TRPC3 is a major contributor to functional heterogeneity of cerebellar Purkinje cells
Figures
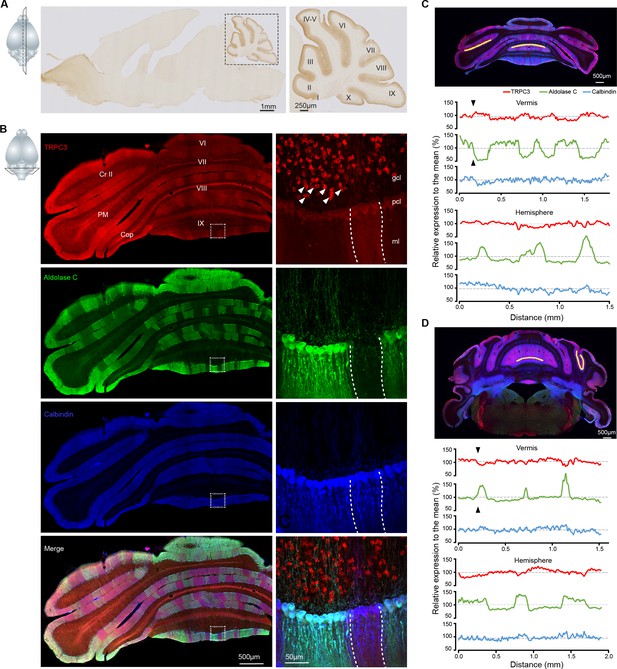
TRPC3 is predominantly expressed in the cerebellum in a zebrin-related pattern.
(A) Representative image and magnification (right) of sagittal cryosection of an adult mouse brain stained with anti-TRPC3. Inset, plane of section. (B) Coronal immunofluorescence images with anti-TRPC3 (red), anti-Zebrin II/Aldolase C (green) and anti-calbindin (blue) staining of the cerebellar cortex (left), with magnifications (right). TRPC3 is expressed in the cerebellar PCs and UBCs (triangles), in a pattern that in the vermis complements that of zebrin and appears more uniform in the hemispheres. Inset, plane of section. (C) Posterior coronal section of the cerebellar cortex (top) used to performed a quantification of the relative intensity of immunofluorescence staining of TRPC3, Zebrin II/Aldolase C and calbindin for PCs in the vermis (ventral lob. VIII, middle) and the hemisphere (ventral PM, bottom) (values normalized to the respective means). (D) Similar analysis of dorsal lob. III (middle) and sulcus of Sim to Crus I (bottom) in anterior section (top). TRPC3 expression is largely complementary to Zebrin II in the vermis and parts of the hemispheres (black arrow heads), but more uniform in other hemispheric areas. In general, TRPC3 expression demonstrates a weaker differentiation between low and high levels than Zebrin II. I-X, cerebellar lobules I-X; Sim, Simplex lobule; Cr II, Crus II; PM, paramedian lobule; Cop, Copula Pyramidis; gcl, granule cell layer; pcl, Purkinje cell layer; ml, molecular layer; D, dorsal; V, ventral; M, medial; L, lateral.
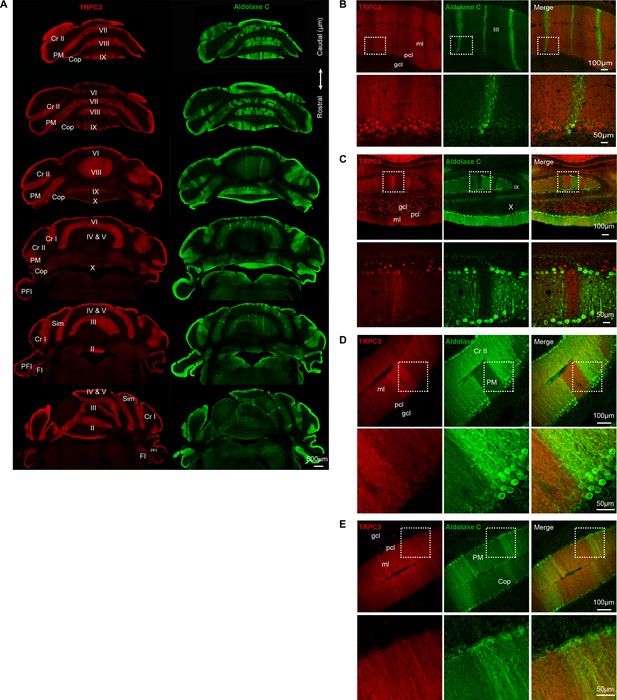
Overview and local patterns of TRPC3 expression.
(A) Confocal immunofluorescent images of coronal sections of wild-type mouse cerebellar cortex, from rostral to caudal, stained with anti-TRPC3 (red) and anti-Zebrin II/Aldolase C (green). TRPC3 is expressed in evident parasagittal bands, and is complementary to the expression of Zebrin II/Aldolase C (left) in the vermis, but more uniform in the hemispheres. (B–D) Immunofluorescent images of coronal sections of wild-type cerebellum, illustrating the distribution of TRPC3 in the anterior vermis (B), posterior vermis (C) and hemispheres (D–E). Demarcated areas in top rows are magnified underneath. Note that TRPC3 immunoreactivity is moderately higher in the Z– PCs than that in the Z+ PCs in the vermis. In the hemispheres the TRPC3-labeled bands are less well defined and can either be complementary to (D) or virtually indistinguishable from (E) Zebrin II bands. Cr II, Crus II; PM, paramedian lobule; Cop, copula of the pyramis; Sim, simple lobule; PFL, paraflocculus; FL, flocculus; ml, molecular layer; gcl, granule cell layer; pcl, Purkinje cell layer.
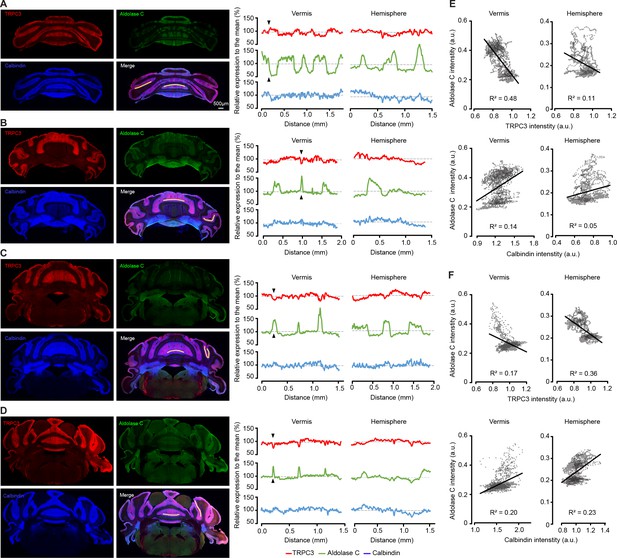
Quantification of TRPC3 expression compared to Zebrin II and calbindin.
(A–D) Left, individual confocal immunofluorescent images of anti-TRPC3, anti-Zebrin II/Aldolase C and anti-calbindin staining, with the quantified section demarcated in the merged image. Examples (A) and (C) are similar to Figure 1C and D, respectively, but here presented with all individual images. Right, analysis of the relative intensity of the fluorescent image in the indicated section for TRPC3 (red), Zebrin II/Aldolase C (green) and calbindin (blue). Intensity values were normalized to their respective means and filtered by a moving average filter over 15 µm to reduce noise. Note that 1) TRPC3 expression appears complementary to Zebrin II in most of the vermis (see arrowheads) and hemispheric parts, while it appears to be expressed more uniformly in other areas of the hemispheres and 2) that Zebrin II expression differentiates the subpopulations more clearly than TRPC3, under these conditions. (E–F) Cross-correlation of staining intensities for ZebrinII/Aldolase C vs. TRPC3 (top) and ZebrinII/Aldolase C vs. Calbindin (bottom) in the vermis (left) and hemispheres (right) for examples in A (E) and in C (F). Whereas ZebrinII/Aldolase C positively correlates to Calbindin intensity (suggesting general fluctuations in staining intensity), the correlation with TRPC3 is consistently negative, that is when ZebrinII/Aldolase C intensity is high, TRPC3 is low and vice versa.
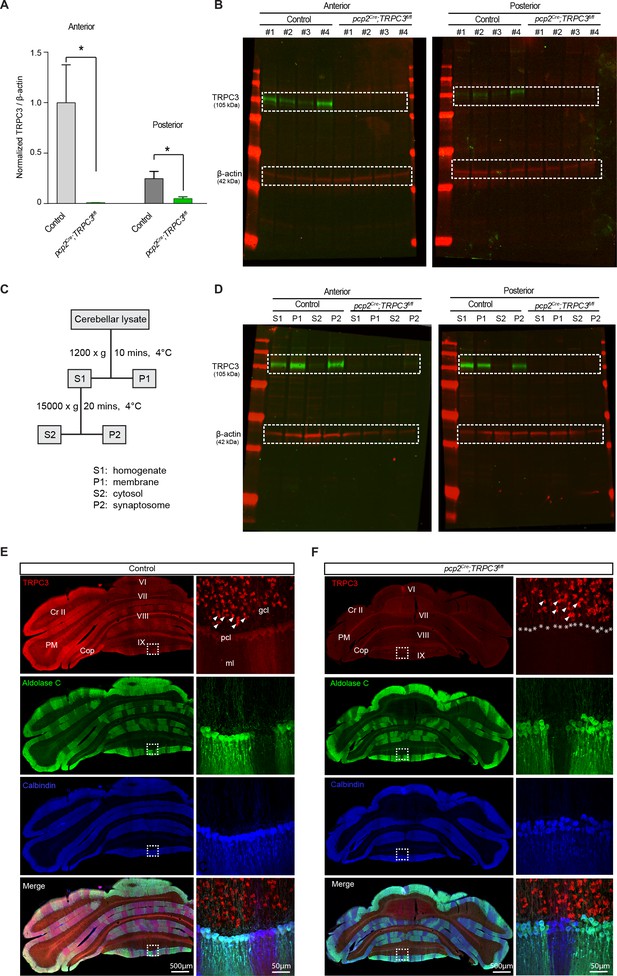
Western blot and immunostaining of pcp2Cre;TRPC3fl/fl mice.
(A–B) Quantification (bottom) of TRPC3 levels in the anterior (left) and posterior (right) cerebellum in pcp2Cre;TRPC3fl/fl mice. The level of TRPC3 was lower in tissue from the anterior part (lobules I-III) (t7 = 2.63, p=0.034) and posterior part (lobules X) (t8 = 2.67, p=0.028) of the cerebellum of pcp2Cre;TRPC3fl/fl mice compared to controls. Note the residual TRPC3 present in the posterior cerebellum, presumably due to the presence of unaffected TRPC3-expressing UBCs, which are virtually absent in the anterior cerebellum. (B) Example images of full-length western blots. (C) Schematic for synaptic protein extraction protocol. (D) Subcellular localizations by western blots in the anterior (top) and posterior (bottom) cerebellum. TRPC3 is abundantly present in the membrane (P1) and synaptosomes (P2), but less so in the cytosol (S2). pcp2Cre;TRPC3fl/fl mice were devoid of TRPC3 completely in both anterior and posterior cerebellar fractions. (E–F) Coronal immunofluorescence images of immunohistochemical analysis of TRPC3 (red) and Zebrin II/Aldolase C (green) expression in the posterior cerebellar cortex of pcp2Cre;TRPC3fl/fl mutants (F) and normal mice (E) with a higher magnifications of the squared areas (right). In contrast to the TPRC3 staining in control mice there is no longer a banding pattern visible for TRPC3 in mutant mice, while aldolase C is still clearly present in bands. The presence of TRPC3 staining in the UBCs, see for example the example indicated by the arrowhead, confirms that the antibody worked and that the loss of TRPC3 is specific for PCs (marked by asterisks).
Light sheet imaging reconstruction of whole-mount immunolabeling for TRPC3 (white signal), cleared with iDISCO protocol and scanned in the horizontal plane of an adult mouse brain from dorsal to ventral (see Materials and methods).
For comparison, a subregion in the anterior vermis is compared side-by-side to the same region in a brain immunolabeled for EAAT4, which has an expression pattern similar to Zebrin II/Aldolase C.
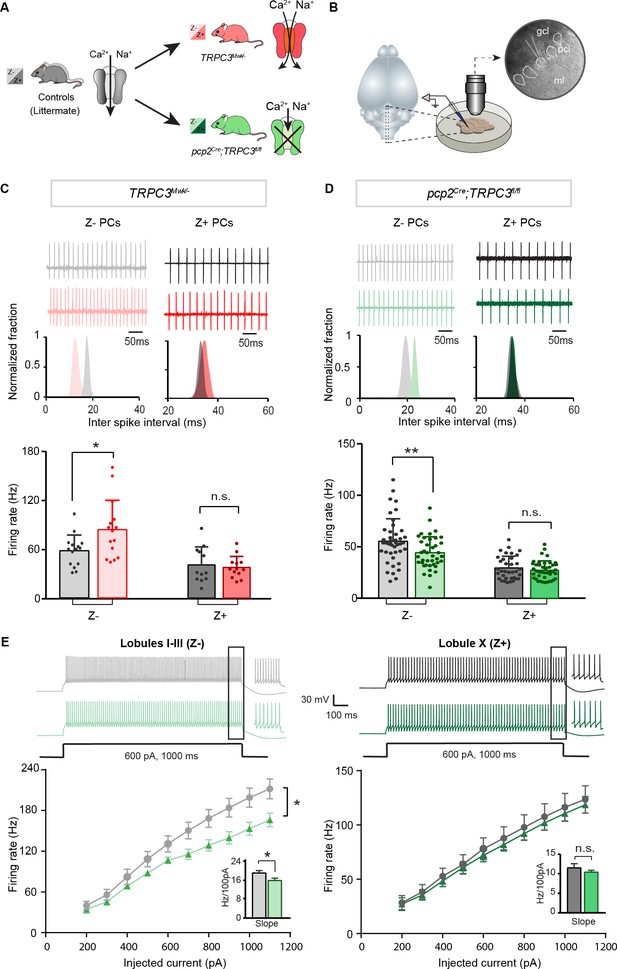
Differential controls of PC firing properties by TRPC3 in vitro.
(A) Schematic drawing of TRPC3 channel function in control (black), gain-of-function (TRPC3Mwk/-, red) and loss-of-function (pcp2Cre;TRPC3fl/fl, green) mice. (B) Schematic approach illustrating of PCs (right circle, dashed lines) recording in vitro, in acute sagittal slices. (C, D) Representative traces of cell-attached PC recordings (top) and corresponding inter spike interval (ISI) distributions (middle) in a Z– PC (left) and a Z+ PC (right) of TRPC3Mwk/- (C) and pcp2Cre;TRPC3fl/fl (D) mice. Z– PCs were affected in TRPC3Mwk/- (C), light-red, n = 15 cells/N = 4 mutant mice vs. n = 15 cells/N = 4 littermate controls, t28 = −2.47, p=0.020 and in pcp2Cre;TRPC3fl/fl mice (D), light-green, n = 40/N = 6 mutants vs. n = 43/N = 5 controls, t81 = 2.69, p=0.009). No differences in the firing rate of Z+ PCs in TRPC3Mwk/- (C), dark-red, n = 13/N = 4 mutants vs. n = 12/N = 4 controls, t18 = 0.419, p=0.680) and pcp2Cre;TRPC3fl/fl mice (D), dark-green, n = 36/N = 10 mutants vs. n = 35/N = 4 controls, t64 = 0.937, p=0.352). (E) Whole-cell patch-clamp recordings in slice from PCs of pcp2Cre;TRPC3fl/fl and control mice were used to test intrinsic excitability, by keeping cells at a holding potential of −65 mV and evoking action potentials by current steps of 100 pA (example, top). Top, exemplary traces evoked by current injection at 600 pA. Bottom, Input-output curves from whole-cell recordings of pcp2Cre;TRPC3fl/fl mice of Z– PCs (left, n = 17/N = 5 mutants vs n = 17/N = 5 controls, t32 = −2.20, p=0.035) and Z+ PCs (right, n = 12/N = 5 mutants vs n = 12/N = 4 controls, t22 = −0.95, p=0.354). gcl, granule cell layer; pcl, Purkinje cell layer; ml, molecular layer. (C–D), data are represented as mean ± s.d.; (E), data are represented as mean ± s.e.m., * means p<0.05 and **p<0.01. For values see Source data.
-
Figure 2—source data 1
Source data for Figure 2 and supplement.
Source data for the in vitro recordings in TRPC3Mwk/- and pcp2Cre;TRPC3fl/fl.
- https://doi.org/10.7554/eLife.45590.009
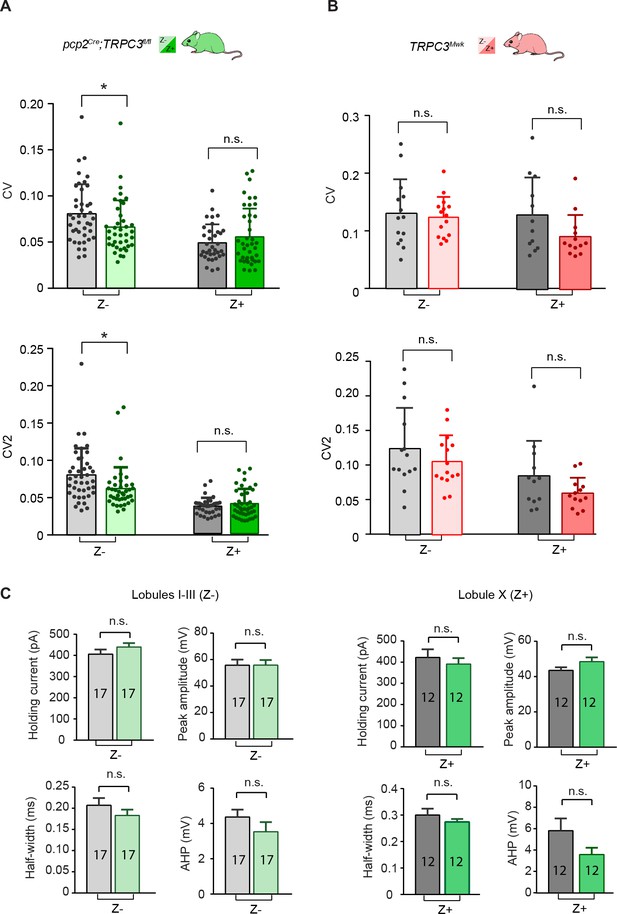
PC firing activity in TRPC3 mutants in vitro.
(A), The coefficient of variation (CV), measure for regularity of the entire trace, and CV2, measure of regularity on short time-scales, of ISIs were both significantly reduced in Z– PCs of pcp2Cre;TRPC3fl/fl mice (light-green, n = 40 cells/N = 6 mutants vs. n = 43/N = 5 controls; CV: t81 = 2.19, p=0.031; CV2: t81 = 2.61, p=0.011), but unaltered in Z+ PCs (dark-green, n = 36/N = 10 vs. n = 35/N = 4, CV: t71 = −1.13, p=0.263; CV2: t67 = −0.977, p=0.332), compared with littermate controls. (B) PCs of TRPC3Mwk/- mice showed no significant differences in vitro in CV and CV2, either in Z– PCs (light-red, n = 15/N = 4 mutants vs. n = 11/N = 2 controls; CV: t24 = 0.34, p=0.735; CV2: t24 = 1.32, p=0.199), or in Z+ PCs (dark-red, n = 13/N = 4 vs. n = 10/N = 2 controls; CV: t21 = 0.985, p=0.336; CV2: t21 = 0.960, p=0.348), compared with littermate controls. (C) Whole-cell patch clamp recordings of PCs of pcp2Cre;TRPC3fl/fl mice, revealed no significant differences in holding current or parameters of first action potential evoked by current injection, including peak-amplitude, half-width and AHP, between mutants and controls. Error bars denote s.d. (A–B) or s.e.m. (C). Lighter colors represent Z– and darker colors represent Z+ PCs, respectively. See Source data for values and statistics, * equals p<0.05, **p<0.01 and ***p<0.001.
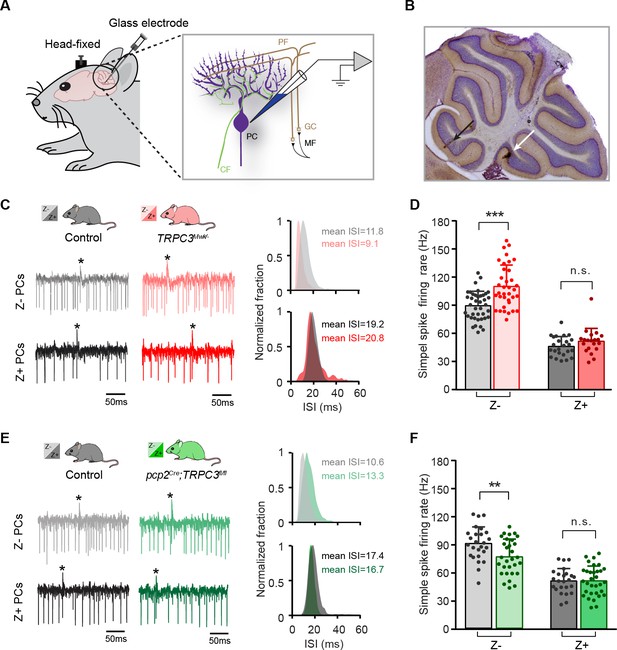
TRPC3 contributes to the in vivo simple spike firing rate of Z–, but not Z+ PCs.
(A) Schematic illustration of extracellular recording configuration in vivo. PF, parallel fiber; CF, climbing fiber; MF, mossy fiber; GC, granule cell. (B) Representative sagittal cerebellar section with recording sites labeled by BDA injection, in lobule II (black arrow) and X (white arrow). (C) Representative example traces (left) and ISI distributions (right) of a Z– PC (top) and a Z+ PC (bottom) in gain-of-function TRPC3Mwk/- mice. (D) PC simple spike firing rate recorded in vivo in TRPC3Mwk/- mice compared to control littermates, for the Z–lobules I-III (light-red, n = 36/N = 7 mutants vs. n = 40/N = 6 controls, t60 = −4.58, p<0.001) and the Z+ lobule X (dark-red, n = 20/N = 6 mutants vs. n = 24/N = 5 controls, t42=-1.47, p=0.148). (E) Representative example traces (left) and ISI distributions (right) in a Z– PC (top) and a Z+ PC (bottom) of loss-of-function pcp2Cre;TRPC3fl/fl mice. (F) PC simple spike firing rate of pcp2Cre;TRPC3fl/fl mice compared to controls, for Z– lobules I-III (light-green, n = 30/N = 7 mutants vs. n = 26/N = 8 controls, t54=2.88, p=0.006) and in Z+ lobule X (dark-green, n = 32/N = 8 mutants vs. n = 24/N = 6 controls, t54 = −0.053, p=0.958). Data are represented as mean ± s.d., for values see Source data, ** means p<0.01 and ***p<0.001.
-
Figure 3—source data 1
Source data for Figure 3 and supplement.
Source data for the in vivo recordings in TRPC3Mwk/- and pcp2Cre;TRPC3fl/fl mice, simple spikes data.
- https://doi.org/10.7554/eLife.45590.012
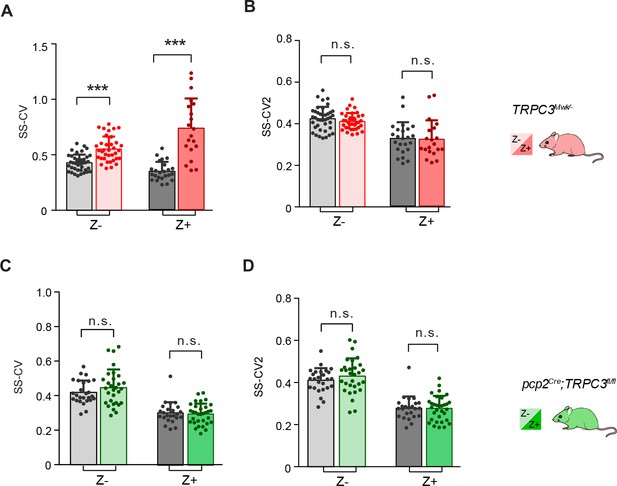
In vivo extracellular recordings of PC simple spike activity in TRPC3Mwk/- and pcp2Cre;TRPC3fl/fl mice.
(A–B) CV, a measure for regularity of the entire trace, for simple spikes (SS-CV) of PCs recorded in TRPC3Mwk/- mice, were significantly increased in both Z– and Z+ PCs. In Z– PCs this can exclusively be attributed to TRPC3 gain-of-function, while in Z+ PCs the loss of the regular input from UBCs (predominantly present in Z+ areas) potentially contributes to the phenotype. CV2, a measure for regularity on short time-scales, for simple spikes (SS-CV2), in both Z– and Z+ PCs of TRPC3Mwk/- mice were unaffected, compared with those of littermate controls. (C–D), In pcp2Cre;TRPC3fl/fl mice, SS-CV, SS-CV2 in both Z– and Z+ PCs do not differ from littermate controls. Error bars denote s.d.. See Source data for values and statistics, * equals p<0.05, **p<0.01 and ***p<0.001.
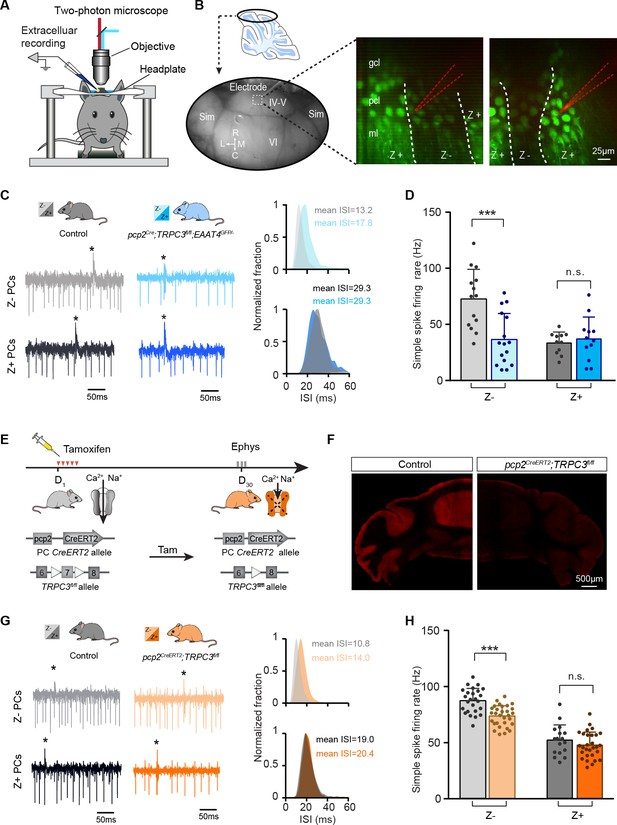
The contribution of TRPC3 to firing rate is dependent on zebrin-identity and independent of developmental changes.
(A) Schematic experimental setup for two-photon imaging-based targeted PC recordings, in vivo. (B) Sagittal view of cerebellum (schematic, top) indicating the recording region in the ellipse (bottom). Representative images (right) show the visualization of Z+ bands (dark green) in an awake pcp2Cre;TRPC3fl/fl;EAAT4GFP/- mouse, with recording electrodes (red) positioned in Z– (left) and Z+ (right) bands. (C) Representative firing traces (left) and ISI distributions (right) in a Z– PC (top) and a Z+ PC (bottom) of loss-of-function pcp2Cre;TRPC3fl/fl;EAAT4GFP/- mice (blue) and control littermates (no Cre; gray). (D) Average simple spike firing rate of PCs recorded from adjacent modules of pcp2Cre;TRPC3fl/fl;EAAT4GFP/- mice and those in controls. Comparison for Z– PCs (light-blue, n = 16/N = 3 mutants vs. n = 14/N = 2 controls, t28 = 3.99, p<0.001), and Z+ PCs (dark-blue, n = 12/N = 3 mutants vs. n = 12/N = 2 controls, t21 = −0.550, p=0.588). (E–F) Intraperitoneal tamoxifen injections for five days (D1-5) to trigger TRPC3 gene ablation solely in PCs in adult pcp2CreERT2;TRPC3fl/fl mice. Open triangles indicate loxP sites. PC in vivo extracellular activity was recorded four weeks later (D29-31) in pcp2CreERT2;TRPC3fl/fl mice (orange). TRPC3 deletion was confirmed post-mortem by confocal imaging following anti-TRPC3 staining (F). (G) Representative firing traces (left) and ISI distributions (right) in a Z– PC (top) and a Z+ PC (bottom) of pcp2CreERT2;TRPC3fl/fl mice. (H) Simple spike firing rate in vivo in pcp2CreERT2;TRPC3fl/fl and control mice (no Cre) recorded in lobules I-III (Z–) and lobule X (Z+) PCs. Comparison for Z– PCs (light-orange, n = 30/N = 4 mutants vs. n = 25/N = 4 controls, t53 = 5.05, p<0.001), and Z+ PCs (dark-orange, n = 29/N = 4 mutants vs. n = 17/N = 3 controls, t44 = 1.21, p=0.234). Sim, simplex lobule; IV-VI, lobules IV-VI, R, rostral, C, caudal; L, lateral, M, medial. Data are represented as mean ± s.d., for values see Source data, *** means p<0.001.
-
Figure 4—source data 1
Source data for Figure 4 and supplement.
Source data for the in vivo recordings in pcp2Cre;TRPC3fl/fl;EAAT4GFP/- and pcp2CreERT2;TRPC3fl/fl mice, simple spikes data.
- https://doi.org/10.7554/eLife.45590.015
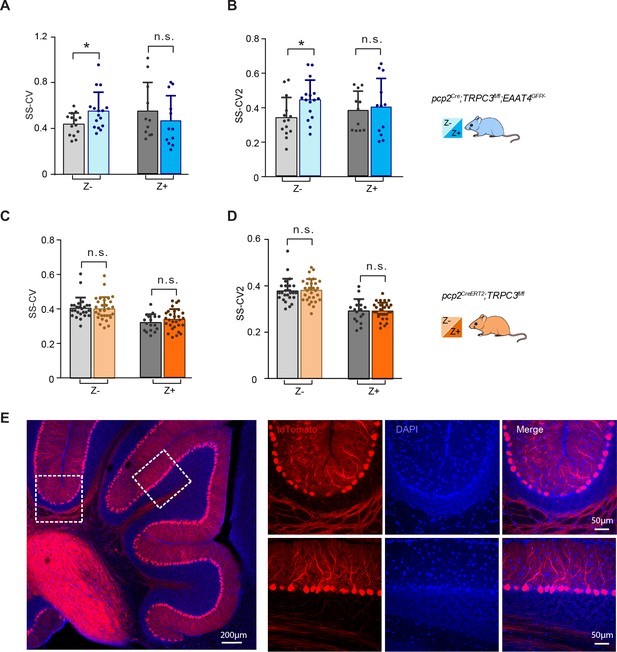
In vivo extracellular recordings of PC simple spike activity in pcp2Cre;TRPC3fl/fl;EAAT4GFP/- and pcp2CreERT2;TRPC3fl/fl mice.
(A–B) In pcp2Cre;TRPC3fl/fl;EAAT4GFP/- mice, the SS-CV and SS-CV2 of Z– PCs were significantly increased compared to those of littermate controls. However, there was no change in those features of the Z+ PCs. (C–D), PCs in pcp2CreERT2;TRPC3fl/fl mice showed, after tamoxifen-induced TRPC3 ablation, no significant differences in SS-CV and SS-CV2 in Z– or Z+ PCs, as compared with those of littermate controls that were also injected with tamoxifen. (E) To verify the efficiency and selectivity of the inducible PC-specific Cre expression line (pcp2CreERT2), these mice were first crossed with Cre-dependent tdTomato expressing (Ai14) mice. Confocal images of a sagittal section of the cerebellar cortex show that labeling is virtually exclusively found in PCs, which are all labeled. Error bars denote s.d.. * equals p<0.05, **p<0.01 and ***p<0.001.
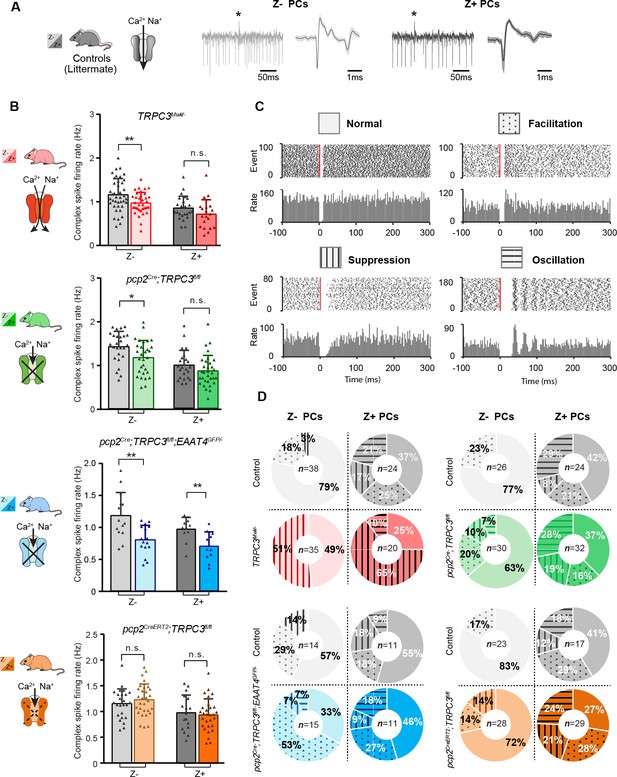
Complex spikes and complex spike - simple spike interaction are affected by TRPC3 mutations.
(A) Representative PC recording traces and complex spikes shape of Z– (light black) and Z+ (dark black) PCs in the control mice. (B) Top half, comparison of complex spike firing rates in TRPC3Mwk/- (red) and pcp2Cre;TRPC3fl/fl (green) mice versus their respective littermate controls for Z– PCs (TRPC3Mwk/-: t68=2.68, p=0.009; pcp2Cre;TRPC3fl/fl: t54=2.50, p=0.016) and Z+ PCs (TRPC3Mwk/-: t42=1.56, p=0.126; pcp2Cre;TRPC3fl/fl: t54=1.41, p=0.164). Bottom half, comparison of complex spike firing rates in pcp2Cre;TRPC3fl/fl;EAAT4GFP/- (blue) and pcp2CreERT2;TRPC3fl/fl (orange) mice versus their respective controls for Z– PCs (pcp2Cre;TRPC3fl/fl;EAAT4GFP/-: t28=3.49, p=0.002; pcp2CreERT2;TRPC3fl/fl: t53=-0.940, p=0.352) and Z+ PCs (pcp2Cre;TRPC3fl/fl;EAAT4GFP/-: t20=3.03, p=0.007; pcp2CreERT2;TRPC3fl/fl: t44=0.448, p=0.656). (C) Raster plots of simple spike activity around the occurrence of each complex spike (−100 to +300 ms). These peri-complex splike time histograms can, based on post-complex spike activity, be divided into one of four types: normal (no change), facilitation, suppression and oscillation. (D) The distribution of post-complex spike response types for Z– and Z+ PCs, in TRPC3Mwk/-, pcp2Cre;TRPC3fl/fl, pcp2Cre;TRPC3fl/fl;EAAT4GFP/- and pcp2CreERT2;TRPC3fl/fl mice. Data are represented as mean ± s.d., for values see Source data, * means p<0.05 and **p<0.01.
-
Figure 5—source data 1
Source data for Figure 5 and supplement.
Source data for the in vivo recordings in TRPC3Mwk/-, pcp2Cre;TRPC3fl/fl, pcp2Cre;TRPC3fl/fl;EAAT4GFP/- and pcp2CreERT2;TRPC3fl/fl mice, complex spikes data.
- https://doi.org/10.7554/eLife.45590.018
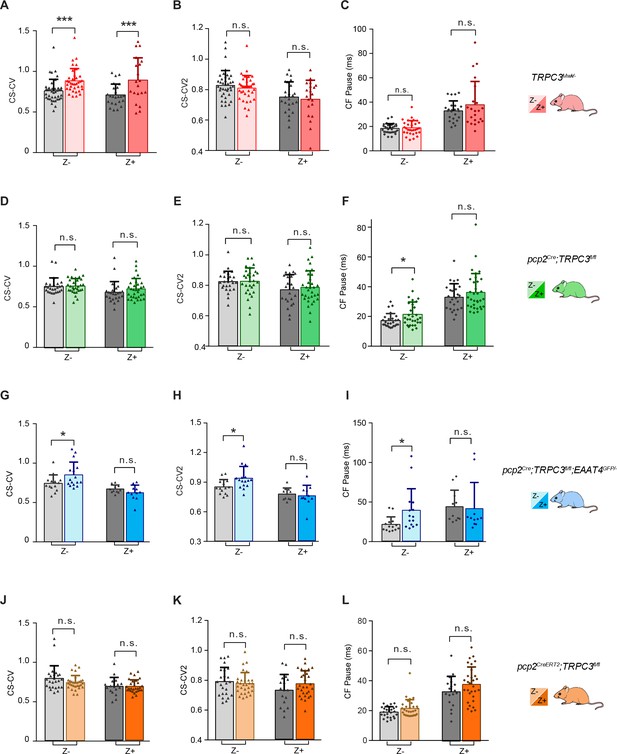
In vivo extracellular recordings of PC complex spike activity in TRPC3Mwk/-, pcp2Cre;TRPC3fl/fl mice, pcp2Cre;TRPC3fl/fl;EAAT4GFP/- and pcp2CreERT2;TRPC3fl/fl mice.
(A–C) CV for complex spikes (CS-CV) of PCs recorded in TRPC3Mwk/- mice, were significantly increased in both Z– and Z+ PCs. CS-CV2 and CF-pause in both Z– and Z+ PCs of TRPC3Mwk/- mice were unaffected, compared with those of littermate controls. (D–F), In pcp2Cre;TRPC3fl/fl mice, CS-CV, CS-CV2 in both Z– and Z+ PCs do not differ from littermate controls. However, CF-pause was significant longer Z– PC (F), left), but unaffected in Z+ PCs. (G–I) In pcp2Cre;TRPC3fl/fl;EAAT4GFP/- mice, all parameters for Z– PCs, including CS-CV, CS-CV2 and CF-pause, were significantly increased compared to those of littermate controls. However, there was no change in those features of the Z+ PCs. (J–L), After tamoxifen-induced TRPC3 ablation, PCs in pcp2CreERT2;TRPC3fl/fl mice showed no significant differences in CS-CV, CS-CV2 and CF-pause in Z– or Z+ PCs, as compared with those of littermate controls that were also injected with tamoxifen. Error bars denote s.d.. * equals p<0.05, **p<0.01 and ***p<0.001.
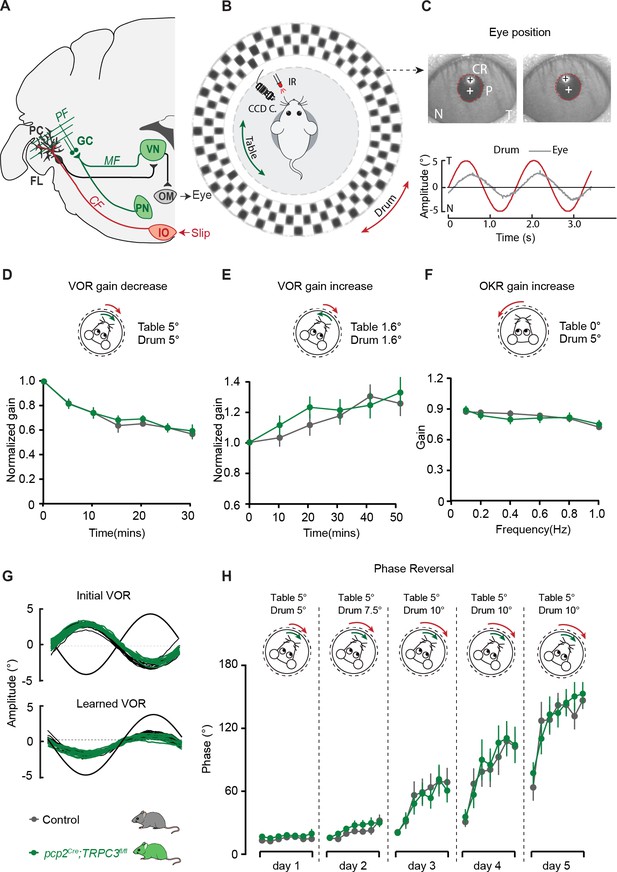
PC-specific deletion of TRPC3 does not affect Z+-dependent VOR adaptation.
(A) Cerebellar circuitry controlling compensatory eye movements and their adaptation. PCs in the flocculus (FL) receive vestibular and visual input via the mossy fiber (MF) - parallel fiber (PF) system (green) and climbing fiber input (CF, red) from the inferior olive (IO), indicating retinal slip. These two inputs converge on PCs, which influence eye movements via the vestibular nuclei (VN) and the oculomotor (OM) neurons. PN, pontine nuclei; GC, granule cell. (B) Schematic illustration of eye movement recording setup. Mice are head-fixed in the center of a turntable for vestibular stimulation and surrounded by a random dotted pattern (‘drum’) for visual stimulation. A CCD camera was used for infrared (IR) video-tracking of the left eye. (C) Top, examples of nasal (N) and temporal (T) eye positions. Red circles, pupil fit; black cross, corneal reflection (CR); white cross, pupil center. Bottom, example trace of eye position (gray) with drum position (red), during stimulation at an amplitude of 5° and frequency of 0.6 Hz. (D) pcp2Cre;TRPC3fl/fl and control mice were subjected to six 5 min training sessions with mismatched in-phase visual and vestibular stimulation (in light, see insets), aimed at decreasing the VOR gain (probed in the dark before, between and after sessions). (E) Similar, but now mice were trained with out-of-phase stimulation, aimed at increasing VOR gain. (F) Re-recording of OKR gain following the VOR phase reversal training (see G–H) to test OKR gain increase (compare to Figure 6—figure supplement 2C, left). (G) Multiple-day training using in-phase mismatch stimulation (see inset in H) aimed at reversing the direction of the VOR (quantified as a reversal of the phase). Representative eye position recordings of VOR before (top) and after (bottom) training. (H) Results of five days of VOR phase reversal training, probed by recording VOR (in the dark before, between and after sessions) with mice kept in the dark in overnight. Data are represented as mean ± s.e.m., N = 11 mutants versus N = 13 controls, all p>0.05, ANOVA for repeated measurements. See Source data for values.
-
Figure 6—source data 1
Source data for Figure 6 and supplements.
Source data for the Erasmus ladder and compensatory eye movement experiments in pcp2Cre;TRPC3fl/fl mice.
- https://doi.org/10.7554/eLife.45590.022
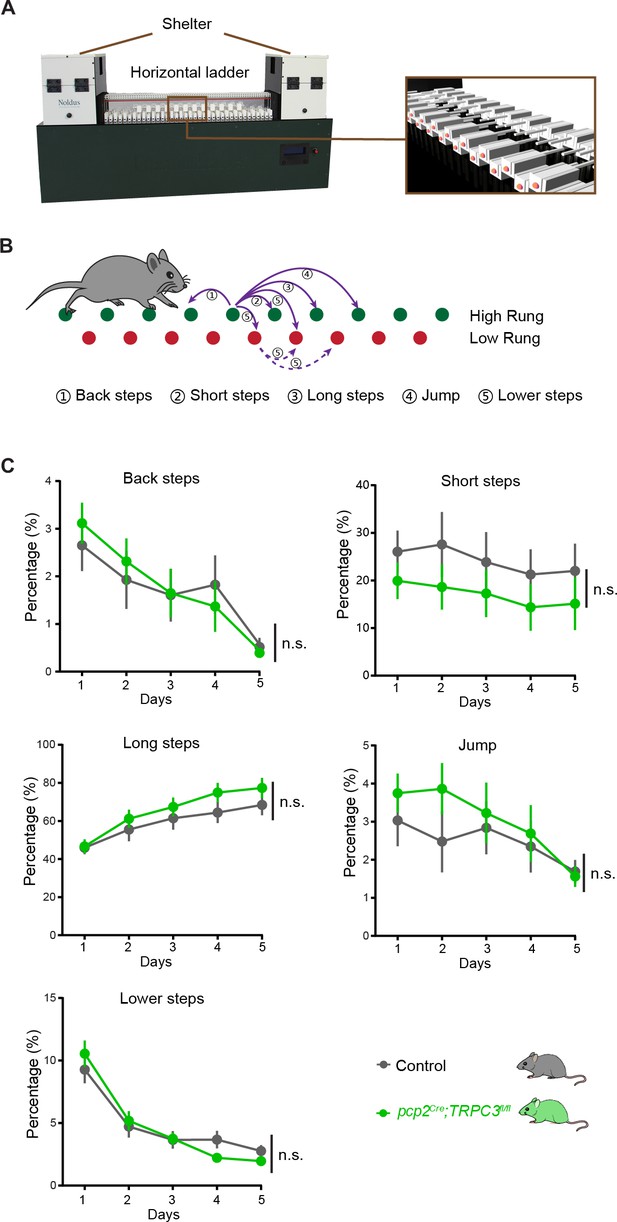
pcp2Cre;TRPC3fl/fl mice show normal Erasmus ladder performance.
(A) The setup of Erasmus Ladder which consists of a horizontal ladder (magnifications on the right) connecting two shelter boxes. (B) Schematic of high rungs (green) and low rungs (red) with purple arrows illustrating the five different step types: ① Back steps, ② Short steps, ③ Long steps, ④ Jumps, ⑤ Lower steps (see methods). (C) The distribution of step types in pcp2Cre;TRPC3fl/fl mice did not differ from their littermate controls over the five days tested. Values are shown as mean ± s.e.m.
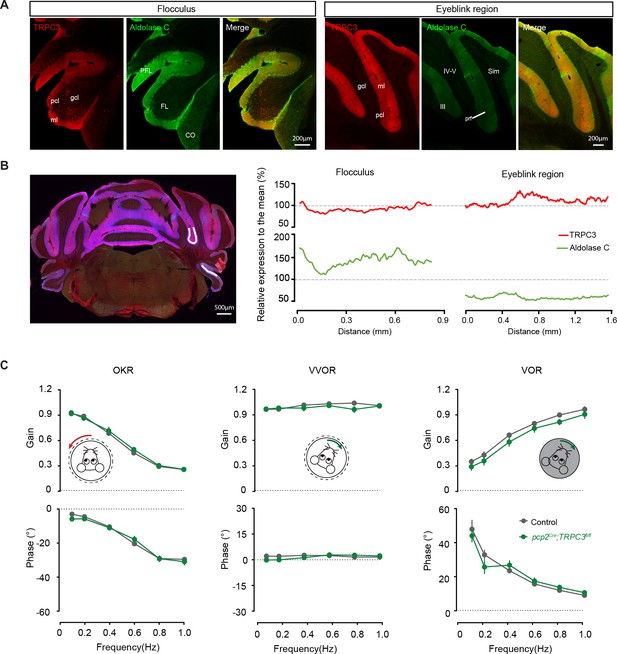
Compensatory eye movements and eyeblink conditioning in pcp2Cre;TRPC3fl/fl mice.
(A) Confocal images of staining for TRPC3 and Zebrin II/Aldolase C in the regions related to compensatory eye movements and eyeblink conditioning. Compensatory eye movements and their adaptation are under control of the flocculus of the cerebellum. Zebrin-staining is more positive in the flocculus (middle), while being less intense for TRPC3 (left). Note that TRPC3 staining is not absent in the flocculus and that UBCs in the granule cell layer of the flocculus also stain positive for TRPC3. The region of eyeblink conditioning, locating at the sulcus between lobule IV-V and VI and related parts of the hemispheres, is largely Z– (middle), and stained positive for TRPC3 (left). (B) Quantification of relative TRPC3 and Zebrin II/Aldolase C intensities in subsections of both regions, indicated by the white line. Values are normalized to the average of the two lines. Note that TRPC3 is expressed in both areas, but levels are higher in the eyeblink regions than the flocculus, and vice versa for Zebrin II. ml, molecular layer; gcl, granule cell layer; pcl, Purkinje cell layer; prf, primary fissure; Sim, simple lobule; PFL, paraflocculus; FL, flocculus; CO, cochlear nucleus. (C) Gain (top) and phase (bottom) of baseline performance of compensatory eye movements: the optokinetic reflex (OKR), the vestibulo-ocular reflex (VOR) and the visually-enhanced VOR (VVOR) were not affected in pcp2Cre;TRPC3fl/fl mice, compared to littermate controls (N = 13 versus N = 11, all p>0.05).
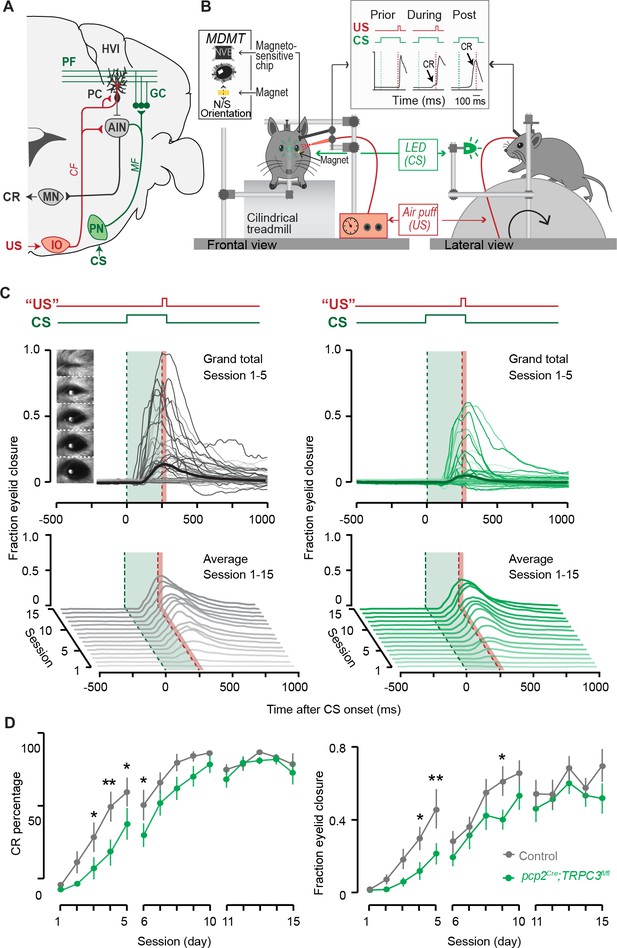
Eyeblink conditioning, linked to Z– modules is delayed in pcp2Cre;TRPC3fl/fl mice.
(A) Cerebellar circuitry controlling eyeblink conditioning. PCs in the paravermal region around the primary fissure receive inputs carrying sensory information from for example the pontine nucleus (PN) through the MF-PF pathway and the error signal from the inferior olive (IO) through the climbing fibers (CF). These PCs in turn influence eyelid muscles via the anterior interposed nucleus (AIN) and motor nuclei (MN). (B) Schematic illustration of eyeblink conditioning setup. Head fixed mice on a freely moving treadmill, are presented a green LED light (conditional stimulus, CS) followed several hundred milliseconds later by a weak air-puff on the eye (unconditional stimulus, US). As a result of repeated CS-US pairings, mice will eventually learn to close their eye in response to the CS, which is called the conditioned response (CR). Eyelid movements were recorded with the magnetic distance measurement technique (MDMT). (C) Comparison of fraction of eyelid closure between controls (left) and pcp2Cre;TRPC3fl/fl mice (right). Top, session averages (thin-lines) per mouse and overall average (thick-lines) for the first 5 days (color intensity increasing from day 1 to 5). Insets: mouse eye video captures show eyelid closure ranging from 0 (fully-open) to 1 (fully-closed). Bottom, waterfall plot of the averaged eyeblink trace during CS-only trials for the 15 daily sessions. (D) The CR percentage and CR amplitude for pcp2Cre;TRPC3fl/fl mice initially have a significantly slower acquisition but eventually reach the same levels as control littermates. Data are represented as mean ± s.e.m., N = 15 mutants versus N = 15 controls, P values were all FDR corrected for multiple comparisons, see Source data for values and statistics, * means p<0.05 and **p<0.01.
-
Figure 7—source data 1
Source data for Figure 7 and supplement.
Source data for the eyeblink conditioning experiments in pcp2Cre;TRPC3fl/fl mice.
- https://doi.org/10.7554/eLife.45590.025
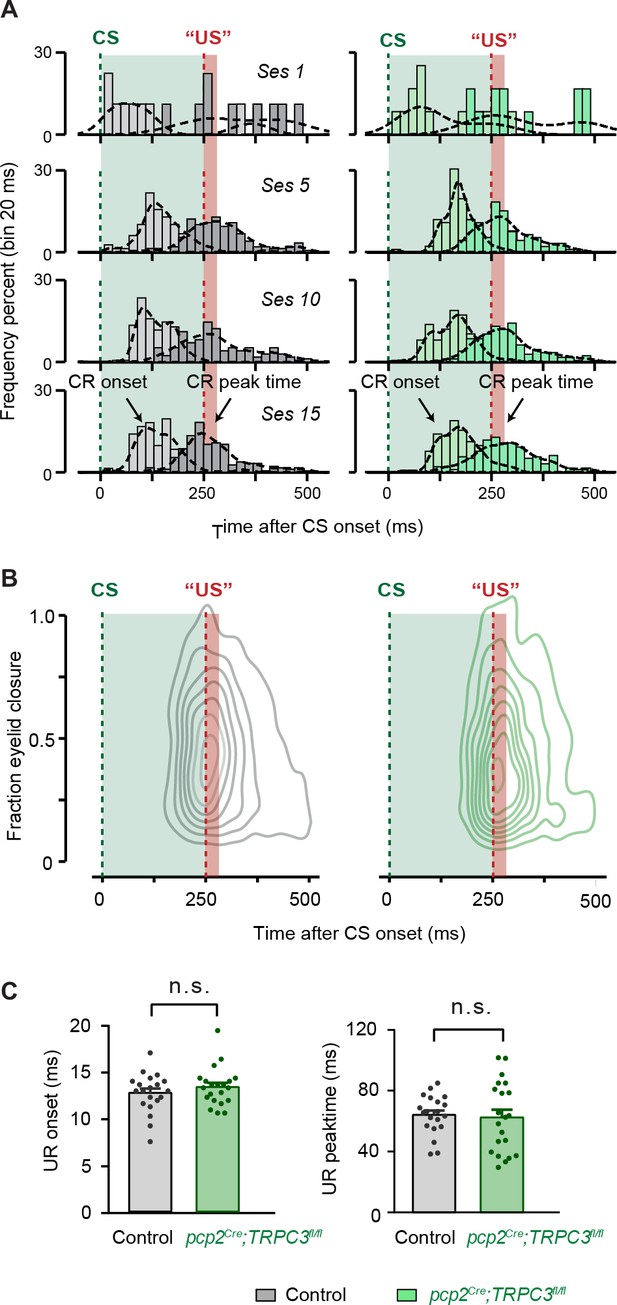
Eyeblink conditioning in pcp2Cre;TRPC3fl/fl mice.
(A) Peri-stimulus histogram plots with a Gaussian kernel density estimate (black dashed line) showing the distribution of CR onset (dark filled bars) and CR peak time (light filled bars) relative to CS and US onset in CS only trials for session 1, 5, 10, and 15. In both groups, there is a clear development in CR onset and peak time: there are no clearly preferred times in the CS-US interval at the start of training (session 1), but during training CR onset values are centered around 100–125 ms after CS onset, and CR peak times are located around the onset of the expected US. Green dashed line is CS onset, red dashed line is US onset; light green and light red fill indicate CS and US duration, respectively. (B) Two-dimensional density plot showing latency to CR peak relative to the fraction eyelid closure over all sessions. Both groups clearly show CRs that are timed around the onset of US.(C) On the first recording day the onset and peak time of the unconditioned response (UR) are analyzed and no difference was found (UR onset: t39 = −0.972, p=0.337; UR peak time t32 = 0.301, p=0.765). Values are shown as mean ± s.e.m.
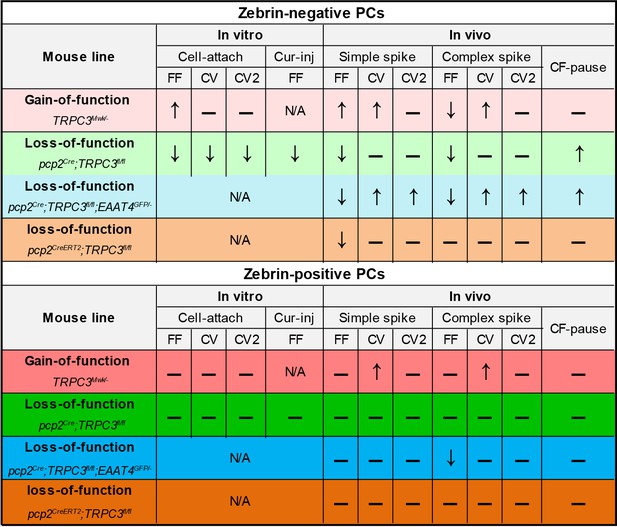
Summary of the electrophysiological changes in the gain- and loss-of-function TRPC3 mutants.
https://doi.org/10.7554/eLife.45590.026Tables
| Reagent type (species) or resource | Designation | Source or reference | Identifiers | Additional information |
|---|---|---|---|---|
| Species Mus musculus | C57BL/6J mice | Charles Rivers | IMSR_JAX:000664 | |
| Species Mus musculus | TRPC3Mwk/- | Becker et al., 2009 | MGI:3689326 | F1 of (original) C3H/HeH and C57BL/6J background |
| Species Mus musculus | TRPC3fl/fl | Hartmann et al., 2008 | MGI:5451202 | C57BL/6Jbackground |
| Species Mus musculus | pcp2Cre | Barski et al., 2000 | MGI:2137515 | C57BL/6Jbackground |
| Species Mus musculus | pcp2CreERT2 | The Institut Clinique de la Souris, www.ics-mci.fr | C57BL/6Jbackground | |
| Species Mus musculus | EAAT4GFP/- | Gincel et al., 2007 | C57BL/6Jbackground | |
| Species Mus musculus | Ai14 | https://www.jax.org/strain/007908 | MGI:3809524 | C57BL/6Jbackground |
| Antibody | Rabbit anti-TRPC3 | Cell Signaling | Cat.#: 77934 | IHC (1:500), WB (1:1000) |
| Antibody | Mouse anti-actin | Millipore | Cat.#: MAB1501 | WB (1:1000) |
| Antibody | Goat anti-Zebrin II/Aldolase C | Santa Cruz Biotechnology | Cat.#: SC-12065 | IHC (1:500) |
| Antibody | Mouse anti-calbindin | Sigma | Cat.#: C9848 | IHC (1:7000) |
| Antibody | Rabbit anti-GFP | Abcam | Cat. # 290 | IHC (1:1000) |
| Chemical compound, drug | Dextran, Biotin, 3000 MW, Lysine Fixable (BDA-3000) | Thermo Fisher Scientific | D7135 | |
| Software, algorithm | MATLAB v2014a | Mathworks | RRID: SCR_001622 | |
| Software, algorithm | Clampfit 10 | Molecular Devices | RRID: BDSC_14352 | |
| Software, algorithm | Patchmaster software (for in vitro recording analysis) | HEKA Electronics | ||
| Software, algorithm | Spiketrain software (for in vivo recording analysis) | Used under Neurasmus license, currently: kai.voges@nus.edu.sg | ||
| Software, algorithm | Erasmus Ladder 2.0 analysis | Noldus, Wageningen, Netherlands | ||
| Software, algorithm | Compensatory eye movements analysis | https://github.com/MSchonewille/iMove | ||
| Software, algorithm | Eyeblink conditioning analysis | Neurasmus BV, Rotterdam, Netherlands | ||
| Software, algorithm | GraphPad Prism 6 | GraphPad | RRID: SCR_002798 | |
| Software, algorithm | SPSS 20.0 | IBM SPSS | RRID: SCR_002865 |
Additional files
-
Supplementary file 1
Summary of the electrophysiological changes in gain- and loss-of-function TRPC3 mutants.
- https://doi.org/10.7554/eLife.45590.027
-
Transparent reporting form
- https://doi.org/10.7554/eLife.45590.028





