Multi-enhancer transcriptional hubs confer phenotypic robustness
Figures
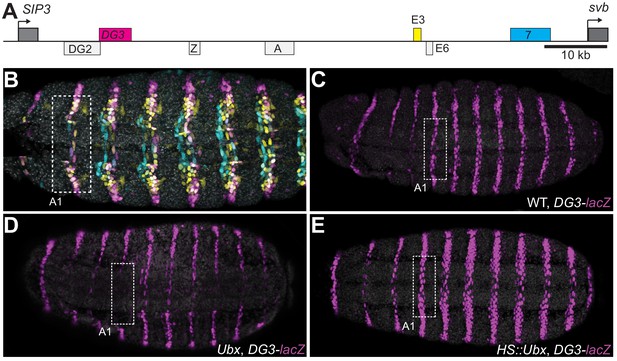
Ubx drives the expression of the DG3 shavenbaby enhancer along the ventral abdominal segments.
(A) The cis-regulatory region of the shavenbaby (svb) gene contains three enhancers expressing stripes on the ventral side of the abdominal segments: DG3, E3 and 7. (B) The expression patterns of the three enhancers are partially overlapping. The color scheme corresponds to (A), where DG3 is magenta, E3 is yellow and 7 is cyan. (C) Expression pattern of a reporter construct with the DG3 enhancer driving LacZ expression in an embryo with wild-type Ubx expression, as visualized using immunofluorescence staining. (D) DG3 reporter in Ubx null mutant shows no expression in A1 and significantly weakened expression in the other abdominal segments. (E) Overexpression of Ubx driven through a heat shock promoter induces overexpression of DG3 reporter in all abdominal segments and ectopic expression on the ventral surface of the thoracic segments T2 and T3.
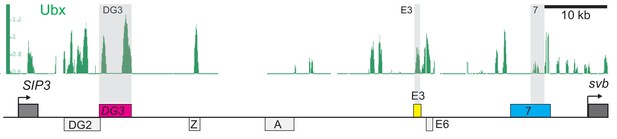
Ubx enrichment around the three ventral svb enhancers.
ChIP experiment of whole embryos between stages 10 and 12 targeting Ubx shows different enrichment profile around the three ventral svb enhancers: DG3, E3 and 7.
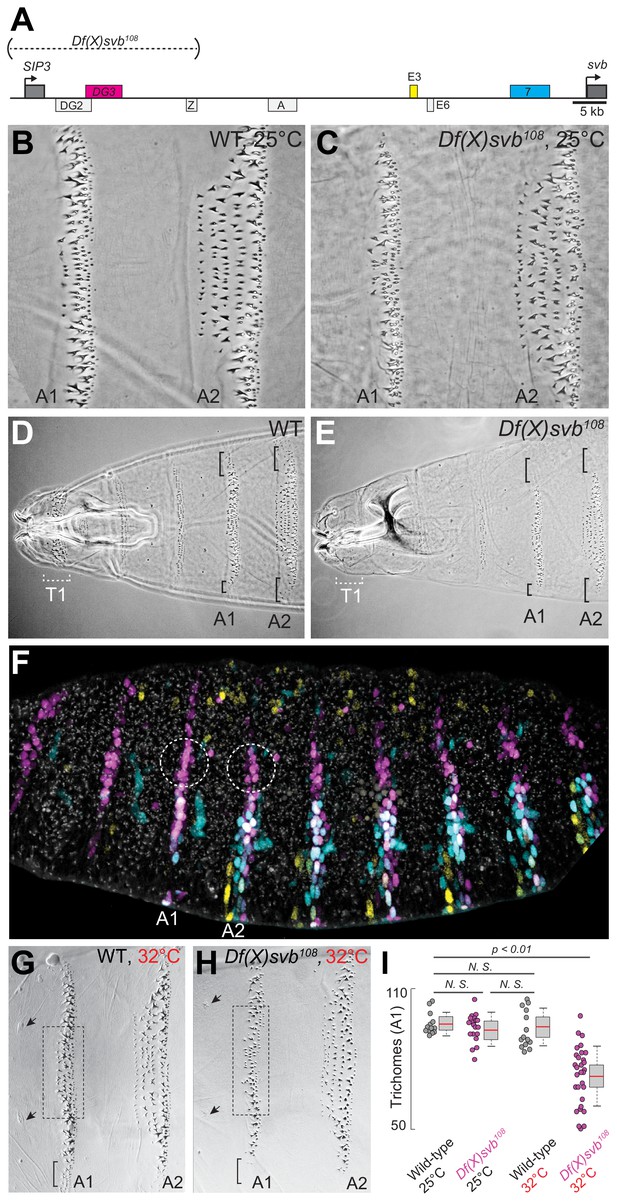
Deletion of a region from the svb locus containing DG3 reduces ventral trichome numbers under heat-induced stress.
(A) The Df(X)svb108 allele contains a deletion in the cis-regulatory of svb spanning three enhancers: DG2, DG3 and Z. Of those, only DG3 expresses on the ventral side. (B) Wild-type phenotype of trichomes along the A1 and A2 segments at 25°C. (C) At 25°C, the Df(X)svb108 deletion allele did not show a mutant phenotype along the A1 and A2 segments. (D) Zoomed out shot of the anterior region of a cuticle preparation of a wild-type (w1118) larva. (E) Zoomed out shot of the anterior region of a cuticle preparation of a larva carrying Df(X)svb108. The lack of trichomes along the T1 segment is a recessive marker used in subsequent experiments to select for embryos/larvae carrying this deletion allele. Even at 25°C, where the overall trichome numbers in A1 and A2 for the Df(X)svb108 deletion mutant is indistinguishable from wild-type svb, the trichomes at the side of the ventral stripe for A1 and A2 were lost, as marked by the black brackets. (F) Within the overall expression pattern of svb enhancers, DG3 provides exclusive coverage in the circled regions in segments A1 and A2. These regions correspond to the black brackets in panels D and E. (G) Wild-type phenotype of trichomes along the A1 and A2 segments at 32°C. (H) At 32°C, the Df(X)svb108 deletion allele showed a mutant phenotype. (I) A1 trichomes in the dashed boxes bounded by the two sensory cells, as indicated by the arrows, as shown in panels G and H, were counted. Deficiencies of the deletion allele only become clear when the animal is subjected to elevated temperature at 32°C, showing reduced trichome numbers in the A1 segment. The number of larvae counted was: 13 for wild-type at 25°C, 19 for Df(X)svb108 at 25°C, 14 for wild-type at 32°C and 30 for Df(X)svb108 at 32°C. Two-tailed t-test was applied for each individual comparison. In box plots, center line is the mean, upper and lower limits are standard deviation and whiskers show 95% confidence intervals.
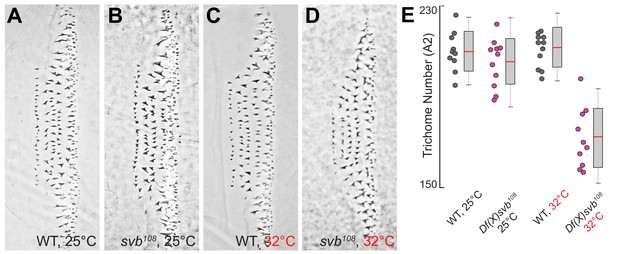
Loss of trichomes in the A2 segment.
(A–C) Phenotype of trichomes along the A2 segment for wild-type and Df(X)svb108 (shown as svb108 in the panels) at 25°C and wild-type at 32°C. (D) At 32°C, the Df(X)svb108 deletion allele showed a mutant phenotype. (E) Deficiencies of the deletion allele become clear when the animal is subjected to elevated temperature at 32°C, showing reduced trichome numbers in the A2 segment (p-value<0.001). The number of larvae counted was: 10 for wild-type at 25°C, 11 or Df(X)svb108 at 25°C, 11 for wild-type at 32°C and 10 for Df(X)svb108 at 32°C. In box plots, center line is mean, upper and lower limits are standard deviation and whiskers show 95% confidence intervals.
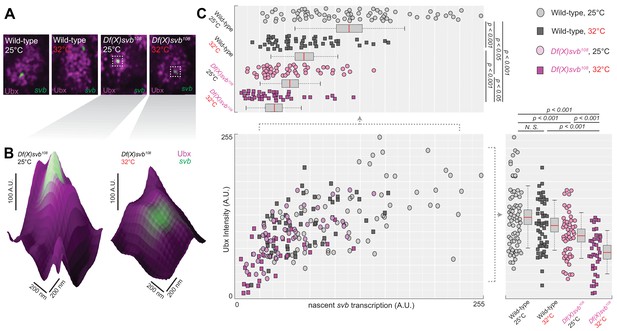
Deletion of the cis-region of svb containing DG3 led to defects in the Ubx microenvironment and svb transcriptional output.
(A) Panels showing a nucleus from embryos with either the wild-type (w1118) or Df(X)svb108 deletion svb allele at either at normal (25°C) or elevated temperature (32°C), imaged using confocal fluorescence microscopy. Ubx (shown in magenta) is stained using immunofluorescence (IF) and the svb transcription sites (shown in green) are stained using fluorescence in situ hybridization (FISH). (B) Zoomed-in panes centered on svb transcription sites, with the height of the surface plots representing the Ubx intensity. (C) Correlation between Ubx and svb transcription intensities at different conditions. The number of transcription sites quantified was: 71 for wild-type at 25°C, 51 for wild-type at 32°C, 50 for Df(X)svb108 at 25°C and 38 for Df(X)svb108 at 32°C. Bottom right panel: Integrating the Ubx intensity surrounding transcription sites shows a small defect in the Ubx concentration around the deletion allele. The drop in Ubx increased at elevated temperature. Upper left panel: The integrated intensity of svb transcriptional output shows that there is a drop in transcriptional output for the deletion allele compared to the wild-type at both 25°C and 32°C. Even the wild-type showed reduced transcriptional output at elevated temperature (32°C). We analyzed four embryos for each genotype/temperature combination. Two-tailed t-test was applied for each individual comparison. In box plots, center line is mean, upper and lower limits are standard deviation and whiskers show 95% confidence intervals.
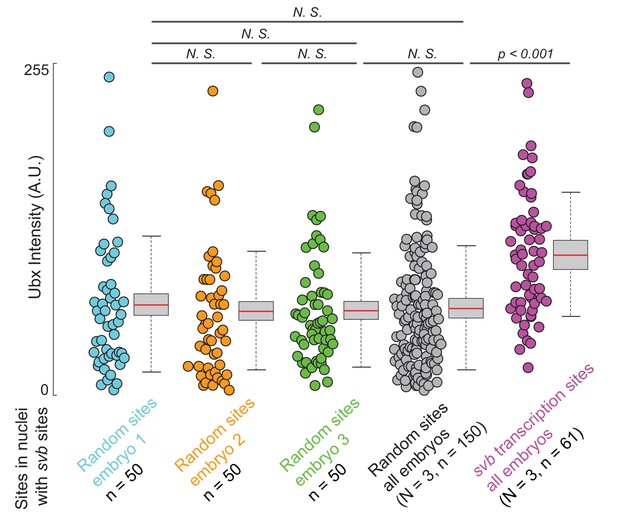
Ubx signal intensities in nuclei outside of transcription sites are consistent.
Fifty random locations are selected from nuclei with svb transcription sites from three wild-type (w1118) embryos raised at 25°C used for analysis in Figure 3 and the Ubx intensity around these sites are measured using the same circle ROI. The Ubx intensity distributions from these sites in each individual embryo (cyan, orange and green) and with all three embryos pooled together (gray) are similar. The average Ubx intensity from actual svb transcription sites (purple, 61 sites) in the same three embryos is higher compared to randomly selected sites, consistent with our previous findings (Tsai et al., 2017). Two-tailed t-test was applied for each individual comparison. In box plots, center line is mean, upper and lower limits are standard deviation and whiskers show 95% confidence intervals.
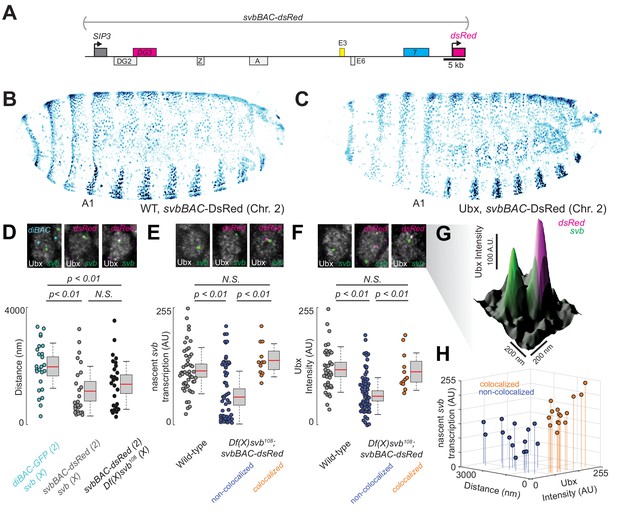
Introduction of the cis-regulatory region of svb on another chromosome rescues the microenvironment deficiencies of the Df(X)svb108 deletion mutant.
(A) The svbBAC construct encompasses the entire cis-regulatory region of svb, driving the expression of dsRed. (B and C) The svbBAC driving the expression of DsRed inserted into the second chromosome drives a similar expression pattern as the wild-type svb locus and responds similarly to Ubx. (D) In nuclei having both svb (for both the wild-type and the Df(X)svb108 allele) and svbBAC-dsRed transcription sites at 32°C, the distances between them areon average closer than between that of svb and a reporter construct of an unrelated gene, diachete (diBAC-gfp, at 25°C), inserted into the same location as svbBAC on the second chromosome. The pairs of distance quantified were: 25 between diBAC-gfp and wild-type svb, 25 between svbBAC-dsRed and wild-type svb and 26 between svbBAC-dsRed and Df(X)svb108. (E) The svb FISH intensity (representing transcriptional output) in Df(X)svb108 x svbBAC-dsRed embryos at 32°C recovered to wild-type levels when the svb transcription site is close to a dsRed transcription site (colocalized). FISH intensity of svb in the same embryos in nuclei without a dsRed transcription site or where svb and dsRed transcription sites were not near each other (non-colocalized) did not recover. The center image panel was stained for dsRed but the particular nucleus does not show dsRed signal. The number of transcription sites quantified was: 49 for wild-type, 53 for Df(X)svb108 not near a dsRed transcription site (including cells where there were no dsRed transcription sites) and 12 for Df(X)svb108 near a dsRed transcription site. (F) At 32°C, Ubx concentration around svb transcription sites recovered to wild-type levels in nuclei containing colocalized svb and dsRed transcription sites (colocalized) in Df(X)svb108 x svbBAC-dsRed embryos. Ubx levels around transcription sites of svb in the same embryos in nuclei without a dsRed transcription site or where svb and dsRed transcription sites were not near each other (non-colocalized) did not recover. The number of transcription sites quantified was: 38 for wild-type, 60 for Df(X)svb108 not near a dsRed transcription site (including cells where there were no dsRed transcription sites) and 12 for Df(X)svb108 near a dsRed transcription site. Two-tailed t-test was applied for each individual comparison. In box plots, center line is mean, upper and lower limits are standard deviation and whiskers show 95% confidence intervals. (G) A surface plot (the height representing Ubx intensity) showing two svb transcription sites in a nucleus, with the one on the right overlapping with a svbBAC-dsRed transcription site and showing higher Ubx concentration. (H) At 32°C, in nuclei having both svb (from the Df(X)svb108 allele) and svbBAC-dsRed transcription sites, the distances between them are plotted against svb FISH intensity and Ubx intensity. There are two clusters separated by a threshold of 360 nm in distance. Co-localized pairs below this distance threshold present higher intensities for both Ubx intensity and nascent svb transcription. The pairs quantified were 15 for colocalized and 14 for non-colocalized between svbBAC-dsRed and Df(X)svb108. We analyzed four embryos for diBAC-gfp x w1118, four embryos for svbBAC-dsRed x Df(X)svb108 and for five embryos for svbBAC-dsRed x w1118.
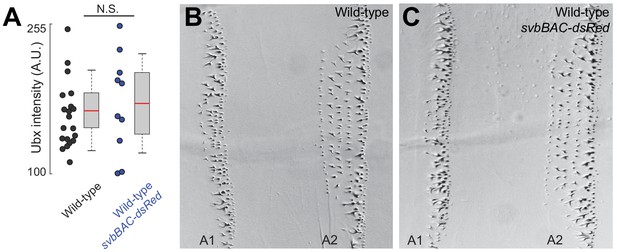
Introduction of svbBAC-dsRed to wild-type (w1118) does not change Ubx microenvironment and phenotype.
(A) Ubx concentrations around svb transcription sites in the ventral region of the A1 segment in wild-type (w1118) x svbBAC-dsRed embryos developed at 32°C did not change compared to wild-type. The number of quantified svb transcription sites was: 20 for wild-type and 10 for wild-type x svbBAC-dsRed. Two-tailed t-test was applied for each individual comparison. We analyzed three embryos for each genotype. In box plots, center line is mean, upper and lower limits are standard deviation and whiskers show 95% confidence intervals. (B and C) The trichome phenotype along A1 and A2 did not change with the introduction of svbBAC-dsRed to wild-type (w1118).
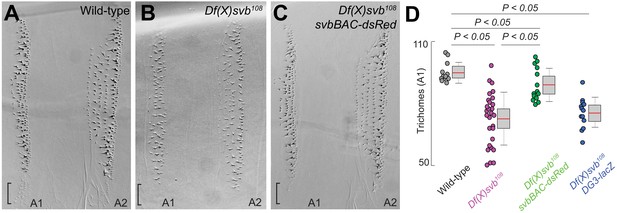
The complete cis-regulatory region of svb rescues trichome number.
(A–C) Cuticle preparations from larvae developed at 32°C with wild-type svb, Df(X)svb108 and Df(X)svb108 x svbBAC-dsRed. The bracket at the edge of the A1 stripe marks a region where trichome growth is exclusively covered by DG3, which disappeared with the deletion of DG3 and did not recover with the introduction of svbBAC-dsRed. (D) The trichome number in larvae developed at 32°C with Df(X)svb108 partially recovered to wild-type levels with the introduction of svbBAC-dsRed. The number of larvae counted was: 12 for wild-type svb, 28 for Df(X)svb108, 14 for Df(X)svb108 x svbBAC-dsRed and 13 for Df(X)svb108 x DG3-lacZ. Two-tailed t-test was applied for each individual comparison. In box plots, center line is mean, upper and lower limits are standard deviation and whiskers show 95% confidence intervals.
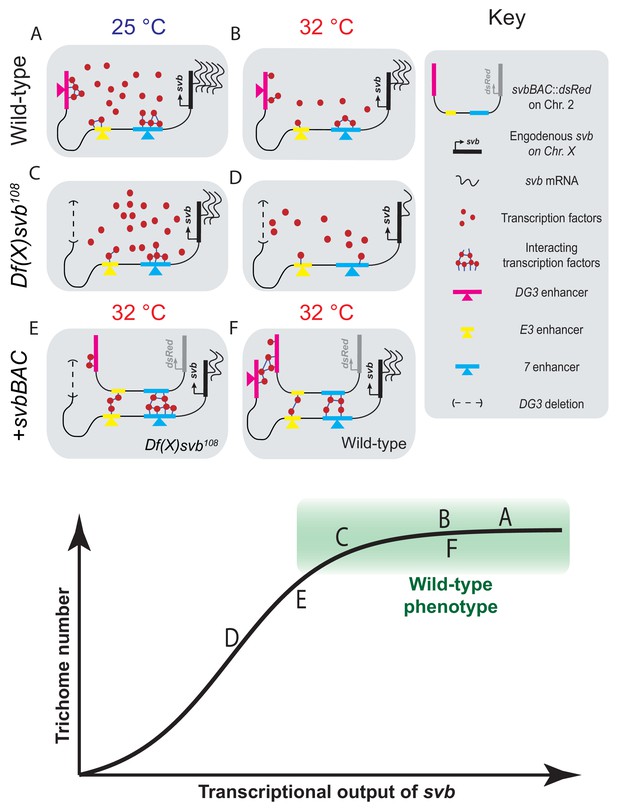
Summary of model including a sigmoidal relationship between svb transcriptional output and the number of cells fated to become trichomes.
(A–F) Schematic representations of the various genotypes, temperatures and rescue conditions as tested in this work. Bottom panel: Schematic of the proposed sigmoidal relationship between svb transcriptional output and the number of cells fated to become trichomes. Note that A and C are at 25°C and the rest are at 32°C. The different conditions we have studied are represented by letters A-F along the sigmoidal curve. The part of the curve corresponding to wild-type phenotype is shaded in green.
Additional files
-
Supplementary file 1
Primers for RNA-probe generation Sequences of primers for amplification of DNA to be used for generation of antisense RNA-probes.
The targets - reporter construct RNA (lacZ, dsRed, gfp) and first intron and second exon (16 kb) of svb - are indicated in the left column. Sequences are indicated for forward or reverse primers of each pair. Reverse primers include a T7 sequence for transcription with T7 RNA polymerase.
- https://doi.org/10.7554/eLife.45325.012
-
Transparent reporting form
- https://doi.org/10.7554/eLife.45325.013





