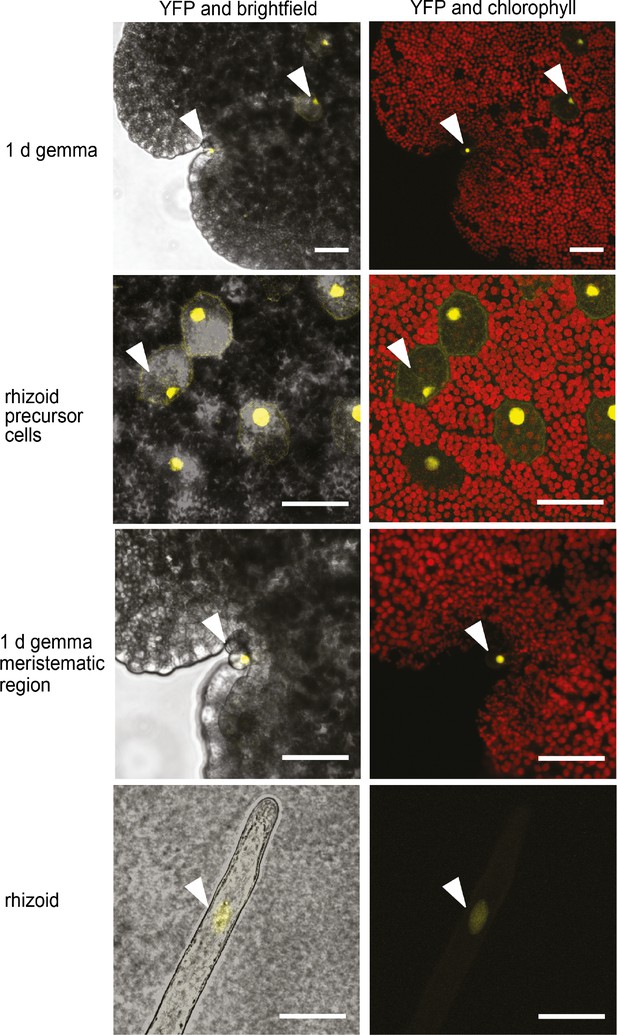
MpFRH1 is expressed in rhizoid precursor cells, epidermal papillae and rhizoids.
Pattern of promoterMpFRH1:3xYFP-NLS expression in 1 day old gemmae. The arrowheads indicate rhizoid precursor cells, epidermal papillae and rhizoids. Scale bar 50 μm.
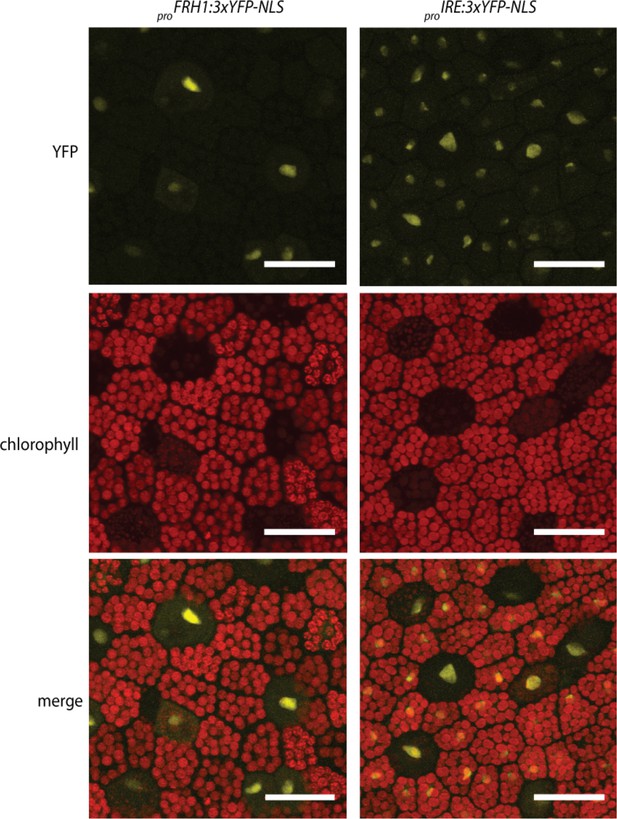
MpFRH1 is expressed in rhizoid precursor cells.
Comparison of MpFRH1 with MpIRE promoter expression pattern in the mid region of 1 day old gemmae. Scale bar 50 µm. Rhizoid precursor cells can be distinguished from non-rhizoid precursor cells based on their strongly reduced red chlorophyll autofluorescence.

The MpFRH1 miRNA target site is conserved in liverwort RSL class I transcripts.
Liverwort RSL class I transcript alignment. The MpFRH1 miRNA target site is circled in black. Longer region of the alignment is provided in Figure 8—figure supplement 1.

The MpFRH1 miRNA target site is conserved in liverwort RSL class I transcripts.
Liverwort RSL class I transcript alignment. The MpFRH1 miRNA target site is circled in red. Black columns indicate 100% conservation. Supplemental data file legends.
Additional files
-
Supplementary file 1
TAIL-PCR band sequences
- https://doi.org/10.7554/eLife.38529.020
-
Supplementary file 2
MpFRH1 transcript sequence
- https://doi.org/10.7554/eLife.38529.021
-
Supplementary file 3
5'RLM-RACE PCR band sequences
- https://doi.org/10.7554/eLife.38529.022
-
Supplementary file 4
Multiple sequence alignments of the predicted MpFRH1 miRNA target mRNAs in M.
polymorpha and their orthologs from other liverworts. The predicted MpFRH1 target site is indicated with a grey arrow. Region around the predicted miRNA target site (top) and overview of the alignment (bottom). (A) Foie gras domain containing protein Mapoly0075s0041.1. (B) Basic helix-loop-helix transcription factor MpRSL1 Mapoly0039s003 (C) Basic helix-loop-helix transcription factor transcript 27676. (D) Nucleotide-rhamnose synthase/epimerase-reductase Mapoly0005s0120.
- https://doi.org/10.7554/eLife.38529.023
-
Supplementary file 5
Promoter analysis
- https://doi.org/10.7554/eLife.38529.024
-
Transparent reporting form
- https://doi.org/10.7554/eLife.38529.025





