The C-terminal tail of the bacterial translocation ATPase SecA modulates its activity
Figures
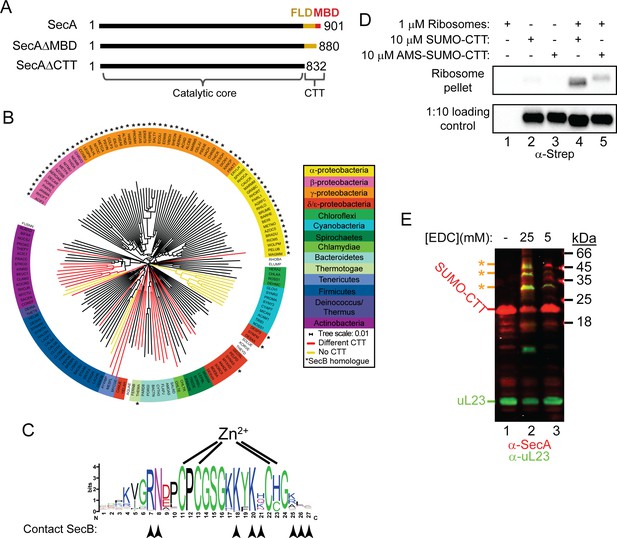
Phylogenetic analysis of the CTT and binding of E. coli CTT to the ribosome.
(A) Schematic diagram of the primary structure of SecA, SecAΔMBD and SecAΔCTT. Structures are oriented with the N-termini to the left, and the amino acid positions of the N- and C-termini are indicated. Residues of the catalytic core and the CTT are indicated below. Catalytic core, black; FLD, yellow; MBD, red. (B) Phylogenetic tree of the SecA proteins of 156 representative species from 155 different bacterial families. Species names are given as the five-letter organism mnemonic in UniProtKB (The UniProt Consortium, 2017). Taxonimical classes are colour-coded according to the legend. Leaves representing SecA proteins with an MBD are coloured black. Those with CTTs lacking a MBD are coloured red, and those that lack a CTT entirely are coloured yellow. Species that also contain a SecB protein are indicated with a star (*). (C) Logo of the consensus sequence of the MBD generated from the 117 species containing the MBD in the phylogenetic analysis. Positions of the metal-coordinating amino acids are indicated above. Amino acids that contact SecB in the structure of the MBD-SecB complex (Zhou and Xu, 2003) (1OZB) are indicated by arrowheads below. (D) Binding reactions containing 1 μM ribosomes, 10 μM SUMO-CTT and 10 μM AMS-modified SUMO-CTT (AMS-SUMO-CTT) were equilibrated at room temperature and layered on a 30% sucrose cushion. Ribosomes were then sedimented through the cushion by ultracentrifugation. Samples were resolved on SDS-PAGE and probed by western blotting against the Strep tag using HRP-coupled Streptactin. (E) 10 μM SUMO-CTT containing an N-terminal Strep(II)-tag was incubated with 1 μM purified ribosomes and treated with 5 mM or 25 mM EDC, as indicated. Samples were resolved by SDS-PAGE and analysed by western blotting by simultaneously probing against SecA (red) and ribosomal protein uL23 (green). The positions of SUMO-CTT, L23 and crosslinking adducts between them (*) are indicated at left.
-
Figure 1—source data 1
Clustal Omega alignment of SecA proteins used to construct phylogenetic tree in Figure 1.
- https://doi.org/10.7554/eLife.48385.005
-
Figure 1—source data 2
Phylogenetic tree data generated by Clustal Omega used to construct Figure 1B and C.
- https://doi.org/10.7554/eLife.48385.006
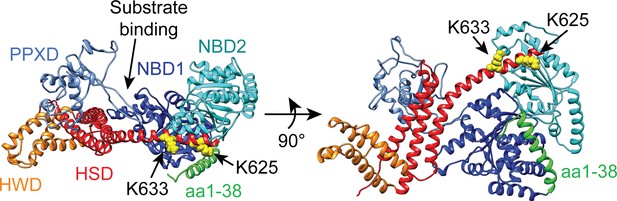
Structural model of the catalytic core of SecA in the ‘closed’ conformation.
Structural model of E. coli SecA from PDB file 2VDA (Gelis et al., 2007) in ribbon diagram. The model is coloured according to domains described in the main text. NBD1, dark blue; NBD2 cyan; PPXD, light blue; HSD, red; HWD, orange. The side chains of lysines 625 (K625) and 633 (K633), which were identified by Huber et al. (2011) to be involved in ribosome binding, are depicted in space-fill. The N-terminal α-helix (aa1-38), which was identified by Singh et al. (2014) to be involved in ribosome binding is coloured green. The CTT is not resolvable in high-resolution structures of SecA and is therefore not depicted.

SUMO-MBD cosediments with ribosomes.
The C-terminal 27 amino acids of SecA were fused to the C-terminus of Strep-tagged SUMO (SUMO-MBD). 5 μM SUMO-MBD was incubated in the presence or absence of 1 μM 70S ribosomes. Binding reactions were layered on a 30% sucrose cushion and subjected to ultracentrifugation to sediment ribosomes. The pellet fractions were then resolved by SDS-PAGE and analysed by western blotting against the Strep tag.
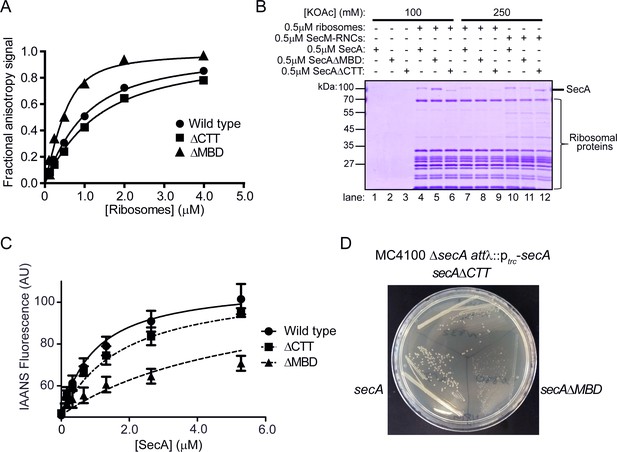
Effect of C-terminal truncations on SecA function in vitro and in vivo.
(A) 900 nM Ru(bpy)2(dcbpy)-labelled SecA (Wild type; circles), SecAΔMBD (ΔMBD; triangles) or SecAΔCTT (ΔCTT; squares) was incubated in the presence of increasing concentrations of purified 70S ribosomes. Because error bars corresponding to one standard deviation obscured the symbols, they were omitted from the graph. The equilibrium dissociation constant (KD) of the complex was determined by fitting the increase in fluorescence anisotropy from the Ru(bpy)2(dcbpy) (lines; Table 1). (B) 0.5 μM SecA, SecAΔMBD or SecAΔCTT was incubated in the absence (lanes 1–3) of ribosomes, in the presence of 0.5 μM vacant 70S ribosomes (lanes 4–9) or in the presence of 0.5 μM RNCs containing nascent SecM peptide (lanes 10–12). Where indicated, binding reactions were incubated in the presence of 100 mM (lanes 1–6) or 250 mM (lanes 7–12) potassium acetate (KOAc). Binding reactions were layered on a 30% sucrose cushion and ribosomes were sedimented through the sucrose cushion by ultracentrifugation. Ribosomal pellets were resolved by SDS-PAGE and stained by Coomassie. (C) 600 nM IAANS-VipB peptide was incubated with increasing concentrations of SecA (Wild type; circles), SecAΔMBD (ΔMBD; triangles) or SecAΔCTT (ΔCTT; squares). Confidence intervals represent one standard deviation. The KD for the SecA-peptide complex was determined by fitting the increase in IAANS fluorescence upon binding to SecA (lines; Table 1). (D) Growth of strains producing SecA (DRH1119; bottom left), SecAΔMBD (DRH1120; bottom right) and SecAΔCTT (DRH1121; top) on LB plates containing 100 μM IPTG.
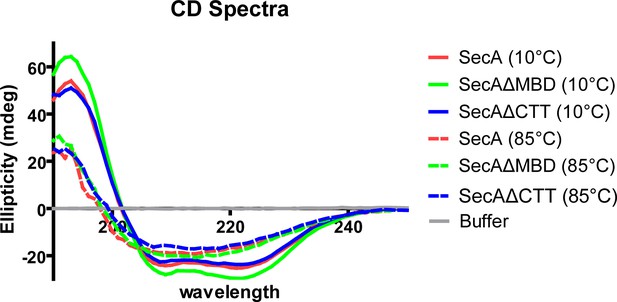
CD spectra of SecA, SecAΔMBD and SecAΔCTT.
Far-UV circular dichroism (CD) spectra of 2 µM solutions of SecA, SecAΔMBD, and SecAΔCTT in 10 mM potassium phosphate (pH 7.5).
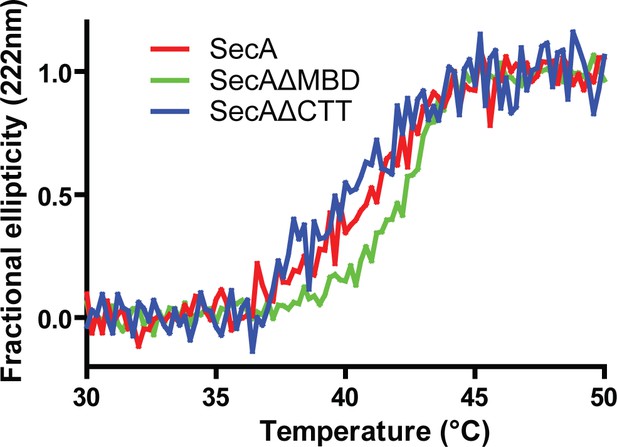
Thermal denaturation plots of SecA, SecAΔMBD and SecAΔCTT.
Representative plot of the thermal denaturation of SecA as determined by CD spectroscopy. The α-helical content of 2 µM solutions of SecA, SecAΔMBD, and SecAΔCTT in 10 mM potassium phosphate (pH 7.5) was determined by measuring molar ellipticity at 222 nm while the temperature of the solution was raised from 30°C to 50°C. The TMs listed in Table 1 were determined by van’t Hoff analysis.

Expression of SecA, SecAΔMBD and SecAΔCTT in strains DRH1119, DRH1120 and DRH1121.
Strains DRH1119, DRH1120 and DRH1121 (relevant genotype: MC4100 ΔsecA attλ-placUV5-secA), which produce SecA, SecAΔMBD and SecAΔCTT, respectively, were grown in LB in the presence of 100 μM IPTG to mid-log phase. Cell lysates were normalised to cell density, resolved by SDS-PAGE and probed by western blotting using antisera against SecA and thioredoxin-1, as a loading control.
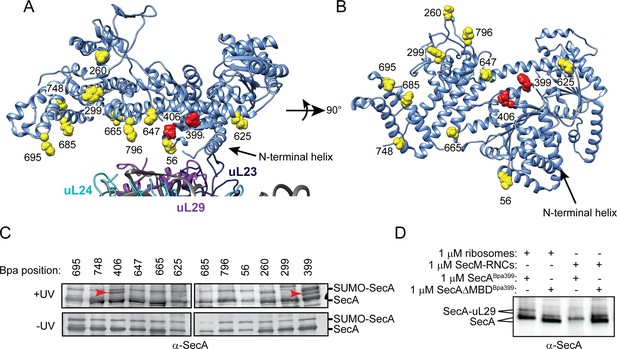
Site-specific crosslinking of SecA to purified ribosomes and ribosome-nascent chain complexes.
(A and B) Sites of incorporation of Bpa in the structure of E. coli SecA. (A) Fit of the high resolution structure of SecA (PDB code 2VDA [Gelis et al., 2007]) and the 70S ribosome (PDB code 4V4Q [Schuwirth et al., 2005]) to the cryoEM structure of the SecA ribosome complex (EMD-2565 [Singh et al., 2014]). (B) View of SecA from the ribosome-interaction surface. Amino acid positions where Bpa was incorporated are represented in space fill (yellow). Positions that crosslink to ribosomal proteins are coloured red. The locations of the N-terminal α-helix of SecA and of ribosomal proteins uL23 (dark blue), uL29 (purple) and uL24 (cyan) are indicated. Structural models were rendered using Chimera v. 1.12 (Pettersen et al., 2004). (C) Bpa-mediated photocrosslinking of SecA variants to vacant 70S ribosomes. 1 μM purified ribosomes were incubated with 1 μM SecA containing BpA at the indicated position and exposed to light at 365 nm (above) or incubated in the dark. Crosslinking adducts consistent with the molecular weight of a covalent crosslink to ribosomal proteins are indicated with red arrowheads. The positions of full-length SecA and uncleaved SUMO-SecA protein are indicated to the right. (D) 1 μM SecABpa399 or SecAΔMBDBpa399 was incubated with 1 μM non-translating 70S ribosomes or 1 μM arrested RNCs containing nascent SecM (SecM-RNCs) and exposed to light at 365 nm. The positions of full-length SecA and the SecA-uL29 crosslinking adduct are indicated. In (C and D), samples were resolved using SDS-PAGE and probed by western blotting using anti-SecA antiserum. LC-MS/MS analysis of the high-molecular weight bands produced by SecABpa399 and SecAΔMBDBpa399 in the presence of vacant 70S ribosomes indicated that they contained both SecA and ribosomal protein uL29.

Crosslinking of SecABpa399 to RNCs containing arrested nascent full-length SecM and MBP.
1 μM SecABpa399 was incubated with 1 μM non-translating 70S ribosomes (vacant) or RNCs containing arrested nascent SecM (SecM-RNCs) or maltose binding protein (MBP-RNCs). Where indicated, samples were exposed to light at 365 nm (UV). Samples were then resolved using SDS-PAGE and probed by western blotting against SecA. The positions of SecA and the crosslinking adduct between SecA and ribosomal protein uL29 are indicated.
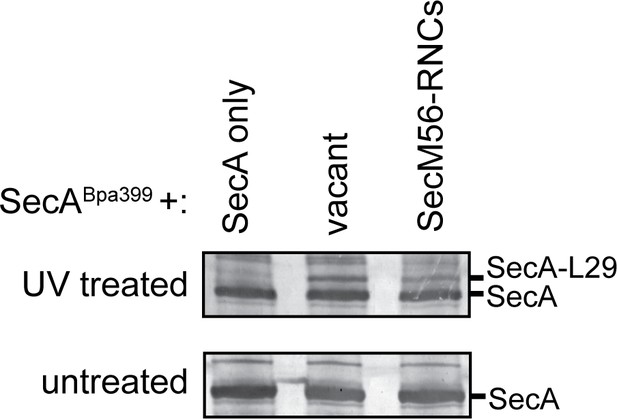
Crosslinking of SecABpa399 to RNCs containing arrested nascent chains with different lengths.
1 μM SecABpa399 was incubated with 1 μM non-translating 70S ribosomes (vacant) or RNCs containing arrested nascent SecM, which was internally truncated between the signal sequence and the translation arrest sequence (SecM56-RNCs). Previous studies indicate that SecM56 does not promote salt-resistant binding of SecA to the ribosome (Huber et al., 2017). After incubation, samples were exposed to light at 365 nm (UV treated) or incubated in the dark (untreated). Samples were then resolved using SDS-PAGE and probed by western blotting against SecA. The positions of SecA and the crosslinking adduct between SecA and ribosomal protein uL29 are indicated.
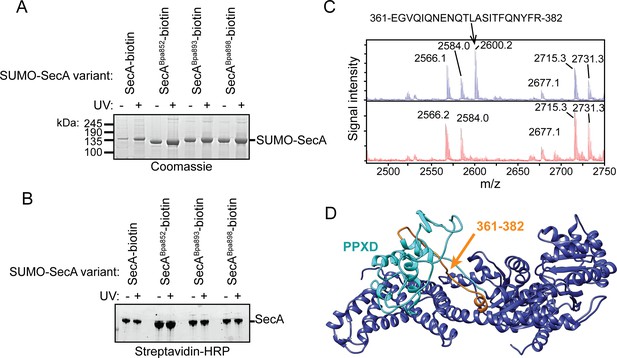
Auto-crosslinking of the CTT to the catalytic core of SecA.
(A and B) 1 μM SUMO-tagged SecA-biotin containing Bpa at position 852, 893 or 898 in the CTT was incubated in the absence (-) or presence (+) of UV light at 365 nm. The protein samples were resolved using SDS-PAGE and visualised by (A) Coomassie staining or (B) western blotting against the C-terminal biotin tag. The positions of full-length SUMO-SecA is indicated. (C) Mass spectra of wild-type SecA-biotin (above, blue) and SecABpa852-biotin (below, red) in the region of 2450–2750 Da region. Wild-type SecA-biotin and SecABpa852-biotin were exposed to light at 365 nm and subsequently digested with trypsin. The masses of the tryptic fragments were determined using MALDI-TOF. (D) Structure of SecA (2VDA [Gelis et al., 2007]). The main body of the catalytic core is coloured blue, the PPXD is coloured cyan and the tryptic peptide that crosslinks to position 852 (amino acids 361–382) is highlighted in orange. The structural model was rendered using Chimera v. 1.12 (Pettersen et al., 2004).

C-terminal purification of SecA-biotin and SecABpa852-biotin by the C-terminal biotin.
Lysates of cells producing SecA-biotin (DRH854) or SecABpa852-biotin (DRH1166) were incubated in the dark (-) or exposed to light at 365 nm (+) for 30 min. The biotinylated protein was purified using streptavidin-coated magnetic beads and resolved on a BioRad Stain-free gel. The position of the faster migrating band that is isolated by N-terminal affinity purification is indicated (*). An additional high molecular weight band, which is consistent with the weight of dimeric SecA, is also indicated (**).
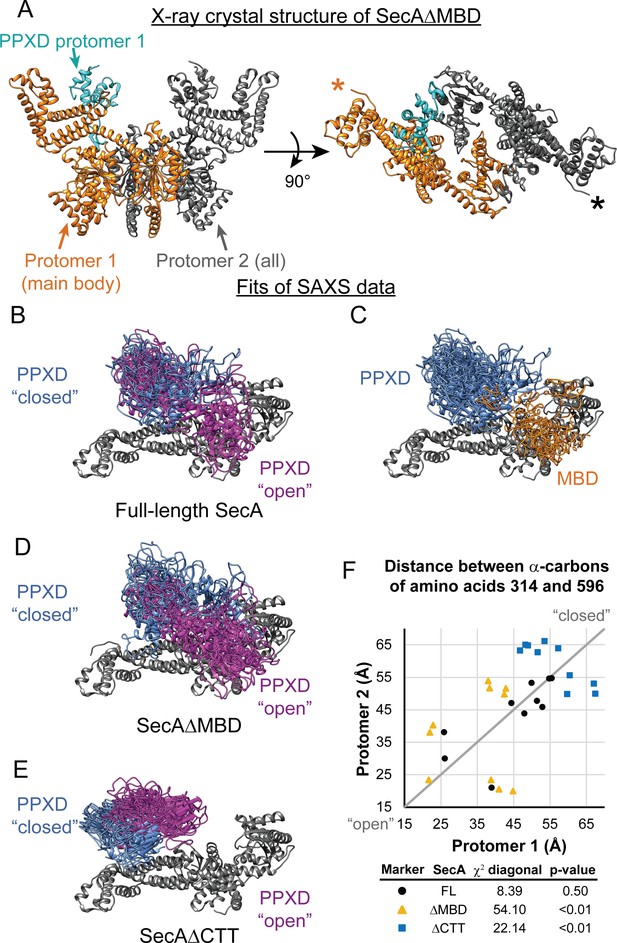
SAXS analysis of SecA truncation variants.
(A) X-ray crystal structure of SecAΔMBD at 3.5 Å solved by molecular replacement. The main body of the catalytic core in the asymmetric unit (Protomer 1) is coloured orange with the PPXD highlighted in cyan. The crystallographic mate (Protomer 2) interacts with promoter one using an interface similar to that found in 2FSG (Papanikolau et al., 2007), suggesting that this is the dimer interface of the purified protein in solution. The position of the most C-terminal residue that could be resolved (proline 834) is noted with an asterisk in the right panel. (B–E) Overlay of 10 independent structural models of SecA (B, C), SecAΔMBD (D) and SecAΔCTT (E) generated from fitting to the SAXS data using CORAL. The main body of the catalytic core is coloured grey, and the flexible residues are not displayed. (B, D, E) To facilitate visualization of the asymmetry in the in the dimeric models, both protomeric partners of the dimer were overlaid and the PPXD was coloured (blue/magenta) according to the protomer. The MBD is not displayed in panel B. (C) To facilitate visualization of the position of the MBD in the full-length protein, both protomeric partners of the dimer were overlaid and the MBD of the dimer pair that was located nearest to position 596 of the depicted protomer (orange) was displayed. In panel C, the PPXDs of two protomers, which occupy the same space as the MBDs, are not displayed. (F) Plot of the position of the PPXD in partners of the SecA dimer predicted by structural modelling. The distance between the α-carbon of amino acid 314, which is located near the centroid of the PPXD, and amino acid 596 in NBD2 was determined for each protomer and plotted against the distance in the second protomer. SecA, black circles (FL); SecAΔMBD, orange triangles (ΔMBD); SecAΔCTT, blue squares (ΔCTT). The grey diagonal line indicates the position of the distances if the dimers were symmetric. χ2 values to the diagonal were calculated and used to determine p-values to test whether the asymmetry in the dimer was statistically significant.
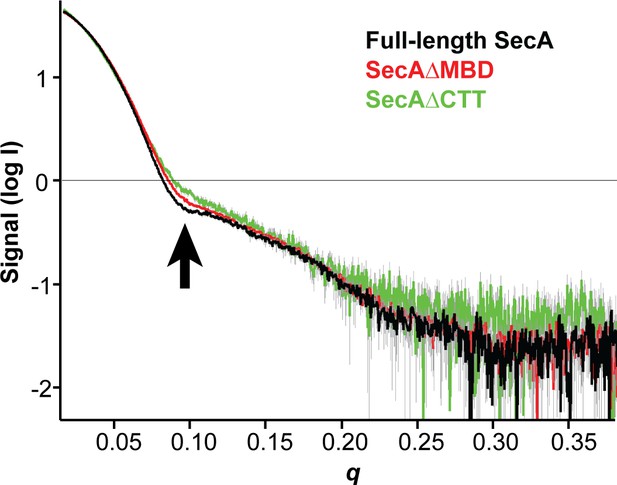
SAXS analysis of the solution structure of SecA, SecAΔMBD and SecAΔCTT.
X-ray scattering plots for SecA (black), SecAΔMBD (red) and SecAΔCTT (green). The region of divergence between the three SAXS traces in the mid-q region is indicated (black arrow).
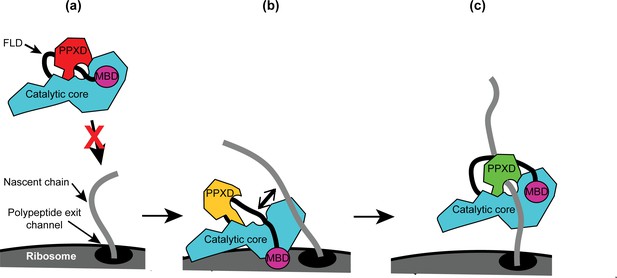
Diagram of the proposed mechanism for recognition of nascent substrate proteins by SecA.
(a) In solution, SecA occupies an autoinhibited conformation with the FLD bound stably in the substrate protein binding site and the PPXD in the open conformation. (b) Binding of both the catalytic core and the MBD to the ribosomal surface causes the PPXD to shift to the open conformation, which destabilises binding of the FLD and allows SecA to sample nascent polypeptides. (c) Binding to the nascent substrate protein displaces the FLD from the substrate protein binding site and the PPXD returns to the open conformation, stabilising this interaction. Binding to nascent substrate releases SecA from the ribosomal surface.
Tables
Biochemical properties of wild-type and mutant SecA proteins.
https://doi.org/10.7554/eLife.48385.011| SecA variant | KD Ribosomes* | KD VipB† | Basal ATPase activity‡ | TM§ |
|---|---|---|---|---|
| Wild type | 640 ± 33 nM | 0.9 μM | 0.053 ± 0.02 s−1 | 40.7 ± 0.09°C |
| SecAΔMBD | 160 ± 35 nM | 1.7 μM | <0.001 s−1 | 42.0 ± 0.08°C |
| SecAΔCTT | 920 ± 38 nM | 5.9 μM | 0.91 ± 0.02 s−1 | 40.0 ± 0.1°C |
| SecAC885A/C887A | ND¶ | >10 μM | <0.001 s−1 | ND¶ |
| SecABpa852 | ND¶ | >10 μM | <0.001 s−1 | ND¶ |
-
*Equilibrium dissociation constant of the complex between SecA and non-translating 70S ribosomes as determined by fluorescence anisotropy. Confidence intervals are the standard error of the fit.
†Equilibrium dissociation constant of the complex between SecA and IAANS-labelled VipB peptide as determined by change in fluorescence.
-
‡Rate of ATP hydrolysis by SecA in the absence of substrate protein and SecYEG
§Denaturation midpoint temperature as determined by the change in circular dichroism at 222 nm.
-
¶not determined
| Reagent type (species) or resource | Designation | Source or reference | Identifiers | Additional information |
|---|---|---|---|---|
| Strain, strain background (Escherichia coli K-12) | MC4100 | Casadaban, 1976 | F- araD139 DlacU169 rpsL150 thi rbsR | |
| Strain, strain background (Escherichia coli K-12) | DRH1119 | This paper | MC4100 ΔsecA::KanR λatt::placUV5-secA | |
| Strain, strain background (Escherichia coli K-12) | DRH1120 | This paper | MC4100 ΔsecA::KanR λatt::placUV5-secAΔMBD | |
| Strain, strain background (Escherichia coli K-12) | DRH1121 | This paper | MC4100 ΔsecA::KanR λatt::placUV5-secAΔCTT | |
| Strain, strain background (Escherichia coli K-12) | DRH663 | This paper | MC4100 ΔsecA::KanR + pDH663 | |
| Strain, strain background (Escherichia coli B) | BL21(DE3) | Lab stock | Escherichia coli Genetic Stock Center (CGSC), Yale University, USA. CGSC#: 12504 http://cgsc2.biology.yale.edu/Strain.php?ID=139459 | MC4100 ΔsecA::KanR + pDH663 |
| Strain, strain background (Escherichia coli B) | DRH584 | This paper | BL21(DE3) containing plasmid pDH584 | |
| Strain, strain background (Escherichia coli B) | DRH1166 | This paper | BL21(DE3) containing plasmid pDH1166 and pSup-Bpa-6TRN | |
| Genetic reagent (phage λ) | lambda InCh | Boyd et al., 2000 | ||
| Antibody | Rabbit anti-SecA antiserum | Huber et al., 2011 | (1:20000) | |
| Antibody | Sheep anti-uL23 antiserum | other | Gift from R. Brimacombe (1:2500) | |
| Antibody | Rabbit anti-thioredoxin-1 antiserum | Sigma-Aldrich (St. Louis, MO, USA) | Catalogue number: T0803 | (1:10000) |
| Antibody | Goat IR700-labelled anti-rabbit | Rockland (Philadelphia, PA, USA) | Catalogue number: 611-130-122 | Discontinued (1:5000) |
| Antibody | Goat IR800-labelled anti-sheep | Rockland (Philadelphia, PA, USA) | Catalogue number: 613-445-002 | (1:10000) |
| Antibody | Donkey HRP-labelled anti-rabbit | GE Healthcare | Catalogue number: NA934V | (1:10000) |
| Recombinant DNA reagent | pCA528 | Andréasson et al., 2010 | pET24 expression vector containing gene encoding His-tagged SUMO protein | |
| Recombinant DNA reagent | pCA597 | Andréasson et al., 2010 | pET24 expression vector containing gene encoding Strep-tagged SUMO protein | |
| Recombinant DNA reagent | pDH543 | This paper | pCA597 containing portion of secA gene corresponding to amino acids829–901 | |
| Recombinant DNA reagent | pDH934 | This paper | pCA597 containing portion of secA gene corresponding to amino acids 875–901 | |
| Recombinant DNA reagent | pDH625 | Huber et al., 2011 | pCA528 containg the secA gene | |
| Recombinant DNA reagent | pDH584 | Huber et al., 2011 | pCA528 containg the secA-biotin gene | |
| Recombinant DNA reagent | pDH1166 | This paper | pDH584 containing amber codon at position corresponding to amino acid 852 in SecA. | |
| Recombinant DNA reagent | pDSW204 | Weiss et al., 1999 | pTrc99a-derived plasmid containing partially disabled trc promoter | |
| Recombinant DNA reagent | pDH692 | Huber et al., 2011 | pDSW204-derived plasmid producing SecA under control of an IPTG-inducible promoter | |
| Recombinant DNA reagent | pDH939 | This paper | pDSW204-derived plasmid producing SecAΔMBD under control of an IPTG-inducible promoter | |
| Recombinant DNA reagent | pDH663 | Huber et al., 2011 | pTrc99b-derived plasmid producing SecA under control of an IPTG-inducible promoter and containing a Spectinomycin resistance gene in place of the bla (ampicillin resistance) gene | |
| Recombinant DNA reagent | pDH787 | Huber et al., 2017 | pCA597 containing full-length secM gene | |
| Recombinant DNA reagent | pDH784 | Huber et al., 2017 | pCA597 containing secM with internal deletion between region encoding signal sequence and the translation arrest sequence | |
| Recombinant DNA reagent | pDH894 | Huber et al., 2017 | pCA528 containing gene encoding malE gene that is translationally fused to sequence encoding the SecM translation arrest sequence | |
| Recombinant DNA reagent | pSup-Bpa-6TRN | Chin et al., 2002 | Plasmid producing orthologous tRNA and tRNA synthetase required for incorporation of Bpa | |
| Peptide, recombinant protein | VipB peptide | Pietrosiuk et al., 2011 | ||
| Peptide, recombinant protein | HRP-coupled Streptactin | IBA Life Sciences (Goettingen, Germany) | Catalogue number: 2-1502-001 | |
| Chemical compound | 4-acetamido-4′-maleimidylstilbene-2,2′-disulfonic acid (AMS) | Invitrogen (Carlsbad, California) | Catalogue number: A485 | |
| Chemical compound, drug | Hydrophilic streptavidin magnetic beads | New England Biolabs (Ipswich, Massachusetts) | Catalogue number: S1421S | |
| Chemical compound, drug | Benzophenylalanine (Bpa) | Bachem (Santa Cruz, CA, USA) | H-p-Bz-Phe-OH Article number:4017646.0005 | |
| Chemical compound, drug | 1-ethyl-3-(3-dimethylaminopropyl) carbodiimide (EDC) | ThermoScientific Pierce | Catalogue number: 22980 | |
| Chemical compound, drug | Ru(bpy)2(dcbpy) | Sigma Aldrich (St Louis, MO, USA) | Product 96632 | Discontinued |
| Software, algorithm | ATSAS v2.8.3 | European Molecular Biology Laboratory (EMBL) Hamburg | https://www.embl-hamburg.de/biosaxs/software.html | |
| Software, algorithm | PyMol v1.8.0.5 | Schrödinger Scientific | https://pymol.org/2/ | |
| Software, algorithm | GROMACS | Schrödinger Scientific | Pronk et al., 2013 | |
| Chemical compound, drug | PEG/Ion Screen 2 #39 | Hampton Research (Aliso Viejo, CA, USA) | Product HR2-126 | |
| Chemical compound, drug | Morpheus | Molecular Dimensions (Newmarket, Suffolk, UK) | Product MD1-46 | |
| Other | Superose 610/300 GL column | GE Healthcare | Product 17517201 | Discontinued |
Additional files
-
Supplementary file 1
Table of SecA tryptic peptides detected by MALDI-TOF.
- https://doi.org/10.7554/eLife.48385.020
-
Supplementary file 2
Table of data collection and refinement statistics for the crystal structure of SecAΔMBD.
- https://doi.org/10.7554/eLife.48385.021
-
Supplementary file 3
Table of SAXS data collection and processing details for SecA, SecAΔMBD and SecAΔCTT.
- https://doi.org/10.7554/eLife.48385.022
-
Supplementary file 4
Table of fitting parameters of models of the E. coli SecA dimer.
- https://doi.org/10.7554/eLife.48385.023
-
Transparent reporting form
- https://doi.org/10.7554/eLife.48385.024





