Cisplatin-induced DNA double-strand breaks promote meiotic chromosome synapsis in PRDM9-controlled mouse hybrid sterility
Figures
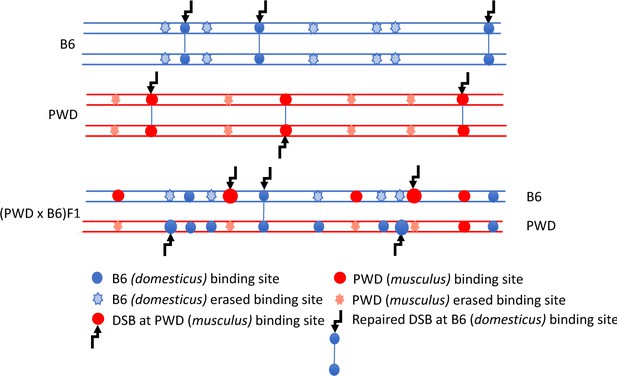
DSB asymmetry model based on historical erosion of PRDM9 binding sites.
A simplified scheme of a pair of homologous chromosomes in PWD (Mus m. musculus) and B6 (Mus m. domesticus) mice and sterile (PWD x B6) intersubspecific male F1 hybrids. Eroded PRDM9B6 binding sites are not recognized or hardly recognized by the PRDM9B6 zinc-finger array in B6 meiosis, but the same sites were saved from erosion during the evolution of the other subspecies. Thus, in (PWD x B6)F1 hybrids PRDM9B6 often binds to the sites on PWD chromosome that are erased on B6 homolog and, vice versa, PRDM9PWD more often binds to the sites on B6 homolog, eroded in PWD. The proportion of such asymmetric sites exceeds 70% of all DSBs in (PWD x B6)F1 hybrid meiosis (Davies et al., 2016) and interferes with chromosome synapsis and meiotic progression. The higher activity of these asymmetric hotspots estimated by DMC1-ChIP-seq is explained by a delay or failure of DSB repair.
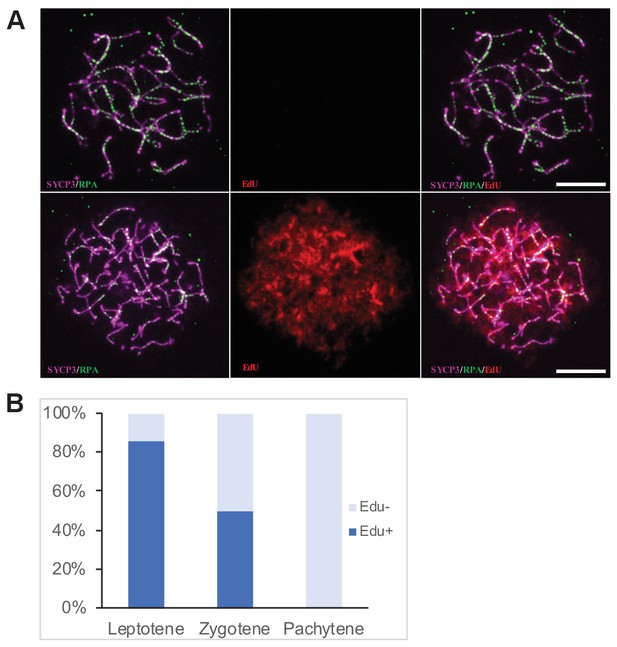
Determination of the cell cycle phase at the time of cisPt injection.
(A) Forty h after EdU and CisPt injection, EdU-negative and positive zygonemas represent cells before and after the premeiotic S phase at the time of injection. Immunostaining of SYCP3 protein (violet) made chromosome axes visible. The RPA foci (green) associate with ssDNA of endogenous, SPO11-induced, and exogenous, cisPt-generated DSBs. Visualization of EdU-labeled DNA is based on the click reaction method (Salic and Mitchison, 2008). Scale bar 10 μM. (B) Proportion of EdU positive cells and EdU-negative cells at three prophase stages 40 hr after EdU treatment of eight males further analyzed in Figures 3 and 4. Numbers of examined cells: leptonemas 126, zygonemas 507, pachynemas 473.
-
Figure 2—source data 1
Distribution of Edu + and EdU- spermatocytes at the first prophase 40 hr after EdU and cisPt injection.
- https://doi.org/10.7554/eLife.42511.004
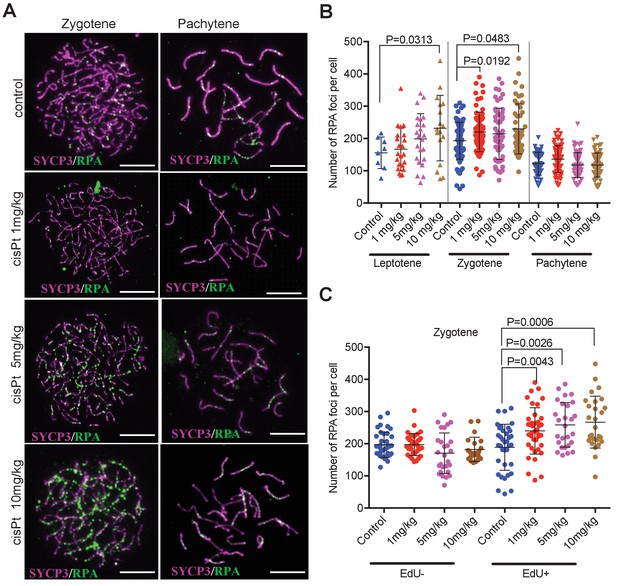
CisPt increases the frequency of exogenous DSBs monitored as RPA foci.
(A) Images of RPA foci during zygotene and pachytene stages of the first meiotic prophase. RPA foci (green) harbored on chromosome axes visualized by immunostaining of SYCP3 protein (violet). Scale bar 10 µM. (B) Numbers of RPA foci per cell 40 hr after CisPt injection. In spite of the large variation of RPA foci between individual cells of the same cohort a significant increase (p<0.05) after cisPt application can be seen in leptotene and zygotene stages, while no indication of RPA foci increase is apparent at pachytene spermatocytes (C). When EdU-positive and -negative zygotene spermatocytes were analyzed separately, the enhancing effect of cisPt on the number of RPA foci was confined to EdU-positive cells. A significant dependence of RPA foci frequency on the dosage of cisPt is shown.
-
Figure 3—source data 1
RPA foci in leptonemas (L), zygonemas (Z) and pachynemas (P) of PBF1 hybrid males treated with cisPt.
- https://doi.org/10.7554/eLife.42511.008
-
Figure 3—source data 2
RPA foci in EdU-negative (E-) and EdU-positive (E+) zygonemas of PBF1 hybrid males treated with cisPt.
- https://doi.org/10.7554/eLife.42511.009
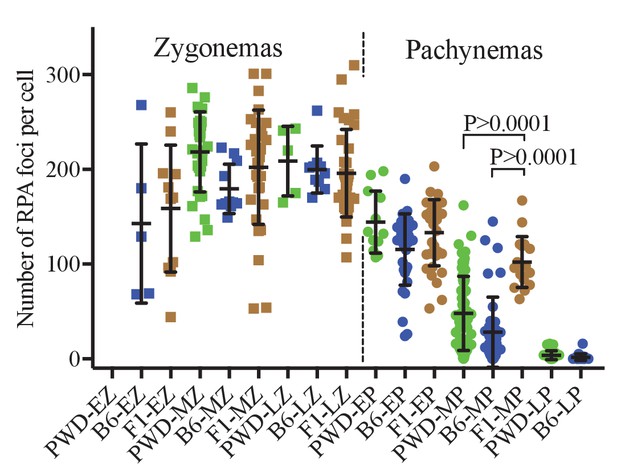
RPA foci in spermatocytes of PWD, B6 and (PWDxB6)F1 males.
The numbers of RPA foci per cell in cisPt untreated males do not show significant differences at early, mid and late (EZ, MZ, LZ) zygotene stage. The extent of DSB repair failure in pachynemas of PBF1 hybrids is reflected by the higher number of RPA foci compared to both parental strains at mid pachytene stage.
-
Figure 3—figure supplement 1—source data 1
RPA foci in spermatocytes of PWD, B6 and PBF1 males.
- https://doi.org/10.7554/eLife.42511.007
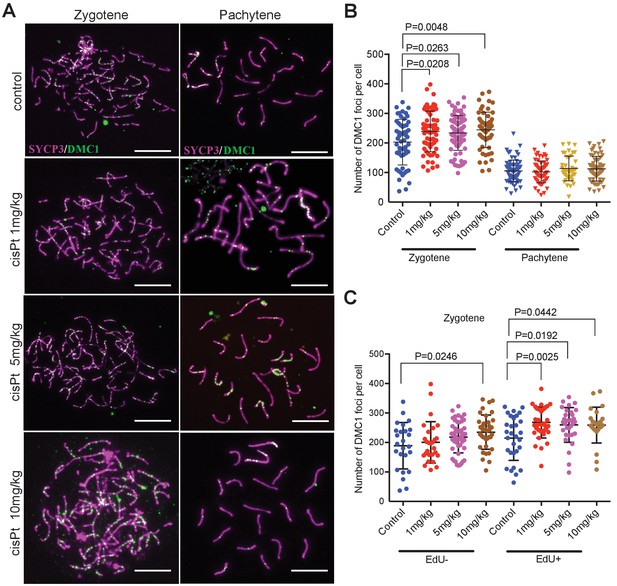
CisPt increases the frequency of exogenous DSBs monitored as DMC1 foci.
(A) Images of DMC1 foci (green) during zygotene and pachytene stages of the first meiotic prophase. Scale bar 10 µM. (B) The numbers of DMC1 foci 40 hr after CisPt injection increase in a dose-dependent manner in zygonemas but do not change in spermatocytes at the pachytene stage. (C) The enhancing effect of cisPt on the number of DMC1 foci at the zygotene stage is barely significant in EdU-negative cells but detectable at all three cisPt doses in EdU-positive zygonemas.
-
Figure 4—source data 1
DMC1 foci in zygonemas (Z) and pachynemas (P) of PBF1 hybrid males treated with cisPt.
- https://doi.org/10.7554/eLife.42511.013
-
Figure 4—source data 2
DMC1 foci in EdU-negative (E-) and EdU-positive (E+) zygonemas of PBF1 hybrid males treated with cisPt.
- https://doi.org/10.7554/eLife.42511.014
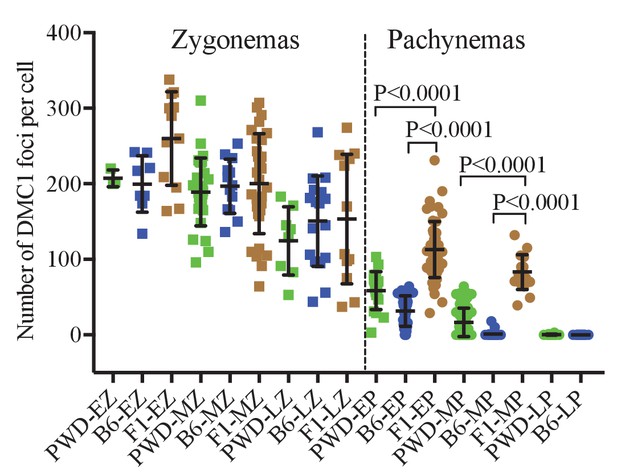
DMC1 foci in spermatocytes of PWD, B6 and (PWDxB6)F1 males.
The numbers of DMC1 foci per cell in cisPt untreated males do not show significant differences at the zygotene stage. The extent of DSB repair failure in early and mid-pachynemas of PBF1 hybrids is reflected by significant persistence of DMC foci compared to both parental strains.
-
Figure 4—figure supplement 1—source data 1
DMC1 foci in spermatocytes of PWD, B6 and PBF1 males.
- https://doi.org/10.7554/eLife.42511.012
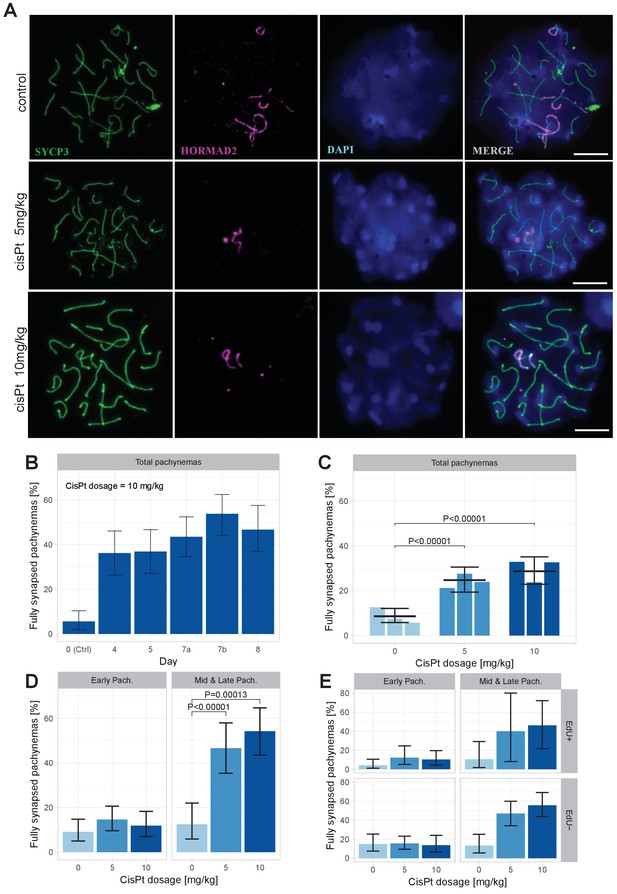
CisPt supports full synapsis of homologous chromosomes at the pachytene stage.
(A) Examples of control and cisPt-treated pachynemas 8 days after cisPt injection. Unsynapsed parts of X and Y chromosomes (5 and 10 mg cisPt/kg) together with unsynapsed autosomal axes (control) were visualized by anti-HORMAD2 antibody (violet). Axial elements of unsynapsed chromosomes and lateral elements of synaptonemal complexes were decorated by anti-SYCP3 antibody (green) and DNA painted by DAPI. The displayed spermatocytes are at early (control and 5 mg/kg) and late (10 mg/kg) pachytene stage. Scale bar 10 µM. (B) Frequency of fully synapsed pachynemas +-S.E. (based on GLMM model), after a single dose of 10 mg/kg of cis Pt; a pilot experiment. Treated males were sacrificed from day 4 to day eight after injection. Each column represents a single male. (C) CisPt dosage-dependent improvement of meiotic chromosome synapsis. Eight days after cisPt injection the percentage of fully synapsed pachynemas significantly increased after cisPt treatment (based on GLMM model and Tukey’s post-hoc test). (D) The effect of cisPt on meiotic synapsis is apparent in mid and late pachytene stages. (E) The meiotic synapsis is slightly enhanced in EdU-negative pachynemas. See text for details.
-
Figure 5—source data 1
The effect of cisPt (10 mg/kg) on chromosome synapsis at pachytene in sterile PBF1 hybrid males 0 to 8 days after treatment.
Pilot experiment, one male per column.
- https://doi.org/10.7554/eLife.42511.016
-
Figure 5—source data 2
Evaluation of meiotic chromosome synapsis at pachytene stage in PBF1 males treated with cisPt.
Syn = apparently complete synapsis of all chromosomes. Asyn = one or more pairs of homologous chromosomes are asynapsed.
- https://doi.org/10.7554/eLife.42511.017
Tables
| Reagent type (species) or resource | Designation | Source or reference | Identifiers | Additional information | |
|---|---|---|---|---|---|
| Strain, strain background (Mus m. domesticus) | C57BL/6J | The Jackson Laboratory | Stock No: 000664 | Black 6 | Laboratory inbred strain, predominantly of Mus m. domesticus origin | |
| Strain, strain background (Mus m. musculus) | PWD/Ph | Institute of Molecular Genetics, ASCR, Prague | N/A | Wild-derived inbred strain of Mus m. musculus origin | |
| Antibody | anti SYCP3 (mouse monoclonal) | Santa Cruz Biotechnology | Santa Cruz: sc-74569; RRID:AB_2197353 | (1:50) | |
| Antibody | anti HORMAD2 (rabbit polyclonal) | gift from Attila Toth | N/A | (1:700) | |
| Antibody | anti HORMAD2 (rabbit polyclonal , C-18) | Santa Cruz Biotechnology | Santa Cruz:sc-82192; RRID:AB_2121124 | (1:500) | |
| Antibody | Anti RPA (rabbit polyclonal) | gift from Willy M. Baarends | N/A | (1:150) | |
| Antibody | Anti DMC1 ((rabbit polyclonal) | Santa Cruz | Santa Cruz: SC-22768; RRID:AB_2277191 | (1:300) | |
| Antibody | anti-rabbit IgG - AlexaFluor568 (goat polyclonal) | Molecular Probes | Molecular Probes: A-11036; RRID:AB_10563566 | (1:500) | |
| Antibody | anti-mouse IgG - AlexaFluor647 (goat polyclonal) | Molecular Probes | Molecular Probes: A-21235; RRID:AB_141693 | (1:500) | |
| Other | normal goat serum from healthy animals | Chemicon | Chemicon: S26-100ML | ||
| Commercial assay or kit | Base-click EdU IV Imaging kit 555S | Baseclick | BaseClick: BCK-EdU555 | ||
| Chemical compound, drug | cisplatin | Sigma-Aldrich- Merck | Sigma-Aldrich: C2210000 | 1, 5, or 10 mg/kg | |
Additional files
-
Transparent reporting form
- https://doi.org/10.7554/eLife.42511.018





