A calcium transport mechanism for atrial fibrillation in Tbx5-mutant mice
Figures
Atrial fibrillation in Tbx5fl/fl;R26CreERT2 mice is associated with altered expression of genes important to cellular calcium handling.
(A) Tbx5fl/fl;R26CreERT2 mice developed spontaneous AF as assessed by surface ECG compared to R26CreERT2. Traces are representative of 15 animals per genotype. (B) Poincaré plot shows irregularly irregular rhythm in Tbx5fl/fl;R26CreERT2, consistent with AF, compared to normal sinus rhythm in R26CreERT2 mice. Poincaré plots are each from one animal, andrepresentative of 15 animals per genotype. (C) Simultaneous AP and [Ca]i recordings show prolonged AP duration and slowed [Ca2+]i transient decay in Tbx5fl/fl;R26CreERT2 atrial cardiomyocytes compared to R26CreERT2. Recordings are representative of simultaneous [Ca]i and Em recordings (myocytes/mice; dual Em and [Ca]i from 5/5 R26CreERT2 and 17/5 Tbx5fl/fl;R26CreERT2, Em only from 23/9 R26CreERT2 and 20/9 Tbx5fl/fl;R26CreERT2, and [Ca]i only from 27/6 R26CreERT2 and 28/6 Tbx5fl/fl;R26CreERT2). (D) Quantitative PCR was performed on RNA isolated from left atrial tissue of 3–5 animals per genotype. mRNA expression of a panel of calcium handling genes potentially important for rhythm regulation was determined. Ryr2 and Atp2a2 expression were decreased and Pln expression was increased in Tbx5fl/fl;R26CreERT2 relative to R26CreERT2 atria. (***p<0.001, **, p<0.01, *, p<0.05).
-
Figure 1—source data 1
qPCR data for Figure 1D.
- https://doi.org/10.7554/eLife.41814.005

PCR primers.
https://doi.org/10.7554/eLife.41814.004
Calcium current blockade dramatically shortened the AP in Tbx5fl/fl;R26CreERT2 atrial cardiomyocytes, consistent with the [Ca]i dependence of AP prolongation following TBX5 loss.
(A) Representative recording of an AP from R26CreERT2 atrial cardiomyocytes before and after 30 µM nifedipine treatment. (B) Representative recording of a Tbx5fl/fl;R26CreERT2 atrial cardiomyocytes before and after nifedipine treatment. (C) Paired APD properties before and after treatment with 30 µM nifedipine (myocytes/mice; n = 8/3Tbx5fl/fl;R26CreERT2 and n = 6/4 R26CreERT2). In R26CreERT2 cardiomyocytes, the effect of nifedipine on APD90 was small, but significant 19 ± 4%. A much larger nifedipine effect was observed in Tbx5fl/fl;R26CreERT2 cardiomyocytes. APD50 decreased by 16 ± 6% and APD90 decreased by 61 ± 6% in the presence of nifedipine. (D) Western blot of atrial tissue in five animals for each genotype showed protein expression for the alpha 1C subunit of the L-type calcium channel (Cav1.2) was unchanged. (normalized to GAPDH) (E,F) Representative ICaL recordings show Peak L-type calcium current was increased in Tbx5fl/fl;R26CreERT2 cardiomyocytes compared to R26CreERT2 (G) Average IV relationship of L-type calcium current (myocytes/mice; n = 22/7 R26CreERT2 and 20/5 Tbx5fl/fl;R26CreERT2). (***p<0.001, **, p<0.01, *, p≤0.05).
-
Figure 2—source data 1
AP and ICaL parameters for Figure 2C,G.
- https://doi.org/10.7554/eLife.41814.010
-
Figure 2—source data 2
Western blot for Figure 2D.
- https://doi.org/10.7554/eLife.41814.011
30 µM Nifedipine blocks L-type calcium current and calcium-induced calcium release in R26CreERT2 and Tbx5fl/fl;R26CreERT2 cardiomyocytes.
https://doi.org/10.7554/eLife.41814.007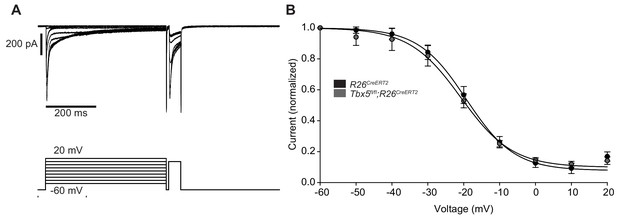
Steady state inactivation of ICaL was unchanged in Tbx5fl/fl;R26CreERT2 atrial cardiomyocytes.
(A) Protocol used to assess steady state inactivation of ICaL (representative trace from Tbx5fl/fl;R26CreERT2 cardiomyocytes) (B) Steady state inactivation of ICaL was the same in R26CreERT2 and Tbx5fl/fl;R26CreERT2 cardiomyocytes. (myocytes/mice; Representative of 4/9 R26CreERT2 and 3/10 Tbx5fl/fl;R26CreERT2).
-
Figure 2—figure supplement 2—source data 1
ICaL parameters for Figure 2—figure supplement 2B.
- https://doi.org/10.7554/eLife.41814.009
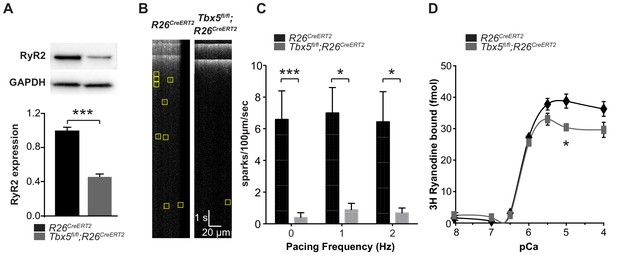
Spark frequency is reduced in Tbx5fl/fl;R26CreERT2 atrial cardiomyocytes.
(A) Western blot from atrial tissue from 10 animals per genotype was used to measure RyR2 expression. RyR2 was significantly decreased in Tbx5fl/fl;R26CreERT2 atria compared to R26CreERT2 atria (normalized to GAPDH). (B) Fluo-4 loaded cardiomyocytes demonstrated reduced spark frequency in Tbx5fl/fl;R26CreERT2 compared to R26CreERT2 atrial cardiomyocytes (representative recordings). (C) Spark frequency was reduced at rest and after steady state pacing at different frequencies (myocytes/mice; n = 12/4 Tbx5fl/fl;R26CreERT2 and n = 12/3 R26CreERT2). (D) Ryanodine binding assay (without normalization) demonstrated no significant difference over the physiologic range of [Ca]i in Tbx5fl/fl;R26CreERT2 compared to R26CreERT2 (Weng et al., 2018). Each measure corresponds to an assay performed on pooled atria from 8 to 10 mice with three independent measures per condition (*p<0.05, **p<0.01, ***p<0.001).
-
Figure 3—source data 1
Western blot for Figure 3A.
- https://doi.org/10.7554/eLife.41814.013
-
Figure 3—source data 2
Spark analysis for Figure 3C.
- https://doi.org/10.7554/eLife.41814.014
-
Figure 3—source data 3
Ryanodine binding assay for Figure 3D.
- https://doi.org/10.7554/eLife.41814.015
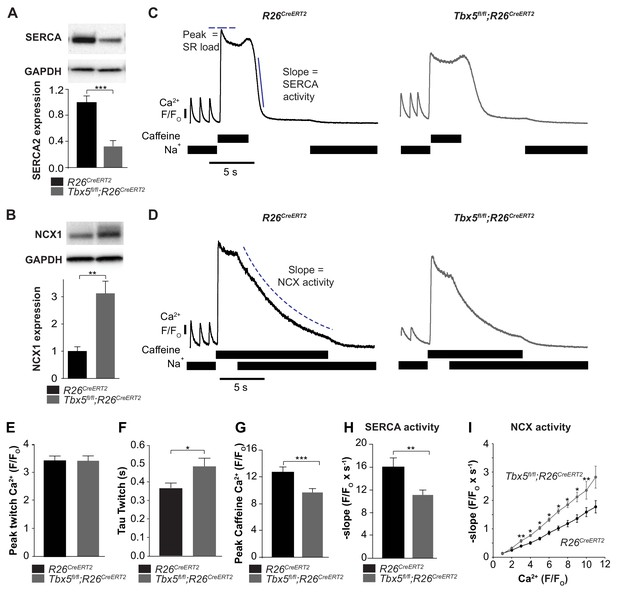
SERCA function is decreased while NCX function is increased in Tbx5fl/fl;R26CreERT2 atrial cardiomyocytes.
(A) Expression of SERCA2 was significantly decreased (normalized to GAPDH) while (B) expression of NCX1 was significantly increased in Tbx5fl/fl;R26CreERT2 atria compared to R26CreERT2 atria as measured by western blot in 10 animals per genotype. (normalized to GAPDH) (C) Application of Na+ free caffeine solution after pacing to steady state at 1 Hz provided a measurement of SR load. In the absence of extracellular Na+, [Ca2+]i plateaued at high levels due to negligible role of non-NCX mediated extrusion in R26CreERT2 and Tbx5fl/fl;R26CreERT2 atrial cardiomyocytes. Removal of caffeine in the absence of external Na+ provided a measure of SERCA mediated SR calcium uptake (representative traces). (D) Restoration of external Na+, in the presence of sustained extracellular caffeine provided a measure of NCX mediated calcium efflux (representative traces). (E) The peak of steady state twitch [Ca2+]i transients was similar but (F) tau of [Ca2+]i decay, determined from twitch [Ca2+]i transients, was increased in Tbx5fl/fl;R26CreERT2 compared to R26CreERT2 cardiomyocytes (myocytes/mice; n = 27/6 R26CreERT2, n = 28/6 Tbx5fl/fl;R26CreERT2). (G) SR load, determined from peak caffeine transients was decreased in Tbx5fl/fl;R26CreERT2 compared to R26CreERT2 cardiomyocytes (myocytes/mice; n = 34/6 R26CreERT2, n = 32/6 Tbx5fl/fl;R26CreERT2). (H) SERCA activity, determined from the maximal rate of calcium decay was diminished in Tbx5fl/fl;R26CreERT2 compared to R26CreERT2 cardiomyocytes (myocytes/mice; n = 29/3 R26CreERT2, n = 32/3 Tbx5fl/fl;R26CreERT2). (I) NCX activity (decay slope), was increased at all levels of calcium in Tbx5fl/fl;R26CreERT2 cardiomyocytes (myocytes/mice; n = 35/3 R26CreERT2, n = 21/3 Tbx5fl/fl;R26CreERT2). (*p<0.05, **p<0.01, ***p<0.001).
-
Figure 4—source data 1
Western blot for Figure 4A,B.
- https://doi.org/10.7554/eLife.41814.019
-
Figure 4—source data 2
Ca2+ handling parameters for Figure 4E–H.
- https://doi.org/10.7554/eLife.41814.020
-
Figure 4—source data 3
NCX activity for Figure 4I.
- https://doi.org/10.7554/eLife.41814.021
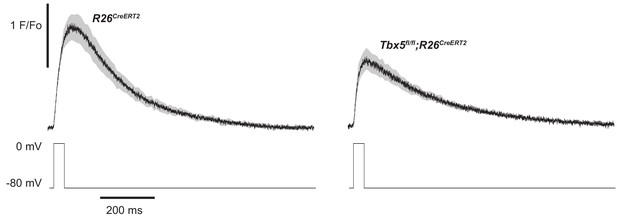
[Ca2+]i transients recorded using 40 ms voltage clamp pulses demonstrate 23 ± 4% (p=0.02) reduction in peak calcium in Tbx5fl/fl;R26CreERT2 compared to R26CreERT2 atrial cardiomyocytes.
While the decay kinetics of Tbx5fl/fl;R26CreERT2 are slowed, there no differences in the initial rate of [Ca2+]i rise (R26CreERT2 rise slope = 107 ± 11 F/Fo*s; Tbx5fl/fl;R26CreERT2 rise slope = 108 ± 18 F/Fo*s, p=0.96). Average of all myocytes is shown in black with grey indicating standard error of mean (myocytes/mice; n = 5/3 R26CreERT2, n = 13/5 Tbx5fl/fl;R26CreERT2).
-
Figure 4—figure supplement 1—source data 1
Ca2+ transients for Figure 4—figure supplement 1.
- https://doi.org/10.7554/eLife.41814.018
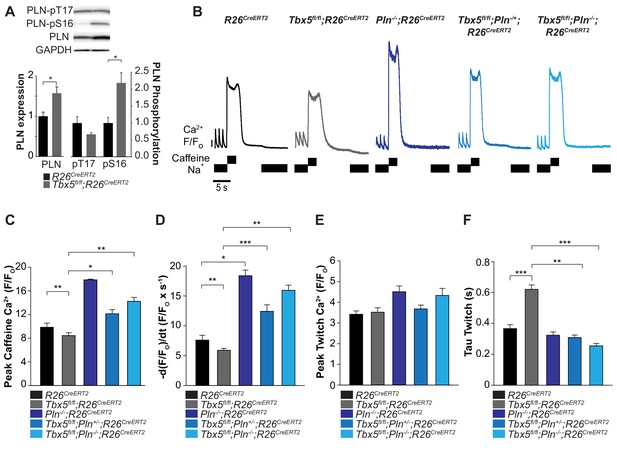
Phospholamban knockout normalized SERCA function in Tbx5fl/fl;R26CreERT2.
(A) PLN expression was increased in Tbx5fl/fl;R26CreERT2 compared to R26CreERT2 as measured by western blot with five animals per genotype. PLN expression was normalized to GAPDH. The proportion of PLN S16, but not T17 phosphorylation was also increased (normalized to PLN). (B) Representative SR load and SERCA measurements in R26CreERT2, Tbx5fl/fl;R26CreERT2, Pln-/-;R26CreERT2, Tbx5fl/fl;Pln-/+;R26CreERT2 and Tbx5fl/fl;Pln-/-;R26CreERT2 atrial cardiomyocytes were collected as described in Figure 4. (C, D) SR load and SERCA function were significantly higher in Tbx5fl/fl;Pln-/+;R26CreERT2 and Tbx5fl/fl;Pln-/-;R26CreERT2 compared to Tbx5fl/fl;R26CreERT2 cardiomyocytes and comparable to R26CreERT2 cardiomyocytes. (E) [Ca2+]i transient peaks were unchanged in Tbx5fl/fl;R26CreERT2, Tbx5fl/fl;Pln-/+;R26CreERT2 and Tbx5fl/fl;Pln-/-;R26CreERT2, but increased in Pln-/-;R26CreERT2 cardiomyocytes. (F) [Ca2+]i transient decay rate in Tbx5fl/fl;Pln-/+;R26CreERT2 and Tbx5fl/fl;Pln-/-;R26CreERT2 cardiomyocytes were normalized to that of R26CreERT2 cardiomyocytes (myocytes/mice; n = 34/3 R26CreERT, n = 36/3 Tbx5fl/fl;R26CreERT2, n = 30/3 Pln-/-;R26CreERT2, n = 21/3 Tbx5fl/fl;Pln-/+;R26CreERT2, n = 27/3 Tbx5fl/fl;Pln-/- atrial cardiomyocytes). (*p<0.05, **p<0.01, ***p<0.001).
-
Figure 5—source data 1
Western blot for Figure 5A.
- https://doi.org/10.7554/eLife.41814.023
-
Figure 5—source data 2
Ca2+ Handling parameters for Figure 5C–F.
- https://doi.org/10.7554/eLife.41814.024
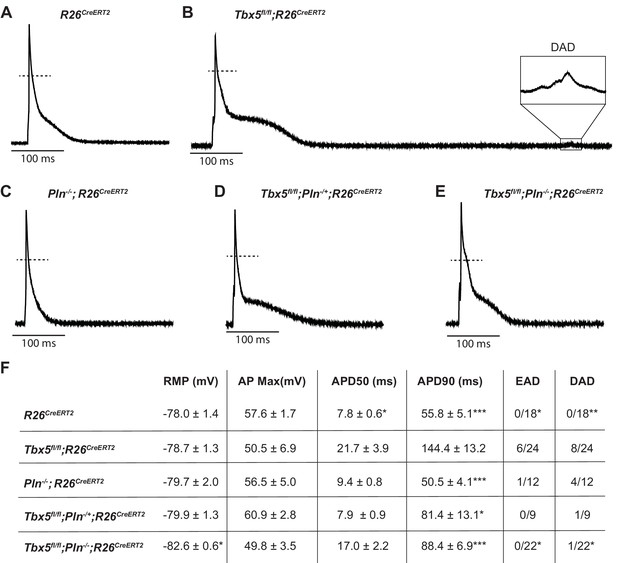
PLN knockout normalized AP duration and prevented triggered activity in Tbx5fl/fl;R26CreERT2.
Representative APs recorded from (A) R26CreERT2, (B)Tbx5fl/fl;R26CreERT2, (C)Pln-/-;R26CreERT2, (D)Tbx5fl/fl;Pln-/+;R26CreERT2, (E)Tbx5fl/fl;Pln-/-;R26CreERT2 atrial cardiomyocytes as described previously in Figure 2. (F) TBX5-loss dependent AP prolongation and frequency of triggered activity was normalized by phospholamban knockout (myocytes/mice: n = 18/7 R26CreERT2, n = 24/12 Tbx5fl/fl;R26CreERT2, n = 12/5 Pln-/-;R26CreERT2, n = 9/3 Tbx5fl/fl;Pln-/+;R26CreERT2, and n = 22/3 Tbx5fl/fl;Pln-/-;R26CreERT2). (*p<0.05, **p<0.01, ***p<0.001).
-
Figure 6—source data 1
AP Parameters for Figure 6F.
- https://doi.org/10.7554/eLife.41814.026
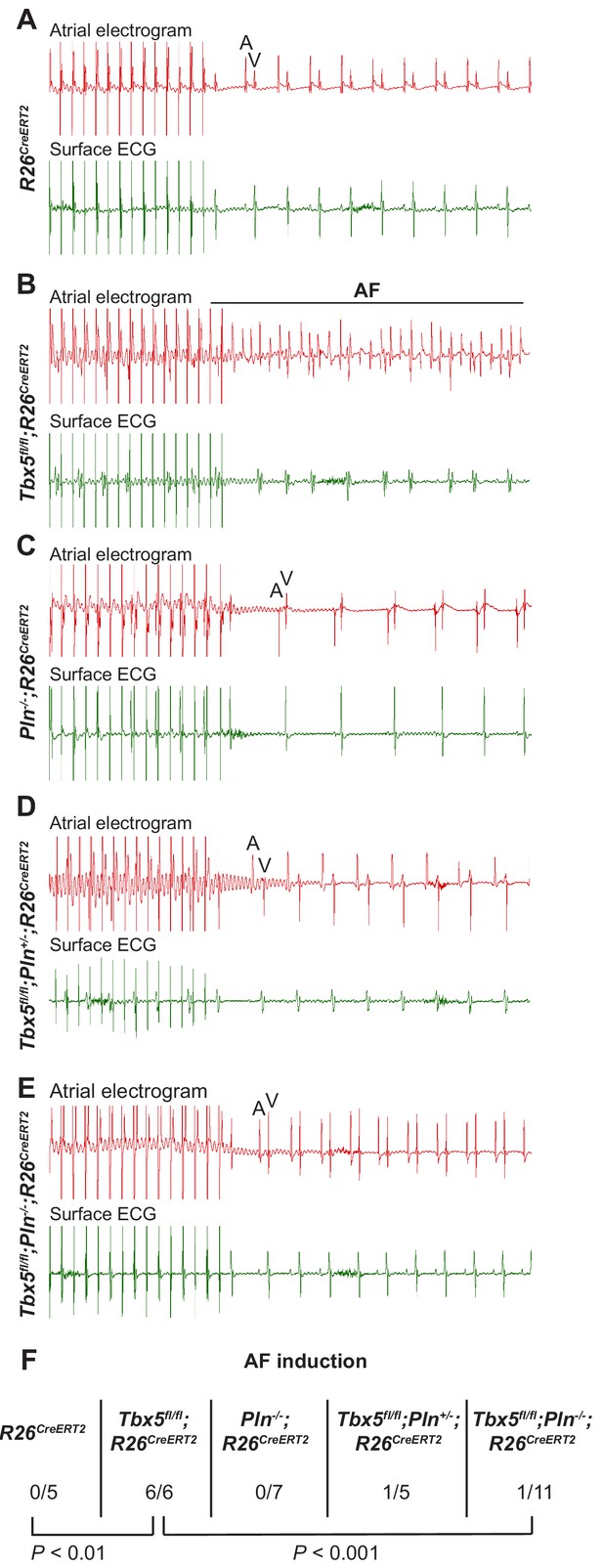
PLN deficiency protected against TBX5-loss associated AF Intra-atrial pacing was used to induce AF.
Representative intracardiac atrial electrogram recordings and corresponding surface ECG are shown from (A) R26CreERT2, (B)Tbx5fl/fl;R26CreERT2, (C)Pln-/-;R26CreERT2, (D)Tbx5fl/fl;Pln-/+;R26CreERT2, (E)Tbx5fl/fl;Pln-/-;R26CreERT2 atrial cardiomyocytes. A, atrial electrical signal; V, far field ventricular electrical signal. (F) AF was reproducibly demonstrated in 6/6 Tbx5 knockouts in contrast to 1/11 Pln/Tbx5 double knockouts, indicating rescue of atrial arrhythmogenesis. P values were determined by Fisher’s exact test (n = 5 R26CreERT2, n = 6 Tbx5fl/fl;R26CreERT2, n = 7 Pln-/-;R26CreERT2, and n = 5 Tbx5fl/fl;Pln-/+;R26CreERT2, n = 11 Tbx5fl/fl;Pln-/- mice).
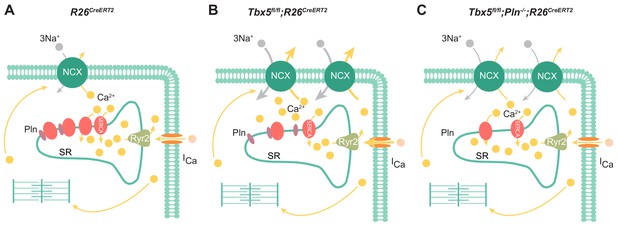
Model of TBX5-dependent calcium regulation in atrial cardiomyocytes.
(A) Excitation-contraction coupling of atrial cardiomyocytes is achieved through regulation of intracellular calcium handling. (B) Adult-specific Tbx5 knockout leads to decreased expression of SERCA2 and increased expression of PLN, leading to decreased SR Ca2+ load. In addition, removal of Tbx5 is associated with increased NCX1 expression and activity, thereby increasing Ca2+ extrusion, which is balanced by increased L-type calcium entry. (C) Combined Tbx5/Pln knockout relieves repression of SERCA2. This results in normalization of SERCA activity and rescue of cardiomyocyte ectopy, triggered activity, and AF observed with Tbx5 deficiency.
Tables
| Reagent type (species) or resource | Designation | Source or reference | Identifiers | Additional information |
|---|---|---|---|---|
| Genetic reagent (M. musculus) | Tbx5fl/fl (Tbx5tm1Jse) | PMID: 11572777, 27582060 | MGI:2387850 | Dr. Jonathan G Seidman (Harvard) |
| Genetic reagent (M. musculus) | Pln-/- (Plntm1Egk) | PMID: 8062415 | MGI:2158357 | Dr. Evangelia Kranias (University of Cincinnati) |
| Genetic reagent (M. musculus) | Rosa26CreERT2(Gt(ROSA)26Sortm1(cre/ERT2)Tyj) | PMID: 17251932, 27582060 | MGI:3790674 | Dr. Tyler Jacks (Massachusetts Insititute of Technology) |
| Antibody | Mouse anti-RyR2 | ThermoFisher | Cat. #: MA3-925 | WB (1:2000) |
| Antibody | Mouse anti-SERCA2 | ThermoFisher | Cat. #: MA3-919 | WB (1:1000) |
| Antibody | Mouse anti-NCX | ThermoFisher | Cat. #: MA3-926 | WB (1:1000) |
| Antibody | Mouse anti-PLN | Badrilla | Cat. #: A010-14 | WB (1:5000) |
| Antibody | Rabbit anti-pT17-PLN | Badrilla | Cat. #: A010-13 | WB (1:5000) |
| Antibody | Rabbit anti-pS16-PLN | Badrilla | Cat. #: A010-12 | WB (1:5000) |
| Antibody | Rabbit anti-Cav1.2 | Alomone | Cat. #: ACC-003 | WB (1:200) |
| Antibody | Mouse anti-GAPDH | Millipore | Cat. #: MAB374 | WB (1:10000) |
| Antibody | Goat anti-mouse-HRP | Thermofisher | Cat. #: 31437 | WB (1:5000) |
| Antibody | Goat anti-rabbit-HRP | Thermofisher | Cat. #: 31463 | WB (1:5000) |
| Chemical compound, drug | Fluo-4 AM | Thermofisher | Cat. #: 14201 | 10 µM x 20 min |
| Chemical compound, drug | Nifedipine | Sigma | Cat. #: N7634 | 30 µM |
| Chemical compound, drug | Collagenase Type 2 | Worthington Biochemical | Cat. # LS004177 | 1 g/L |
| Chemical compound, drug | Tamoxifen | MP Biomedicals | Cat#: 156738 | 2 mg/injection x three doses |
| Chemical compound, drug | Laminin | Invitrogen | Cat. #: 2039175 | 0.5 mg/ml |
| Chemical compound, drug | [3H]ryanodine | PerkinElmer | Cat. #: NET950250UC | |
| Chemical compound, drug | TRIzol | Invitrogen | Cat. #: 15596026 | |
| Chemical compound, drug | Ryanodine | MP Biomedicals | SKU #:0215377001 | |
| Chemical compound, drug | Caffeine | Sigma | Cat. #: C0750 | 10 mM |
| Software, algorithm | Clampex/Clampfit Data acquisition and analysis | Molecular Devices | Version 10.3.2.1 | |
| Software, algorithm | LabChart for electrophysiology studies | ADInstruments | Version 5 and 8 | |
| Software, algorithm | Buffering analyses using MaxChelator | Stanford | WEBMAXCLITE v1.15 | Chris Patton, Stanford University |
| Software, algorithm | Western Blot quantification ImageJ | NIH | Version 1.48 | |
| Software, algorithm | Hierarchical Statistical technique using R | R Core Team | Script from PMID: 29016722 | Ken Macleod, Imperial College London |
| Commercial Assay or Kit | qScript cDNA synthesis kit | Quanta | ||
| Commercial Assay or Kit | Power SYBR Green PCR Master Mix | Applied Biosystems |
Additional files
-
Transparent reporting form
- https://doi.org/10.7554/eLife.41814.030