Targeting the vascular-specific phosphatase PTPRB protects against retinal ganglion cell loss in a pre-clinical model of glaucoma
Figures
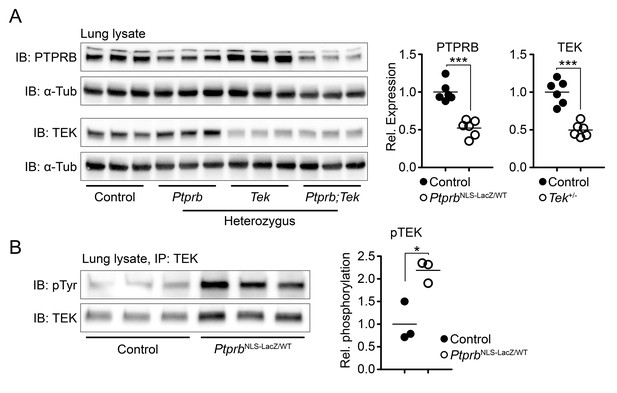
Deletion of one Ptprb allele leads to increased TEK phosphorylation.
(A) Western blot of lung lysate from P5 Control, Tek+/-, PtprbNLS-LacZ/WT and Tek+/-;PtprbNLS-LacZ/WT mice revealed a 50% reduction in PTPRB expression in PtprbNLS-LacZ/WT (heterozygous) and Tek+/-;PtprbNLS-LacZ/WT mice. Likewise, TEK expression was reduced approximately 50% in Tek+/- and Tek+/-;PtprbNLS-LacZ/WT mice. (B) Immunoprecipitation of lung lysates from adult control and PtprbNLS-LacZ/WT using anti-TEK antibody followed by western blotting with anti-phospho tyrosine antibody revealed a marked elevation of TEK phosphorylation in PtprbNLS-LacZ/WT animals compared to littermate controls. Horizontal lines indicate population means. *p<0.05, ***p<0.001 as determined by Student’s t-test.
Uncropped images corresponding to the western blots presented in Figure 1A.
Uncropped images corresponding to the western blots presented in Figure 1B.
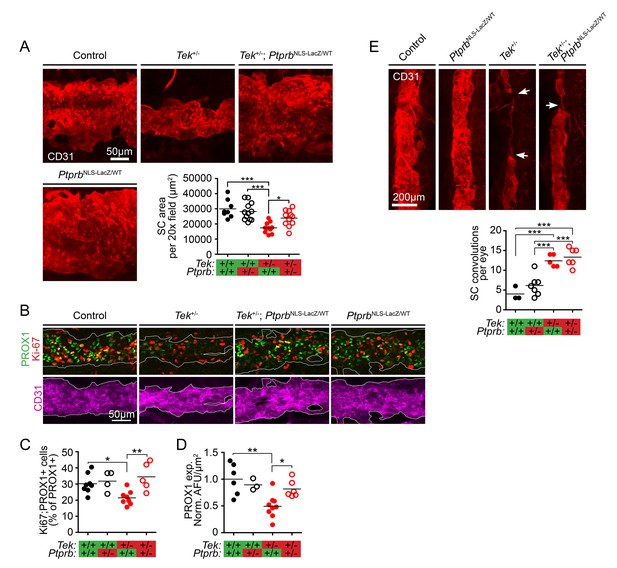
TEK signaling has a dose-dependent effect on Schlemm’s canal (SC) area and development.
(A) Confocal microscopy of whole mount eyes revealed reduced CD31+ SC area in adult Tek+/- haploinsufficient mice. This phenotype was blunted in Tek+/-;PtprbNLS-LacZ/WT double heterozygous animals, confirming the importance of TEK activation in canal development. PtprbNLS-LacZ/WT heterozygous controls had normal SC area (n = 8 WT, 12 PtprbNLS-LacZ/WT, 11 Tek+/- and 11 Tek+/-;PtprbNLS-LacZ/WT mice). 20x fields shown represent an area of 65,536 μm2. Images were captured as 10-frame Z stacks with a step size of 1.67 μm and a pinhole of 1.2 Airy units, and are shown as maximum intensity projections. (B, quantified in C) At postnatal day 5 (P5), confocal microscopy of the developing SC in eye whole mounts revealed reduced numbers of proliferating Ki-67-positive SC ECs (Ki67-PROX1 double positive cells) in Tek haploinsufficient animals compared to littermate WT or PtprbNLS-LacZ/WT controls. Normal proliferation was observed in Tek+/-;PtprbNLS-LacZ/WT animals. (D) Compared to control and PtprbNLS-LacZ/WT mice, PROX1 expression was reduced in Tek+/- littermate eyes. Expression was normal in Tek+/-;PtprbNLS-LacZ/WT double heterozygotes. n = 8 (WT), 4 (PtprbNLS-LacZ/WT), 8 (Tek+/-) and 5 (Tek+/-;PtprbNLS-LacZ/WT) Shown are maximum intensity projections from 8-frame confocal Z stacks captured using a 20x objective, step size of 1 μm and pinhole of 1.2 Airy units. Norm. AFU: Normalized, background subtracted arbitrary fluorescence units. (E) Compared to control and PtprbNLS-LacZ/WT littermates, confocal analysis of adult SC revealed a marked increase in the number of focal convolutions and narrowings in the eyes of Tek+/- and Tek+/-;PtprbNLS-LacZ/WT mice. N = 4 (WT), 7 (PtprbNLS-LacZ/WT), 5 (Tek+/-) and 6 (Tek+/-;PtprbNLS-LacZ/WT). Horizontal lines indicate population means. *p<0.05, **p<0.01, ***p<0.001 as determined by 1-way ANOVA followed by Bonferroni’s correction.
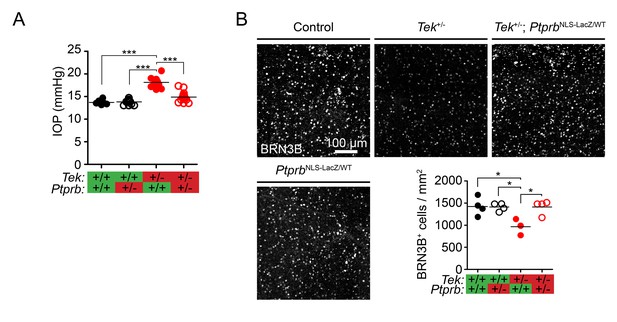
Ptprb heterozygosity prevents ocular hypertension and RGC loss in Tek haploinsufficient mice.
(A) Elevated intraocular pressure (IOP) was observed in Tek+/- haploinsufficient mice at 30 weeks of age when measured by rebound tonography. As in Figure 2, this phenotype was prevented in Tek+/-;PtprbNLS-LacZ/WT double heterozygous animals, confirming the importance of TEK activation in IOP homeostasis (n = 6 WT, 14 PtprbNLS-LacZ/WT, 12 Tek+/- and 14 Tek+/-;PtprbNLS-LacZ/WT mice). (B) BRN3B staining in retinal flat-mounts from a second group of mice revealed loss of retinal ganglion cells by 19 weeks in Tek+/- mice. Littermate Tek+/-;PtprbNLS-LacZ/WT animals were protected, correlating with the reduced IOP observed (n = 4 WT, 4 PtprbNLS-LacZ/WT, 3 Tek+/- and 4 Tek+/-;PtprbNLS-LacZ/WT mice). Horizontal lines indicate population means. *p<0.05, ***p<0.001 as determined by 1-way ANOVA followed by Bonferroni’s correction.
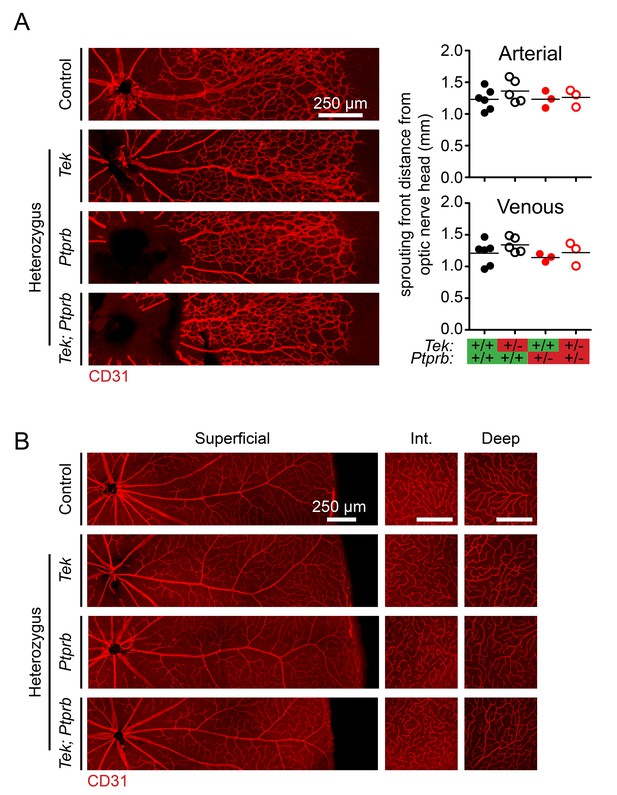
Retinal vasculature was normal in Tek and Ptprb haploinsufficient mice.
(A) Confocal microscopy of P5 retinas stained with anti-CD31 antibody reveal normal progression of the superficial vasculature at both arterial and venous sprouting fronts. n = 6 (WT), 5 (Tek+/-), 3 (PtprbNLS-LacZ/WT), and 3 (Tek+/-;PtprbNLS-LacZ/WT). Horizontal lines indicate population means. Lack of significance was determined by 1-way ANOVA followed by Bonferroni’s correction. (B) Normal, fully developed retinal vasculature was observed in all three plexus layers in mice of all genotypes at P20. Scale bars indicate 250 μm in all panels.
Tables
| Resource | Designation | Source or reference | Identifiers | Additional information |
|---|---|---|---|---|
| Genetic Reagent (M. musculus) | PtprbNLS-LacZ | Bäumer et al., 2006 | Maintained on a mixed background | |
| Genetic Reagent (M. musculus) | Tek+/- | Thomson et al., 2017 | Tektm1.1Vlcg; MGI:5544795 | Maintained on a mixed background |
| Antibody | anti-PTPRB (Rabbit polyclonal) | Nawroth et al., 2002 | Western blot: 1:2000 | |
| Antibody | Anti-TEK (Rabbit polyclonal) | Santa Cruz Biotech | sc-324 | Western blot 1:2500 |
| Antibody | anti-αTubulin (Mouse monoclonal) | Santa Cruz Biotech | sc-32293 | Western blot: 1:10,000 |
| Antibody | 4G10 Platinum anti-phosphotyrosine (Mouse monoclonal) | Millipore | 05–1050 | Western blot: 1:2000 |
| Antibody | anti-CD31 MEC13.3 (Rat monoclonal) | BD Biosciences | 55337 | IF: 1:100 |
| Antibody | anti-PROX1 (Goat polyclonal) | R and D Systems | AF2727 | IF: 1:200 |
| Antibody | anti-Ki-67 (Rabbit monoclonal) | ThermoFisher | MA5-14520 | IF: 1:200 |
| Antibody | anti-BRN3b (Goat polyclonal) | Santa Cruz Biotech | sc-6026 | IF: 1:1000 |
| Software, algorithm | ImageJ Fiji | Schindelin et al., 2012 | Version 1.52 p | Used for all image analysis |
| Software, algorithm | Graphpad Prism | Graphpad.com | Version 5.0 | Used for statistical analysis and graph generation |
| Software, algorithm | Adobe Indesign | Adobe.com | Version 14.01 × 64 | Used for figure creation |





