Response to short-term deprivation of the human adult visual cortex measured with 7T BOLD
Figures
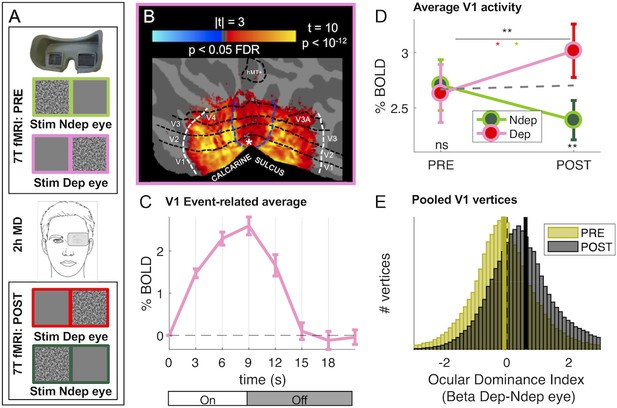
Monocular deprivation modulates 7T BOLD responses in early visual cortex.
(A) Schematic illustration of the methods. The icons show a band-pass noise stimulus shown to either eye through the MR compatible goggles. Before and after the Pre- and Post-deprivation scans, outside the bore, we also measured binocular rivalry. (B) BOLD responses evoked by our band-pass noise stimulus with peak frequency 2.7 cycles per degree (cpd), presented in the deprived eye PRE-deprivation, mapped on the flattened cortical surface, cut at the calcarine sulcus. T-values are obtained by aligning GLM betas for each subject and hemisphere to a left/right symmetric template hemisphere, excluding vertices for which preferred eccentricity was not adequately estimated or smaller than 1 (the same criterion used for al analyses), then evaluating the distribution of betas in each vertex against 0 (one-sample t-test) and FDR correcting across the entire cortical surface. Black dashed lines show the approximate average location of the regions of interest V1 through MT, which were mapped on the individual subject spaces (see Materials and methods); white and blue lines represent the outer limits of the representation of our screen space (24 × 32 deg) and the foveal representation (≤1 deg, where eccentricity could not be mapped accurately) respectively. (C) BOLD modulation during the 3 TRs of stimulus presentation (from 0 to 9 s) and the following four blank TRs, for the 2.7 cpd noise stimuli delivered to the deprived eye before deprivation. The y-axis show the median percent BOLD signal change in V1 vertices relative to the signal at stimulus onset, averaged across subjects. Error bars give s.e. across participants. Note the small between-subject variability of the response (given that the response of each subject was computed for just two blocks of stimulation-blank). D: Average BOLD response to the band-pass noise stimulus with peak frequency 2.7 cpd, in each of the four conditions, computed by taking the median BOLD response across all V1 vertices then averaging these values across participants (after checking that distributions do not deviate from normality, Jarque-Bera hypothesis test of composite normality, all p > 0.06). The top black star indicates the significance of the ANOVA interaction between factors time (PRE, POST deprivation) and eye (deprived, non-deprived); the other stars report the results of post-hoc t-tests: red and green stars give the significance of the difference POST minus PRE, for the deprived and non-deprived eye respectively; bottom black stars give the significance of the difference deprived minus non-deprived eye before and after deprivation. *p < 0.05; **p < 0.01; ***p < 0.001; ns non-significant. E: Histograms of Ocular Drive Index: the difference between the response (GLM beta) to the deprived and non-deprived eye, computed for each vertex, separately before and after deprivation. Yellow and black lines give the median of the distributions, which are non-normal (logistic) due to excess kurtosis.
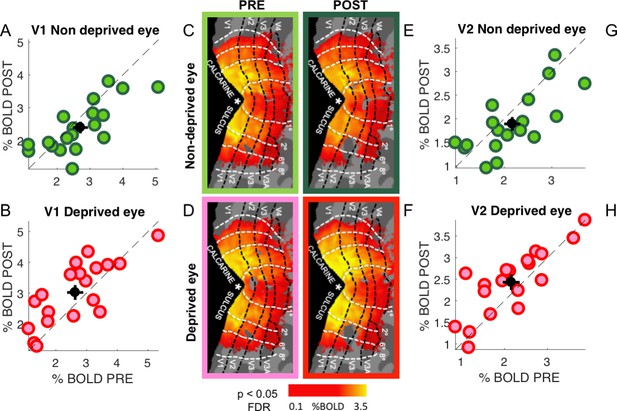
Effects of deprivation across the visual cortex Monocular deprivation had strong and opposite effects on the response to the 2.7 cpd stimulus in the two eyes.
Panels in the top row report responses to the non-deprived eye, those in the bottom row to the deprived eye. The central panels map the average %BOLD response to stimuli presented in either eye, before and after monocular deprivation (isoeccentricty lines are taken from the pRF mapping shown in Figure 5A main text), showing that suppression of the non-deprived eye and enhancement of the deprived eye are largely homogeneous within each ROI. Panel A and G compare % BOLD responses in each of our 19 individual participants (each point shows the average across the two hemispheres), to stimuli in the non-deprived eye, before vs. after deprivation; the majority of points lie below the bisection of the axes, implying a reduction of responses to the non-deprived eye after monocular deprivation. The same comparison for stimuli in the deprived eye in panels B and H shows an increase of BOLD responses: most point lie above the bisection line, implying a boost of responses to the deprived eye after deprivation.
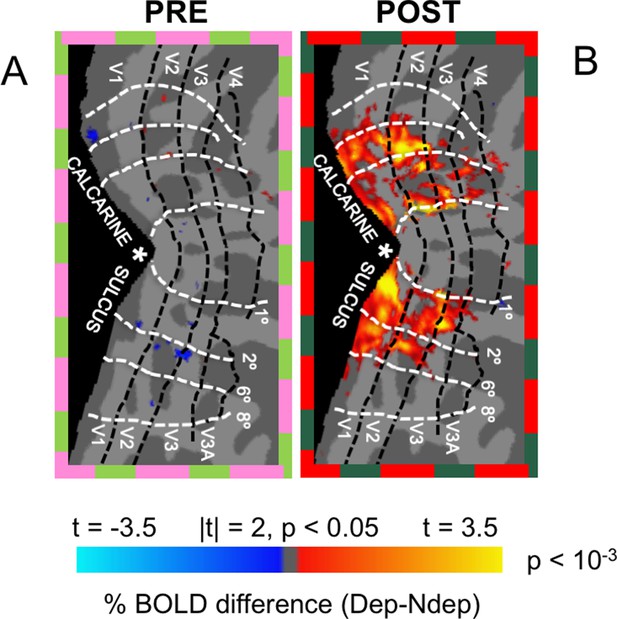
Change of ocular preference after deprivation.
Panels (A) and (B) map the difference of the %BOLD response to the Deprived – Non deprived eye, before and after deprivation, respectively. While there is no organized preference for either eye in any visual area before deprivation, there is a clear preference for the deprived eye after deprivation (the net result of the boost of the deprived eye response and the suppression of the non deprived eye response), which spreads across most of the areas activated by our stimulation.
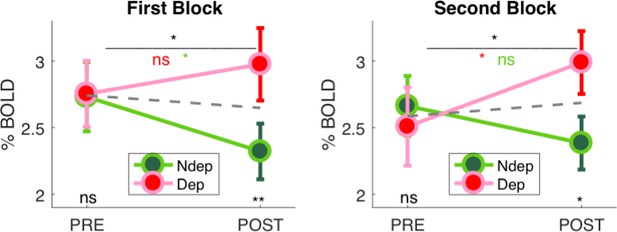
Split-half reliability of the deprivation effect in V1.
Average V1 BOLD response to the band-pass noise stimulus with peak frequency 2.7 cpd, same as in Figure 1D but computed separately for each of the two stimulus repetitions that occurred in each scan (PRE and POST, to either eye). This essentially splits the dataset in half, and we show that both halves reveal a significant interaction between the factors time (PRE, POST deprivation) and eye (deprived, non-deprived; interaction term: first block F(1,18) = 7.53470, p = 0.01332, second block F(1,18) = 7.11116, p = 0.01572).
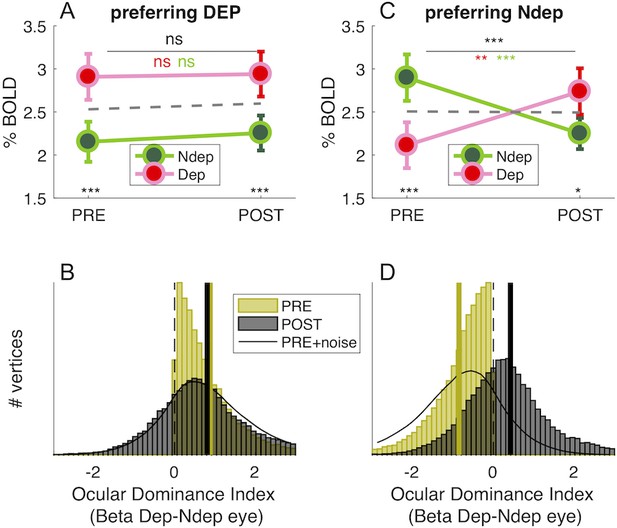
Monocular deprivation shifts 7T BOLD Ocular Dominance in V1.
(A) and (C) Average BOLD responses with the same conventions as in Figure 1D but analysing data from two sub-regions of V1. (A) only vertices that, before deprivation, respond preferentially to the deprived eye. (C) only vertices that, before deprivation, respond preferentially to the non-deprived eye. (B) and (D) Histograms of Ocular Dominance Index (as for Figure 1E), in the two sub-regions of V1, computed before and after deprivation. The black curve simulates the result of adding random noise to the distribution obtained before deprivation; only in B does this approximate the distribution observed after deprivation.
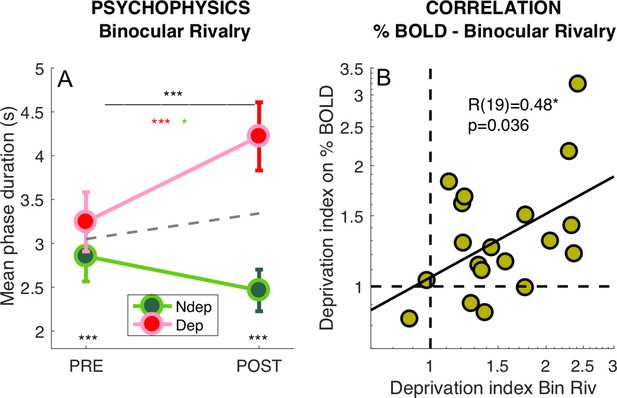
Deprivation effects on BOLD and on psychophysics are correlated.
(A) Effect of deprivation on Binocular Rivalry dynamics. Average phase duration for the deprived and non-deprived eye, before and after deprivation, same conventions as in Figure 1D. Mean phase duration distributions do not deviate from normality (Jarque-Bera hypothesis test of composite normality, all p > 0.171) (B) Correlation between the deprivation index (the POST to PRE- ratio for the deprived eye divided by the same ratio for the non-deprived eye, Equation 6 in Materials and method) computed for the binocular rivalry mean phase duration and for the BOLD response to our band-pass noise stimulus with peak frequency 2.7 cpd. Text insets show the Pearson’s correlation coefficient and associated p-value..
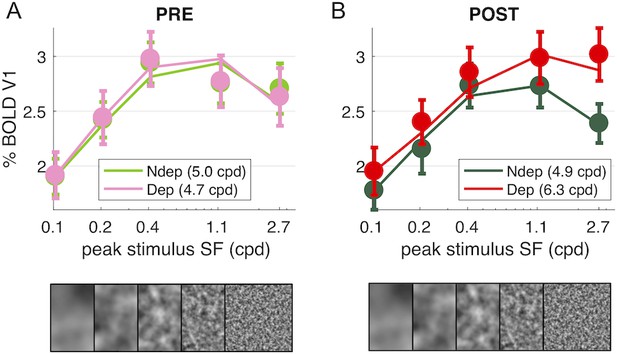
Deprivation affects spatial frequency selectivity in V1.
V1 BOLD responses to all five of our band-pass noise stimuli (with peaks at 0.1, 0.2, 0.4, 1.1 and 2.7 cpd, see spectra in Figure 4—figure supplement 1); (A) response to stimuli in either eye, before deprivation; (B) response to stimuli in either eye, after deprivation. Responses are computed as medians across all V1 vertices (like in Figure 1D), averaged across subjects (error bars report s.e.m.). Continuous lines show the response of the best-fit population Spatial Frequency tuning (with the one parameter, the high spatial frequency cut-off, indicated in the legend), estimated by applying to the average V1 BOLD response the same model used to predict individual vertex responses (fitting procedure illustrated in Figure 5—figure supplement 1)..
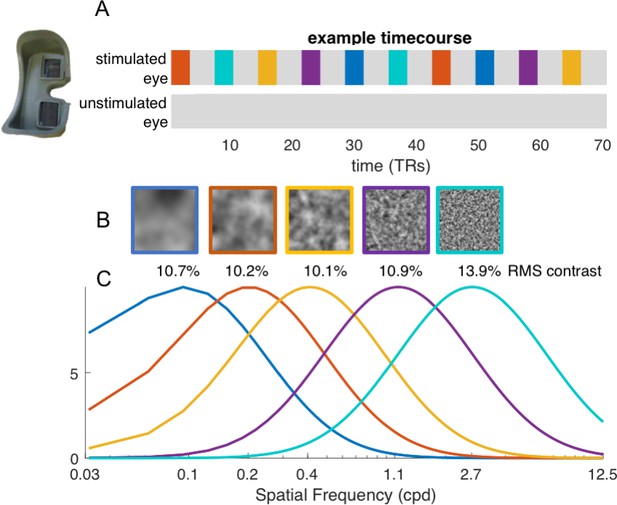
Bandpass noise stimuli.
(A) example time-course of stimulation, showing the blocked presentation of the five spatial frequency stimuli. Blocks were presented in pseudo-random order, twice per run, for a total of 70 TRs. In each run, stimuli were only presented to one eye, while the other was shown a mid-level gray screen. Each eye was tested once, before and after deprivation. (B) example of the 2D bandpass noise stimuli, with their effective RMS contrast. (C) Normalized spectra of the bandpass filter that, multiplied by white noise, generated the five bandpass noise stimuli.
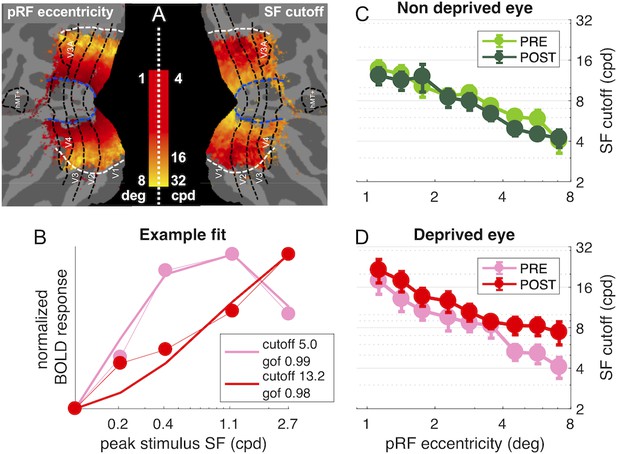
population Spatial Frequency Tuning in V1.
(A) Maps of pRF eccentricity and best fit spatial frequency cut off (for the deprived eye before deprivation) after aligning the parameter estimates for all hemispheres to a common template and averaging them across subjects and hemispheres, after excluding vertices for which the average preferred eccentricity was not adequately estimated or smaller than 1 (the same exclusion criteria used for analyses). (B) Predicted and observed BOLD activity in one example vertex, elicited in response to our bandpass noise stimuli in the deprived eye PRE (pink) and POST deprivation (red), with best fit spatial frequency cut off (reported in the legend). (C-D) Best fit spatial frequency cut-off, averaged in sub-regions of V1 defined by pRF eccentricity bands, and estimated separately for the two eyes and PRE/POST deprivation.
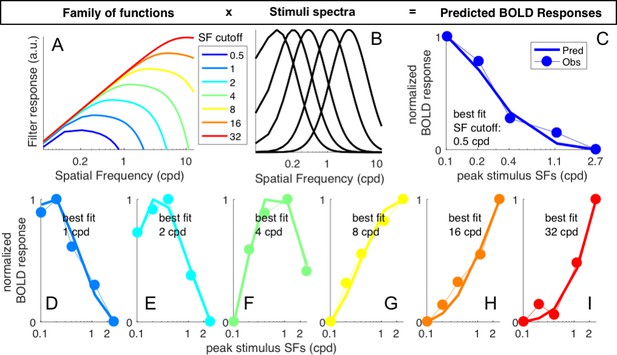
population Spatial Frequency Tuning estimation.
(A) family of functions used to model spatial frequency sensitivity in individual vertices. Different curves are generated by manipulating a single parameter, which is linearly related to the high spatial frequency cut-off of the function. (B) spatial frequency tuning curves in A were multiplied by the five spatial frequency spectra defining our band-pass noise stimuli, yielding a five-element vector that predicts the BOLD response to the stimuli. (C-I) The BOLD response observed in each vertex (or pool of vertices, for Figure 4) was fit with the model, by varying the spatial frequency cut-off and finding the value for which the predicted BOLD response correlates best with the observed BOLD response. For the example vertex in panel (C), the best fit cut-off is 0.5 cpd.; panels (D-I) show individual vertices for which the best fit cut-off is 1, 2, 4, 8, 16 or 32 cpd (see text insets).
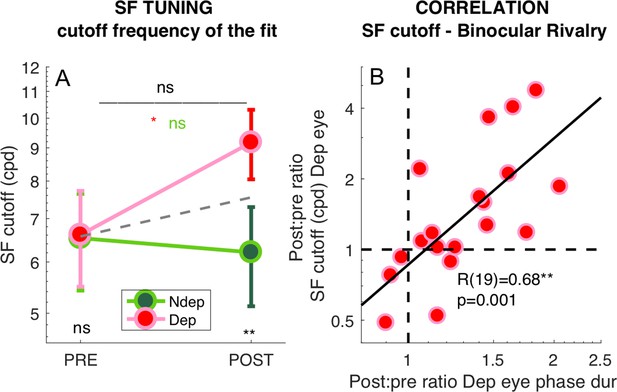
Deprivation effects on the deprived eye population Spatial Frequency Tuning and binocular rivalry phase duration are correlated.
(A) Effect of deprivation on spatial frequency cut off values. Average cut-off across all V1 vertices (pooled across eccentricities) for the deprived and non-deprived eye, before and after deprivation, same conventions as in Figure 1D. Distributions of the log-values do not deviate from normality (Jarque-Bera hypothesis test of composite normality, all p > 0.285). (B) Correlation between the POST/PRE ratio (Equation 7 in the Materials and methods) computed for the binocular rivalry mean phase duration and for the spatial frequency cut off for the deprived eye.
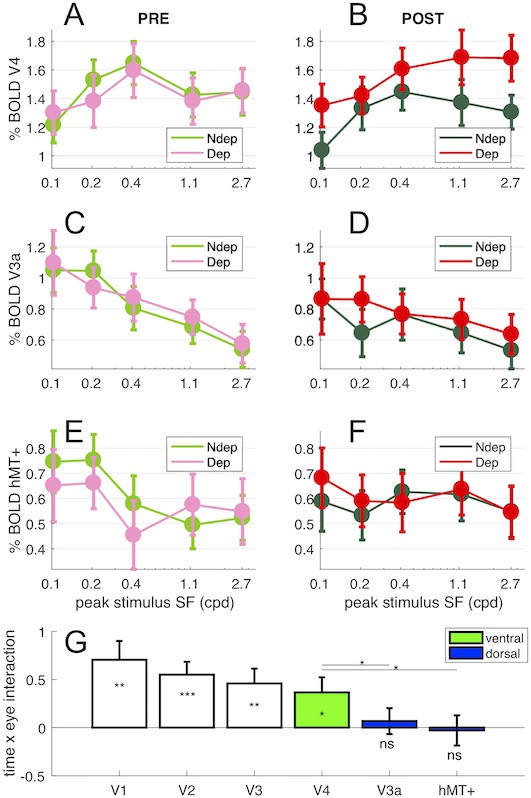
Deprivation effects are stronger in ventral than in dorsal stream areas.
Panels (A-B) show V4 responses across spatial frequency stimuli presented to each eye (colored lines) before (A) and after deprivation; panels (C-D) show V3a responses and panels (E-F) show hMT+ responses. Each data point is computed by taking the median BOLD response across vertices in the region of interest for each stimulus and subject, then averaging across subjects (errorbar report s.e.m.). Panel (G) summarizes the effect of deprivation measured for the highest spatial frequency stimulus in the V1, V2, V3/VP, V4, V3a and hMT+ region of interest, computing the interaction term (POST-PRE difference of BOLD response for the deprived eye, minus the same value for the non-deprived eye) for individual participants and the 2.7 cpd stimulus. Values around 0 indicate no effect of deprivation and values larger than 0 indicate a boost of the deprived eye after deprivation. One-sample t-tests comparing this value against 0 give a p-value equivalent to that associated with the interaction term of the ANOVA (Figure 1D); the significance of the resulting t-value is given by the stars plotted below each errorbar. Stars plotted above the lines show the results of paired t-tests comparing interaction terms in V4 and V3a/hMT+. *** = p < 0.001; ** = p < 0.01; * = p < 0.05; ns = p ≥ 0.05. Green and Blue highlight the assignment of the higher tier areas to the ventral and dorsal stream respectively.
Tables
| Reagent type (species) or resource | Designation | Source or reference | Identifiers | Additional information |
|---|---|---|---|---|
| Software, algorithm | Freesurfer v6.0.0 | (Fischl et al., 2002) | SCR_001847 | |
| Software, algorithm | SPM | (Friston, 2007) | SCR_007037 | |
| Software, algorithm | BrainVoyager | (Goebel et al., 2006) | SCR_006660 | |
| Software, algorithm | FSL | (Jenkinson et al., 2012) | SCR_002823 | |
| Software, algorithm | MATLAB | MathWorks | SCR_001622 | |
| Software, algorithm | PsychToolbox | (Brainard, 1997) | SCR_002881 |
Additional files
-
Transparent reporting form
- https://doi.org/10.7554/eLife.40014.015





