The readily-releasable pool dynamically regulates multivesicular release
Figures
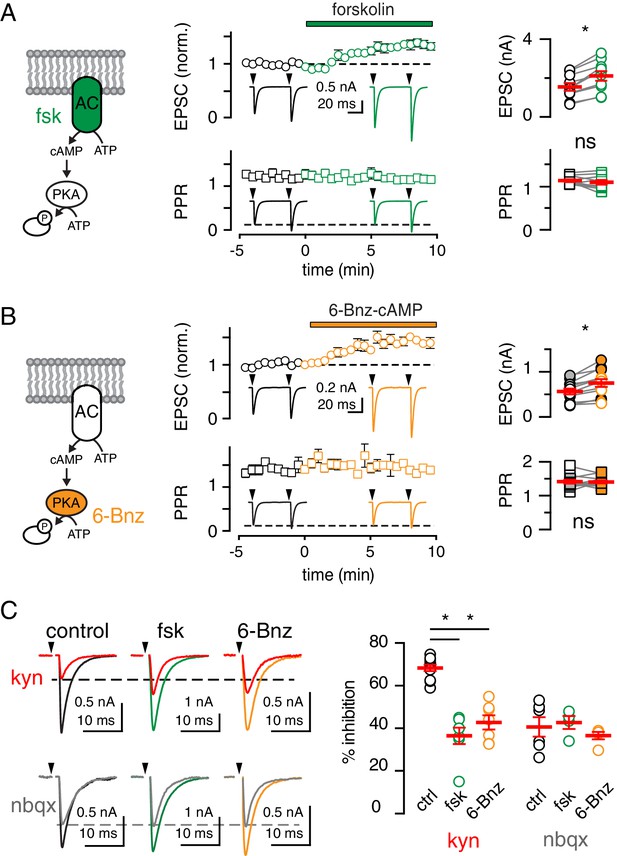
cAMP/PKA activation shifts vesicle release mode from UVR to MVR.
(A and B, left). Fsk (50 µM) was used to stimulate cAMP production by adenylyl cyclase (AC, green) and the cAMP analog 6-Bnz (20 µM) was used to activate protein kinase A (PKA, orange). (A and B, middle) Time course of CF-PC EPSC amplitude (top: normalized, circles) and paired pulse ratio (bottom: PPR with an inter-stimulus interval = 50 ms, squares) following bath application of fsk (green) or 6-Bnz (orange), respectively. Insets: representative and normalized traces show the increase in amplitude and lack of change in PPR. (A and B, right) Fsk increased the EPSC amplitude (from 1.6 ± 0.2 nA to 2.1 ± 0.3 nA; p<0.0001, paired t-test) without changing the PPR (from 1.1 ± 0.03 to 1.1 ± 0.05, n = 10 each; p=0.27). Likewise, 6-Bnz increased the EPSC amplitude (from 0.6 ± 0.1 nA to 0.7 ± 0.1 nA; p=0.0009, paired t-test) without changing the PPR (from 1.4 ± 0.07 to 1.4 ± 0.05, n = 10 each; p=0.68). Filled symbols indicate inclusion of the PKA inhibitory peptide PKi (1 µM) in the patch pipette. For this data set: EPSC amplitude (from 0.6 ± 0.1 nA to 0.8 ± 0.1 nA; p=0.02 and PPR from 1.4 ± 0.03 to 1.4 ± 0.07, n = 6 each; p=0.51, paired t-test). (C, left) Superimposed EPSCs (0.5 mM Ca2+/10 mM Mg2+) before and after bath application of 250 µM KYN (top) or 100 nM NBQX (bottom) in the absence or presence of fsk or 6-Bnz. (C, right) Both fsk and 6-Bnz reduced % EPSC inhibition by kyn from 68.3 ± 1.5% in control (n = 11) to 36.5 ± 3.8% in fsk (n = 7; p<0.0001) and to 42.8 ± 3.3% in 6-Bnz (n = 7; p<0.0001; ANOVAs followed by Holm-Sidak post-tests). In contrast, neither fsk nor 6-Bnz affected %EPSC inhibition by NBQX (control: 40.7 ± 4.6%, n = 6; fsk: 42.7 ± 3.0%, n = 4, p=0.98; 6-Bnz: 36.6 ± 1.7%, n = 5; p=0.92, ANOVA). Asterisks denote statistical significance.
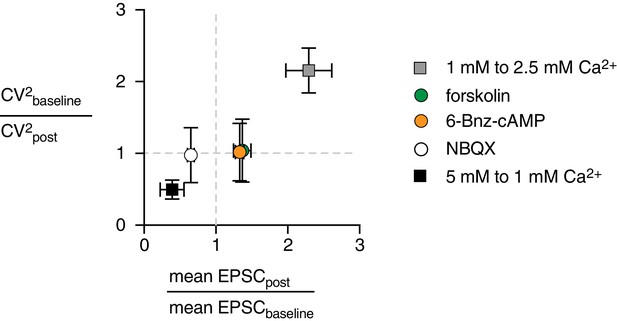
CF-PC EPSCs CV analysis.
CV analysis, plotted as the ratio of CV2 versus the mean EPSC amplitude ratio, normalized to baseline. For increases or decreases in mean amplitude (vertical dotted line is the mean ratio = 1), a deviation of the CV2 ratio from 1 (horizontal dotted line) reflects a change in Pr that occurs with shifts in extracellular Ca2+ (from 5 to 1 mM Ca2+ or from 1 to 2.5 mM Ca2+, filled and gray squares, respectively). In contrast, the CV2 ratio remains ~1 with NBQX (250 nM, open circle), fsk (50 µM, green circle), or 6-Bnz (10 µM, orange circle).
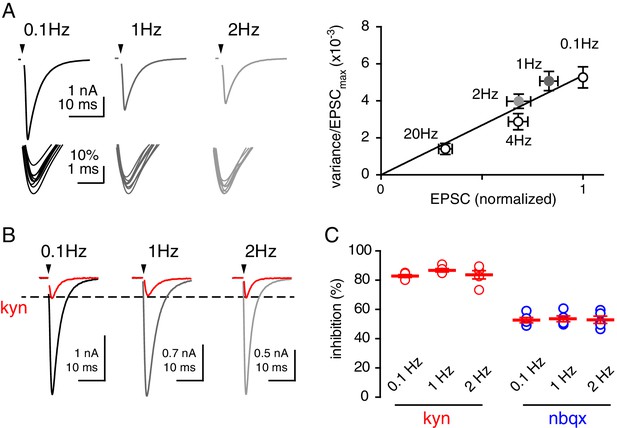
Reducing release site number does not alter KYN block.
(A) Increasing stimulation frequency between 0.1 and 20 Hz reduced EPSCs due to decrease the number of active release sites, confirmed by variance/mean analysis (Foster and Regehr, 2004). (B and C) The LAA, kynurenic acid (KYN; 1 mM), inhibited EPSCs by 82.2 ± 0.9% at 0.1 Hz, 86.7 ± 1.2% at 1 Hz, 83.7 ± 2.8% at 2 Hz; n = 5 each; p=0.3, ANOVA). The high-affinity antagonist NBQX (100 nM) that is insensitive to cleft glutamate concentration blocked EPSCs by 52.7 ± 1.8% at 0.1 Hz, 53.6 ± 1.9% at 1 Hz, 52.9 ± 2.4% at 2 Hz, n = 5 each; p=0.9, ANOVA).
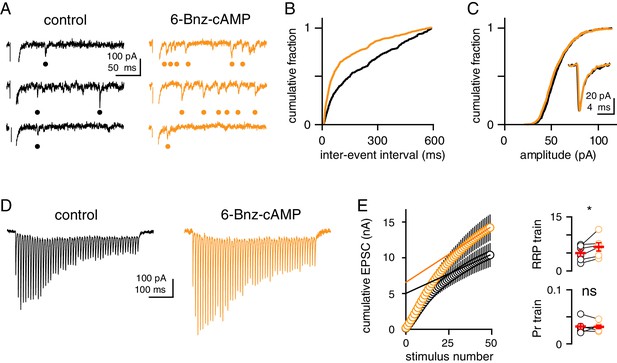
cAMP/PKA activation does not change quantal size but increases the RRP.
(A) Representative sweeps of Sr2+-evoked asynchronous EPSCs (aEPSCs) before (black) and after (orange) application of 6-Bnz (20 µM). Bullets denote detected events. (B) Distribution of aEPSC inter-event intervals before and after bath application of 6-Bnz (n = 6; p<0.0001, KS test). (C) Distribution of aEPSC amplitudes in control conditions (black) and 6-Bnz (20 µM, orange), compared with the (n = 6; p>0.99, KS test). Inset shows average aEPSCs. (D) Representative EPSCs recorded in response to CF stimulation at 100 Hz for 500 ms before (black) and after (orange) 6-Bnz treatment. (E, left) Cumulative EPSC amplitude plotted as a function of stimulus number before (black) and after (orange) 6-Bnz treatment. A line was fit to the final 5 EPSCs in each condition and the y-intercept of this line was used to estimate the RRP. (E, right).
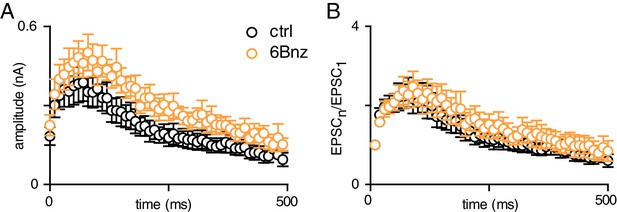
cAMP activation does not affect short-term plasticity.
(A) Time course of EPSC amplitudes in response to CF stimulation at 100 Hz for 500 ms before (black) and after (orange) 6-Bnz treatment. (B) Normalized EPSC amplitudes during train stimulation before (black) and after (orange) 6-Bnz treatment. The facilitating (fac) and depressing (dep) time course of EPSC amplitudes showed no differences between control and 6-Bnz treatment (τfac = 18.9 ± 2.0 ms and 19.4 ± 2.9 ms, p=0.9; τdep = 309.8 ± 18.7 ms and 340 ± 52.5 ms, p=0.4, paired t-tests).
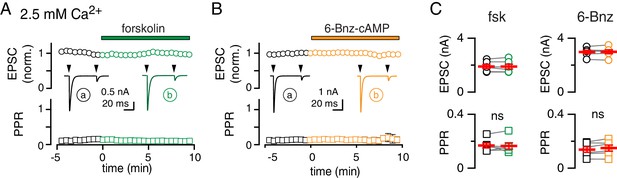
cAMP/PKA activation is occluded when MVR is prevalent.
(A and B) Time course of CF-PC EPSC amplitude (top: normalized, open circles) and PPR (bottom: open squares) during bath application of fsk (green) or 6-Bnz (orange). Insets: representative traces showing the lack of change in amplitude or PPR. Recordings performed in 2.5 mM Ca2+ and 1.3 mM Mg2+. (C) Fsk had no effect on EPSC amplitude (1.9 ± 0.2 nA in control versus 1.9 ± 0.2 nA in fsk; n = 6, p=0.87, paired t-test) or the PPR (0.17 ± 0.02 in control versus 0.17 ± 0.02 in fsk, n = 6, p=0.82, paired t-test). Likewise, 6-Bnz had no effect on EPSC amplitude (3.0 ± 0.2 nA in control versus 3.0 ± 0.1 nA in 6-Bnz; n = 6, p=0.64, paired t-test) or PPR (0.14 ± 0.02 in control versus 0.15 ± 0.03 n=6; p=0.08, paired t-test).
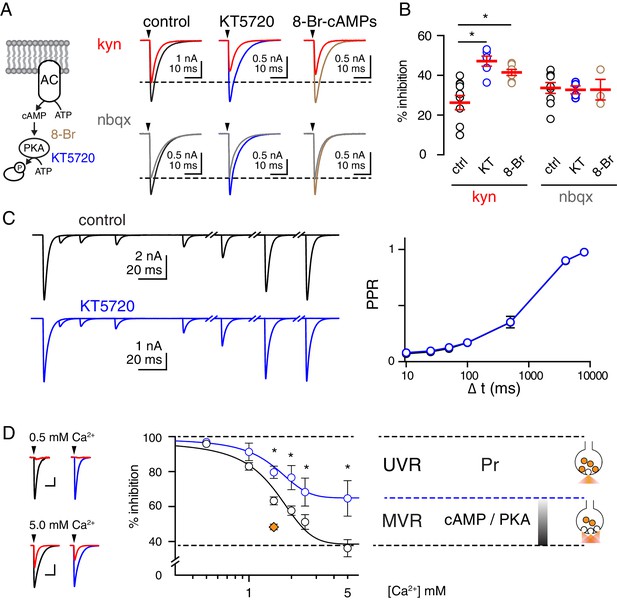
PKA-inhibition shifts vesicle release mode from MVR to UVR.
(A, left) The inactive cAMP analog 8-Br-cAMPs (8-Br, brown) was used to prevent activation of PKA and KT5720 (blue), a small molecule that occupies PKA’s ATP-binding site, was used to inhibit phosphorylation of substrates by PKA. (A, right) Superimposed EPSCs before and after bath application of kynurenic acid (1 mM kyn, top) or NBQX (100 nM, bottom) in control slices and those incubated with KT5720 (1 µM) or 8-Br-cAMPs (50 µM) for 90–120 min. Experiments were performed in 2.5 mM Ca2+ and 1.3 mM Mg2+. Membrane potential has held between −10 and −20 mV to reduce the amplitude of CF-EPSCs. (B) Incubation with KT5720 or 8-Br-cAMPs increased the %inhibition by kyn (control: 26 ± 4%, KT5720: 47 ± 3%, 8-Br-cAMPs: 42 ± 2%; n = 10, 6, and 7, p=0.0004 and p=0.0048, ANOVA and Holm-Sidak post-tests). Incubation with KT5720 or 8-Br-cAMPs had no effect on % inhibition by NBQX (control: 34 ± 3%, KT5720: 33 ± 2%, 8-Br-cAMPs: 33 ± 5%; n = 9, 4, and 4; p=0.97, ANOVA). Asterisks denote statistical significance. (C, left) Representative traces in control (black) and following KT5720 incubation (blue). There was no change in the PPR (C, right) at any interstimulus intervals (Δt). PPR in control 0.07 ± 0.009, 0.09 ± 0.01, 0.12 ± 0.01, 0.17 ± 0.02, 0.35 ± 0.03, 0.90 ± 0.01, 0.98 ± 0.01 and in KT5720 0.08 ± 0.005, 0.1 ± 0.01, 0.13 ± 0.01, 0.17 ± 0.02, 0.35 ± 0.05, 0.90 ± 0.01, 0.98 ± 0.01 for interstimulus intervals of 10, 25, 100, 500, 4000, 8000 ms, respectively; n = 8 for each; p=0.8, repeated measures ANOVA. (D, left) Superimposed EPSCs before and after bath application of KYN (3 mM, red) in the indicated extracellular Ca2+ in control slices (black) or those incubated with KT5720 (blue). Scale bars: 10 pA, 10 ms; and 1 nA, 10 ms. (D, right) Semi-log plot of the inhibition by KYN (3 mM) as a function of extracellular Ca2+ in control (black) and KT5720-treated slices (blue). The Ca2+-insensitive inhibition by KYN determined from the best fit to a four-parameter dose response curve was 38.5% in control and 64.9% in KT5720-treated slices suggesting that cAMP/PKA signaling controls 43% of the total KYN inhibition. The orange diamond (1.5 mM Ca2+) denotes inhibition by 3 mM KYN in the presence of the PKA activator 6-Bnz.
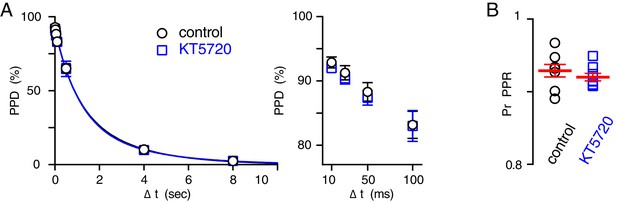
No change in PPR using a simple depletion model.
(A, left) Paired pulse depression (PPD = (1-PPR) x 100)) plotted as a function of ΔT in control (black) and KT5720-treated (blue) slices. Both data sets were fit with two-phase exponential decays. The fast decay time constant (28.2 ± 12.3 ms and 21.3 ± 10.2 ms, p=0.7), percent of fast decay (12.1 ± 2.4% and 12.6 ± 2.3%, p=0.9), and the slow decay time constant (1.9 ± 0.12 s and 1.8 ± 0.19 s, p=0.6) were similar in control and KT5720-treated slices (n = 8 each). (A, right) PPD at 10, 25, 50, and 100 ms ISIs. PPD in control: 92.9 ± 0.9, 91.3 ± 1.1, 88.3 ± 1.4, 83.2 ± 2.1, and in KT5720: 91.9 ± 0.5, 90.4 ± 0.9, 87.4 ± 1.1, 83.0 ± 2.4 10, 25, 50, and 100 ms interstimulus intervals, respectively, n = 8 for each. (B) Vesicular release probability (PrPPR) in control and KT 5720-treated slices estimated from PPR at 10 ms (0.93 ± 0.09 and 0.92 ± 0.05, respectively; p=0.4).
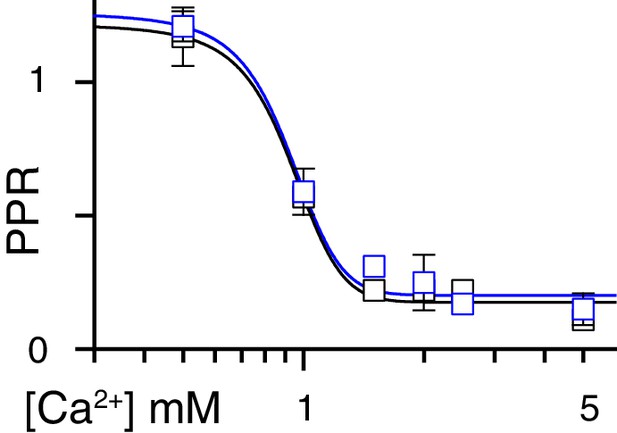
PKA inhibition and PPR changes with extracellular Ca2+.
The PPR of CF-PC EPSCs (50 ms ISI) was highly sensitive extracellular Ca2+ reflecting changes in Pr; however, PPR was not affected by KT5720 (p=0.0001 for changes with Ca2+ and p=0.3 for PKA inhibition, 2-way ANOVA). PPR in control: 1.17 ± 0.1, 0.57 ± 0.03, 0.22 ± 0.03, 0.22 ± 0.03, 0.22 ± 0.03, 0.11 ± 0.02 and in KT5720: 1.21 ± 0.04, 0.57 ± 0.05, 0.31 ± 0.01, 0.25 ± 0.06, 0.17 ± 0.01, 0.15 ± 0.03 for extracellular [Ca2+] of 0.5, 1, 1.5, 2, 2.5, 5 mM, respectively, n = 2–5).
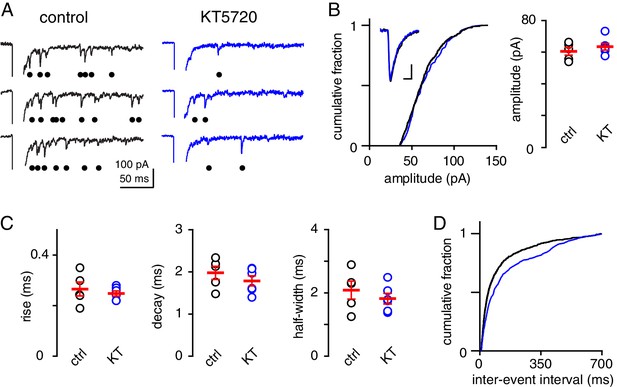
Inhibition of cAMP/PKA does not affect quantal content.
(A) Representative sweeps of Sr2+-evoked asynchronous EPSCs (aEPSCs) recorded from control (black) and KT5720-treated slices (blue). Bullets denote detected events. aEPSCs were recorded in ACSF containing 0.5 mM Ca2+, 3.3 mM Mg2+ and 1.0 mM Sr2+. (B, left) Lack of difference in the cumulative distribution of aEPSC amplitudes in control (black) and KT 5720-treated slices (blue; KS test, p=0.48). Inset shows superimposed example aEPSCs for each condition; scale bar: 20 pA and 2 ms and (B, right) is the summary data of average aEPSC amplitudes. Control: 60.6 ± 2.5 pA (n = 5) and KT5720: 63.3 ± 2.2 pA (n = 6; p=0.39). (C) Summary of the average aEPSC rise time (0.27 ± 0.3 ms and 0.25 ± 0.01 ms, n = 5 and 6; p=0.54), decay time constant (2.0 ± 0.2 ms and 1.8 ± 0.1 ms; p=0.35), and half-width (2.1 ± 0.3 ms and 1.8 ± 0.2 ms; p=0.4) in control (black) and KT5720-treated (blue) slices. (D) Distribution of aEPSC inter-event intervals (from 50 ms to 600 ms) in control (black) and KT5720-treated (blue) slices (p=0.0001, KS test).
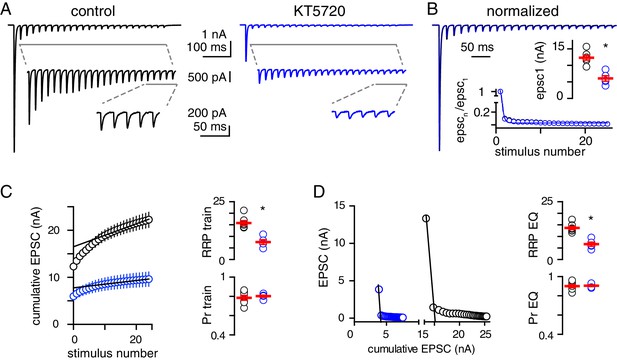
PKA inhibition reduces the size of the RRP.
(A) Representative EPSCs recorded in response to CF stimulation at 50 Hz for 500 ms in control (black) and KT5720-treated (blue) slices. To relieve receptor saturation, 3 mM KYN was added in a recording solution with 2.5 mM Ca2+; Vm = −60 mV). (B) Superimposed train responses from control (black) and KT5720-treated (blue) slices, normalized to the first EPSC. Inset1: Average EPSC1 amplitude in control and KT5720-treated slices. Inset2: Normalized EPSC amplitude plotted as a function of stimulus number. The time constants were similar between control (23.3 ± 0.6 ms, n = 7) and KT5720-treated slices (22.9 ± 0.4 ms, n = 5, p=0.6, unpaired t-test). (C, left) Cumulative EPSC amplitude plotted as a function of stimulus number for control (black) and KT5720-treated (blue) slices. A line was fit to the final 5 EPSCs in each condition. (C, right) The RRPtrain (15.8 ± 1.0 nA and 7.5 ± 1.0 nA, n = 7 and 5; p=0.0002, unpaired t-test) and Prtrain (0.78 ± 0.03 and 0.80 ± 0.05, n = 7 and 5; p=0.61, unpaired t-test) were calculated from this plot. (D, left) Representative plots of EPSC amplitude versus the cumulative EPSC in control (black) and KT5720-treated (blue) recording with linear regressions to the initial portion of each data set. (D, right) The RRPeq (13.7 ± 0.7 nA and 6.6 ± 0.9 nA, n = 7 and 5; p=0.0001, unpaired t-test) and Preq (0.90 ± 0.02 and 0.90 ± 0.01, n = 7 and 5; p=0.85, unpaired t-test) were calculated from this plot.
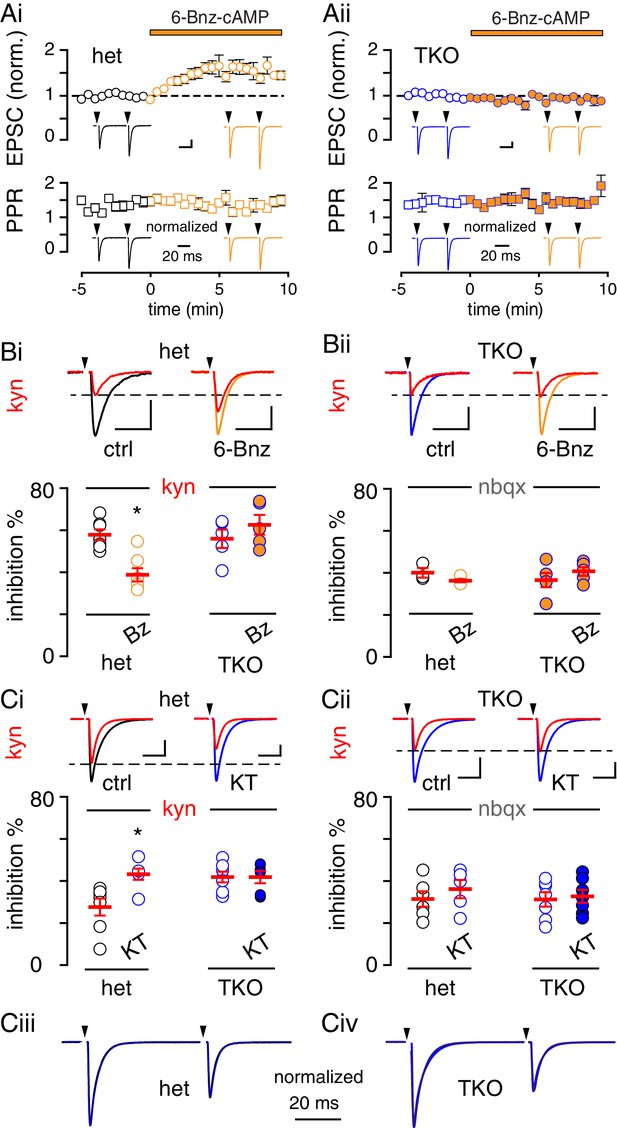
PKA-mediated regulation of MVR is absent in synapsin TKO mice.
(A) Time course of CF-PC EPSC amplitude (top: normalized, circles) and paired pulse ratio (bottom: PPR with an inter-stimulus interval = 50 ms, squares) following bath application of 6-Bnz (orange) from slices of synapsin triple het (Ai, black) or TKO (Aii, brown) mice recorded in 0.5 mM Ca2+. Insets: representative and normalized traces show the effects on amplitude and lack of change in PPR. Het-slices: EPSC from 0.91 ± 0.12 nA to 1.22 ± 16 nA, n = 7; p=0.008, paired t-test. PPR from 1.46 ± 0.07 to 1.42 ± 0.06; n = 7, p=0.54. TKO-slices EPSC from 0.96 ± 0.21 nA to 0.89 ± 0.22 nA, n = 6; p=0.82, paired t-test. PPR from 1.42 ± 0.09 to 1.49 ± 0.11, n = 6; p=0.06. Scale bars: 200 pA, 10 ms. (B) Top, blockade of EPSCs by KYN (red) in control (black) and 6-Bnz (orange) in slices from synapsin triple het (Bi, black) or TKO (Bii, blue) mice. Recordings were made in 0.5 mM Ca2+. Bottom, 6-Bnz reduced the KYN (0.25 mM) block in het slices (control: 57.9 ± 2.5%, n = 7; 6-Bnz: 38.2 ± 3.2, n = 7; p=0.003) but not in slices from TKO (control: 55.9 ± 4.4%, n = 5; 6-Bnz: 62.6 ± 4.7%, n = 5; p=0.56, ANOVA with Holm-Sidak post-test). NBQX was similar in all conditions (het slices: control: 40.7 ± 2.4% and 6-Bnz: 36.9 ± 0.8, n = 3 each; p=0.87. TKO slices: control: 37.2 ± 3.4% and 6-Bnz: 41.2 ± 2.2, n = 5 each; p=0.84. Scale bars: 1 nA, 10 ms (hets) and 500 pA, 10 ms (TKO). (C) Top, blockade of EPSCs by KYN (red) in control (black) and KT 5720-treated slices (blue) from synapsin triple het (Ci) or TKO (Cii) mice. Recordings were made in 2.5 mM Ca2+. Bottom, KT5720 increased the KYN (1 mM) block in het slices (control: 27.6 ± 4.0% and KT5720: 43.2 ± 2.7, n = 6 and 7, p=0.02), but there was no difference in KYN block in slices from TKO mice (control: 41.8 ± 2.5% and KT5720: 41.8 ± 2.9%, n = 6 each; p=0.99, 2-way ANOVA). NBQX blocked EPSCs to a similar extent in all conditions (het slices: control: 31.3 ± 3.6% and KT5720: 36.1 ± 4.3, n = 5 and 6, p=0.93; TKO slices: control: 31.2 ± 3.4% and KT5720: 32.7 ± 3.1, n = 7 each; p=0.4, ANOVA). Scale bars: 2 nA, 10 ms. (Ciii and Civ) KT5720 incubation had no effect on PPR in slices from either het mice (control: 0.67 ± 0.02, n = 11; KT5720: 0.62 ± 0.02, n = 8; p=0.75) or in TKO mice (control: 0.65 ± 0.04, n = 6; KT5720: 0.64 ± 0.03, n = 8; p=0.96).
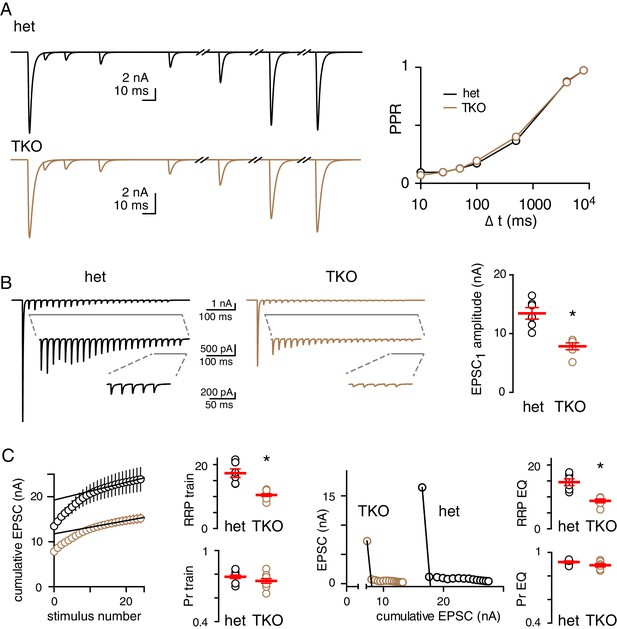
Synaptic release in TKO mice mimics inhibition of PKA signaling, with a smaller RRP but no change in Pr.
(A) The PPR of CF-EPSCs at multiple ISIs was unchanged in synapsin TKO mice. PPR in hets (black): 0.09 ± 0.02, 0.1 ± 0.01, 0.13 ± 0.01, 0.17 ± 0.02, 0.37 ± 0.01, 0.88 ± 0.02, 0.97 ± 0.01 and in TKOs (brown): 0.07 ± 0.01, 0.1 ± 0.01, 0.13 ± 0.02, 0.19 ± 0.02, 0.4 ± 0.02, 0.87 ± 0.03, 0.97 ± 0.01 for an ISI of 10, 25, 100, 500, 4000, 8000 ms, respectively; n = 8–10; (p=0.4, repeated measures ANOVA). Experiments were done in 2.5 mM Ca2+ with 3 mM KYN at Vm = −60 mV. (B) High frequency train stimulation (50 Hz, 25 stimuli) generated similar depression both genotypes (left), however, the peak EPSC amplitude was smaller in TKO mice was smaller compared to their het littermates (hets: 13.5 ± 1.0 nA and TKO: 7.8 ± 0.6 nA; p=0.0006, n = 6). (C) Two methods of estimating the RRP (as described in Figure 5) both suggested smaller RRP in TKO mice with no change in Pr. RRPtrain: hets 17.3 ± 1.3 nA and TKO 10.5 ± 0.6 nA, n = 6, p=0.0009; RRPEQ: hets 14.6 ± 1 nA and TKO 8.7 ± 0.6, n = 6; p=0.0006. Prtrain: hets 0.77 ± 0.02 and TKO 0.74 ± 0.01 nA, n = 6, p=0.39; PrEQ: hets 0.92 ± 0.01 and TKO 0.89 ± 0.01, n = 6; p=0.15.
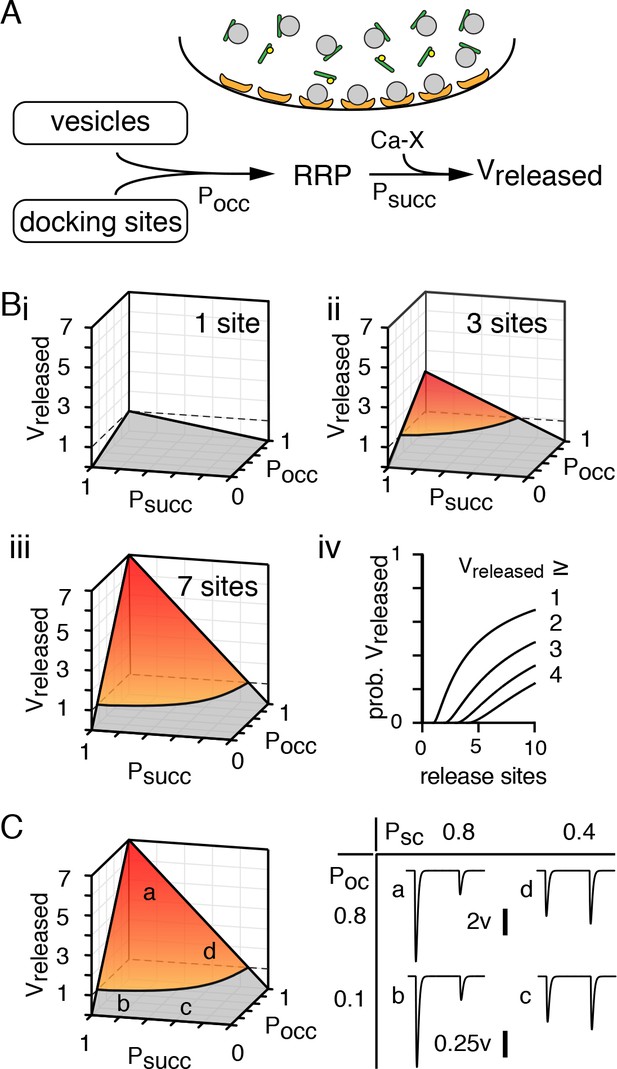
Synapsin-mediated regulation of the RRP controls MVR independent of Pr.
(A) Cartoon and schematic of short-term plasticity model showing how synapsin controls the number of release-competent sites per active zone. Synaptic vesicles (gray) are bound by synapsin (green) restricting the size of the readily-releasable pool of vesicles that is limited by the number of release sites (orange). Synapsin phosphorylation (yellow) allows vesicles to become part of the RRP upstream from subsequent Ca2+-dependent facilitation and recovery from depression as in Dittman et al. (2000) that dictate Vreleased. Although, we have illustrated that synapsin phosphorylation controls vesicle docking, its influence can also be exerted by priming already docked vesicles. (Bi - iii) The number of vesicles released per active zone (Vreleased) is shown with increasing number of release sites per active zone and is the product of the probability of site occupancy (Pocc), the probability that a competent vesicle will release (Psucc = F*D), and number of release sites (NT). The product of these parameters steepens with increasing number of release sites. (Biv) The probability of observing more than 1–4 vesicles follow a logarithmic function (see Materials and methods). (C) Vreleased and simulated EPSCs in four conditions (points a - d) show that the number of vesicles released (Vreleased) does not require a high probability that a competent vesicle will release (Psucc) and that short-term plasticity does not predict release mode.
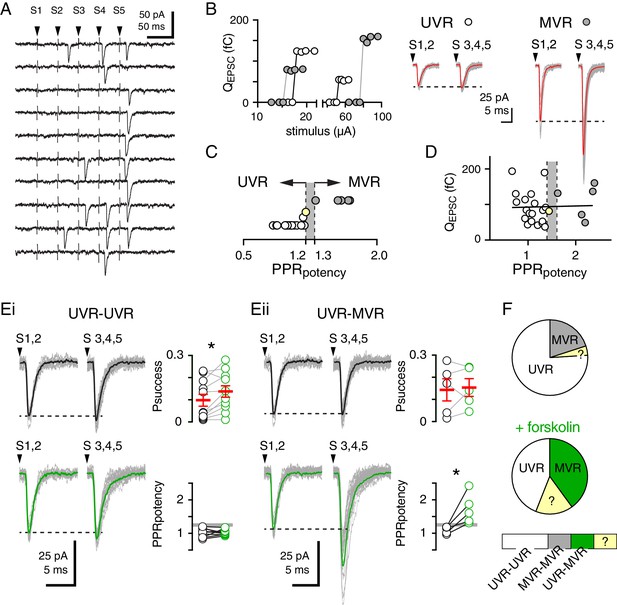
Fsk increases MVR at PF-MLI synapses independent of Pr.
(A) Unitary responses following five parallel fiber stimuli (S1– S5) delivered at 50 Hz. (B left) Individual parallel fibers were identified by a sharp threshold between failures and successes (S1 and S2) with increasing stimulus intensity. Four representative experiments demonstrating that all-or-none responses at UVR (open circles) or MVR (grayed circles) synapses. Classification as UVR does not depend on stimulus intensity or synaptic charge (also see (D)). (B middle) The potency of UVR synapses in final three stimuli (S3, 4, 5) was similar to that of the first two stimuli (S1, 2). (B right) The potency of MVR synapses increased later in the train. Superimposed individual successful responses are shown in gray and average responses are shown in red. (C) The ratio of the potencies of S3, 4, 5 to S1, 2 (PPRpotency) was used to classify synapses as UVR (0.8–1.2) or MVR (>1.3). Synapses with PPRpotencies between 1.2 and 1.3 (gray area) were excluded from further analysis. (D) Lack of correlation between release mode (PPRpotency) and the size of the response to S1,2 (R2 = 0.001; p=0.9). (Ei) In a subset of cells, fsk (50 µM; green) increased the probability of success (Psuccess of S1-2 stimuli: 0.10 ± 0.03 vs. 0.14 ± 0.03; p=0.01, paired t-test, n = 11) of UVR synapses with no effect on release mode (PPRpotency: 1.03 ± 0.04 vs 1.03 ± 0.2; p=0.9, paired t-test, n = 11). Individual responses are shown in gray and average responses before or following fsk are shown in black or green, respectively. (Eii) In another subset of cells, fsk increased PPR potency from 1.1 ± 0.05 to 1.7 ± 0.2 (p=0.03, paired t-test, n = 5), changing their classification from UVR to MVR without altering Psuccess (0.14 ± 0.05 vs. 0.15 ± 0.04, p=0.74, paired t-test, n = 5). Individual responses are shown in gray and average responses before or following fsk are shown in black or green, respectively. (F) Summary data showing that in control conditions (top pie chart), 19/25 synapses were classified as UVR, 5/25 were MVR, and 1/25 could not be classified. Following fsk (bottom pie chart), 11/25 synapses were UVR, 10/25 were MVR, and four could not be classified. Grouped data (bottom) showing the classification of synaptic responses before and after fsk treatment: 11/25 were unchanged from UVR (UVR-UVR), 5/25 were converted from UVR to MVR, five were unchanged from MVR (MVR-MVR), and four could not be classified in one or both conditions because PPRpotency was neither <1.2 nor >1.3.
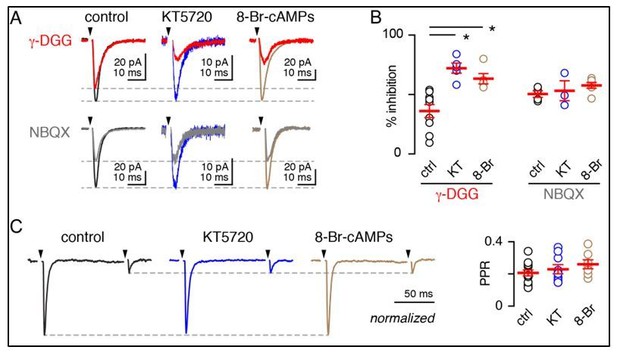
PKA inhibition reduces the glutamate transient underlying spillover response onto MLIs in mice aged P21-P28.
(A) Superimposed CF-MLI spillover EPSCs before and after bath application of γ-DGG (300 µM) or NBQX (200 nM) in control slices and after incubation with KT5720 (1 µM) or 8-Br-cAMPs (50 µM) for 90-120 min. (B) KT5720 and 8-BrcAMPs increased sensitivity to γDGG (control: 36.1% ± 5.5, n = 9; KT5720: 72.1% ± 4.2, n = 5, p = 0.0006; Rp-8-Br-cAMPs: 63.6% ± 4.3, n = 5, p = 0.0044), but not NBQX (control: 50.4 ± 2.8, n = 4; KT5720: 53.2 ± 8.4, n = 3, p = 0.71; Rp-8-Br-cAMPs: 57.78% ± 2.5, n = 5, p = 0.49). One-way ANOVA and Holm-Sidak post-tests. (C, left) Normalized representative EPSCs in response to paired stimuli (100 ms) in control-, KT5720-, and 8-Br-cAMPs-treated slices. (C, right)Summary of PPR in control- (0.21 ± 0.02, n = 12), KT5720- (0.23 ± 0.03, n = 7, p > 0.99) and 8-BrcAMPs-treated slices (0.26 ± 0.03, n = 7, p >0.99). One-way ANOVA and Holm-Sidak post-tests.
Tables
| Reagent type (species) or resource | Designation | Source or reference | Identifiers | Additional information |
|---|---|---|---|---|
| Gnetic reagent (M. musculus) | wildtype; WT; control | Jackson Laboratories | RRID:IMSR_JAX:000664 | |
| Genetic reagent (M. musculus) | synapsin triple knockout, TKO | MMRC | RRID:MMRRC_041434-JAX | |
| Genetic reagent (M. musculus) | synapsin triple het; het | this paper | WT x TKO cross | |
| Peptide, recombinant protein | PKA inhibitory fragment (6-22) amide, PKi | Tocris | Cat#: 1904; CAS: 121932-06-7 | |
| Chemical compound, drug | 6-Bnz-cAMP; 6-Bnz | BioLog via Axxora | Cat#: B009; CAS: 30275-80-0 | |
| Chemical compound, drug | 8-Br-cAMPs; 8-Br | Santa Cruz | Cat#: B009; CAS: 30275-80-0 | |
| Chemical compound, drug | forskolin; fsk | HelloBio | Cat#: HB1348; CAS: 66575-29-9 | |
| Chemical compound, drug | KT5720; KT | Tocris | Cat#: 1288; CAS: 108068-98-0 | |
| Chemical compound, drug | kynurenic acid, KYN | Abcam | Cat#: ab120256; CAS: 494-27-3 | |
| Chemical compound, drug | NBQX | Abcam | Cat#: ab120045; CAS: 118876-58-7 | |
| Chemical compound, drug | Picrotoxin | Abcam | Cat#: ab120315; CAS: 124-87-8 | |
| Chemical compound, drug | QX-314 | Abcam | Cat#: ab120118; CAS: 5369-03-9 | |
| Software, algorithm | Axograph X, version 1.5.4 | AxoGraph Scientific | https://axograph.com/ | |
| Software, algorithm | Mathematica 11 | Wolfram | http://www.wolfram.com/mathematica/ | |
| Software, algorithm | pCLAMP 10 | Molecular Devices | https://www.moleculardevices.com/ | |
| Software, algorithm | Prism | Graphpad | https://www.graphpad.com/ |
Parameters used in FD2 model.
https://doi.org/10.7554/eLife.47434.018| Symbol | Definition | |
|---|---|---|
| CaXF0 | Concentration of Ca2+-bound site XF | 0 |
| CaXD0 | Concentration of Ca2+-bound site XD | 0 |
| ΔF | Incremental increase in CaXF after a stimulus | 5 |
| ΔD | Incremental increase in CaXD after a stimulus | 0.001 |
| τF | Decay time constant of CaXF after an action potential | 0.1 sec−1 |
| τD | Decay time constant of CaXD after an action potential | 0.05 s |
| KF | Affinity of CaXF for site | 2 |
| KD | Affinity of CaXD for site | 2 |
| k0 | Baseline rate of recovery from recovery state | 0.7 sec−1 |
| kmax | Maximal rate of recovery from refractory state | 20 sec−1 |
| D | Fraction of sites that are release-ready | 1 |
| F | Facilitation probability | (0–1) |
| NT | Total number of sites | (1 - 10) |
| Pocc | Probability of site occupancy | (0–1) |
| RRP | Readily releasable pool | NT * Pocc |
| Psucc | Probability that a competent vesicle will release | F * D |
Additional files
-
Transparent reporting form
- https://doi.org/10.7554/eLife.47434.019





