Calcium-dependent electrostatic control of anion access to the pore of the calcium-activated chloride channel TMEM16A
Figures
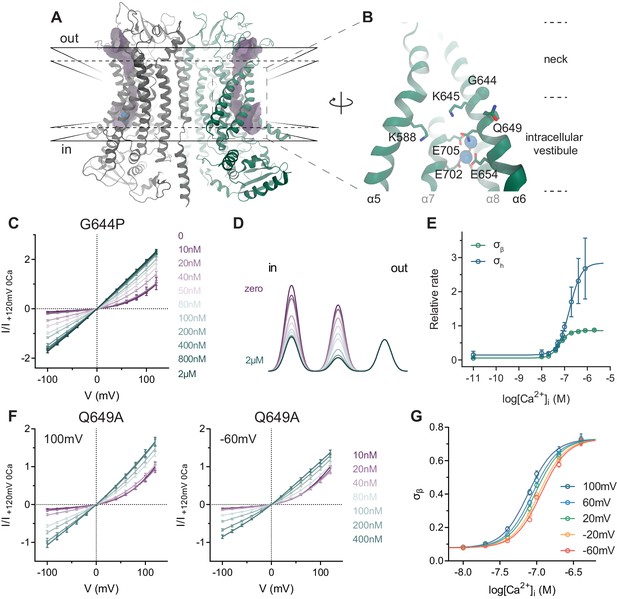
TMEM16A structure and conduction properties of constitutively active mutants.
(A) Ribbon representation of the Ca2+-bound mouse TMEM16A channel viewed from within the membrane (PDB: 5OYB), subunits are shown in unique colors. Black solid lines, outer- and innermost membrane boundaries, black dashed lines, boundaries of the hydrophobic core; blue spheres, Ca2+ ions; violet surface, molecular surface of the pore (generated in HOLE [Smart et al., 1996] with a solvent probe radius of 0.7 Å). (B) Close-up of the intracellular vestibule with selected residues displayed as sticks and labelled. Green sphere, Cα of Gly 644. α3 and α4 are removed for clarity. (C) Instantaneous I-V relations of the constitutively active mutant G644P from pre-pulses at 80 mV in the presence of the indicated intracellular Ca2+ concentrations. Solid lines are fits to Equation 2. Data were normalized to the fitted amplitude factor (A in Equation 2) and were subsequently normalized to the current amplitude at 120 mV at zero Ca2+ (I/I +120mV 0Ca). Data are mean values of normalized I-V plots from 5 to 13 individual patches, errors are s.e.m. (D) Relative energy profiles (at 0 mV) of the ion conduction path at the indicated intracellular Ca2+ concentrations (colors as in C). Barriers are visualized as a sum of three Gaussians with the peaks and amplitudes indicating their locations and relative barrier heights respectively. (E) Relative rate of barrier crossing at the indicated location (σh and σβ) as a function of intracellular Ca2+ concentration. Solid lines are fits to the Hill equation. Data are best-fit values and errors are 95% confidence intervals. (F) Instantaneous I-V relations of the mutant Q649A from pre-pulses at 100 mV (left) and −60 mV (right) in the presence of the indicated intracellular Ca2+ concentrations. Solid lines are fits to Equation 2. Data were normalized as in C and are mean values of normalized I-V plots from five individual patches, errors are s.e.m. (G) Relative rate of barrier crossing at the intracellular pore entrance (σβ) as a function of both intracellular Ca2+ concentration and voltage. Solid lines are fits to the Hill equation.
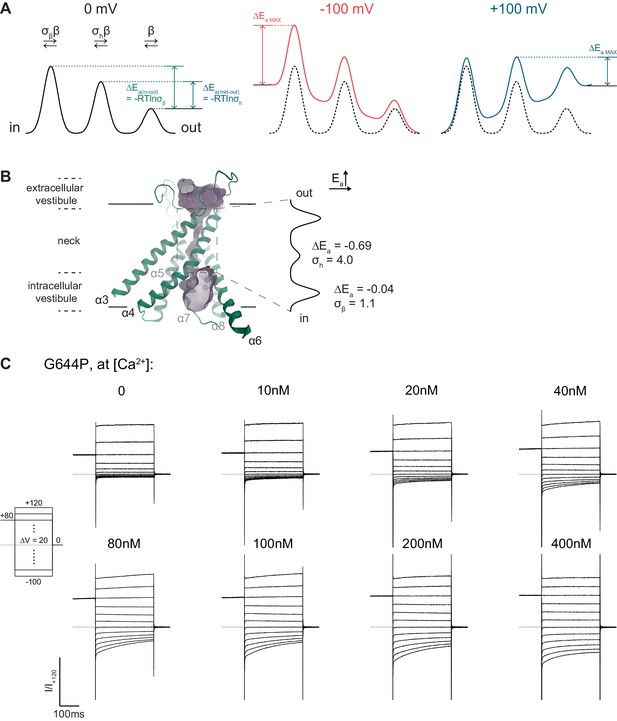
Permeation model and G644P currents.
(A) Energy profile indicating the relationship between the relative rate constants (σh and σβ) and the energy difference between the middle or inner barrier and the outer barrier at 0 mV (ΔEa (mid-out) and ΔEa (in-out), left). The effect of a constant electric field across the channel is shown (center, right). (B) Relationship between the pore and the energy profile of WT. The molecular surface of the pore is shown in violet. Blue spheres, calcium ions. Selected helices are shown and are labelled. View is from within the membrane. (C) Rundown-corrected and normalized current responses of G644P used to construct the I-V relations shown in Figure 1C.
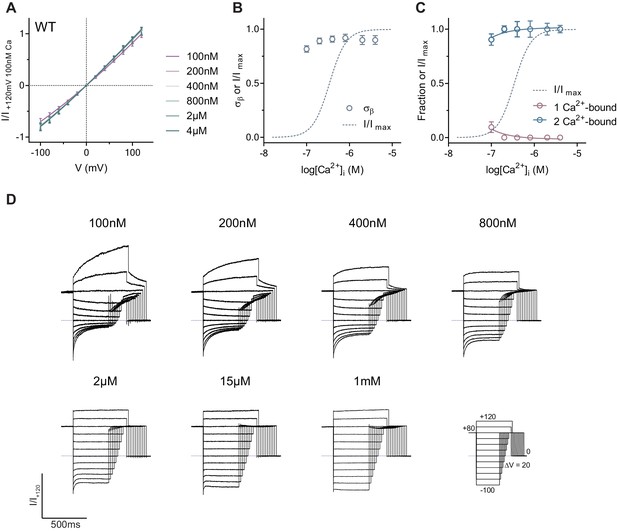
Relative contribution of open states with various Ca2+occupancy to the activation of WT.
(A) Instantaneous I-V relations from pre-pulses at 80 mV in the presence of the indicated intracellular Ca2+ concentrations for WT. Solid lines are fits to Equation 2. Data were processed as in Figure 1C and are mean values of normalized I-V plots from 7 to 8 individual patches, errors are s.e.m. (B) Relative rate of barrier crossing at the intracellular pore entrance (σβ) of WT as a function of intracellular Ca2+ concentration. Dashed lines are the concentration-response relation of WT at 80 mV. Data are best-fit values and errors are 95% confidence intervals. (C) Relative contribution of open states with various Ca2+ occupancy during activation. Data are best-fit values obtained from fitting the data in A to Equation 7, errors are 95% confidence intervals. Solid lines describe the trend and have no theoretical meaning. (D) Rundown-corrected and normalized current responses of WT used to construct the I-V relations shown in A. .
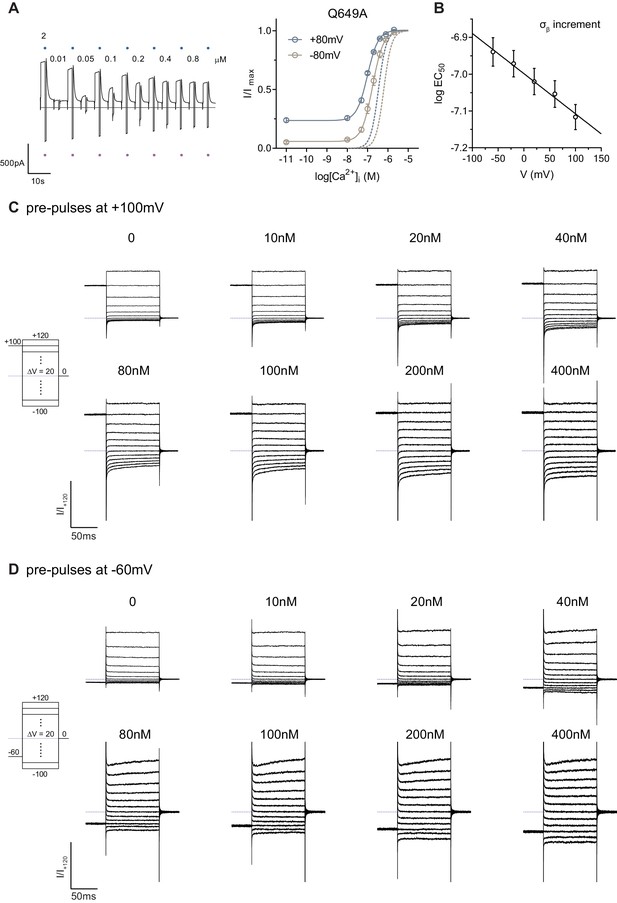
Characterization of the mutant Q649A.
(A) Representative current trace (left) and concentration-response relations (right) of Q649A recorded at +/-80 mV using a rundown-correction protocol. Solid lines are fits to the Hill equation. Dashed lines are the relations of WT. Data are mean values of normalized concentration-response relations from 6 individual patches, errors are s.e.m. Blue and violet dots on the current trace indicate the reference pulses used for rundown correction. (B) EC50 for σβ increment (Figure 1G) as a function of voltage. Solid line is a fit to Equation 1 . Data are best-fit values and errors are 95 % confidence intervals. The electrical distance () was estimated to be 0.032 ± 0.007. C-D. Rundown-corrected and normalized current responses of Q649A with pre-pulses at, (C), 100 mV and, (D), -60 mV.
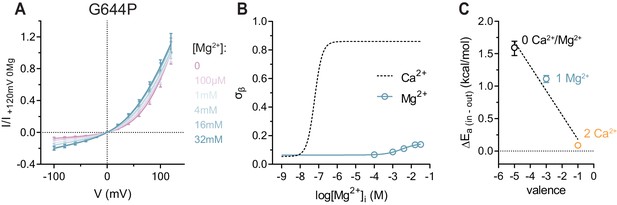
Conduction properties of the constitutively active mutant G644P in the presence of intracellular Mg2+.
(A) Instantaneous I-V relations of the constitutively active mutant G644P from pre-pulses at 80 mV in the presence of the indicated intracellular Mg2+ concentrations. Solid lines are fits to Equation 2. Data were normalized to the fitted amplitude factor (A in Equation 2) and were subsequently normalized to the current amplitude at 120 mV at zero Mg2+ (I/I +120mV 0Mg). Data are mean values of normalized I-V plots from 9-11 individual patches, errors are s.e.m. (B) Relative rate of barrier crossing at the intracellular pore entrance (σβ) as a function of intracellular Mg2+ concentration. Solid line is a fit to the Hill equation for one binding site. The relation with Ca2+ is shown as dashed line for comparison. Data are best-fit values and errors are 95% confidence intervals. (C) Experimental relationship between the assumed net charge of the Ca2+ binding site (valence) and anion conduction energetics (ΔEa (in-out)). Data are transforms of σβ, using Equation 3, at zero and saturating Mg2+ and Ca2+ concentrations. Dashed Line is a fit to Equation 4. The relative permittivity () for occupancy by divalent cations was estimated to be 64.8 ± 35.1.
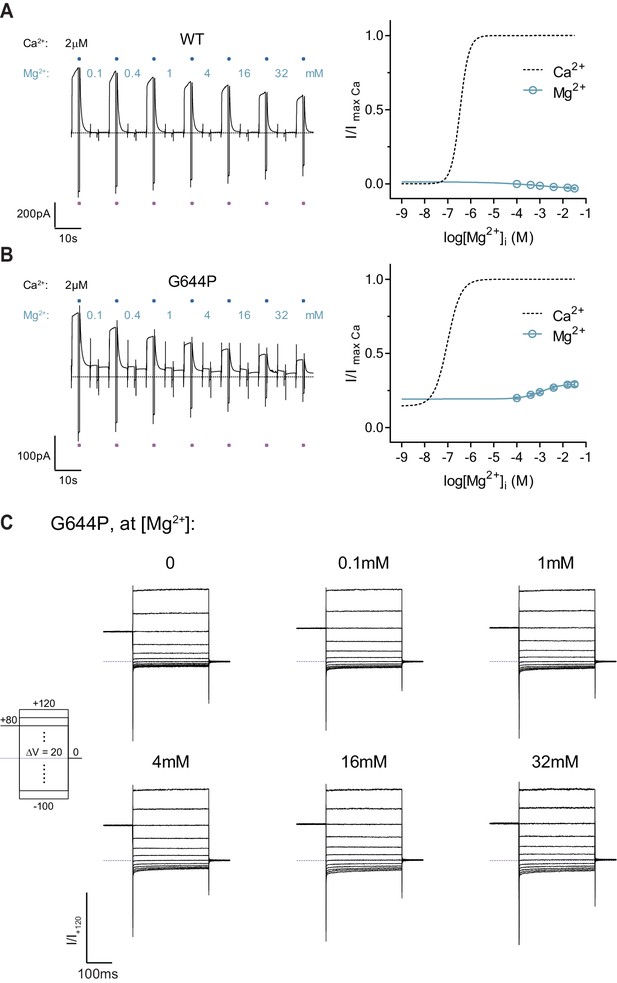
Mg2+concentration-response relations of WT and G644P.
A-B, Representative current traces (left) and concentration-response relations (right) of WT (A) and G644P (B) recorded at ±80 mV using a rundown-correction protocol. Solid lines are fits to the Hill equation for one binding site. Dashed lines are the relations with Ca2+ shown for comparison. Data are mean values of normalized concentration-response relations from 5 and 7 individual patches respectively, errors are s.e.m. Blue and violet dots on the current traces indicate the reference pulses used for rundown correction. (C) Rundown-corrected and normalized current responses of G644P at the indicated Mg2+ concentrations used to construct the I-V relations shown in Figure 2A.
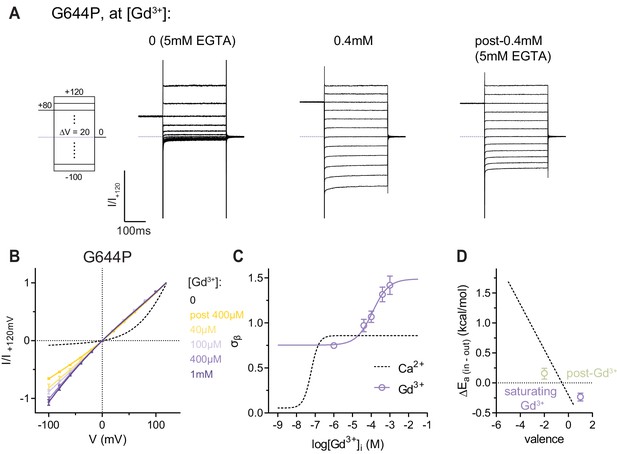
Conduction properties of G644P in the presence of intracellular Gd3+.
(A) Rundown-corrected and normalized current responses of G644P at the indicated Gd3+ concentrations. A slowly dissociating occupancy persists upon a jump to 5 mM EGTA post-0.4 mM Gd3+. (B) Instantaneous I-V relations of G644P from pre-pulses at 80 mV in the presence of the indicated intracellular Gd3+ concentrations. Solid lines are fits to Equation 2. Data were normalized to the respective current amplitudes at 120 mV (I/I+120). Data are mean values of normalized I-V plots from 3 to 9 individual patches, errors are s.e.m. (C) Relative rate of barrier crossing at the intracellular pore entrance (σβ) as a function of intracellular Gd3+ concentration. Solid line is a fit to the Hill equation for one binding site. The relation with Ca2+ is shown as dashed line for comparison. Data are best-fit values and errors are 95% confidence intervals. (D) Anion conduction energetics (ΔEa (in-out)) at saturating Gd3+ concentrations and after a jump to 5 mM EGTA post-0.4mM Gd3+. Data are transforms of σβ, using Equation 3, at the two plateaus determined in C. Dashed line indicates the relation between divalent occupancy and anion conduction energetics determined with Mg2+ and Ca2+ from Figure 2C. The extra valence due to Gd3+ binding were assigned as 3 and 6, which might not necessarily be the case as a mixed occupancy by Gd3+ and other trace di/trivalent cations cannot be ruled out under the recording conditions.
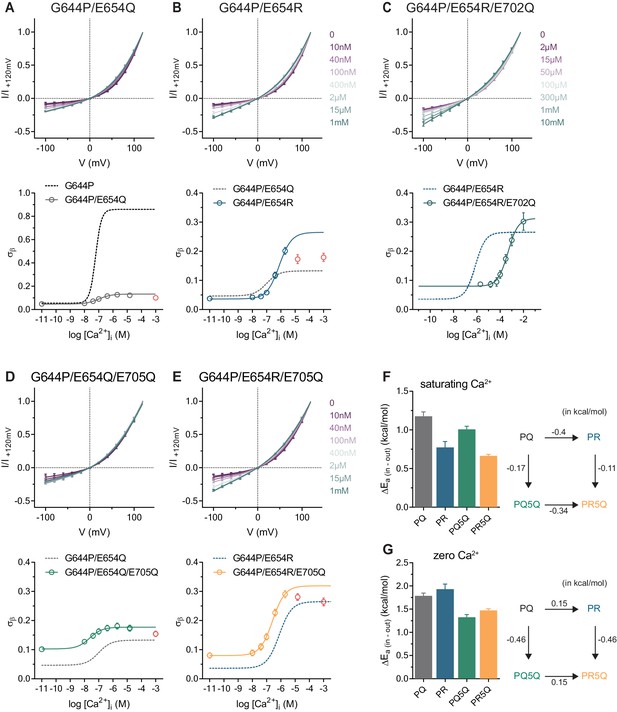
Conduction properties of the constitutively active mutant G644P with additional mutations at the Ca2+binding site.
A-E, Top, instantaneous I-V relations from pre-pulses at 80 mV in the presence of the indicated intracellular Ca2+ concentrations for mutants G644P/E654Q (A), G644P/E654R (B), G644P/E654R/E702Q (C), G644P/E654Q/E705Q (D) and G644P/E654R/E705Q (E). Solid lines are fits to Equation 2. Data were normalized to the current amplitude at +120 mV of each curve and are mean values of normalized I-V plots from 6, 7, 5–10, 5 and 7 individual patches respectively, errors are s.e.m. Bottom, relative rate of barrier crossing at the intracellular pore entrance (σβ) as a function of intracellular Ca2+ concentration for mutants G644P/E654Q (A), G644P/E654R (B) G644P/E654R/E702Q (C), G644P/E654Q/E705Q (D) and G644P/E654R/E705Q (E). Solid lines are fits to the Hill equation for one binding site. Data in red were omitted from the fit (see Materials and methods). Dashed lines are the relations of the indicated mutant shown for comparison. Data are best-fit values and errors are 95% confidence intervals. F-G, Left, relative activation energies at the intracellular pore entrance (ΔEa (in-out)) for the indicated mutants at saturating Ca2+ concentrations (F) and zero Ca2+ (G). Data are transforms of σβ, using Equation 3, at saturating and zero Ca2+ concentrations obtained from the fits shown in A-E. Right, energetic changes from the parent mutant G644P/E654Q (PQ) in a double-mutant cycle for the indicated mutants at saturating Ca2+ concentrations (F) and zero Ca2+ (G).
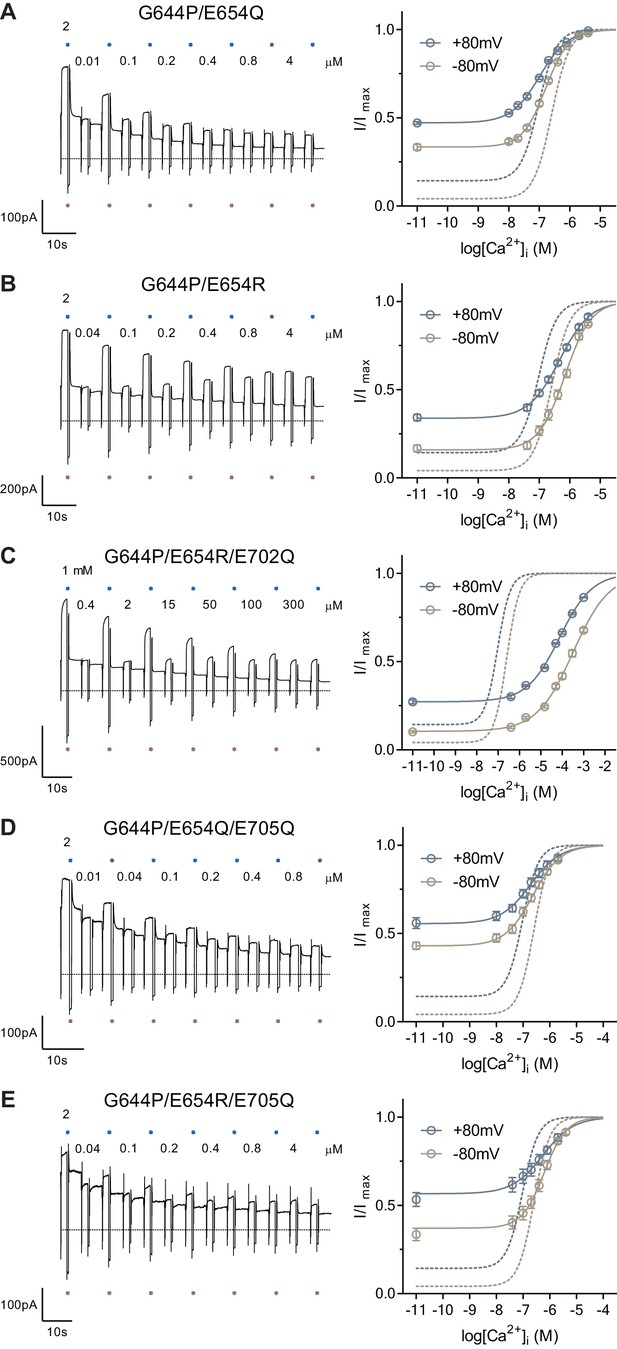
Concertation-response relations of mutants.
Representative current traces (left) and concentration-response relations (right) of mutants G644P/E654Q (A), G644P/E654R (B) G644P/E654R/E702Q (C), G644P/E654Q/E705Q (D) and G644P/E654R/E705Q (E) recorded at ±80 mV using a rundown-correction protocol. Solid lines are fits to the Hill equation. Dashed lines are the relations of G644P shown for comparison. Data are mean values of normalized concentration-response relations from 4 to 12, 8 – 11, 5 – 7, 6 – 7 and 10 individual patches respectively, errors are s.e.m. Blue and violet dots on the current traces indicate the reference pulses used for rundown correction.
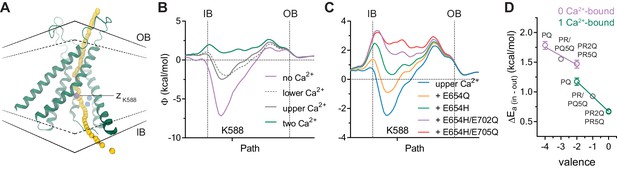
Electrostatic profiles.
(A) System in which the electrostatic profile of the pore was calculated. Selected helices are shown and view is from within the membrane on one of the two pores in the dimeric protein. Black solid lines, outer- and innermost membrane boundaries (OB and IB, respectively); black dashed lines, boundaries of the hydrophobic core; blue spheres, Ca2+ ions; yellow spheres, points at which the electrostatic potential (Φ) was plotted. Magenta sphere corresponds to the z position of the nitrogen atom of Lys 588. B-C. Electrostatic potential along the pore in the Ca2+-bound structure containing the indicated number of Ca2+ ions (B) and carrying the indicated in silico mutations with only the upper Ca2+ ion bound (C). Vertical dashed lines indicate the membrane boundaries. The position of Lys 588 at the intracellular pore entrance is indicated. (D) Experimental relationship between the assumed net charge of the Ca2+ binding site (valence) and anion conduction energetics (ΔEa (in-out)). Data are transforms of σβ, using Equation 3, at saturating and zero Ca2+ concentrations for all the mutants on the G644P background (Figure 2). Lines are fits to Equation 4. The relative permittivity () for the 0 Ca2+-bound and 1 Ca2+-bound states were estimated to be 162.7 ± 127.2 and 96.7 ± 6.3 respectively.
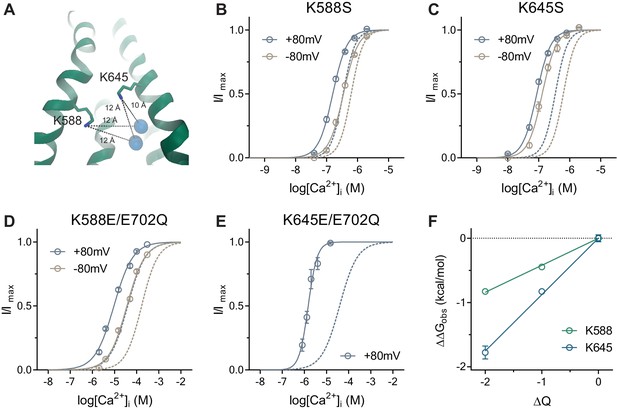
Activation properties of pore mutants.
(A) Location of the pore residues Lys 588 and 645 relative to the Ca2+ binding site (distances in Å). B-E. Concentration-response relations of K588S (B), K645S (C), K588E/E702Q (D) and K645E/E702Q (E) recorded at +/-80 mV using a rundown-correction protocol. Solid lines are fits to the Hill equation. Dashed lines are the relations of WT (B–C) and E702Q (D–E). Data are mean values of normalized concentration-response relations from 8-11, 7-8, 10 and 5-7 individual patches respectively, errors are s.e.m. (F) Relationship between the electrostatic potential of the intracellular pore entrance (ΔQ) and Ca2+ binding energetics (ΔΔGobs) for the indicated residues. Data are transforms of EC50 mutant/EC50 background at +80 mV using Equation 6. Solid lines are fits to Equation 5. The relative permittivity () for Lys 588 and 645 were estimated to be 131.9 ± 12.3 and 71.2 ± 6.1 respectively.
Current traces of mutants at the neck region.
Representative current traces of mutants K588S (A), K645S (B), K588E/E702Q (C) and K645E/E702Q (D). Blue and violet dots indicate the reference pulses used for rundown correction.
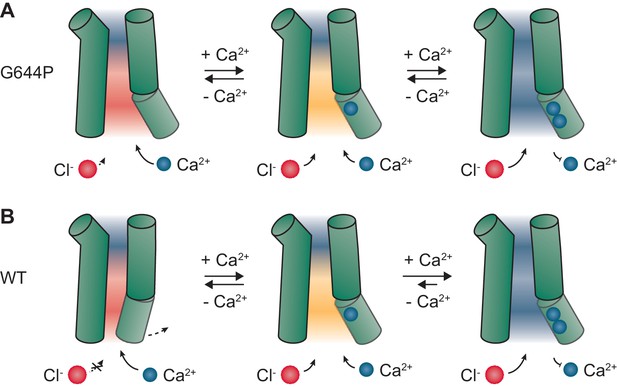
Mechanism.
Schematic depiction of gating in G644P (A) and WT (B). In the apo state of the mutant G644P, the negative electrostatic potential strongly favors the access of Ca2+ over Cl-. In contrast, the positive electrostatic potential in the doubly occupied state strongly favors the access of Cl-. A similar mechanism is expected to occur in WT, which requires Ca2+ for the activation of α6 and the opening of a steric gate located above the intracellular vestibule. When activated, the major conducting state in WT is the open state with two Ca2+ ions bound. Green cylinders, selected helices delimiting the pore; blue spheres, Ca2+ ions; red spheres, Cl- ions. Red, orange and blue backgrounds depict negative, mildly negative and positive electrostatic potential in the pore.
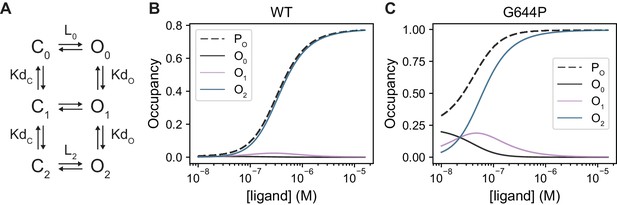
Contribution of apo-, singly- and doubly occupied states in WT and G644P in a model of activation.
(A) Minimal model for TMEM16A activation. C and O indicate closed and open states, respectively; the subscript indicates the number of bound Ca2+ ions., equilibrium dissociation constant;, forward equilibrium constant of gating transitions. Microscopic reversibility requires that. B-C. Occupancy of open states in relation to the open probability (PO) as a function of Ca2+ concentration. = 5 x 10−3, = 3.5 and = 6 x 10−7 M for WT (B) and = 0.3, = 210 and = 6 x 10−7 M for G644P (C). The gating constants were set 60 times larger in (C) while the affinity was kept unchanged. Parameters were chosen to account for the qualitative behaviour of the constructs and may not be quantitatively exact. Nonetheless, this model is consistent with the degree of cooperativity observed in steady-state responses and our experimental estimates on the relative contribution of open states with various occupancy in WT.
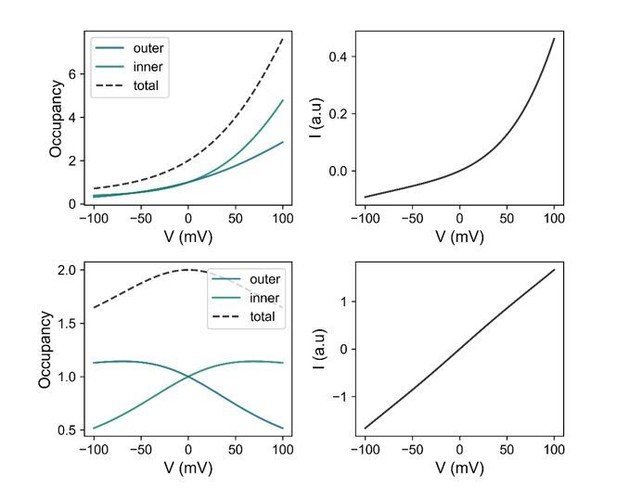
Model calculations based on the energy profile of G644P at zero Ca2+ (top) and a WT-like channel with a totally symmetric energy profile (bottom).
Left, Ion occupancy in the model channel relative to that at the outermost wells ( or ) as a function of voltage at the indicated locations. Right, Shape of the I-V relation at steady state. At zero voltage, the relative occupancy is 1 for all cases because our model assumes no binding affinity when no voltage gradient is present. The curves on the left panels were calculated using
and those on the right panels using
where
under symmetrical ionic conditions. The number of barriers is 3 in this model. This has been determined to describe the conduction properties of mutants in our previous study (Paulino et al., 2017). is the voltage-dependent rate constant of the outermost barrier. The rate constant of the innermost barrier is defined as and that of the middle barrier as where both fitted parameters and are independent of voltage. ’s are the probabilities of occupying the ith wells (from to ). is a proportionality factor that has a dimension of volume and may be interpreted as the hypothetical volume for outermost well at the channel entrance. is the membrane potential, is the valence of the ion, and , and have their usual meanings.
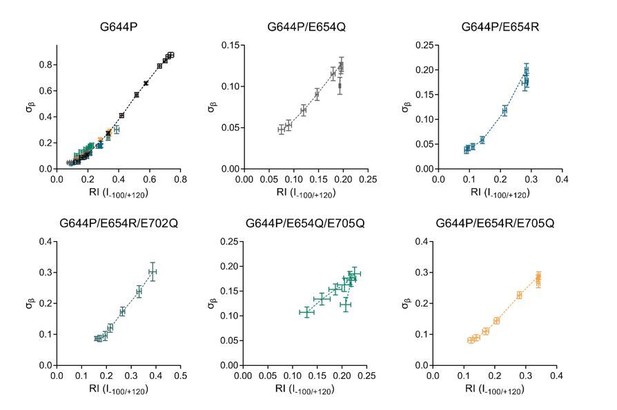
Relationship between experimental and RI (I-100/+120) for the G644P family of constructs.
Horizontal error bars indicate S.E.M and vertical error bars indicate the 95% confidence interval.
Tables
| Reagent type (species) or resource | Designation | Source or reference | Identifiers | Additional information |
|---|---|---|---|---|
| Gene (Mus musculus) | TMEM16A or Ano1 splice variant ac | DOI: 10.1038/ nature13984 | UniProt identifier: Q8BHY3-1 | |
| Cell line (Homo sapiens) | HEK293T | ATCC | ATCC CRL-1573 | Obtained directly from ATCC; tested negative for myc oplasma contamination |
| Transfected construct (Mus musculus) | G644P | this paper | generated using a modified QuikChange protocol as described in Methods | |
| Transfected construct (Mus musculus) | Q649A | this paper | as G644P | |
| Transfected construct (Mus musculus) | G644P/E654Q | this paper | as G644P | |
| Transfected construct (Mus musculus) | G644P/E654R | this paper | as G644P | |
| Transfected construct (Mus musculus) | G644P/E6 54R/E702Q | this paper | as G644P | |
| Transfected construct (Mus musculus) | G644P/E6 54Q/E705Q | this paper | as G644P | |
| Transfected construct (Mus musculus) | G644P/E654R/E705Q | this paper | as G644P | |
| Transfected construct (Mus musculus) | K588S | this paper | as G644P | |
| Transfected construct (Mus musculus) | K645S | this paper | as G644P | |
| Transfected construct (Mus musculus) | K588E/E702Q | this paper | as G644P | |
| Transfected construct (Mus musculus) | K645E/E702Q | this paper | as G644P | |
| Recombinant DNA reagent | modified pcDNA3.1 vector | Invitrogen, DOI: 10.1085/jgp.201611650 | bearing a 5′ untranslated region (UTR) of hVEGF (from pcDNA4/ HisMax; Invitrogen) | |
| Software, algorithm | Clampex 10.6 | Molecular devices | ||
| Software, algorithm | Clampfit 10.6 | Molecular devices | ||
| Software, algorithm | Prism 5/6 | GraphPad | ||
| Software, algorithm | Excel | Microsoft | ||
| Software, algorithm | NumPy | http://www.numpy.org/ | ||
| Software, algorithm | CHARMM | https://www. charmm.org/ | ||
| Software, algorithm | VMD | https://www.ks.uiuc.edu/Research/vmd/ |
Additional files
-
Transparent reporting form
- https://doi.org/10.7554/eLife.39122.016





