The Natural History of Model Organisms: From molecular manipulation of domesticated Chlamydomonas reinhardtii to survival in nature
Abstract
In the mid-20th century, the unicellular and genetically tractable green alga Chlamydomonas reinhardtii was first developed as a model organism to elucidate fundamental cellular processes such as photosynthesis, light perception and the structure, function and biogenesis of cilia. Various studies of C. reinhardtii have profoundly advanced plant and cell biology, and have also impacted algal biotechnology and our understanding of human disease. However, the 'real' life of C. reinhardtii in the natural environment has largely been neglected. To extend our understanding of the biology of C. reinhardtii, it will be rewarding to explore its behavior in its natural habitats, learning more about its abundance and life cycle, its genetic and physiological diversity, and its biotic and abiotic interactions.
https://doi.org/10.7554/eLife.39233.001Introduction
Chlamydomonas reinhardtii is a single-celled green alga found in temperate soil habitats (Figure 1). It has proven to be such a powerful model for dissecting fundamental processes in biology that investigators have dubbed it the 'green yeast' (Goodenough, 1992; Rochaix, 1995). Ehrenberg described the genus Chlamydomonas in 1833, and Dangeard the species C. reinhardtii in 1888 (Harris et al., 2009). Chlamydomonas was found suitable for genetic studies in the early 20th century (Harris, 2001), while the development of C. reinhardtii as a model organism dates to the 1950s when the first mutants were generated (Harris, 2009).
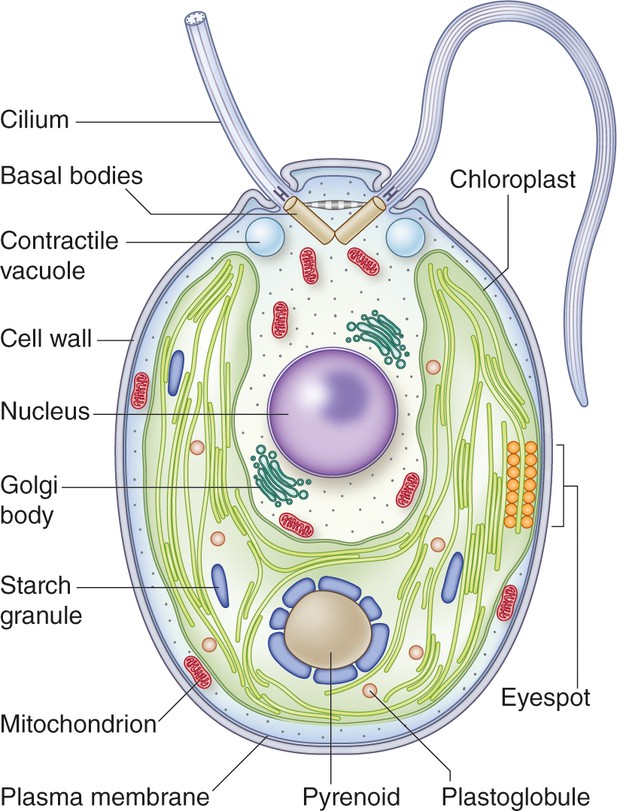
Structure of a vegetative Chlamydomonas reinhardtii cell.
This cell has a 5-10 µm diameter (Gallaher et al., 2015). The two anterior cilia possess a 9+2 microtubule structure characteristic of motile cilia of eukaryotes. The cilia are critical for mating processes and confer motility to the cell (Harris, 2001). A single cup-shaped chloroplast occupies a large proportion of the cell's volume. This organelle houses the machinery for oxygenic photosynthesis and contains the pyrenoid, a structure in which Rubisco is concentrated; the pyrenoid is a component of the carbon concentrating mechanism (CCM) which functions to concentrate inorganic carbon in the cell against a concentration gradient (Mackinder et al., 2016). Close to the cell equator, at the edge of the chloroplast, is the eyespot. This primordial visual system allows the cells to orient their swimming toward or away from the light (phototaxis). Under hypoosmotic conditions, the cytoplasmic water content is maintained by pumping water out of the cell through contractile vacuoles positioned at the cell’s anterior (Komsic-Buchmann et al., 2014). At the base of the cilia are the basal bodies, which are responsible for ciliary assembly (Dutcher and O'Toole, 2016). Other features of the cell include a centrally located nucleus, a proteinaceous cell wall, Golgi bodies within the cup-shaped region formed by the chloroplast, and mitochondria. Image credit: Debbie Maizels.
Various features make C. reinhardtii an excellent laboratory species. It grows vegetatively as a haploid, which allows mutant phenotypes to be expressed immediately. Under optimal conditions, C. reinhardtii grows so rapidly that its numbers can double approximately every 8 hours (Harris, 2001). The fact that it can grow in the dark on acetate-containing medium while retaining a functional photosynthetic apparatus, has allowed even light-sensitive photosynthesis mutants to be isolated (Levine, 1969; Spreitzer and Mets, 1980). The motile cilia of this photosynthetic eukaryote share the same structure and many of the same constituent proteins as those of mammals, and so research into its motility prompted studies that greatly advanced our understanding of cilium dysfunctions in humans (Brown and Witman, 2014). Furthermore, C. reinhardtii can be induced to sexually reproduce in the laboratory, making it easy to introduce multiple traits into a single haploid strain (e.g. to generate double or triple mutants). The power of C. reinhardtii as a model organism was further elevated by the advent of genetic transformation (Boynton et al., 1988; Kindle, 1990; Remacle et al., 2006), the establishment of a full nuclear genome sequence (Merchant et al., 2007), the construction of a genome-wide library of mapped, indexed insertional mutants (Li et al., 2016) and CRISPR-mediated targeted gene disruptions (Ferenczi et al., 2017 and references therein).
Studies of C. reinhardtii have enabled numerous landmark discoveries and advances. One remarkable example is the discovery of intraflagellar transport of granule-like particles (Kozminski et al., 1993) and the roles of motor proteins in the process (Prevo et al., 2017 and references therein). Furthermore, structural analyses of wild type and mutants with defective cilia have massively contributed to our knowledge of the building blocks of these structures, their organization and their function (Goodenough and Heuser, 1985; Silflow and Lefebvre, 2001; Nicastro et al., 2006). These analyses also led to classic studies that demonstrated that abnormal cilia could cause human genetic diseases such as polycystic kidney disease (Pazour et al., 2000; Li et al., 2004). Additionally, acetate-requiring mutants (often unable to perform photosynthesis) have immensely advanced our understanding of photosynthesis, especially the ordering of electron carriers in the photosynthetic electron transport chain (e.g. Gorman and Levine, 1965). Two core proteins of photosystem II (D1 and D2) were first identified in C. reinhardtii (Chua and Gillham, 1977) and later proposed to be key components of this photosystem’s reaction centers (Deisenhofer et al., 1985; Trebst, 1986; Satoh, 2003). More recently, a central role of the STT7 kinase in photosynthetic state transitions (Depège et al., 2003), and a key function of the xanthophyll cycle in nonphotochemical quenching were first established in C. reinhardtii (Niyogi et al., 1997).
The field of optogenetics experienced a recent quantum leap with the discovery of channelrhodopsins in C. reinhardtii. When expressed in other cells, these gated ion channels can be stimulated with light to activate various processes, including neuronal activity (Hegemann and Nagel, 2013). Sophisticated genetic, biochemical and cell biological analyses of C. reinhardtii are currently being performed to understand the cell cycle (Cross and Umen, 2015), basal bodies/centrioles function (Dutcher and O'Toole, 2016), pyrenoid structure (Freeman Rosenzweig et al., 2017), mechanisms associated with photoreceptor function and light acclimation (Minagawa and Tokutsu, 2015; Petroutsos, 2017) and organismal interactions in ecosystems (Thrane et al., 2016). Finally, C. reinhardtii is being exploited to study the evolution of multicellularity, especially with respect to multicellular algal species of the order Volvocales (Hallmann, 2011).
Habitats and biogeography
C. reinhardtii can unambiguously be identified by sequencing internal transcribed spacers (ITS) or various phylogenetically informative genes (Pröschold et al., 2005). Yet many ecological studies have relied on light microscopy to identify Chlamydomonas species (sensu lato – see Box 1). Typically, two anterior cilia and a cup-shaped chloroplast harboring a pyrenoid have been sufficient criteria for a cell to be considered a Chlamydomonas sp. This morphology-based identification may be reliable to the level of genus, but rarely to the species level since many species look very similar. For these reasons, at times we omit species designations and simply note the organism as Chlamydomonas sp. A routine use of genetic taxonomic markers in the future would improve our knowledge of the geographical distribution of C. reinhardtii and related species and allow for more precise classifications.
Box 1.
Taxonomic and laboratory history of C. reinhardtii
Based on traditional taxonomic criteria, the genus Chlamydomonas (sensu lato) contains more than 500 species. In the course of taxonomic revisions, which are still in progress, Chlamydomonas (sensu stricto) is comprised of three species (Pröschold et al., 2018). Accordingly, the taxonomy of some species mentioned in this article, such as C. nivalis or C. euryale, may be revised in the future. Furthermore, our use of the designations 'Chlamydomonas sp.' and 'Chlamydomonas spp.' refers to one or more Chlamydomonas species, respectively, which were typically not classified to the level of species and may not be C. reinhardtii. The majority of the contemporary C. reinhardtii laboratory strains were derived from a single zygote isolated from a potato field in Massachusetts in 1945 (Harris, 2009). The sequencing of 39 common laboratory strains shows that they fall into five genetically distinct lineages from two parents or haplotypes (Gallaher et al., 2015). Under laboratory conditions, mutations accumulate at a rate of ~0.03 division-1 genome-1, corresponding to one mutation every 30 generations (Gallaher et al., 2015). In addition, removal of C. reinhardtii from its natural environment, including cultivation in the laboratory or cryopreservation, may unintentionally select for specific traits. For example, C. reinhardtii is often grown on medium containing ammonium as a nitrogen source, which allowed for the evolution of mutants (nit1, nit2) unable to utilize nitrate (Harris, 2009; Gallaher et al., 2015). For these reasons, the isolates domesticated for decades in the laboratory may only loosely correspond to wild C. reinhardtii strains. Furthermore, we do not know if the laboratory strains are still capable of surviving in the wild. To examine the ecological significance of laboratory findings, it will be important to isolate additional wild C. reinhardtii strains and characterize their behavior both in the field and in culture.
https://doi.org/10.7554/eLife.39233.003While Chlamydomonas spp. (not identified at the species level) occur widely in temperate, subtropical and tropical soils (Starks et al., 1981), confirmed C. reinhardtii has only been found in temperate soils in Northern America and Japan (Pröschold et al., 2005; Nakada et al., 2010). It occurs in cultivated fields but appears absent from many other habitats, suggesting it prefers nutrient-rich, disturbed soils (Sack et al., 1994). Most contemporary laboratory strains have emanated from a single soil isolate collected in 1945 (Box 1). Light typically penetrates only millimeters into the soil, depending on factors such as the soil structure and moisture content (Tester and Morris, 1987; Ciani et al., 2005). Therefore, photosynthetic microbes are generally most abundant in the upper few millimeters where they can harvest light energy, although in some instances they can be present in soil layers where there is essentially no light (Metting, 1981). Chlamydomonas spp. are even present in biological soil crusts where they help stabilize the surface of drylands, contribute to primary production and potentially act as pioneer species (Büdel et al., 2009).
All unambiguously identified C. reinhardtii isolates were collected from soil habitats (T. Pröschold, personal communication), yet Chlamydomonas spp. are also commonly found in the pelagic zone of lakes, where they sometimes form spring blooms (Similä, 1988; Krivtsov et al., 2000). The term 'pelagic zone' refers to the water column of lakes and oceans not on or near the lake or ocean bottom. Chlamydomonas spp. are usually motile, and although this has an energetic cost, it gives them a competitive advantage in lakes that have stratified into distinct layers as a consequence of seasonal changes in temperature (Striebel et al., 2009). Under conditions of stratification, motile algae often ascend toward the lake surface during the day to optimize their exposure to sunlight. During the night, they tend to descend to access the nutrient-rich environment below the surface. Indeed, this pattern of vertical movement has been observed for the population of Chlamydomonas sp. in a small Finnish lake (Jones, 1988).
Environmental conditions and the availability and distribution of natural resources differ substantially in soils and lakes (Sommer et al., 2012; Coleman et al., 2017). Phosphorous, for example, is likely to be limiting to the growth of organisms in lakes and geologically old soils, while nitrogen limitation is more common in young soils (Schindler, 1977; Vitousek and Howarth, 1991). Light availability and grazing pressure by predators represent additional key environmental differences between soil and lake habitats. Consequently, these two habitats require distinct adaptations and life history strategies to optimize fitness, and it is still an open question as to whether specific pelagic strains of C. reinhardtii exist in lakes (Box 2).
Chlamydomonas spp. other than C. reinhardtii are adapted to a wide range of habitats. For example, Chlamydomonas eustigma is an acidophilic species isolated from acid mine drainage (Hirooka et al., 2017), Chlamydomonas euryale is found in temperate marine environments (Burch et al., 2015), Chlamydomonas spp. have been isolated from Antarctic ice (Liu et al., 2006), and some members of the genus Chlamydomonas are carotenoid-rich organisms present on the surface of snow, giving it a red appearance (Remias et al., 2005). A Chlamydomonas sp. has even been identified in the air at 1,100 meters above the ground: this and other algae can be dispersed by wind over extended distances (Brown et al., 1964). Taken together, several reports provide information on the biogeographical distribution of C. reinhardtii and other Chlamydomonas spp. However, there is little knowledge of the abundance and variations of Chlamydomonas spp. in different soil types, the dynamics of these natural populations over daily or seasonal cycles, and their physiological capabilities.
Genomics
The chloroplast and mitochondrial genomes of C. reinhardtii have been sequenced and are 206 and 15.8 kb, respectively (Vahrenholz et al., 1993; Maul et al., 2002; Gallaher et al., 2018). Since the sequence of the nuclear genome was first published (Merchant et al., 2007), the scientific community has focused some effort on elevating the quality of the genome sequence and improving its assembly and annotation (Blaby et al., 2014). The current version 5.5 nuclear genome is 111 Mb, which is similar in size to the genome of the model land plant Arabidopsis thaliana (Blaby et al., 2014). Recently, whole-genome sequences for more than 50 additional laboratory strains and field isolates were generated (Flowers et al., 2015; Gallaher et al., 2015). The sequences of 12 field isolates confirmed earlier reports that with a nucleotide diversity (π) of ~3%, the C. reinhardtii genome is among the most polymorphic of all eukaryotes (Flowers et al., 2015). The field strains, isolated from various locations in the United States and Canada, genetically group into three distinct populations, with gene flow between populations sufficiently low to allow the populations to adapt to their local environments. The low ratio of genome-wide non-synonymous to synonymous substitutions (0.58) further indicates that natural selection efficiently eliminates C. reinhardtii alleles of low fitness (Flowers et al., 2015). Whole-genome and epigenome sequencing has also been used to examine adaptation in the laboratory under changing environmental conditions (Kronholm et al., 2017).
Life cycle and its role in nature
Forming zygotes likely allows C. reinhardtii to survive when conditions become harsh (Harris, 2001; Goodenough et al., 2007). In the laboratory, gametogenesis can be induced by nitrogen starvation (Treier et al., 1989) in conjunction with specific light conditions; both signals may inform the cell of deteriorating environmental conditions (see below). The fusion of haploid gametes results in diploid zygotes that can develop over several days into highly resistant, dormant zygospores (Figure 2). When nitrogen is added back to the medium, the zygotes germinate in the light, undergo meiosis and typically release four haploid cells that resume vegetative growth (Harris, 2001). Dormant zygospores can remain viable in soil for many years (Harris, 2001) and survive freezing (Suzuki and Johnson, 2002), desiccation (Heimerl et al., 2018) and probably other forms of harsh environmental conditions. This extraordinary resistance is associated with the multilayered cell wall of the zygospores, which contains a durable lipid polymer structurally similar to those found in million-year-old microfossils (described for Chlamydomonas monoica; Blokker et al., 1999). Furthermore, sexual reproduction can increase the rate of adaptation of C. reinhardtii to new or changing environmental conditions, particularly if the population and genetic diversity within the population are large (Colegrave, 2002).
Box 2.
Outstanding questions about the natural history of C. reinhardtii
What are the geographic origins of C. reinhardtii? What are its current geographic and vertical distributions? How do populations of C. reinhardtii quantitatively change over time and what factors impact these changes? What are the major mechanisms of C. reinhardtii dispersal? For example, are aquifers common routes for the transport of C. reinhardtii over long distances?
What is the genetic variability within and between C. reinhardtii populations? What are the relationships among populations of the various Chlamydomonas species?
Do specific pelagic strains of C. reinhardtii exist in lakes? If so, do they have major differences in their life histories, physiologies and genome sequences compared to soil-dwelling strains?
What are the typical division rates of vegetative C. reinhardtii cells in the wild? How frequently does sexual reproduction occur in natural populations? How common are dormant zygospores in the environment, and where do they occur? Are zygospores typical overwintering forms, and do they also have an increased resistance to challenging biotic interactions?
What are the most common biotic interactions of C. reinhardtii in the environment (competing photosynthetic microbes, grazers, bacteria, fungi)? In what ways does C. reinhardtii communicate with its neighbors (e.g. infochemical signals)? What is the metabolic significance of these interactions?
How often and under what situations do cells shed their cilia in nature? Is there a selective advantage of deciliation in response to stress?
Does C. reinhardtii associate with biofilms on soil particles and, if so, how are the algal cells organized within the biofilm community?
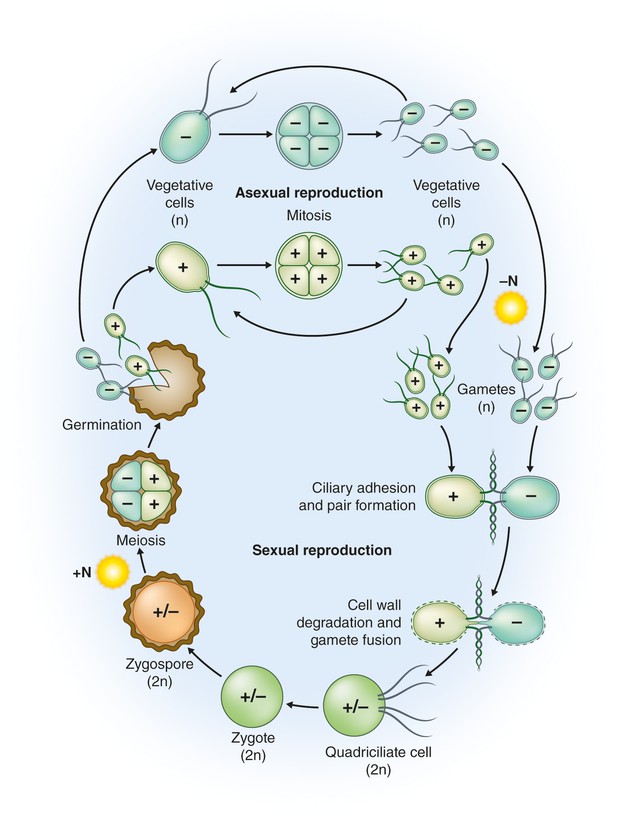
Life cycle of C.reinhardtii.
Haploid (n) vegetative cells occur as two mating types, mt+ and mt-, that divide by mitosis (“Asexual reproduction”; Harris, 2001, Goodenough et al., 2007). Gametogenesis can be induced by nitrogen starvation (-N) in the presence of light, and gametes of opposite mating types can fuse to form diploid (2n) zygotes (“Sexual reproduction”). Within a few hours of fertilization, zygotes resorb their four cilia to become immotile. Over the course of several days these zygotes are remodeled into highly resistant, dormant zygospores. In this process, a strong, multilayered cell wall is formed, and chlorophyll is degraded (Harris, 2001; Goodenough et al., 2007). As a result, mature zygospores appear orange, which reflects their carotenoid content (Lohr, 2009). When environmental conditions improve, the zygote undergoes meiosis to release four haploid cells (sometimes 8 and 16 when mitosis also occurs within the zygote wall; “Germination”). The haploid cells then resume vegetative growth. In the laboratory, zygote germination is induced by the addition of nitrogen (+N) to the medium in the light (Harris, 2001); nitrogen also causes reprogramming of gametes to vegetative cells (Pozuelo et al., 2000). Image credit: Debbie Maizels.
Following gamete fusion, a pair of homeodomain transcription factors initiates the genetic program for zygote development (Kurvari et al., 1998; Lee et al., 2008). The first zygote-specific genes are induced within minutes of gamete fusion, with hundreds of additional genes activated over the next few hours (Lopez et al., 2015; Joo et al., 2017). A gene encoding a polyketide synthase is induced two days after zygote formation and is critical for the zygote-to-zygospore transition, probably because it participates in the biosynthesis of the cell wall lipid polymer (Heimerl et al., 2018). Several stages of the sexual cycle, including gamete formation and maintenance and zygote germination, depend on light and involve regulation by three different photoreceptors (Huang and Beck, 2003; Müller et al., 2017; Zou et al., 2017).
Similar to Saccharomyces cerevisiae (Liti, 2015), we know little about the life cycle of C. reinhardtii in its natural environment. For example, there is no quantitative data on the frequency of sexual reproduction relative to vegetative growth. Yet, nitrogen is thought to become limited more commonly in soils than lakes (Schindler, 1977; Vitousek and Howarth, 1991; Coleman et al., 2017). This notion is congruent with nitrogen limitation being a major cue for zygospore formation in nature, but we are not aware of any data on zygospore induction in natural soil environments. The occurrence of clonal cultures of opposite mating types that are derived from a single zygospore isolated from dry soil provides additional evidence for a critical role of zygospores during desiccation (Harris, 2009). Freezing resistance of zygospores and their more efficient germination under long-day conditions compared to short-day conditions suggests that zygospore formation is an overwintering strategy (Suzuki and Johnson, 2002). If true, the question arises as to whether or not nitrogen limitation and day length are adequate cues to herald the approach of winter, or whether, for example, a decrease in soil moisture content or temperature can also induce zygospore formation in C. reinhardtii.
Physiological and metabolic capabilities
C. reinhardtii not only orients itself with respect to light, but can also swim upward in complete darkness. This negative gravitaxis may facilitate orientation and movement of the cells at night or in the soil environment, potentially helping the alga locate areas with more favorable conditions of illumination following daybreak (Bean, 1977). Furthermore, vegetative cells are attracted to ammonium, nitrite and nitrate (Ermilova and Zalutskaya, 2014 and references therein). Chemotaxis towards ammonium is strongest during the night, whereas phototaxis towards the light is strongest during the day, with both processes regulated by the circadian clock (Bruce, 1970; Byrne et al., 1992). Finally, the hypothesis that the circadian clock depends on gravity or a magnetic field was refuted by experiments performed with C. reinhardtii on a space shuttle under microgravity conditions (Mergenhagen and Mergenhagen, 1987).
Cilia enable C. reinhardtii to swim in an aqueous medium, and also glide on solid surfaces. Gliding motility may be important when C. reinhardtii resides within a thin water film that coats soil particles (Mitchell, 2000). The gliding speed of C. reinhardtii is ~1 µm s-1 (Shih et al., 2013) whereas the average forward swimming speed is 100-200 µm s-1 (Rüffer and Nultsch, 1985). Under various stress conditions, such as acidification of the medium, C. reinhardtii loses or sheds its cilia when a specific break point near the base of the cilium is activated (Quarmby, 2009). When conditions improve, the cilia regenerate. The biological reason for deciliation is still a mystery, but various hypotheses have been put forth (Quarmby, 2009). Deciliation is observed in a wide range of different cell types; for example, inhalation of irritant chemicals can lead to deciliation of respiratory epithelial cells in mammals (Buckley et al., 1984). Therefore, it seems likely that a predetermined break point is an ancient and inherent property of every cilium (Quarmby, 2009). Consequently, deciliation may not confer a selective advantage, but might be a consequence of pathological conditions that cause over-stimulation of the ciliary disassembly process. On the other hand, the ciliary membrane of C. reinhardtii is in direct contact with the environment (not protected by cell wall) and therefore, deciliation may reduce the entrance of noxious compounds into cells. Deciliation may also allow cells to escape when their cilia are stuck to the surface of a predator (Quarmby, 2009). Studying deciliation in the natural environment holds the promise of new insights into selection pressures that led to its evolution.
The ability of C. reinhardtii to grow under heterotrophic and fermentative conditions might be an adaptation to soil environments where there can be both low light and low oxygen. Anoxic/hypoxic conditions are likely to mostly occur at night when there is no photosynthesis to release oxygen and the soil microbes are respiring. Under anoxic conditions, C. reinhardtii can use glycolysis to yield energy, which is sustained by fermentation metabolism and the release of reduced organic compounds (Catalanotti et al., 2013). C. reinhardtii has recently been shown to activate a variety of different pathways that result in the formation of many fermentation products including formate, lactate, acetate, acetyl-CoA, succinate, hydrogen and glycerol (Atteia et al., 2013; Catalanotti et al., 2013; Yang et al., 2015). While some regulatory elements involved in anoxic metabolism are known (Hemschemeier et al., 2013; Huwald et al., 2015; Düner et al., 2018), little is understood about what controls the various pathways associated with fermentation and the ways in which these pathways are integrated.
Biotic interactions
In nature, C. reinhardtii is continuously in contact with other organisms, including competitors, predators, pathogens, parasites, commensals or mutualists. Most molecular details concerning these interactions, which likely involve chemical signaling, nutrient exchange and receptor-mediated processes, have not been examined. In lakes, the various Chlamydomonas spp. successfully compete with many other pelagic algal species for light and nutrients; rapid growth of Chlamydomonas spp. likely compensates for severe grazing losses, such as during periods of rapid proliferation of filter feeders, like water fleas (cladocerans). High rates of algal growth demand high nutrient levels. The concentrations of dissolved nutrients during the growing season are usually highest after spring mixing (Sommer et al., 2012), and therefore, the abundance of Chlamydomonas spp. in temperate lakes often shows a strong peak in spring or summer (Dembowska, 2015). In addition, the absence of filter feeders and the presence of more selective feeders in the soil may result in lower grazing losses and less seasonal differences in abundance patterns.
Predation of C. reinhardtii by zooplankton such as Daphnia, a highly efficient filter feeder (Van Donk et al., 1997), rotifers (Lurling and Beekman, 2006), and protists such as Tetrahymena (Taub and McKenzie, 1973) or Peranema (Figure 3), has been shown to occur in the laboratory. These predators either live exclusively in the pelagic zone of lakes, or at least more commonly in this habitat compared to soils. In the soil, animals such as earthworms or springtails, and protists are typical predators of microscopic algae (Schmidt et al., 2016; Seppey et al., 2017), but there is currently little specific information on predators of C. reinhardtii. The formation of large aggregates of C. reinhardtii cells is a general and probably non-specific defense strategy by which the alga may avoid ingestion. For example, the rotifer Brachionus calyciflorus triggers the formation of so-called palmelloid colonies (Lurling and Beekman, 2006). These colonies are aggregates of C. reinhardtii that may form as a consequence of the failure of the mitotically dividing mother cell to release the daughter cells from its encapsulating cell wall (Khona et al., 2016). This phenomenon may be triggered by stress under conditions in which zygospore formation is not possible (Khona et al., 2016). On the other hand, C. reinhardtii can actively aggregate in the presence of the predatory protist Peranema trichophorum (Sathe and Durand, 2016). A P. trichophorum culture filtrate was able to induce algal aggregation, suggesting that C. reinhardtii senses an unidentified substance (a kairomone) that is released by the predator (Sathe and Durand, 2016).
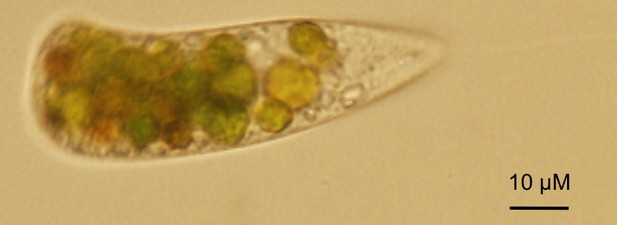
C. reinhardtii ingested by the predatory protist Peranema trichophorum.
Image credit: Santosh Sathe and Pierre Durand.
C. reinhardtii is also a prey for soil bacteria. The bacterium Pseudomonas protegens can surround and immobilize algal cells (Video 1); it secretes a cyclic lipopeptide that triggers an increase in calcium levels inside C. reinhardtii cells with subsequent deciliation (Aiyar et al., 2017). This antagonistic interaction inhibits algal growth and probably leads to the death of most of the algal cells; the bacteria may acquire trace metals from the dying cells (Aiyar et al., 2017). Furthermore, small molecules from C. reinhardtii activate quorum sensing in Pseudomonas aeruginosa (Rajamani et al., 2008). It will be important to determine if algal cells also produce quorum-sensing mimics that influence P. protegens. Finally, while no viral pathogens of C. reinhardtii have been reported, it seems likely that they exist. The areas of algal-bacterial and algal-viral interactions are fertile for more probing basic research.
C. reinhardtii surrounded by the harmful bacteria Pseudomonas protegens (Aiyar et al., 2017)
Video credit: Prasad Aiyar, Severin Sasso and Maria Mittag.
Several beneficial interactions of C. reinhardtii have been described, including interactions with growth-promoting bacteria and even mutualism (e. g. Nikolaev et al., 2008; Lörincz et al., 2010; Kim et al., 2014). These findings provide the basis for future studies that address regulatory mechanisms and identify specific compounds that impact the biology of C. reinhardtii in nature. One compound synthesized by prokaryotes and used by many algae is vitamin B12. Although C. reinhardtii does not depend on vitamin B12 to grow, it can obtain the compound from bacteria and use it as a cofactor in a pathway for methionine biosynthesis that is thermal tolerant (Kazamia et al., 2012; Xie et al., 2013). Indeed, under elevated temperatures, B12-providing bacteria increase the fitness of the alga (Xie et al., 2013). A mutualism was also observed between C. reinhardtii and S. cerevisiae in sealed microtiter plates, with the algae trading reduced nitrogen for CO2 (Hom and Murray, 2014). While the significance of these interactions may be uncertain, they, and many yet to be discovered, likely shape the ways in which C. reinhardtii navigates in a complex biosphere.
Conclusions
Although C. reinhardtii has been studied in the laboratory for many decades, we do not know the extent to which results from the laboratory reflect growth, life cycle and behavior of this alga in nature (Box 2). As a model system, C. reinhardtii is almost exclusively grown as a pure culture, a situation almost never encountered in the 'wild'. Returning a laboratory strain of C. reinhardtii to its native habitat would reveal whether domestication caused it to lose its ability to survive within the dynamic fabric of nature. Molecular analyses of the reintroduced strain could also reveal changes in the cells' physiology that underlie the loss of fitness in natural habitats, as well as other changes potentially associated with its adaptation to laboratory conditions.
Field surveys are often hampered by difficulties in assessing the metabolic state of the cells and in establishing key inter-organismal interactions. However, harnessing the full potential of meta-omics and single-cell technologies could provide a fuller appreciation of the physiological status of cells as they experience environmental fluctuations and the dominant interactions that shape the life of C. reinhardtii. Expanding this understanding will require time-resolved data on the geographical occurrence of C. reinhardtii in different habitats, its genetic potential and population genetics, and dissection of biotic and abiotic interactions. Such studies could then be extended to include analyses performed under controlled laboratory conditions that closely align with conditions encountered in the field, using innovative methods such as microfluidics to mimic conditions of the soil and other complex environments (Stanley et al., 2016).
References
-
Anaerobic energy metabolism in unicellular photosynthetic eukaryotesBiochimica et Biophysica Acta 1827:210–223.https://doi.org/10.1016/j.bbabio.2012.08.002
-
Geotactic behavior of ChlamydomonasThe Journal of Protozoology 24:394–401.https://doi.org/10.1111/j.1550-7408.1977.tb04759.x
-
The Chlamydomonas genome project: a decade onTrends in Plant Science 19:672–680.https://doi.org/10.1016/j.tplants.2014.05.008
-
The biological clock in Chlamydomonas reinhardiThe Journal of Protozoology 17:328–334.https://doi.org/10.1111/j.1550-7408.1970.tb02380.x
-
Respiratory tract lesions induced by sensory irritants at the RD50 concentrationToxicology and Applied Pharmacology 74:417–429.https://doi.org/10.1016/0041-008X(84)90295-3
-
Environmental manipulation of growth and energy carrier release from freshwater and marine Chlamydomonas speciesJournal of Applied Phycology 27:1127–1136.https://doi.org/10.1007/s10811-014-0433-0
-
Fermentation metabolism and its evolution in algaeFrontiers in Plant Science 4:150.https://doi.org/10.3389/fpls.2013.00150
-
The sites of synthesis of the principal thylakoid membrane polypeptides in Chlamydomonas reinhardtiiThe Journal of Cell Biology 74:441–452.https://doi.org/10.1083/jcb.74.2.441
-
Light penetration in soil and particulate mineralsEuropean Journal of Soil Science 56:561–574.https://doi.org/10.1111/j.1365-2389.2005.00688.x
-
Seasonal variation in phytoplankton and aquatic plants in floodplain lakes (lower Vistula River, Poland)Wetlands Ecology and Management 23:535–549.https://doi.org/10.1007/s11273-015-9408-4
-
High-throughput sequencing of the chloroplast and mitochondrion of Chlamydomonas reinhardtii to generate improved de novo assemblies, analyze expression patterns and transcript speciation, and evaluate diversity among laboratory strains and wild isolatesThe Plant Journal 93:545–565.https://doi.org/10.1111/tpj.13788
-
Substructure of inner dynein arms, radial spokes, and the central pair/projection complex of cilia and flagellaThe Journal of Cell Biology 100:2008–2018.https://doi.org/10.1083/jcb.100.6.2008
-
Sex determination in ChlamydomonasSeminars in Cell and Developmental Biology 18:350–361.https://doi.org/10.1016/j.semcdb.2007.02.006
-
Evolution of reproductive development in the volvocine algaeSexual Plant Reproduction 24:97–112.https://doi.org/10.1007/s00497-010-0158-4
-
Chlamydomonas as a model organismAnnual Review of Plant Physiology and Plant Molecular Biology 52:363–406.https://doi.org/10.1146/annurev.arplant.52.1.363
-
The Chlamydomonas SourcebookIntroduction to Chlamydomonas and its laboratory use, The Chlamydomonas Sourcebook, Oxford, Academic Press.
-
From channelrhodopsins to optogeneticsEMBO Molecular Medicine 5:173–176.https://doi.org/10.1002/emmm.201202387
-
Mutualistic interactions between vitamin B12-dependent algae and heterotrophic bacteria exhibit regulationEnvironmental Microbiology 14:1466–1476.https://doi.org/10.1111/j.1462-2920.2012.02733.x
-
Changes in the elemental composition of Asterionella formosa during the diatom spring bloomJournal of Plankton Research 22:169–184.https://doi.org/10.1093/plankt/22.1.169
-
Epigenetic and genetic contributions to adaptation in ChlamydomonasMolecular Biology and Evolution 34:2285–2306.https://doi.org/10.1093/molbev/msx166
-
A gamete-specific, sex-limited homeodomain protein in ChlamydomonasThe Journal of Cell Biology 143:1971–1980.https://doi.org/10.1083/jcb.143.7.1971
-
The analysis of photosynthesis using mutant strains of algae and higher plantsAnnual Review of Plant Physiology 20:523–540.https://doi.org/10.1146/annurev.pp.20.060169.002515
-
BookCarotenoidsIn: Harris EH, Stern DB, Witman GB, editors. The Chlamydomonas Sourcebook. Amsterdam: Academic Press. pp. 799–817.
-
Palmelloids formation in Chlamydomonas reinhardtii: defence against rotifer predators?Annales de Limnologie - International Journal of Limnology 42:65–72.https://doi.org/10.1051/limn/2006010
-
The biological clock of Chlamydomonas reinhardtii in spaceEuropean Journal of Cell Biology 43:203–207.
-
The systematics and ecology of soil algaeThe Botanical Review 47:195–312.https://doi.org/10.1007/BF02868854
-
Dynamic regulation of photosynthesis in Chlamydomonas reinhardtiiThe Plant Journal 82:413–428.https://doi.org/10.1111/tpj.12805
-
Chlamydomonas flagellaJournal of Phycology 36:261–273.https://doi.org/10.1046/j.1529-8817.2000.99218.x
-
Recharacterization of Chlamydomonas reinhardtii and its relatives with new isolates from JapanJournal of Plant Research 123:67–78.https://doi.org/10.1007/s10265-009-0266-0
-
Effect of bacterial satellites on Chlamydomonas reinhardtii growth in an algo-bacterial communityMicrobiology (Mikrobiologiya) 77:78–83.https://doi.org/10.1134/S0026261708010116
-
BookChlamydomonas photoreceptors: Cellular functions and impact on physiologyIn: Hippler M, editors. Chlamydomonas: Biotechnology and Biomedicine. Cham (Switzerland): Springer. pp. 1–19.
-
Intraflagellar transport: mechanisms of motor action, cooperation, and cargo deliveryThe FEBS Journal 284:2905–2931.https://doi.org/10.1111/febs.14068
-
BookDeflagellationIn: Harris EH, Stern DB, Witman GB, editors. The Chlamydomonas Sourcebook. Amsterdam: Academic Press. pp. 43–69.
-
The vitamin riboflavin and its derivative lumichrome activate the LasR bacterial quorum-sensing receptorMolecular Plant-Microbe Interactions 21:1184–1192.https://doi.org/10.1094/MPMI-21-9-1184
-
Photosynthesis, pigments and ultrastructure of the alpine snow alga Chlamydomonas nivalisEuropean Journal of Phycology 40:259–268.https://doi.org/10.1080/09670260500202148
-
Chlamydomonas reinhardtii as the photosynthetic yeastAnnual Review of Genetics 29:209–230.https://doi.org/10.1146/annurev.ge.29.120195.001233
-
Cellular aggregation in Chlamydomonas (Chlorophyceae) is chimaeric and depends on traits like cell size and motilityEuropean Journal of Phycology 51:129–138.https://doi.org/10.1080/09670262.2015.1107759
-
The identification of the photosystem II reaction center: a personal storyPhotosynthesis Research 76:233–240.https://doi.org/10.1023/A:1024933610778
-
Beyond the plankton ecology group (PEG) model: mechanisms driving plankton successionAnnual Review of Ecology, Evolution, and Systematics 43:429–448.https://doi.org/10.1146/annurev-ecolsys-110411-160251
-
Carbon sequestration and stoichiometry of motile and nonmotile green algaeLimnology and Oceanography 54:1746–1752.https://doi.org/10.4319/lo.2009.54.5.1746
-
Photoperiodic control of germination in the unicell ChlamydomonasNaturwissenschaften 89:214–220.https://doi.org/10.1007/s00114-002-0302-6
-
Continuous cultures of an alga and its grazerBulletins from the Ecological Research Committee 17:371–377.
-
The penetration of light through soilPlant, Cell and Environment 10:281–286.https://doi.org/10.1111/j.1365-3040.1987.tb01607.x
-
The topology of the plastoquinone and herbicide binding peptides of photosystem II in the thylakoid membraneZeitschrift für Naturforschung 41c:240–246.https://doi.org/10.1515/znc-1986-1-235
-
Gametic differentiation in Chlamydomonas reinhardtii: light dependence and gene expression patternsArchives of Microbiology 152:572–577.https://doi.org/10.1007/BF00425489
-
Altered cell wall morphology in nutrient-deficient phytoplankton and its impact on grazersLimnology and Oceanography 42:357–364.https://doi.org/10.4319/lo.1997.42.2.0357
-
Nitrogen limitation on land and in the sea: how can it occur?Biogeochemistry 13:87–115.https://doi.org/10.1007/BF00002772
-
Algae after dark: mechanisms to cope with anoxic/hypoxic conditionsThe Plant Journal 82:481–503.https://doi.org/10.1111/tpj.12823
-
An animal-like cryptochrome controls the Chlamydomonas sexual cyclePlant Physiology 174:1334–1347.https://doi.org/10.1104/pp.17.00493
Decision letter
-
Stuart RF KingReviewing Editor; eLife, United Kingdom
-
Peter A RodgersSenior Editor; eLife, United Kingdom
In the interests of transparency, eLife includes the editorial decision letter and accompanying author responses. A lightly edited version of the letter sent to the authors after peer review is shown, indicating the most substantive concerns; minor comments are not usually included.
Thank you for submitting your article to eLife for consideration as a Feature Article. Your article has been reviewed by two peer reviewers, and the evaluation has been overseen by Stuart King as an Associate Features Editor and Peter Rodgers as the Senior Editor. The reviewers have opted to remain anonymous.
The reviewers have discussed the reviews with one another and the Associate Features Editor has drafted this decision to help you prepare a revised submission.
Summary:
This essay is being considered as part of a series of articles on "The Natural History of Model Organisms" (https://elifesciences.org/collections/8de90445/the-natural-history-of-model-organisms). Each article should explain how our knowledge of the natural history of a model organism has informed recent advances in biology, and how understanding its natural history can influence/advance future studies.
There is much that is interesting in this overview of the single-celled green alga Chlamydomonas reinhardtii. The reviewers recognize the difficulty of writing such a wide-ranging article and commend the authors on a thought-provoking piece. They believe that this article will serve the purpose of stimulating new questions in this area of research. However, a number of details should be attended to prior to publication.
Essential revisions:
1) The current title ("Hatched to be wild: Chlamydomonas reinhardtii in nature") does not represent the précis of the article. It is also inconsistent with the other articles in series. The editor will contact you separately to provide some more guidance in this area.
2) While the previous literature has often used "flagella", most working in this area have now migrated to the term "cilia". The "flagella" of C. reinhardtii are synonymous with the cilia or other eukaryotes and not at all with the flagella of bacteria. Given that this is an enormous contribution of C. reinhardtii research in recent decades, this should be properly developed in the opening paragraph. "Cilia" should then be used throughout the rest of the article. In the Abstract, please also add "biogenesis" or "assembly" (to "structure and function of cilia").
3) Figure 1 is a good addition to the article, yet the two panels are largely unrelated to each other and referenced in distant sections of the text. Please split this figure into two, such that each part can positioned closer to the relevant text for the benefit of the reader. In relation to point above, please also replace uses of "flagellum" with "cilium" etc.
4) While it is included as part of a list, referring to C. reinhardtii as a "preferred" model organism "for dissecting photosynthetic function […]" (Introduction) is somewhat of an understatement. Most of the key components were discovered in Chlamydomonas through the collection of acetate-requiring mutants. These components were subsequently re-discovered in Arabidopsis, often without proper attribution to the original decades-earlier discoveries. The history of the photosynthesis discoveries should not be lost by weak language. Later in the text, the contribution of the Hegemann lab to the field of optogenetics also seems understated with the term "leveraged" (Introduction).
5) In the paragraph that begins, "Studies of C. reinhardtii have enabled numerous landmark discoveries" (Introduction), the discoveries chosen are somewhat selective and, in the opinion of the reviewers, some may not be as "landmark" as others that are not cited. The reviewers would encourage the authors to consider which of their mentioned examples represent paradigm shifts or truly big leaps in the understanding of a process. In addition, the reviewers believe that following discoveries merit being considered for this section: the discovery of two related PSII reaction centre proteins (D1, D2); the placement of electron carriers in the Z-scheme by genetic analysis of specific mutants using absorption spectroscopy; and any of the dozens of papers on the centriole or the cell cycle (where C. reinhardtii made significant contributions to the field of cell biology).
6) The reviewers had several other comments related to citations. The authors acknowledge that: "Because of space limitations, we were unable to cite many worthy studies", but the following revisions could help to balance and complete the reference list. These revisions are mentioned below in the order they arise in the manuscript:
6a) There are too many citations for "CRISPR-mediated targeted gene disruptions" (Introduction) compared to all the other technologies in the preceding text.
6b) In the section on Genomics, it may be better to cite the revised chloroplast genome published by Gallaher et al., 2017, rather than the earlier 2002 genome. The Gallaher article also includes the transcriptome and updated gene models.
6c) There was a concern that the literature supporting the current section on chemotaxis is limited (subsection “Physiological and metabolic capabilities”). The general feeling was that findings based on, at best, a single published paper were not substantive contributions and did not warrant inclusion.
6d) No references are included to support the following statements: "the ability to grow under heterotrophic and fermentative conditions is clearly an adaptation to soil environments" (subsection “Physiological and metabolic capabilities”); "the number of competing algal species in the soil is likely to be fewer than that of lakes" (subsection “Biotic interactions”); "In the soil, ciliates such as Paramecium may represent a typical predator" (subsection “Biotic interactions”); that the palmelloid aggregates are a result of "failure" to hatch (subsection “Biotic interactions”). In each case, if suitable citations cannot be found, please consider softening the claims or providing more rationale for including these claims over alternative explanations (for example, couldn't heterotrophy and fermentation equally be adaptations to lake sediments? Why couldn't the palmelloid aggregates instead result from biofilm formation? Etc.)
7) It is worth noting that it is not just the "ability to grow in the dark" (Introduction), but rather to grow in the dark and retain a functional photosynthetic apparatus that made some aspects of the studies possible. Most plants will become yellow in the dark, because there is a photochemical requirement in the chlorophyll biosynthetic pathway. Chlamydomonas (and other algae as well as evergreens) have a second enzyme – protochlorophyllide oxidoreductase – that catalyses the same reaction without a requirement for a photon. This trait is important and should be mentioned in the text.
8) The fact that C. reinhardtii can "form gametes as part of the sexual reproduction cycle" is seemingly put forward as an advantage of this alga as a model organism (Introduction). The text should be revised to make it clearer that this trait is not unique, but rather universal among sexual organisms [which include almost all eukaryotes]). It is also not obvious what the authors mean by the phrase: "which allows for introduction of multiple traits (e.g. mutant alleles) in a single haploid strain". Does it refer to making stable diploids? Or the generating double and triple mutants through classical genetic crosses? Or something else? Please revise to make the meaning clearer.
9) There were some concerns that little is known about the snow algae and that the field remains more confused than this article would suggest (subsection “Habitats and biogeography of C. reinhardtii”). One reviewer noted that Chlamydomonas nivalis is perhaps better considered a cryotolerant mesophile, rather than a strict cryophile/psychrophile (see https://doi.org/10.2478/botcro-2013-0012 and https://doi.org/10.1111/1574-6941.12299), and that it is possibly not even a single species. Overall, the reviewers recommend that the authors acknowledge the confusion here and minimize the discussion, perhaps just to refer that some members of the genus are psychrophilic/cryotolerant and recognized occasionally by the patches of watermelon color on snow. [Notably, the related Chloromonas nivalis is more likely a true psychrophile but, because it belongs to a different genus, it is beyond the scope of this essay].
10) Some of the section on deflagellation is counter to the conclusion of the cited Quarmby, 2009 chapter (subsection “Physiological and metabolic capabilities”). The potential adaptions discussed in this essay are introduced in the book chapter, but the conclusion of the chapter is that they are less likely than deflagellation being a consequence of pathological over-stimulation of a disassembly pathway. The reviewers request that the authors re-read the chapter, and then revise this section (including its citations) accordingly.
11) It would be great if the authors could provide a video of their Figure 2B (like those in https://doi.org/10.1038/s41467-017-01547-8 or similar). These show the wolf pack feeding behavior and the deflagellation response very nicely.
12) There was much debate in the reviewer consultation about whether the word "pelagic" can refer to organisms living in freshwater lakes (in part because of its root from the Greek "pelagikos", which relates to the sea). Please revise the text to make it clearer whether sections are referring to oceanic/marine algae or lake-dwelling algae. If "pelagic" is still used, it should be clearly defined for the benefit of unfamiliar readers. Please consider something like "organisms that live in the water column of lakes and oceans, but not on or near the bottom of the lake or ocean".
13) In the text in Box 1. Taxonomy and laboratory history of C. reinhardtii, please add "from two parents or haplotypes" to the end of the sentence that reads "The sequencing of 39 common laboratory strains show that they fall into five genetically distinct lineages". This should be mentioned; otherwise it may sound like there are 5 haplotypes.
14) The concluding paragraph could be strengthened. For example, the sentence: "It would be interesting to examine the fitness and adaptation of a domesticated laboratory strain to its native environment." Why would this be interesting? Which strain? What questions would be addressed?
https://doi.org/10.7554/eLife.39233.010Author response
Essential revisions:
1) The current title ("Hatched to be wild: Chlamydomonas reinhardtii in nature") does not represent the précis of the article. It is also inconsistent with the other articles in series. The editor will contact you separately to provide some more guidance in this area.
The title has been changed. It is now: "The Natural History of Model Organisms: From molecular manipulation of domesticated Chlamydomonas reinhardtii to survival in nature".
2) While the previous literature has often used "flagella", most working in this area have now migrated to the term "cilia". The "flagella" of C. reinhardtii are synonymous with the cilia or other eukaryotes and not at all with the flagella of bacteria. Given that this is an enormous contribution of C. reinhardtii research in recent decades, this should be properly developed in the opening paragraph. "Cilia" should then be used throughout the rest of the article. In the Abstract, please also add "biogenesis" or "assembly" (to "structure and function of cilia").
The term "flagella" has been replaced with "cilia" throughout the article, except for "intraflagellar transport", which appears to have been retained in the current literature. The biogenesis of cilia is now mentioned in the abstract. In the Introduction, the importance of ciliary research has been stressed; the relevant section is now: "Studies of C. reinhardtii have enabled numerous landmark discoveries and advances. One remarkable example is the discovery of intraflagellar transport of granule-like particles (Kozminski et al., 1993) and the roles of motors in the process (Prevo et al., 2017 and references therein). Furthermore, structural analyses of wild type and mutants defective for ciliary function/assembly (Goodenough and Heuser, 1985, Silflow and Lefebvre, 2001, Nicastro et al., 2006) have massively contributed to our knowledge of the building blocks of a cilium and their organization and function, and led to classic studies that demonstrated that aberrant ciliary function can cause human genetic diseases such as polycystic kidney disease (Pazour et al., 2000, Li et al., 2004)."
3) Figure 1 is a good addition to the article, yet the two panels are largely unrelated to each other and referenced in distant sections of the text. Please split this figure into two, such that each part can positioned closer to the relevant text for the benefit of the reader. In relation to point above, please also replace uses of "flagellum" with "cilium" etc.
This has been done.
4) While it is included as part of a list, referring to C. reinhardtii as a "preferred" model organism "for dissecting photosynthetic function […]" (Introduction) is somewhat of an understatement. Most of the key components were discovered in Chlamydomonas through the collection of acetate-requiring mutants. These components were subsequently re-discovered in Arabidopsis, often without proper attribution to the original decades-earlier discoveries. The history of the photosynthesis discoveries should not be lost by weak language. Later in the text, the contribution of the Hegemann lab to the field of optogenetics also seems understated with the term "leveraged" (Introduction).
As suggested, we have strengthened the language. The phrasing "preferred model" has been changed into "enormously powerful model" (Introduction). The sentence on optogenetics now reads: "The field of optogenetics experienced a quantum leap with the discovery of C. reinhardtii channelrhodopsins […]". Some other sentences in the Introduction have also been strengthened, too.
5) In the paragraph that begins, "Studies of C. reinhardtii have enabled numerous landmark discoveries" (Introduction), the discoveries chosen are somewhat selective and, in the opinion of the reviewers, some may not be as "landmark" as others that are not cited. The reviewers would encourage the authors to consider which of their mentioned examples represent paradigm shifts or truly big leaps in the understanding of a process. In addition, the reviewers believe that following discoveries merit being considered for this section: the discovery of two related PSII reaction centre proteins (D1, D2); the placement of electron carriers in the Z-scheme by genetic analysis of specific mutants using absorption spectroscopy; and any of the dozens of papers on the centriole or the cell cycle (where C. reinhardtii made significant contributions to the field of cell biology).
The following topics have been added to the section on landmark discoveries and current research areas (last paragraph of Introduction): the discovery of IFT (Kozminski et al., 1993), the elucidation of ciliary structure (Goodenough and Heuser, 1985, Silflow and Lefebvre, 2001, Nicastro et al., 2006), the placement of electron carriers in the Z-scheme (e.g. Gorman and Levine, 1965), the discovery of D1 and D2 (Chua and Gillham, 1977) and their inferred function as reaction centers (Deisenhofer et al., 1985; Trebst, 1986; Satoh et al., 2003), elucidation of the cell cycle (Cross and Umen, 2015) and the structure/function of basal bodies/centrioles (Dutcher and O'Toole, 2016).
6) The reviewers had several other comments related to citations. The authors acknowledge that: "Because of space limitations, we were unable to cite many worthy studies", but the following revisions could help to balance and complete the reference list. These revisions are mentioned below in the order they arise in the manuscript:
6a) There are too many citations for "CRISPR-mediated targeted gene disruptions" (Introduction) compared to all the other technologies in the preceding text.
CRISPR-related references are now cited as follows: "(Ferenczi et al., 2017 and references therein)".
6b) In the section on Genomics, it may be better to cite the revised chloroplast genome published by Gallaher et al., 2017, rather than the earlier 2002 genome. The Gallaher article also includes the transcriptome and updated gene models.
We have kept the original references but added the recent work by Gallaher et al., 2018.
6c) There was a concern that the literature supporting the current section on chemotaxis is limited (subsection “Physiological and metabolic capabilities”). The general feeling was that findings based on, at best, a single published paper were not substantive contributions and did not warrant inclusion.
There are a number of papers on chemotaxis in C. reinhardtii. Chemotaxis to ammonium was described by Byrne et al., (1992); to nitrite by Ermilova and Zalutskaya, (2014); and to nitrate by Ermilova et al., (2009). To cover these three references more accurately, we have changed the citation to "Ermilova and Zalutskaya, 2014 and references therein".
6d) No references are included to support the following statements: "the ability to grow under heterotrophic and fermentative conditions is clearly an adaptation to soil environments" (subsection “Physiological and metabolic capabilities”); "the number of competing algal species in the soil is likely to be fewer than that of lakes" (subsection “Biotic interactions”); "In the soil, ciliates such as Paramecium may represent a typical predator" (subsection “Biotic interactions”); that the palmelloid aggregates are a result of "failure" to hatch (subsection “Biotic interactions”). In each case, if suitable citations cannot be found, please consider softening the claims or providing more rationale for including these claims over alternative explanations (for example, couldn't heterotrophy and fermentation equally be adaptations to lake sediments? Why couldn't the palmelloid aggregates instead result from biofilm formation? Etc.)
These issues have been addressed as follows:
The first sentence has been toned down. It now reads: "The ability of C. reinhardtii to grow under heterotrophic and fermentative conditions might be an adaptation to soil environments […]"
The sentence "the number of competing algal species in the soil […]" has been deleted.
The sentence on predators in the soil has been broadened. It now reads: "In the soil, animals such as earthworms or springtails, and protists are typical predators of microscopic algae (Schmidt et al., 2016, Seppey et al., 2017), but there is currently little specific information on predators of C. reinhardtii."
The sentence on palmelloids has been toned down, and a reference has been added: "These colonies are aggregates of C. reinhardtii that may form as a consequence of… (Khona et al., 2016)."
7) It is worth noting that it is not just the "ability to grow in the dark" (Introduction), but rather to grow in the dark and retain a functional photosynthetic apparatus that made some aspects of the studies possible. Most plants will become yellow in the dark, because there is a photochemical requirement in the chlorophyll biosynthetic pathway. Chlamydomonas (and other algae as well as evergreens) have a second enzyme – protochlorophyllide oxidoreductase – that catalyses the same reaction without a requirement for a photon. This trait is important and should be mentioned in the text.
This aspect has been rephrased as follows: "Another outstanding advantage of C. reinhardtii is its ability to grow in the dark on acetate-containing medium while retaining a functional photosynthetic apparatus, which enabled isolation of photosynthesis mutants even if they are light-sensitive (Levine, 1969, Spreitzer and Mets, 1980)." To keep the Introduction short and crisp, we have not included protochlorophyllide oxidoreductase, which seemed to us beyond the scope of this review.
8) The fact that C. reinhardtii can "form gametes as part of the sexual reproduction cycle" is seemingly put forward as an advantage of this alga as a model organism (Introduction). The text should be revised to make it clearer that this trait is not unique, but rather universal among sexual organisms [which include almost all eukaryotes]). It is also not obvious what the authors mean by the phrase: "which allows for introduction of multiple traits (e.g. mutant alleles) in a single haploid strain". Does it refer to making stable diploids? Or the generating double and triple mutants through classical genetic crosses? Or something else? Please revise to make the meaning clearer.
We have rephrased this sentence as follows: "Furthermore, sexual reproduction can be induced in the laboratory, which allows for the introduction of multiple traits in a single haploid strain (e.g. generating double or triple mutants)." The universality of sex in eukaryotes was not included at this point because the introductory section is quite long already, and we feel it is a general point that most people reading the manuscript will already know.
9) There were some concerns that little is known about the snow algae and that the field remains more confused than this article would suggest (subsection “Habitats and biogeography of C. reinhardtii”). One reviewer noted that Chlamydomonas nivalis is perhaps better considered a cryotolerant mesophile, rather than a strict cryophile/psychrophile (see https://doi.org/10.2478/botcro-2013-0012 and https://doi.org/10.1111/1574-6941.12299), and that it is possibly not even a single species. Overall, the reviewers recommend that the authors acknowledge the confusion here and minimize the discussion, perhaps just to refer that some members of the genus are psychrophilic/cryotolerant and recognized occasionally by the patches of watermelon color on snow. [Notably, the related Chloromonas nivalis is more likely a true psychrophile but, because it belongs to a different genus, it is beyond the scope of this essay].
This statement has been rephrased and is more general. It now reads: "[…] and some members of the genus Chlamydomonas are carotenoid-rich organisms present on the surface of snow, giving it a red appearance (Remias et al., 2005)."
10) Some of the section on deflagellation is counter to the conclusion of the cited Quarmby 2009 chapter (subsection “Physiological and metabolic capabilities”). The potential adaptions discussed in this essay are introduced in the book chapter, but the conclusion of the chapter is that they are less likely than deflagellation being a consequence of pathological over-stimulation of a disassembly pathway. The reviewers request that the authors re-read the chapter, and then revise this section (including its citations) accordingly.
This section has been revised. It now reads: "The biological reason for deciliation is still a mystery, but various hypotheses have been put forth (Quarmby, 2009). Deciliation is observed in a wide range of different cell types; for example, inhalation of irritant chemicals can lead to deciliation of respiratory epithelial cells in mammals (Buckley et al., 1984). Therefore, it seems likely that a predetermined break point is an ancient and inherent property of every cilium (Quarmby, 2009). Consequently, deciliation may not confer a selective advantage, but might be a consequence of pathological conditions that cause over-stimulation of the ciliary disassembly process. On the other hand, the ciliary membrane of C. reinhardtii is in direct contact with the environment (not protected by cell wall) and therefore, deciliation may reduce the entrance of noxious compounds into cells. Deciliation may also allow cells to escape when their cilia are stuck to the surface of a predator (Quarmby, 2009). Studying deciliation in the natural environment holds the promise of new insights into selection pressures that led to its evolution."
11) It would be great if the authors could provide a video of their Figure 2B (like those in https://doi.org/10.1038/s41467-017-01547-8 or similar). These show the wolf pack feeding behavior and the deflagellation response very nicely.
We have now reused the video previously published by Aiyar et al., (2017). This is now Video 1.
12) There was much debate in the reviewer consultation about whether the word "pelagic" can refer to organisms living in freshwater lakes (in part because of its root from the Greek "pelagikos", which relates to the sea). Please revise the text to make it clearer whether sections are referring to oceanic/marine algae or lake-dwelling algae. If "pelagic" is still used, it should be clearly defined for the benefit of unfamiliar readers. Please consider something like "organisms that live in the water column of lakes and oceans, but not on or near the bottom of the lake or ocean".
We have included an explanatory sentence as suggested: "The term 'pelagic zone' refers to the water column of lakes and oceans not on or near the lake or ocean bottom." In several cases, we have clarified that we are referring to the pelagic zone of lakes, not oceans.
13) In the text in Box 1. Taxonomy and laboratory history of C. reinhardtii, please add "from two parents or haplotypes" to the end of the sentence that reads "The sequencing of 39 common laboratory strains show that they fall into five genetically distinct lineages". This should be mentioned; otherwise it may sound like there are 5 haplotypes.
This has been added.
14) The concluding paragraph could be strengthened. For example, the sentence: "It would be interesting to examine the fitness and adaptation of a domesticated laboratory strain to its native environment." Why would this be interesting? Which strain? What questions would be addressed?
This sentence mentioned has been changed. It now reads: "Returning a C. reinhardtii laboratory strain to its native habitat would reveal whether domestication caused it to lose its ability to survive within the dynamic fabric of nature and allow for molecular analyses that could reveal changes in the cells' physiology responsible for the loss of their fitness in natural habitats, as well as other changes that may be associated with adaptation to laboratory conditions." Other parts of the concluding section have been revised as well.
https://doi.org/10.7554/eLife.39233.011Article and author information
Author details
Funding
Deutsche Forschungsgemeinschaft (SFB 1127)
- Severin Sasso
- Maria Mittag
U.S. Department of Energy (DE-FG02-07ER64427)
- Arthur R Grossman
U.S. Department of Energy (DE-FG02-12ER16338)
- Arthur R Grossman
Deutsche Forschungsgemeinschaft (SA 2453/1-1)
- Severin Sasso
National Science Foundation (NSF-MCB 0951094)
- Arthur R Grossman
The funders had no role in study design, data collection and interpretation, or the decision to submit the work for publication.
Acknowledgements
We thank Dr. Thomas Pröschold for helpful comments on this manuscript, Debbie Maizels for preparing Figures 1 and 2, Drs. Santosh Sathe and Pierre Durand for providing Figure 3, and Prasad Aiyar for providing Video 1. Because of space limitations, we were unable to cite many worthy studies relevant to the topic of this article; we apologize to all of our colleagues whose work we did not discuss.
Publication history
- Received:
- Accepted:
- Version of Record published:
Copyright
© 2018, Sasso et al.
This article is distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use and redistribution provided that the original author and source are credited.
Metrics
-
- 54,923
- views
-
- 2,508
- downloads
-
- 193
- citations
Views, downloads and citations are aggregated across all versions of this paper published by eLife.
Citations by DOI
-
- 193
- citations for umbrella DOI https://doi.org/10.7554/eLife.39233







