A mechanistic model for long-term immunological outcomes in South African HIV-infected children and adults receiving ART
Figures
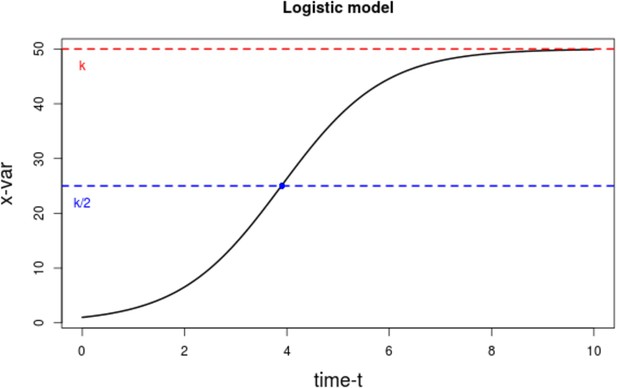
Plot of the logistic growth model.
The dotted red line represents that carrying capacity k, while the dotted blue line is at the inflexion point k/2.
-
Figure 2—source data 1
Data source to reproduce the plot for the logistic growth model.
- https://cdn.elifesciences.org/articles/42390/elife-42390-fig2-data1-v2.txt
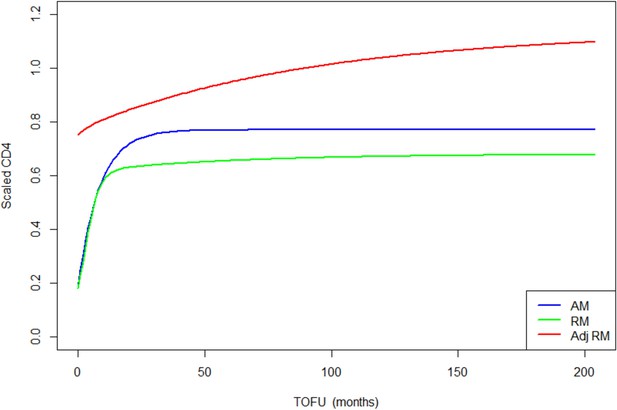
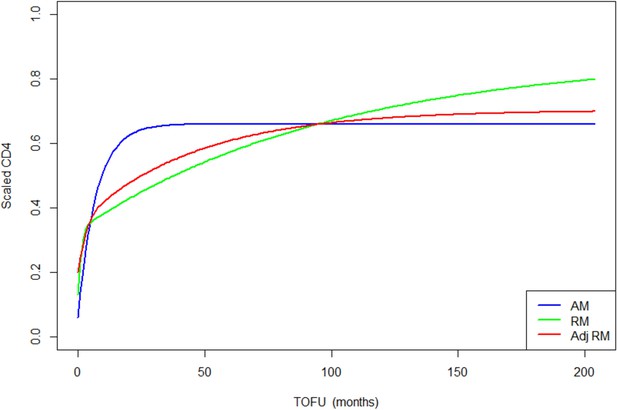
Children population-level CD4 trajectory, as estimated by the unadjusted ratio and asymptotic models, and the adjusted ratio model.
Simulation of population-level CD4+ count trajectory for children, from unadjusted fixed estimates of the asymptotic model (AM) in blue and the ratio model (RM) in green. The red line represents simulation from the adjusted population-level RM estimates. Parameters used for the AM are presented in Supplementary file 3 – Table 1, scenario 1. Those used for the RM are estimated fixed effect for the null model (not shown in the paper): K = 3.4, Q = 0.9, r = 0.35, s = 0.017, z0 = 0.18. Fixed effect presented in Table 3 (scenario 1) are used for the adjusted ratio model (Adj RM), for baseline covariates z-score BMI, age, log viral load; and sex and suppression of viral load within 12 months of starting therapy. Convergence plots for the Adj RM are given in Figure 3—figure supplement 1, and simulation of individual fits in Figure 3—figure supplement 2.
-
Figure 3—source data 1
Data source to reproduce the population-level CD4 trajectories plot for children.
- https://cdn.elifesciences.org/articles/42390/elife-42390-fig3-data1-v2.txt
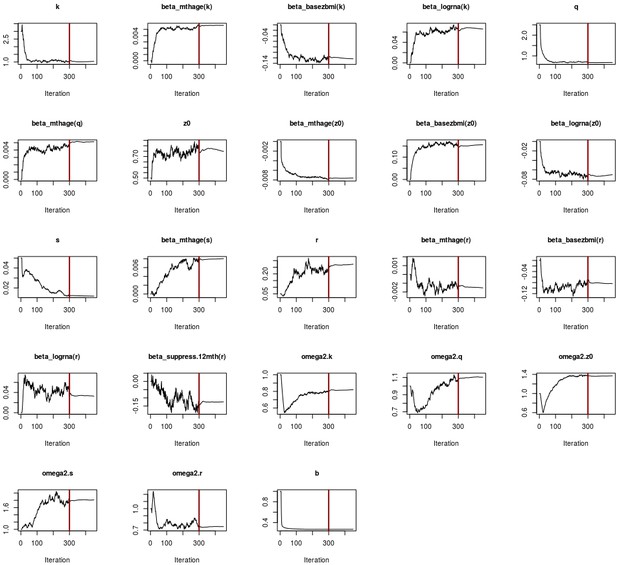
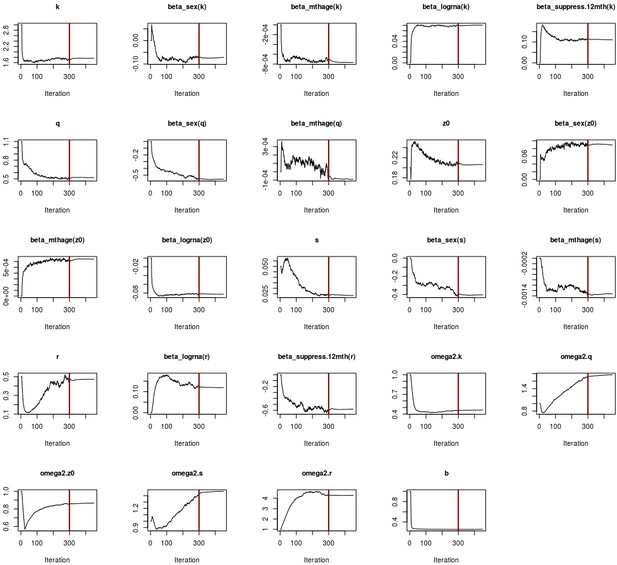
Convergence plots for children adjusted ratio model.
The x-axis represents the number of iterations, while the y-axis is the parameter value. The red vertical line indicates the end of the 300 iterations, where the algorithm explores freely the parameter space. The second phase has 150 iterations, where the step size is gradually decreased in order to ensure convergence.
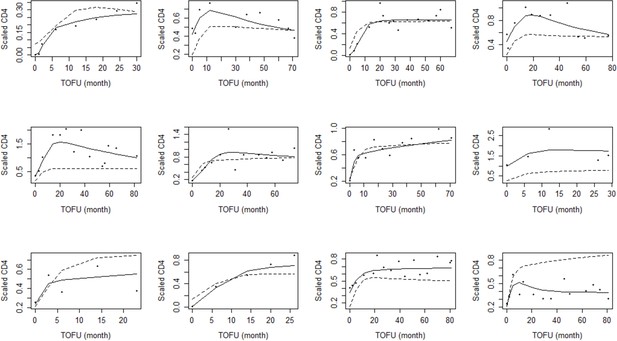
Sample of individual children plots for the adjusted ratio model.
TOFU on the x-axis stands for the time of follow-up since ART initiation. The points are individual scaled CD4+ counts. The dashed line and the solid line represent the population-level and individual-level fits respectively.
-
Figure 3—figure supplement 2—source data 1
Data source to reproduce the individual-level CD4 trajectories plot for children.
Data source to reproduce the individual-level CD4 trajectories plot for adults.
- https://cdn.elifesciences.org/articles/42390/elife-42390-fig3-figsupp2-data1-v2.txt
Adults population-level CD4 trajectory, as estimated by the unadjusted ratio and asymptotic models, and the adjusted ratio model.
Simulation of population-level CD4+ count trajectory for adults, from unadjusted fixed estimates of the asymptotic model (AM) in blue, and the ratio model (RM) in green. The red line represents simulation from the adjusted population-level RM estimates. Parameters used for the AM are presented in Supplementary file 3 – Table 1, scenario 1. Those used for the RM are estimated fixed effect for the null model (not shown in the paper): K = 2.54, Q = 0.38, r = 1.23, s = 0.01, z0 = 0.13. Fixed effect presented in Table 3 (scenario 1) are used for the adjusted ratio model (Adj RM), for baseline covariates sex, age, log viral load, and suppression of viral load within 12 months of starting therapy. Convergence plots for the Adj RM are given in Figure 4—figure supplement 1, and simulation of individual fits in Figure 4—figure supplement 2.
-
Figure 4—source data 1
Data source to reproduce the population-level CD4 trajectories plot for adults.
- https://cdn.elifesciences.org/articles/42390/elife-42390-fig4-data1-v2.txt
Convergence plots for adults adjusted ratio model.
The x-axis represents the number of iterations, while the y-axis is the parameter value. The red vertical line indicates the end of the 300 iterations, where the algorithm explores freely the parameter space. The second phase has 150 iterations, where the step size is gradually decreased in order to ensure convergence.
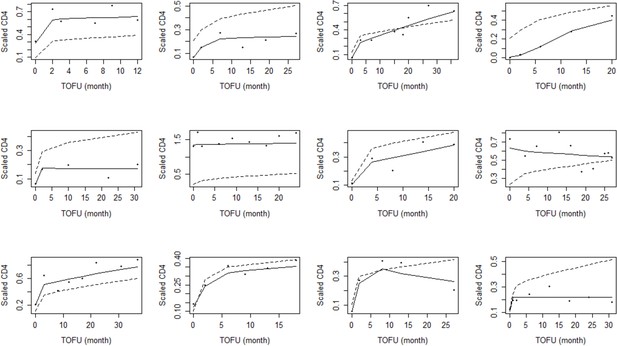
Sample of individual adult plots for the adjusted ratio model.
TOFU on the x-axis stands for the time of follow-up since ART initiation. The points are an individual scaled CD4+ counts. The dashed line and the solid line represent the population-level and individual-level fits, respectively. The table below indicates the clinical details about the patients represented above, in their respective index location.
-
Figure 4—figure supplement 2—source data 1
Data source to reproduce the individual—level CD4 trajectories plot for adults.
- https://cdn.elifesciences.org/articles/42390/elife-42390-fig4-figsupp2-data1-v2.txt
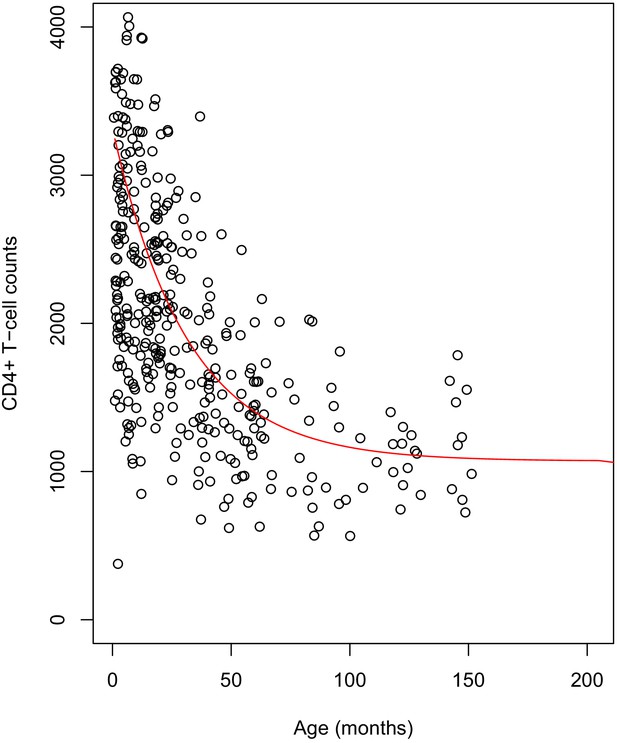
Plot of the simulated reference values for children.
The dots represent the cross-sectional data for healthy children. The fitted red line shows the age-dependent reference values used in the scaling of CD4+ counts of HIV-infected children.
-
Appendix 1—figure 1—source data 1
Simulated reference values for children.
- https://cdn.elifesciences.org/articles/42390/elife-42390-app1-fig1-data1-v2.csv
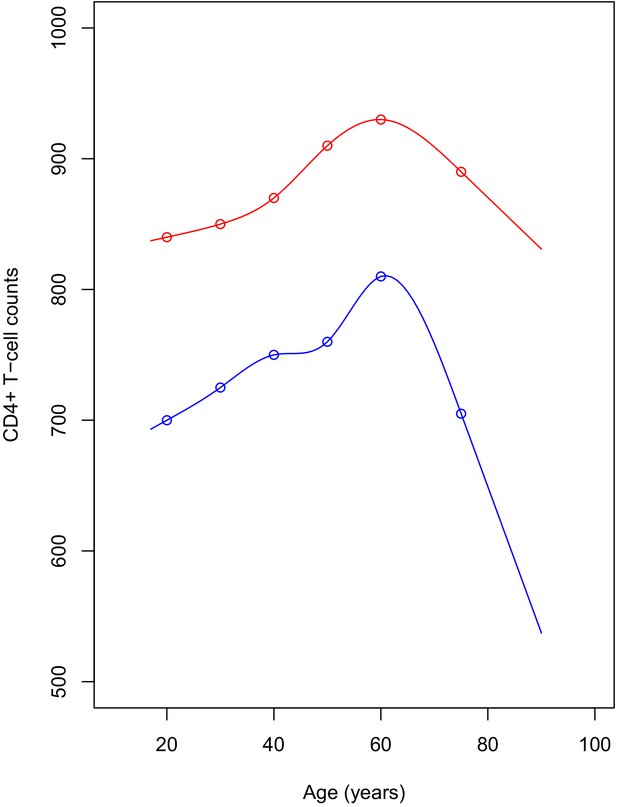
Plot of the simulated reference values for adults.
The points represent the published median values. The red line shows the CD4+ count for women, blue line is for men. CD4+ reference values were simulated yearly, for ages ranging between 17 and 95.
-
Appendix 1—figure 2—source data 1
Simulated reference values for adults.
- https://cdn.elifesciences.org/articles/42390/elife-42390-app1-fig2-data1-v2.csv
Tables
Patient demographics.
| Category | Variable, unit | Category | Full data set | Sample for scenario 1 |
|---|---|---|---|---|
| Children | ||||
| Demographic | Number of patients | All | 19,060 | 1312 |
| Median age, years (IQR) | All | 4.4 (1.1,8.8) | 4.5 (1.4,7.9) | |
| Baseline WHO stage (% relative to all) | Stage I | 693 (3.6%) | 31 (2.3%) | |
| Stage II | 1232 (6.4%) | 108 (8.2%) | ||
| Stage III | 3968 (20.8%) | 614 (46.7%) | ||
| Stage IV | 2756 (14.4%) | 431 (32.8%) | ||
| Median BMI z-scores | All | −0.85 (-2.2,0.2) | −0.76 (-1.98,0.26) | |
| Gender | Female | 9606 (50.4%) | 674 (51.3%) | |
| Male | 9454 (49.6%) | 638 (48.6%) | ||
| Median time on ART, years (IQR) | All | 2.0 (0.0,4.0) | 4.0 (3.0,5.0) | |
| Year of ART initiation (IQR) | 2004–2012 | 2004–2012 | ||
| Clinical characteristics | CD4+ T-cell count at baseline, count/µL (IQR) | 493 (229,890) | 404 (159,706) | |
| Median scaled CD4+ T-cell count at baseline, (IQR) | 0.30 (0.15,1.50) | 0.24 (0.09,0.42) | ||
| Median viral load at baseline, per 1000 copies/mL (IQR) | 150 (20,7342) | 155 (29,670) | ||
| Median log viral load at baseline, copies/mL (IQR) | 5.1 (4.3,6.8) | 5.1 (4.4,5.8) | ||
| Number of patients that suppressed viral load within 12 months of treatment initiation (% relative to non-missing) | Yes | 1673 (26%) | 479 (36.5%) | |
| No | 4764 (74%) | 833 (63.5%) | ||
| Adults | ||||
| Demographic | Number of patients | All | 189,647 | 12,238 |
| Median age, years (IQR) | All | 35 (29,42) | 36 (30.6,42.9) | |
| Baseline WHO stage (% relative to all) | Stage I | 20,711 (10.9%) | 1588 (12.9%) | |
| Stage II | 15,815 (8.3%) | 817 (6.6%) | ||
| Stage III | 42,393 (22.3%) | 4050 (33%) | ||
| Stage IV | 11,466 (6%) | 1336 (10.9%) | ||
| Gender | Female | 124,006 (65.4%) | 6944 (56.7%) | |
| Male | 65,641 (34.6%) | 5294 (43.2%) | ||
| Median time on ART, years (IQR) | All | 1.0 (0.0,3.0) | 3.0 (2.0,5.0) | |
| Year of ART Initiation IQR | 2004–2012 | 2004–2012 | ||
| Clinical characteristics | Median CD4+ T-cell count at baseline, count/µL (IQR) | 140 (67,206) | 128 (62,196) | |
| Median scaled CD4+ T-cell count at baseline (IQR) | 0.18 (0.08,0.26) | 0.16 (0.07,0.24) | ||
| Median viral load at baseline, per 1000 copies/mL (IQR) | 27 (0.8,132.3) | 39 (2.3, 151) | ||
| Median log viral load at baseline, per CD4+ category, copies/mL (IQR) | 4.4 (2.9,5.1) | 4.5 (3.3,5.1) | ||
| Number of patients that suppressed viral load within 12 months of treatment initiation (% relative to relative non missing) | Yes | 10,746 (36.2%) | 5011 (40.9%) | |
| No | 18,945 (63.8%) | 7227 (59.0%) | ||
-
Note: A z-score BMI of −1 indicates that the child’s body mass index is at one standard deviation below the body mass of a healthy child, while a z-score BMI of 0 means that the child has normal body mass for his/her age.
BIC comparison of the unadjusted ratio and asymptotic models, under different scenarios for baseline scaled CD4+ T-cell counts .
| Scenario 1: estimated | Scenario 2: as a predictor | ||||
|---|---|---|---|---|---|
| Ratio model | Asymptotic model | Ratio model | Asymptotic model | ||
| Adults | Sample size | 12,238 | 14,542 | ||
| BIC | −134,016.7 | −126,716.2 | −178,702.3 | −186,244.2 | |
| Children | Sample size | 1312 | 1616 | ||
| BIC | −2,137.1 | −1,523.1 | −6,028.8 | −7,969.7 | |
Ratio model estimated parameters for children and adults.
*** means significant at 99%, and ** significant at 95%.
| Children | Adults | |||
|---|---|---|---|---|
| Model | Scenario 1: estimated (1312 subjects) BIC = −2,457.019 | Scenario 2: as a predictor (1616 subjects) BIC = −6,156.762 | Scenario 1: estimated (12,238 subjects) BIC = −136,284.7 | Scenario 2: as a predictor (14,542 subjects) BIC = −180,557.8 |
| Variable | Estimate (95% CI) | Estimate (95% CI) | Estimate (95% CI) | Estimate (95% CI) |
| Scaled carrying capacity post ART (K) | 1.03 (0.73,1.34) | 1.13 (0.84, 1.41) | 1.75 (1.59, 1.90) | 1.69 (1.57, 1.80) |
| Sex, ref is male | - | - | −0.06 (−0.09,–0.03)*** | - |
| Age, month | 0.005 (0.004, 0.007)*** | 0.0039 (0.002, 0.004)*** | −0.0007 (−0.008,–0.0006)*** | −0.0006 (−0.0007,–0.0004)*** |
| BMI | −0.12 (−0.15,–0.08)*** | −0.09 (−0.12,–0.06)*** | - | - |
| Log viral load | 0.06 (0.04, 0.08)*** | 0.07 (0.05, 0.09)*** | 0.069 (0.065, 0.073)*** | 0.061 (0.058, 0.065)*** |
| Suppress, ref is no | - | 0.001 (−0.096, 0.092)** | 0.11 (0.08, 0.14)*** | 0.12 (0.10, 0.15)*** |
| Scaled carrying capacity healthy individuals (Q) | 0.68 (0.57, 0.78) | 2.02 (1.59, 2.44) | 0.49 (0.47, 0.52) | 0.46 (0.36,0.56) |
| Sex, ref is male | - | - | −0.55 (−0.63,–0.47)*** | −0.54 (−0.63,–0.46)*** |
| Age, month | −0.005 (0.003, 0.007)*** | - | - | 0.0002 (−0.0001, 0.0006) |
| Scaled CD4+ T-cell count at ART initiation () | 0.74 (0.49, 1.00) | - | 0.20 (0.18, 0.22) | - |
| Sex, ref is male | - | - | 0.08 (0.05, 0.12)*** | - |
| Age, month | −0.007 (−0.009,–0.006)*** | - | 0.0006 (0.0004,0.0008)*** | - |
| BMI | 0.15 (0.11, 0.19)*** | - | - | - |
| Log viral load | −0.070 (−0.096,–0.044)*** | - | −0.081 (−0.087,–0.076)*** | - |
| Rate of growth of CD4+ healthy individuals, cells per μL per day (s) | 0.011 (0.007, 0.016) | 0.002 (0.001, 0.003) | 0.022 (0.016, 0.028) | 0.02 (0.01,0.03) |
| Sex, ref is male | - | - | −0.40 (−0.53,–0.28)*** | −0.50 (−0.62,–0.37)*** |
| Age, month | 0.008 (0.004, 0.017)*** | 0.015 (0.010, 0.020)*** | −0.0012 (−0.0017,–0.0007)*** | −0.0016 (−0.0022,–0.0010)*** |
| Rate of growth of CD4+ post ART, cells per μL per day (r) | 0.27 (0.160,0.32) | 0.17 (0.12, 0.21) | 0.49 (0.39, 0.60) | 0.79 (0.60, 0.97) |
| Sex, ref is male | - | −0.19 (−0.33,–0.04)** | ||
| Age, month | −0.001 (−0.003, 0.0001)** | - | - | - |
| BMI | −0.08 (−0.12,–0.04)*** | −0.07 (−0.11,–0.03)*** | - | - |
| Log viral load | 0.03 (0.00, 0.62)** | 0.05 (0.03, 0.08)*** | 0.11 (0.09, 0.13)*** | 0.11 (0.09, 0.13)*** |
| Suppress, ref is no | −0.12 (-−0.27, 0.02) | −0.15 (−0.28,–0.03)*** | −0.57 (−0.70,–0.44)*** | −0.62 (−0.76,–0.47)*** |
Correlations of individual random effects.
Scenario 1 below the diagonal. Scenario 2 above.
| K | Q | s | r | |||
|---|---|---|---|---|---|---|
| Scaled carrying capacity post ART – K | Adults | Scenario 2 | 0.40 | - | 0.48 | −0.28 |
| Children | Scenario 1 | −0.03 | - | 0.57 | 0.01 | |
| Scaled carrying capacity healthy ind – Q | Adults | 0.30 | Scenario 2 | - | −0.41 | |
| Children | 0.10 | Scenario 1 | - | −0.24 | ||
| Baseline scaled CD4+ T-cell count - | Adults | −0.67 | 0.27 | Scenario 2 | - | - |
| Children | −0.85 | 0.38 | Scenario 1 | - | - | |
| Rate of growth of CD4+ healthy ind – s | Adults | 0.36 | −0.30 | Scenario 2 | ||
| Children | 0.45 | −0.52 | Scenario 1 | |||
| Rate of growth of CD4+ post ART – r | Adults | −0.23 | −0.33 | −0.30 | 0.23 | Scenario 2 |
| Children | −0.03 | −0.40 | −0.25 | 0.32 | Scenario 1 |
-
: Opposite direction.
Underlined: Difference of correlation between children and adults.
Sample of simulated healthy children’s CD4+ count values.
| Age (months)/ CD4+ T-cell counts/µL | Age (years)/CD4+ T-cell counts/µL | Age (years)/CD4+ T-cell counts/µL | Age (years)/CD4+ T-cell counts/µL | Age (years)/CD4+ T-cell counts/µL | |||||
|---|---|---|---|---|---|---|---|---|---|
| 0 | 3244 | 1 | 2554 | 5 | 1392 | 9 | 1108 | 13 | 1085 |
| 3 | 3046 | 2 | 2083 | 6 | 1290 | 10 | 1118 | 14 | 1080 |
| 6 | 2866 | 3 | 1761 | 7 | 1220 | 11 | 1102 | 15 | 1077 |
| 9 | 2703 | 4 | 1557 | 8 | 1172 | 12 | 1095 | 16 | 1075 |
Studies that evaluated normal CD4+ T-cells references ranges normal ranges in healthy African adult populations.
| Reference | Value (range) by gender | Country | Age-related comments | |
|---|---|---|---|---|
| Male | Female | |||
| Malaza et al., 2013, IQR | 683 (542–849) | 833 (660–1038) | Durban, South Africa | Median age for women is 35 years vs 23 years for men. CD4+ count increase slightly with age till 64 years old |
| Lawrie et al., 2009, 2.5th and 97.5th percentile | (503–1807) | (561–2051) | Gauteng, South Africa | Average age of 41 years for all |
| Ngowi et al., 2009, mean ± sd | 665.6 ± 246.8 | 802 ± 250.2 | Tanzania | Mean age for women is 30.9 years vs 35.2 for men |
| Lugada et al., 2004, 5th and 95th percentile | 754 (362–1376) | 894 (454–1485) | Uganda | Decrease with age from birth to 18 years. All aged 24 were lumped together |
| Institute of Human Virology/Plateau State Specialist Hospital AIDS Prevention in Nigeria Study Team et al., 2005, mean ± sd | 838 ± 193 | 818 ± 213 | Nigeria | No age effect found |
| Oladepo et al., 2009, median | 746 (351–1455) | 892 (383–1654) | Nigeria | Decreasing with age |
| Asymptote | Log of the rate of increase | Intercept | BIC | |
|---|---|---|---|---|
| Min 3 observations# 17241 adults | 0.632CV 0.70% | 0.071CV 1.88% | 0.142CV 0.77% | -148917 |
| Min 4 observations14,542 | 0.58CV 0.63% | 0.11CV 2.0% | 0.14CV 0.81% | -136244 |
| Min 5 observations12,238 | 0.66CV 0.77% | 0.06CV 2.11% | 0.14CV 0.91% | -126716 |
Additional files
-
Supplementary file 1
Range of the ratio model parameters.
- https://cdn.elifesciences.org/articles/42390/elife-42390-supp1-v2.docx
-
Supplementary file 2
Comparison of parameter estimates for adults, obtained using different fitting scenarios.
- https://cdn.elifesciences.org/articles/42390/elife-42390-supp2-v2.docx
-
Supplementary file 3
Evaluation of the variance–covariance matrix for the ratio and asymptotic models.
- https://cdn.elifesciences.org/articles/42390/elife-42390-supp3-v2.docx
-
Supplementary file 4
Additional estimated parameters.
- https://cdn.elifesciences.org/articles/42390/elife-42390-supp4-v2.docx
-
Transparent reporting form
- https://cdn.elifesciences.org/articles/42390/elife-42390-transrepform-v2.docx
-
Appendix 1—figure 1—source data 1
Simulated reference values for children.
- https://cdn.elifesciences.org/articles/42390/elife-42390-app1-fig1-data1-v2.csv
-
Appendix 1—figure 2—source data 1
Simulated reference values for adults.
- https://cdn.elifesciences.org/articles/42390/elife-42390-app1-fig2-data1-v2.csv