Extracellular matrix-inducing Sox9 promotes both basal progenitor proliferation and gliogenesis in developing neocortex
Figures
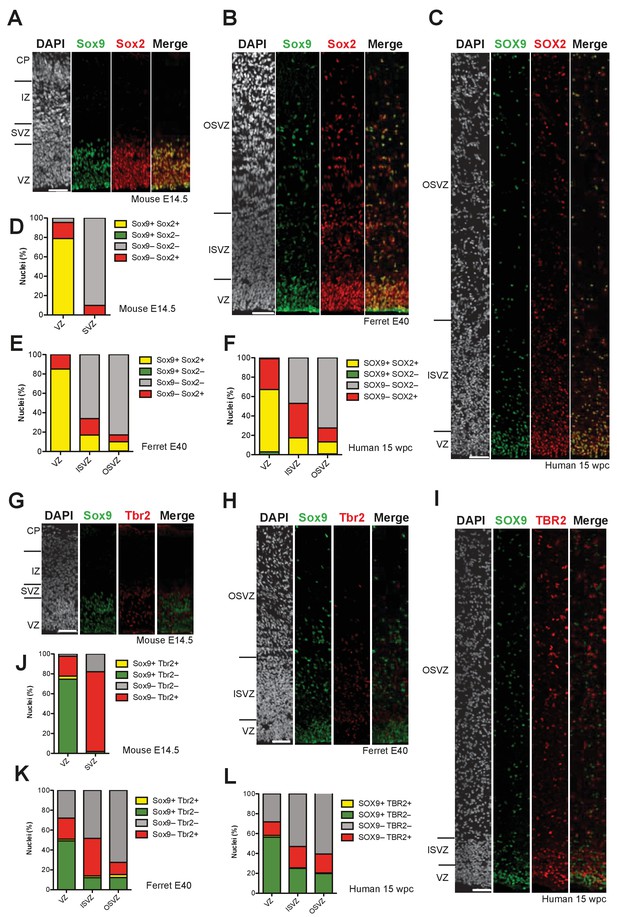
Sox9-expressing BPs occur in the SVZ of embryonic ferret and fetal human but not embryonic mouse neocortex.
(A–C) Double immunofluorescence for Sox9 (green) and Sox2 (red), combined with DAPI staining (white), of mouse E14.5 (A), ferret E40 (B) and human 15 wpc (C) neocortex. (D–F) Quantification of the percentage of nuclei (identified by DAPI staining) that are Sox9 plus Sox2 double-positive (yellow), Sox9-positive only (green), Sox2-positive only (red), and Sox9 plus Sox2 double-negative (gray), in mouse E14.5 (D), ferret E40 (E) and human 15 wpc (F) neocortex. (G–I) Double immunofluorescence for Sox9 (green) and Tbr2 (red), combined with DAPI staining (white), of mouse E14.5 (G), ferret E40 (H) and human 15 wpc (I) neocortex. (J–L) Quantification of the percentage of nuclei (identified by DAPI staining) that are Sox9 plus Tbr2 double-positive (yellow), Sox9-positive only (green), Tbr2-positive only (red), and Sox9 plus Tbr2 double-negative (gray), in mouse E14.5 (J), ferret E40 (K) and human 15 wpc (L) neocortex. (A–C, G–I) Ventricular surface is down. Upper margins of images in (A, B, G, H) correspond to the pial surface (A, G) and the basal boundary of the OSVZ (B, H); in (C, I) most but not all of the OSVZ is shown due to space constraints. Scale bars, 50 µm.
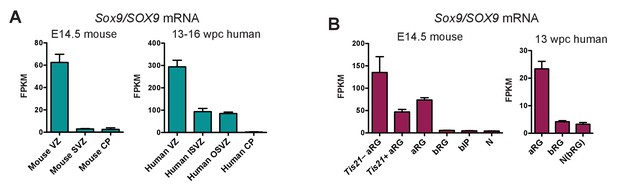
Sox9 mRNA is expressed in fetal human, but not embryonic mouse, BPs.
(A) FPKM values reflecting the Sox9 mRNA levels in the germinal zones of mouse E14.5 (left) and human 13–16 wpc (right) neocortex, as determined previously (Fietz et al., 2012). (B) FPKM values reflecting the Sox9 mRNA levels in distinct mouse E14.5 (left) and human 13 wpc (right) neocortical cell subpopulation, as determined previously (Florio et al., 2015). Tis21 –/+, Tis21-GFP–negative/positive; bIP, mouse cell subpopulation containing bIPs and a minor proportion of other cell types that do not express prominin-1 and Tubb3 and are not labeled by DiI (e.g. endothelial cells); N, mouse neurons; N(bRG), human neuron fraction containing bRG in G1. Data are the mean of 5 (A) and 4 (B) mouse transcriptomes and of 6 (A, one 13 wpc, two 14 wpc, one 15 wpc and two 16 wpc determination(s)) and 4 (B) human transcriptomes. Error bars represent SD.
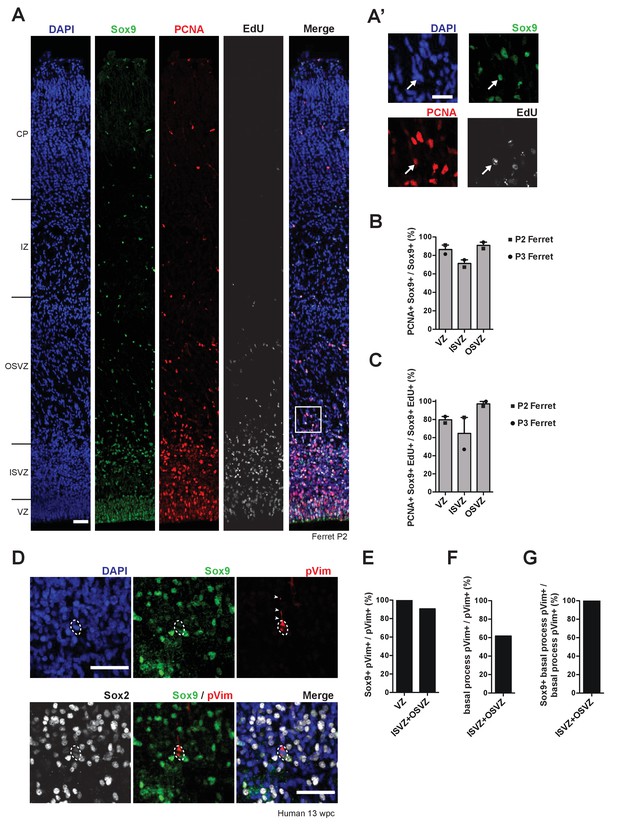
Sox9-expressing BPs in the SVZ of developing ferret and human neocortex are capable of cell cycle re-entry and include bRG.
(A) Triple (immuno)fluorescence for Sox9 (green), PCNA (red) and EdU (white), combined with DAPI staining (blue), of P2 ferret neocortex. EdU was administrated at P0. Boxed area in (A) is shown in higher magnification in (A’). White arrows indicate a nucleus that is triple-positive for Sox9, PCNA and EdU, that is a Sox9-positive cycling BP in the OSVZ. (B) Quantification of the percentage of Sox9-positive nuclei that are PCNA-positive, that is the percentage of Sox9-positive cells that are cycling, in the indicated germinal zones of P2 (squares) and P3 (circles) ferret neocortex. Bars indicate the range between the individual values at P2 and P3. (C) Quantification of the percentage of Sox9 and EdU double-positive nuclei that are PCNA-positive, that is the percentage of Sox9-positive cNPCs that have re-entered the cell cycle, in the indicated germinal zones of P2 (squares) and P3 (circles) ferret neocortex. EdU was administrated at P0. Bars indicate the range between the individual values at P2 and P3. (D) Triple immunofluorescence for Sox9 (green), phospho-vimentin (pVim, red) and Sox2 (white), combined with DAPI staining (blue), in the OSVZ of 13 wpc human neocortex. Arrowheads indicate the basal process, and dashed lines delineate the mitotic cell body, of a bRG. (E) Quantification of the percentage of pVim-positive cells, that is of mitotic cNPCs, that are Sox9-positive, in the indicated germinal zones of 13 wpc human neocortex. (F) Quantification of the percentage of pVim-positive cells that bear a basal process, that is the percentage of mitotic BPs that are bRG, in the ISVZ plus OSVZ of 13 wpc human neocortex. (G) Quantification of the percentage of basal process-bearing pVim-positive cells, that is of mitotic bRG, that are Sox9-positive, in the ISVZ plus OSVZ of 13 wpc human neocortex. (A, A', D) Scale bars, 50 µm (A, D) and 25 µm (A'). In (A), ventricular surface is down.
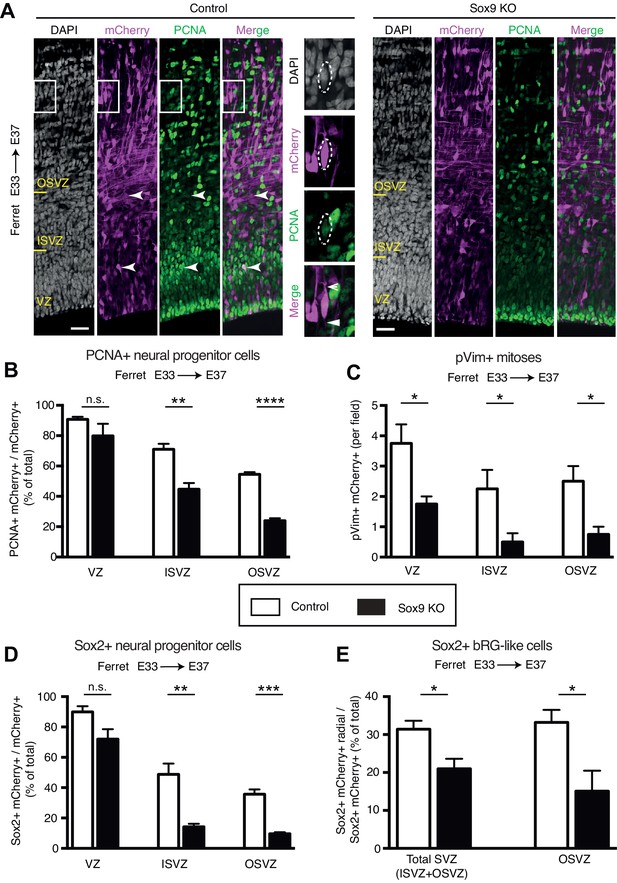
CRISPR/Cas9-mediated KO of Sox9 in embryonic ferret neocortex leads to a reduction in BP proliferation.
Ferret E33 neocortex was electroporated in utero with a plasmid encoding mCherry together with a complex of recombinant Cas9 protein with gRNAs targeting either Sox9 (Sox9 KO) or LacZ (Control), followed by analysis at E37. (A) Double immunofluorescence for mCherry (magenta) and PCNA (green), combined with DAPI staining (white). Images are single optical sections. Scale bars, 30 µm. Boxes (35 × 50 µm) indicate mCherry-positive bRG in the OSVZ, shown at higher magnification on the right. Insets: dashed lines, cell body contour; arrow, a bifurcated basal process; arrowhead, an apically-directed process. (B) Percentage of mCherry-positive cells in the VZ, ISVZ and OSVZ that are PCNA-positive in control (white) and Sox9 KO (black). Note that in the two PCNA immunostainings shown in (A), the brightest cells are equally bright for both the control and the Sox9 KO, as is obvious when comparing (i) the PCNA+ cells at the apical surface, and (ii) the brightest cells in the OSVZ. In contrast, in the basal region of the VZ, the intensity of immunostaining of the PCNA-positive cells is less in the Sox9 KO than the control. Importantly, such differences in PCNA immunostaining intensity did not affect the quantification shown, because even only weakly PCNA-positive cells were scored as PCNA+. (C) Quantification of mCherry-positive mitotic cells in the VZ, ISVZ and OSVZ, as revealed by pVim immunofluorescence (see Figure 3—figure supplement 1 for the immunofluorescence images), in a 100 µm-wide field of the cortical wall, in control (white) and Sox9 KO (black). (D) Percentage of mCherry-positive cells in the VZ, ISVZ and OSVZ that are Sox2-positive in control (white) and Sox9 KO (black) (see Figure 3—figure supplement 2 for the immunofluorescence images). (E) Percentage of mCherry-positive cells in total SVZ and separately OSVZ that are Sox2-positive with a radial orientation of the nucleus and presence of at least one radial process (bRG-like cells) in control (white) and Sox9 KO (black). (B–E) Data are the mean of 4 embryos from three different litters. Error bars indicate SD; *, p<0.05; **, p<0.01; ***, p<0.001; ****, p<0.0001; n.s., not statistically significant; Student’s t-test.
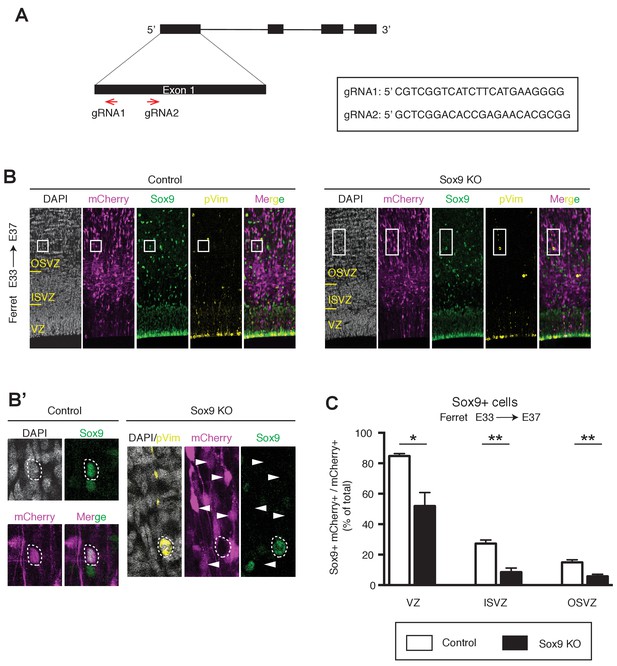
CRISPR/Cas9-mediated KO of Sox9 in embryonic ferret neocortex ablates Sox9 expression in vivo.
(A) Genome editing strategy for KO of Sox9 in ferret. Genomic representation of the Sox9 locus in ferret; black, exons; arrows, guide RNAs (gRNAs). Box shows sequences of the two gRNAs used. (B–C) E33 ferret neocortex was electroporated in utero with a plasmid encoding mCherry together with a complex of recombinant Cas9 protein with gRNAs targeting either Sox9 (Sox9 KO) or LacZ (Control), followed by triple immunofluorescence for mCherry (magenta), Sox9 (green) and pVim (yellow), combined with DAPI staining (white), at E37. (B) Boxes (control, 35 × 40 µm; Sox9 KO, 40 × 95 µm) indicate areas shown at higher magnification in (B'). (B') Left, high magnification of a Sox9-positive BP in control; right, high magnification of an mCherry-positive, Sox9-negative mitotic bRG in Sox9 KO. Arrowheads, mCherry-positive cells that do not express Sox9 in KO. (C) Percentage of mCherry-positive cells in the VZ, ISVZ and OSVZ that are Sox9-positive in control (white) and Sox9 KO (black). Data are the mean of 4 embryos from three different litters. Error bars indicate SD; *, p<0.05; **, p<0.01; Student’s t-test.
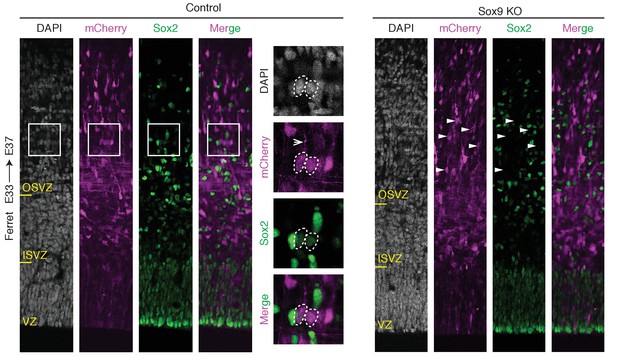
CRISPR/Cas9-mediated KO of Sox9 in embryonic ferret neocortex reduces abundance of Sox2-positive BPs.
E33 ferret neocortex was electroporated in utero with a plasmid encoding mCherry together with a complex of recombinant Cas9 protein with gRNAs targeting either Sox9 (Sox9 KO) or LacZ (Control), followed by double immunofluorescence for mCherry (magenta) and Sox2 (green), combined with DAPI (white), at E37. Control, boxes (50 × 50 µm) indicate areas shown at higher magnification on the right. Insets, dashed lines, cell body contour (bRG, left; multipolar BP, right); arrow, basal process of a bRG. Sox9 KO, arrowheads, mCherry-positive cells in the OSVZ that lack Sox2 immunoreactivity.
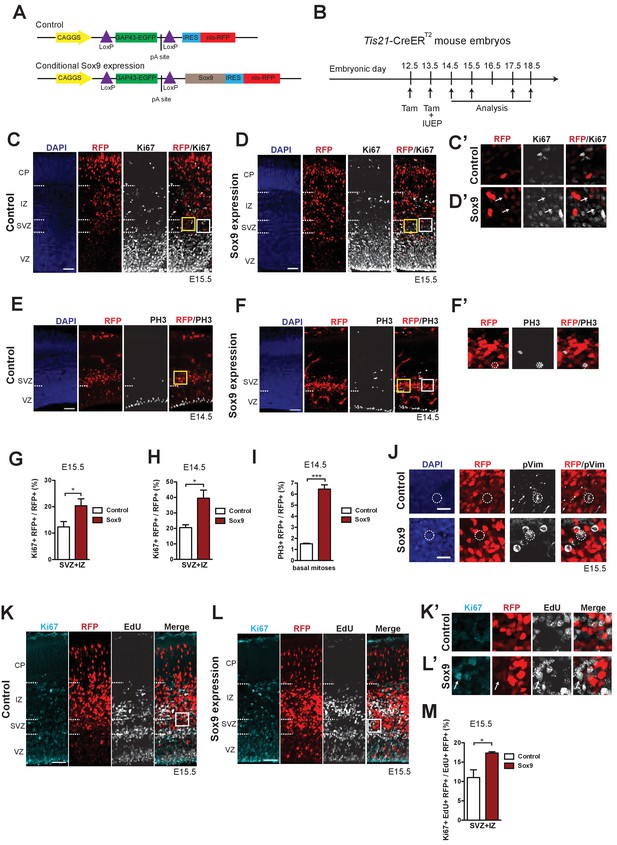
Conditional Sox9 expression in BPs of embryonic mouse neocortex increases their proliferation and cell cycle re-entry.
(A) Constructs used to conditionally express nuclear RFP without (top, control construct) and with (bottom, conditional Sox9 expression construct) Sox9 in mouse BPs and their progeny using the Tis21-CreERT2 line (see B). (B) Workflow of tamoxifen administration (Tam) at E12.5 and E13.5, in utero electroporation (IUEP) at E13.5, and immunostaining analyses of the neocortex at the indicated time points (arrows) yielding the data shown in this figure and subsequent figures, using heterozygous Tis21-CreERT2 mouse embryos. (C, D) Double immunofluorescence of neocortex for RFP (red) and Ki67 (white), combined with DAPI-staining (blue), 48 hr after electroporation of control construct (C) or conditional Sox9 expression construct (D). Dashed lines indicate the borders between VZ, SVZ, IZ and CP. White boxed areas of the SVZ in (C) and (D) are shown at higher magnification in (C’) and (D’), respectively; arrows indicate RFP-positive nuclei that are Ki67-positive. Yellow boxed areas of the SVZ in (C) and (D) are shown at higher magnification in Figure 7A, top row and bottom row, respectively. (E, F) Double immunofluorescence of neocortex for RFP (red) and phosphohistone H3 (PH3, white), combined with DAPI staining (blue), 24 hr after electroporation of control construct (E) or conditional Sox9 expression construct (F). Dashed lines indicate the border between VZ and SVZ. White boxed area in (F) is shown at higher magnification in (F’); dashed circles delineate a basal, PH3 and RFP double-positive mitosis. Yellow boxed areas of the SVZ in (E) and (F) are shown at higher magnification in Figure 7B, top row and bottom row, respectively. (G, H) Quantifications in the neocortical SVZ plus IZ, upon electroporation of control construct (white columns) or conditional Sox9 expression construct (red columns). (G) Quantification of the percentage of RFP-positive nuclei that are Ki67-positive, 48 hr after electroporation. (H) Quantification of the percentage of RFP-positive nuclei that are Ki67-positive, 24 hr after electroporation. Related representative images are not shown. (I) Quantification of the percentage of abventricular RFP-positive cells in neocortex that undergo basal mitosis as revealed by PH3 immunofluorescence, 24 hr after electroporation of control construct (white column) or conditional Sox9 expression construct (red column). (J) Double immunofluorescence of neocortex for RFP (red) and phospho-vimentin (pVim, white), combined with DAPI staining (blue), of the SVZ, 48 hr after electroporation of control construct (white column) or conditional Sox9 expression construct (red column). Dashed circles delineate the cell body of RFP and pVim double-positive cells. (K, L) Triple (immuno)fluorescence of neocortex for Ki67 (cyan), RFP (red) and EdU (white) 48 hr after electroporation of control construct (K) or conditional Sox9 expression construct (L). A single pulse of EdU was administered at E14.5, that is 24 hr after electroporation and 24 hr prior to analysis. Dashed lines indicate the borders between VZ, SVZ, IZ and CP. White boxed areas of the SVZ in (K) and (L) are shown at higher magnification in (K’) and (L’), respectively; arrows indicate RFP-positive nuclei that are Ki67- and EdU-positive. (M) Quantification of the percentage of RFP and EdU double-positive nuclei in the neocortical SVZ plus IZ that are Ki67-positive, that is the percentage of RFP+ BPs that have re-entered the cell-cycle, 48 hr after electroporation of control construct (white column) or conditional Sox9 expression construct (red column) and 24 hr after EdU administration at E14.5. (C–F, J–L) Scale bars, 50 μm (C–F, K, L), 20 μm (J). (G–I, M) Two-tailed, unpaired t-test: *p<0.05, **p<0.01, ***p<0.001. Data are the mean of six embryos electroporated with control construct and six embryos electroporated with conditional Sox9 overexpression construct (G), four embryos electroporated with control construct and four embryos electroporated with conditional Sox9 overexpression construct (H) and three embryos electroporated with control construct and three embryos electroporated with conditional Sox9 overexpression construct (I, M), each from a different litter; for each embryo, two microscopic fields, each of 200–µm apical width, were counted, and the values obtained were averaged. Error bars represent SEM.
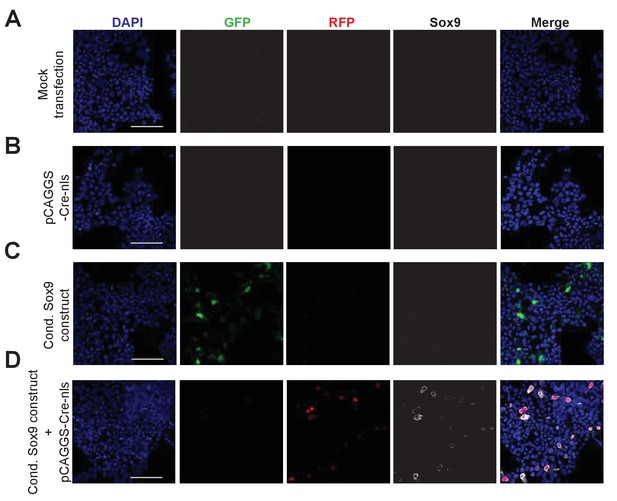
In vitro validation of the functionality of the conditional Sox9 expression construct using HEK293T cells.
Triple immunofluorescence for GFP (green), RFP (red) and Sox9 (white), combined with DAPI staining (blue), of HEK293T cells subjected to mock transfection (A) or transfected with pCAGGS-Cre-nls (B), the conditional (cond.) Sox9 expression construct (C), or the conditional (cond.) Sox9 expression construct plus pCAGGS-Cre-nls (D), followed by 48 hr of culture. Scale bars, 100 µm.
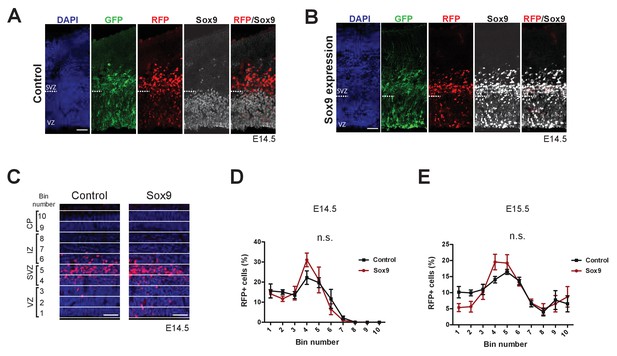
Validation of the Sox9 expression, elicited specifically in BP-genic aRGs and BPs upon in utero electroporation of the conditional Sox9 expression construct into the neocortex of Tis21-CreERT2 mouse embryos.
Heterozygous Tis21-CreERT2 mouse embryos received tamoxifen at E12.5 and E13.5, were subjected to in utero electroporation of the neocortex at E13.5 with either control construct or conditional Sox9 expression construct, and subjected to immunostaining analyses at E14.5 and E15.5. (A, B) Triple (immuno)fluorescence of neocortex for GFP (green), RFP (red) and Sox9 (white), combined with DAPI staining (blue), 24 hr after electroporation of control construct (A) or conditional Sox9 expression construct (B). Dashed lines indicate the border between VZ and SVZ. Scale bar, 50 µm. (C) Immunofluorescence for RFP (red), combined with DAPI staining (blue), in the neocortex 24 hr after electroporation of control or conditional Sox9 expression construct (see Figure 4A). For the quantification of the distribution of RFP-positive nuclei (see D) and E), images were divided into ten bins of equal size (numbered 1–10 from apical to basal) that span the entire cortical wall. (D) Quantification of the percentage of the RFP-positive nuclei in the neocortex that are found in each bin (see C), 24 hr after electroporation of control construct (black line) or conditional Sox9 expression construct (red line). (E) Quantification of the percentage of the RFP-positive nuclei in the neocortex that are found in each bin, 48 hr after electroporation of control construct (black line) or conditional Sox9 expression construct (red line). (A–C) Scale bars, 50 µm. (D, E) Statistical analysis performed separately for each bin of equal size, two-tailed, unpaired t-test: n.s. not significant. Data are the mean of 4 embryos, each from a different litter; for each embryo, two microscopic fields, each of 200 µm width, were counted, and the values obtained were averaged. Error bars represent SEM.
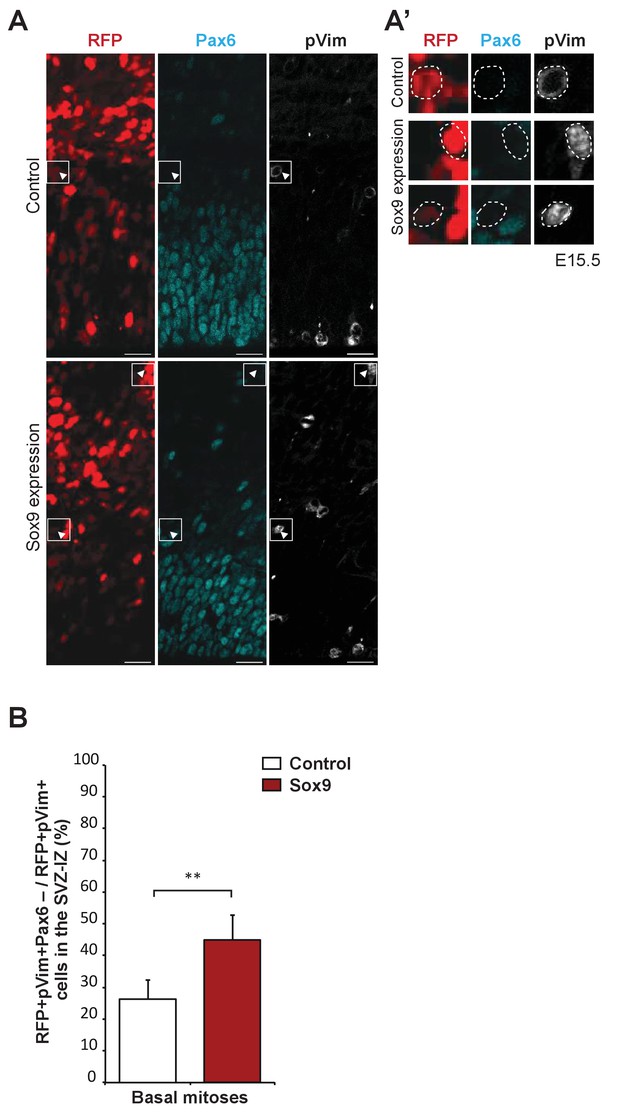
Analysis of Pax6-negative mouse BPs upon conditional Sox9 expression.
Heterozygous Tis21-CreERT2 mouse embryos received tamoxifen at E12.5 and E13.5, were subjected to in utero electroporation of the neocortex at E13.5 with either control construct or conditional Sox9 expression construct, and subjected to immunostaining analysis at E15.5. (A, A') Triple immunofluorescence of neocortex for RFP (red), Pax6 (cyan) and phospho-vimentin (pVim, white). Arrowheads indicate RFP-positive nuclei that are negative for Pax6; white boxed areas of the SVZ in (A) are shown at higher magnification in (A'). Scale bars, 20 μm. (B) Quantification of the percentage of pVim and RFP double-positive cells in the SVZ that are negative for Pax6. Student’s t-test: **p<0.01. Data are the mean of four embryos, each from a different litter; for each embryo, two microscopic fields, each of 350 μm apical width, were counted, and the values obtained were averaged. Error bars represent SD.
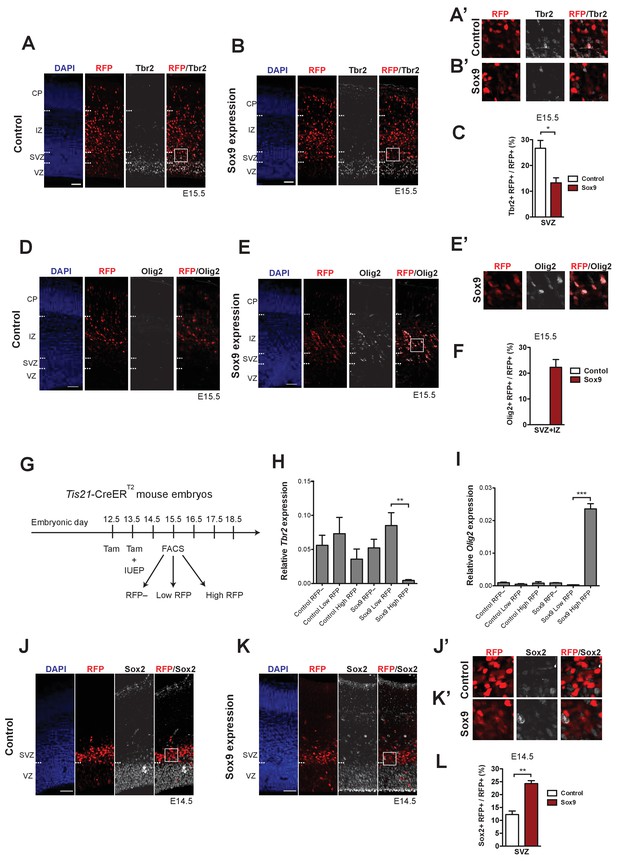
Conditional Sox9 expression in mouse BPs represses Tbr2 expression and induces premature gliogenesis in a dose-dependent manner.
Heterozygous Tis21-CreERT2 mouse embryos received tamoxifen administration at E12.5 and E13.5 and were subjected to in utero electroporation of the neocortex at E13.5 with either control construct or conditional Sox9 expression construct, followed by immunostaining analyses either 24 hr (J–L) or 48 hr (A–F) later. (A, B) Double immunofluorescence for RFP (red) and Tbr2 (white), combined with DAPI staining (blue), 48 hr after electroporation of control construct (A) or conditional Sox9 expression construct (B) (see Figure 4A). Dashed lines indicate the borders between VZ, SVZ, IP and CP. Boxed areas of the SVZ in (A) and (B) are shown at higher magnification in (A’) and (B’) respectively. (C) Quantification of the percentage of RFP-positive nuclei in the SVZ that are Tbr2-positive, 48 hr after electroporation of control construct (white column) or conditional Sox9 expression construct (red column). (D, E) Double immunofluorescence for RFP (red) and Olig2 (white), combined with DAPI staining (blue), 48 hr after electroporation of control construct (D) or conditional Sox9 expression construct (E). Dashed lines indicate the borders between VZ, SVZ, IP and CP. Boxed area of IZ in (E) is shown in higher magnification in (E’). (F) Quantification of the percentage of RFP-positive nuclei in the SVZ plus IZ that are Olig2-positive, 48 hr after electroporation of control construct (white column) or conditional Sox9 expression construct (red column). (G) Workflow of tamoxifen administration (Tam) at E12.5 and E13.5, in utero electroporation (IUEP) of the neocortex at E13.5, and FACS at E15.5 followed by qPCR analyses of RFP-positive cells yielding the data shown in (H) and (I), using heterozygous Tis21-CreERT2 mouse embryos. (H, I) Quantification of Tbr2 (H) and Olig2 (I) mRNA levels relative to the Gapdh mRNA level by qPCR analysis, in RFP-negative, low-level RFP-expressing and high-level RFP-expressing cell populations isolated by FACS (see G) 48 hr after electroporation of control construct (left three columns) or conditional Sox9 expression construct (right three columns). (J, K) Double immunofluorescence for RFP (red) and Sox2 (white), combined with DAPI staining (blue), 24 hr after electroporation of control construct (J) or conditional Sox9 expression construct (K). Dashed lines indicate border between VZ and SVZ. Boxed areas in SVZ in (J) and (K) are shown at higher magnification in (J’) and (K’), respectively. (L) Quantification of the percentage of RFP-positive nuclei in the SVZ that are Sox2-positive, 24 hr after electroporation of control construct (white column) or conditional Sox9 expression construct (red column). (A, B, D, E, J, K) Scale bars, 50 µm. (C, H, I, L) Two-tailed, unpaired t-test: *p<0.05, **p<0.01, ***p<0.001. Data are the mean of 3 (C, F, L), 5 (H) and 4 (I) embryos, each from a different litter; for each embryo in (C, F, L), two microscopic fields, each of 200 µm apical width, were counted, and the values obtained were averaged. Error bars represent SEM.
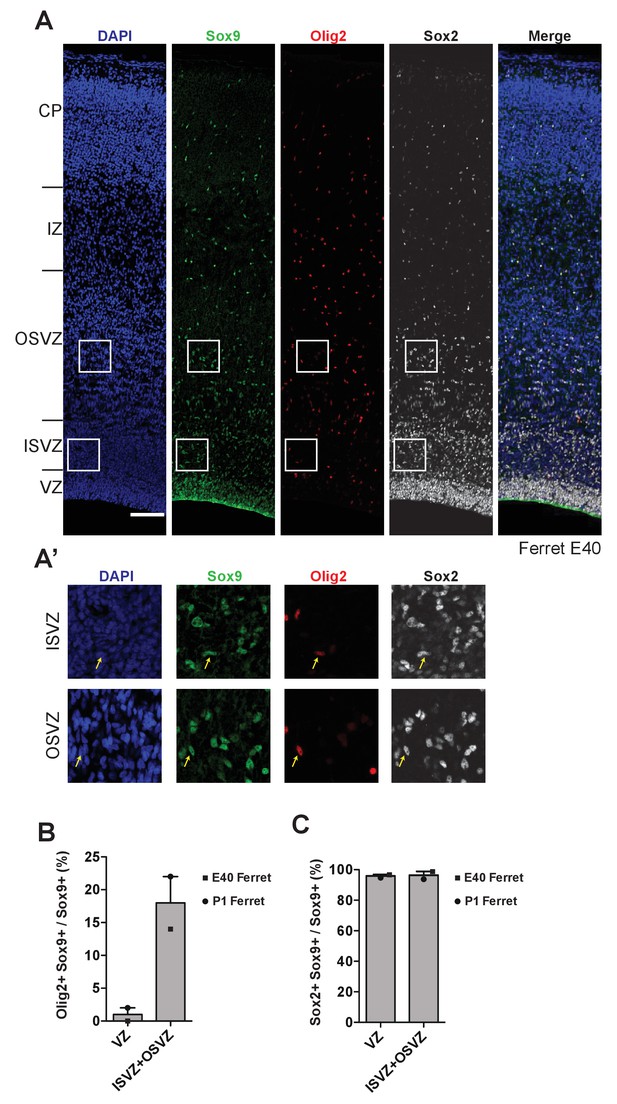
Gliogenic potential of Sox9-expressing cNPCs in the germinal zones of developing ferret neocortex.
(A, A') Triple immunofluorescence for Sox9 (green), and Olig2 (red) and Sox2 (white), combined with DAPI staining (blue), of E40 ferret neocortex. Ventricular surface is down, upper margins of images correspond to the pial surface. Scale bar, 100 µm. Boxed areas in ISVZ and OSVZ in (A) are shown at higher magnification in (A’). Arrows indicate nuclei that are triple-positive for Sox9, Olig2 and Sox2, that is Sox9-positive gliogenic BPs in the ISVZ and OSVZ. (B, C) Quantification of the percentage of the Sox9-positive nuclei in the VZ, and of the Sox9-positive nuclei in the ISVZ plus OSVZ, that are Olig2-positive (B) or Sox2-positive (C), in the E40 (squares) and P1 (circles) ferret neocortex. Bars indicate the range between the individual values at E40 and P1.
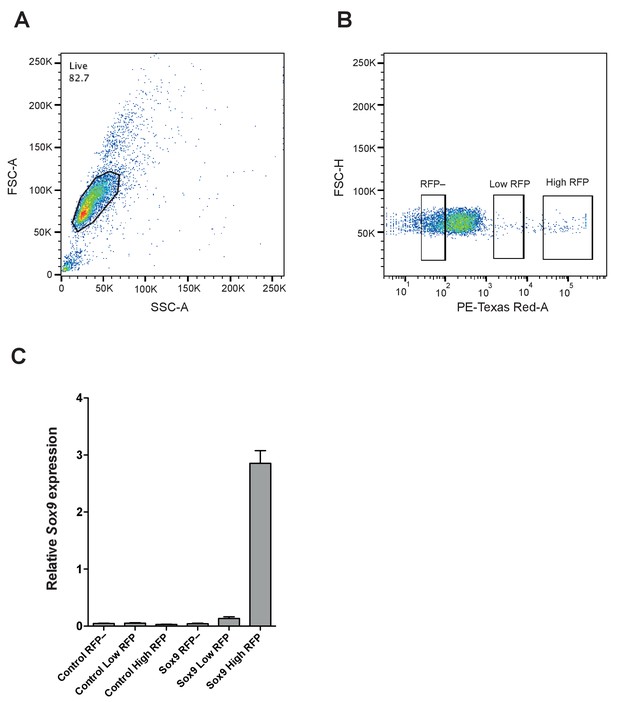
Isolation of RFP-negative, low-level RFP-expressing and high-level RFP-expressing cNPC subpopulations by FACS and determination of Sox9 mRNA levels in the six subpopulations.
(A) FSC/SSC dot-plot showing the distribution of dissociated cells, obtained from neocortices electroporated with control construct or conditional Sox9 expression construct (see Figure 5G), based on their size and shape. A gate for live cells was set as indicated by the polygon. (B) FSC-H/PE-Texas Red-A dot-plot of the live cells (see A) showing the distribution of cells based on their RFP fluorescence intensity. Three gates were set in order to sort RFP-negative (left rectangle), low-level RFP-expressing (middle rectangle) and high-level RFP-expressing (right rectangle) subpopulations. (C) Quantification of Sox9 mRNA levels relative to the Gapdh mRNA level by qPCR analysis in RFP-negative, low-level RFP-expressing and high-level RFP-expressing cell subpopulations isolated by FACS (see A, B) 48 hr after electroporation of control construct (left three columns) or conditional Sox9 expression construct (right three columns) (see Figure 5G).
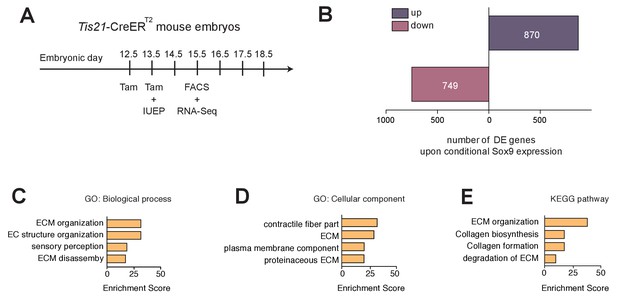
mRNAs upregulated upon conditional Sox9 expression in mouse BPs are mainly related to ECM.
(A) Workflow of tamoxifen administration (Tam) at E12.5 and E13.5, in utero electroporation (IUEP) of the neocortex at E13.5 with either control construct or conditional Sox9 expression construct, and FACS at E15.5 followed by RNA-Seq analysis of high-level RFP-expressing cells yielding the data shown in (B–E), using heterozygous Tis21-CreERT2 mouse embryos. (B) Number of mRNAs of protein-encoding genes differentially expressed (DE) 48 hr after electroporation of conditional Sox9 expression construct as compared to control construct (see Figure 3A); up, upregulated; down, downregulated. (C–E) Gene ontology (GO) term enrichment analyses for biological process (C) and cellular component (D), and KEGG pathway analysis (E), using as input the set of 870 genes upregulated upon conditional Sox9 expression. The top four enriched terms/pathways (p<0.01) are shown.
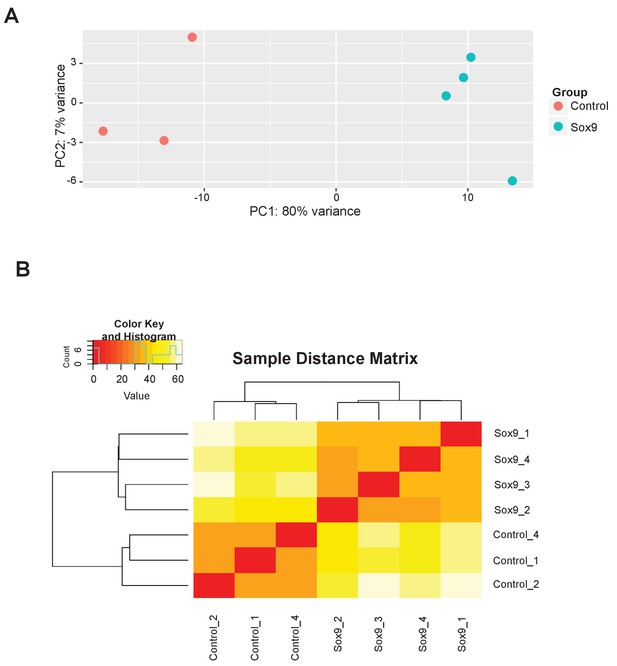
Principal component analysis and hierarchical clustering of electroporated cNPCs.
(A) Principal component (PC) analysis of the high-level RFP-expressing cell subpopulations upon electroporation with control construct (three replicates, red) and conditional Sox9 expression construct (four replicates, turquoise) (see Figure 6A). (B) Hierarchical clustering of the high-level RFP-expressing cell subpopulations upon electroporation with control construct (three replicates) and conditional Sox9 expression construct (four replicates) (see Figure 6A).
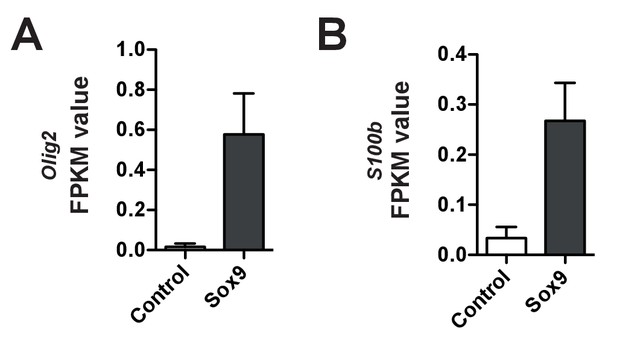
mRNAs of gliogenesis markers are upregulated upon conditional Sox9 expression.
FPKM values for Olig2 mRNA (A) and S100b mRNA (B) in the high-level RFP-expressing cells at E15.5 upon electroporation of heterozygous Tis21-CreERT2 mouse embryos with control and conditional Sox9 expression constructs at E13.5 (for details, see Figure 6A and legend). Data are the mean of transcriptomes of high-level RFP+ cells from 3 (control electroporated) and 4 (Sox9 electroporated) mouse embryonic neocortices, respectively. Error bars represent SEM.
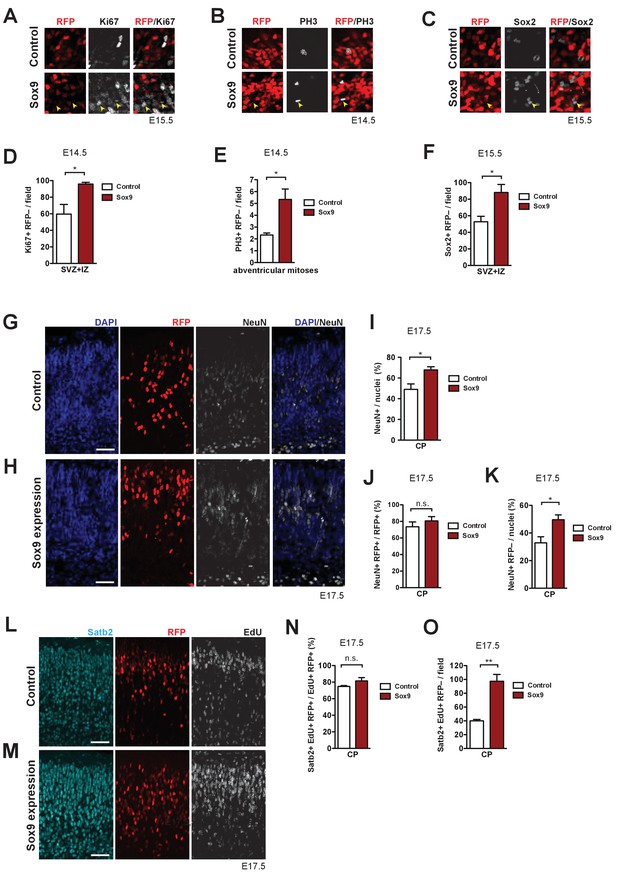
Conditional Sox9 expression in mouse BPs induces cell non-autonomous proliferation and neuron production.
Heterozygous Tis21-CreERT2 mouse embryos received tamoxifen at E12.5 and E13.5, were subjected to in utero electroporation of the neocortex with either control construct or conditional Sox9 expression construct (see Figure 4A) at E13.5, and subjected to immunostaining analyses of the neocortex at the time points indicated in (A–O). (A–C) High-magnification images of the SVZ showing double immunofluorescence for Ki67 (white) and RFP (red) (see yellow boxed areas in Figure 4C, (D) (A), phosphohistone H3 (PH3, white) and RFP (red) (see yellow boxed area in Figure 4E, (F) (B), and Sox2 (white) and RFP (red), 48 hr after electroporation of control or conditional Sox9 expression construct (C). Yellow arrowheads indicate BPs that are negative for RFP, but positive for Ki67 (A), PH3 (B) or Sox2 (C). (D) Quantification of the number of Ki67-positive, RFP-negative nuclei in the SVZ plus IZ per microscopic field of 200 µm apical width, 24 hr after electroporation of control construct (white column) or conditional Sox9 expression construct (red column). (E) Quantification of the number of abventricular, PH3-positive, RFP-negative mitoses per microscopic field of 200 µm apical width, 24 hr after electroporation of control construct (white column) or conditional Sox9 expression construct (red column). (F) Quantification of the number of Sox2-positive, RFP-negative nuclei in the SVZ plus IZ per microscopic field of 200 µm apical width, 48 hr after electroporation of control construct (white column) or conditional Sox9 expression construct (red column). (G, H) Double immunofluorescence for RFP (red) and NeuN (white), combined with DAPI staining (blue), in the CP 4 days after electroporation of control construct (G) or conditional Sox9 expression construct (H). (I) Quantification of the percentage of nuclei (identified by DAPI staining) in the CP that are NeuN-positive, 4 days after electroporation of control construct (white column) or conditional Sox9 expression construct (red column). (J) Quantification of the percentage of RFP-positive nuclei in the CP that are NeuN-positive, 4 days after electroporation of control construct (white column) or conditional Sox9 expression construct (red column). (K) Quantification of the percentage of nuclei (identified by DAPI staining) in the CP that are NeuN-positive and RFP-negative, 4 days after electroporation of control construct (white column) or conditional Sox9 expression construct (red column). (L, M) Triple (immuno)staining for Satb2 (cyan), RFP (red) and EdU (white) in the CP, 4 days after electroporation of control construct (L) or conditional Sox9 expression construct (M). A single pulse of EdU was administered at E14.5, that is 24 hr after electroporation and 3 days prior to analysis. (N) Quantification of the percentage of EdU and RFP double-positive nuclei in the CP that are Satb2-positive, that is the percentage of Satb2-positive neurons in the progeny of the targeted, EdU-labeled cNPCs, 4 days after electroporation of control construct (white column) or conditional Sox9 expression construct (red column) and 3 days after EdU administration at E14.5. (O) Quantification of the number of Satb2 and EdU double-positive, RFP-negative nuclei in the CP per microscopic field of 200 µm width, 4 days after electroporation of control construct (white column) or conditional Sox9 expression construct (red column) and 3 days after EdU administration at E14.5. (G, H, L, M) Scale bars, 50 µm. (D–F, I–K, N, O) Two-tailed, unpaired t-test: *p<0.05, **p<0.01, n.s. not significant. Data are the mean of 3 (E, F, I–K, N, O) and 4 (D) embryos, each from a different litter; for each embryo, two microscopic fields, each of either 200 µm (D–F, N, O) or 150 µm (I–K) width were counted, and the values obtained were averaged. Error bars represent SEM.
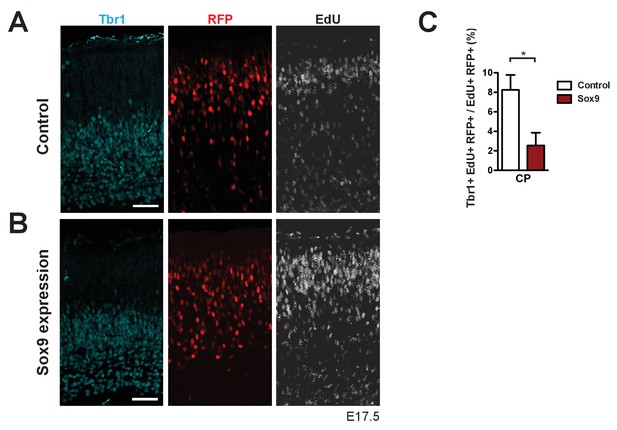
Conditional Sox9 expression results in a decrease of Tbr1-positive neurons in the progeny of the targeted, EdU-labeled mouse BPs.
Heterozygous Tis21-CreERT2 mouse embryos received tamoxifen at E12.5 and E13.5, and the neocortex was in utero electroporated at E13.5, followed by triple (immuno)staining for Tbr1 (cyan), RFP (red) and EdU (white) of the CP, 4 days after electroporation of either control construct (A) or conditional Sox9 expression construct (B) (see Figure 4A). A single pulse of EdU was administered at E14.5, that is 24 hr after electroporation and 3 days prior to analysis. (C) Quantification of the percentage of EdU and RFP double-positive nuclei in the CP that are Tbr1-positive, that is the percentage of Tbr1-positive neurons in the progeny of the targeted, EdU-labeled mouse BPs, 4 days after electroporation of control construct (white column) or conditional Sox9 expression construct (red column) and 3 days after EdU administration at E14.5. (A, B) Scale bars, 50 µm. (C) Two-tailed, unpaired t-test: *p<0.05. Data are the mean of 3 embryos, each from a different litter; for each embryo, two microscopic fields, each of 200 µm apical width, were counted, and the values obtained were averaged. Error bars represent SEM.
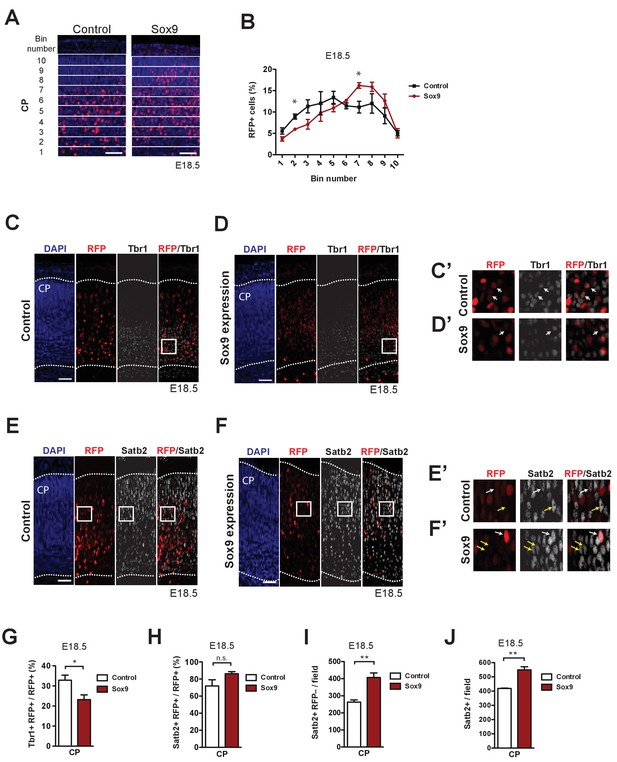
The neurons derived from mouse BPs targeted by conditional Sox9 expression are preferentially destined to the upper layers of the neocortex.
Heterozygous Tis21-CreERT2 mouse embryos received tamoxifen at E12.5 and E13.5, were subjected to in utero electroporation of the neocortex at E13.5 with either control construct or conditional Sox9 expression construct, and subjected to immunostaining analyses at E18.5. (A) Immunofluorescence for RFP (red), combined with DAPI staining (blue), in the CP 5 days after electroporation of control or conditional Sox9 expression construct (see Figure 3A). For the quantification of the distribution of RFP-positive nuclei (see B), images were divided into ten bins of equal size (numbered 1–10 from apical to basal) that span the entire CP. (B) Quantification of the percentage of the RFP-positive nuclei in the CP that are found in each bin (see A), 5 days after electroporation of control construct (black line) or conditional Sox9 expression construct (red line). (C, D) Double immunofluorescence for RFP (red) and Tbr1 (white), combined with DAPI staining (blue), in the CP 5 days after electroporation of control (C) or conditional Sox9 expression (D) construct. Dotted lines delineate the apical and basal borders of the CP. White boxed areas of the CP in (C) and (D) are shown at higher magnification in (C’) and (D’), respectively. (E, F) Double immunofluorescence for RFP (red) and Satb2 (white), combined with DAPI staining (blue), in the CP 5 days after electroporation of control (E) or conditional Sox9 expression (F) construct. Dotted lines delineate the apical and basal borders of the CP. White boxed areas of the CP in (E) and (F) are shown at higher magnification in (E’) and (F’), respectively. (G, H) Quantification of the percentage of the RFP-positive nuclei in the CP that are Tbr1-positive (G) and Satb2-positive (H), 5 days after electroporation of control construct (white columns) or conditional Sox9 expression construct (red columns). (I) Quantification of the number of Satb2-positive, RFP-negative nuclei in the CP per microscopic field of 200 µm width, 5 days after electroporation of control construct (white column) or conditional Sox9 expression construct (red column). (J) Quantification of the total number of Satb2-positive nuclei in the CP per microscopic field of 200 µm width, 5 days after electroporation of control construct (white column) or conditional Sox9 expression construct (red column). (A, C–F) Scale bars, 50 µm. (B) One-way ANOVA: p<0.0001; two-tailed, unpaired t-test performed separately for each bin pair: *p<0.05. Data are the mean of 3 embryos, each from a different litter; for each embryo, two microscopic fields, each of 200 µm width, were counted, and the values obtained were averaged. Error bars represent SEM. (G–J) Two-tailed, unpaired t-test: *p<0.05, **p<0.01, n.s. not significant. Data are the mean of 3 embryos, each from a different litter; for each embryo, two microscopic fields, each of 150 µm width, were counted, and the values obtained were averaged. Error bars represent SEM.
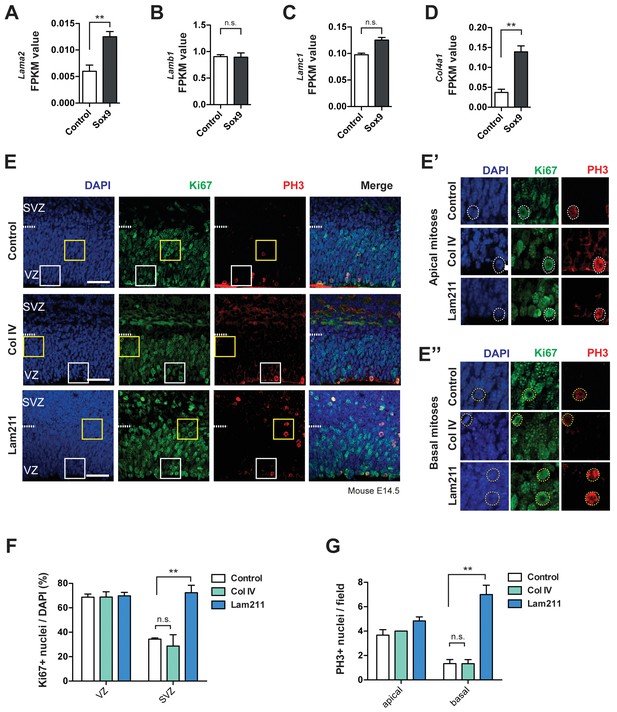
Laminin 211 induces BP proliferation in embryonic mouse neocortex.
(A–D) FPKM values for Lama2 mRNA (A), Lamb1 mRNA (B), Lamc1 mRNA (C) and Col4a1 mRNA (D) in the high-level RFP-expressing cells at E15.5 upon electroporation of heterozygous Tis21-CreERT2 mouse embryos with control and conditional Sox9 expression constructs at E13.5 (for details, see Figure 6A and legend). Two-tailed, unpaired t-test: **p<0.01, n.s. not significant. Data are the mean of transcriptomes of high-level RFP+ cells from 3 (control electroporated) and 4 (Sox9 electroporated) mouse embryonic neocortices, respectively. Error bars represent SEM. (E, E’, E’’) Double immunofluorescence for Ki67 (green) and phosphohistone H3 (PH3, red), combined with DAPI staining (blue), of mouse E14.5 organotypic slices of neocortex cultured for 24 hr without (control) or with collagen IV (Col IV) or laminin α2β1ɣ1 (Lam211). Ventricular surface is down, dashed lines indicate the border between the VZ and SVZ. Scale bars, 50 µm. White and yellow boxed areas are shown at higher magnification in (E’) and (E’’) and present examples of apical mitoses and basal mitoses, respectively; dashed circles indicate Ki67- and PH3-positive mitotic cNPCs. (F) Quantification of the percentage of the nuclei (identified by DAPI staining) that are Ki67-positive, in the VZ and SVZ of 24 hr control (white columns), Col IV-treated (green columns) and Lam211-treated (blue columns) E14.5 mouse neocortical organotypic slice cultures. (G) Quantification of the number of ventricular (apical) and abventricular (basal) PH3-positive mitoses, per microscopic field of 200 µm apical width, in 24 hr control (white columns), Col IV-treated (green columns) and Lam211-treated (blue columns) E14.5 mouse neocortical organotypic slice cultures. (F, G) Two-tailed, unpaired t-test, **p<0.01. Data are the mean of 3 embryos, each from a different litter; for each embryo, two microscopic fields, each of 200 µm apical width, were counted, and the values obtained were averaged. Error bars represent SEM. Note that the three data for apical mitoses upon collagen IV treatment were identical.
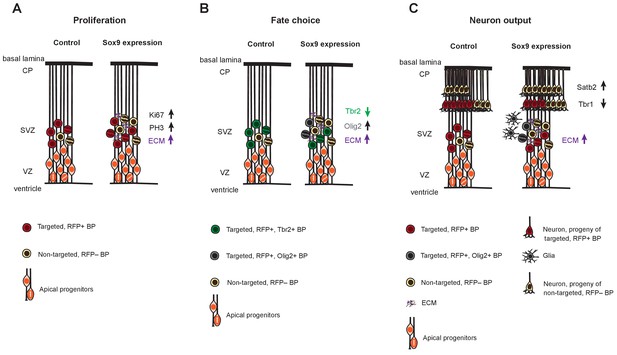
Cartoon illustrating the effects of conditional Sox9 expression in the mouse BP lineage on BP proliferation, fate choice and neuron output.
For details, see text.
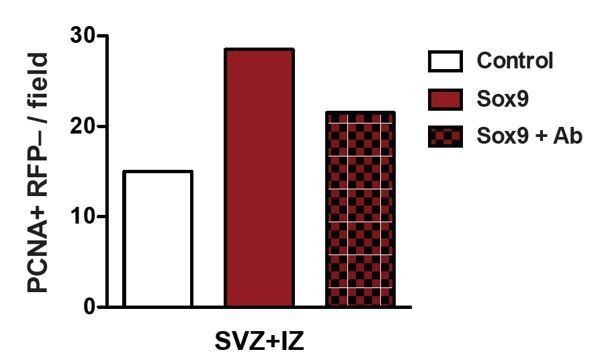
Heterozygous Tis21-CreERT2 mouse embryos received tamoxifen at E12.5 and E13.5 and were subjected to in utero electroporation of the neocortex at E13.5 with either control construct or conditional Sox9 expression construct.
Embryos were harvested 24 hours later, and neocortices were incubated with or without an integrin β1 blocking antibody in HERO culture for 24 hours, followed by immunostaining for PCNA. Quantification of the number of PCNA-positive, RFP-negative nuclei in the SVZ plus IZ per microscopic field of 150-µm apical width. White column, control construct, no integrin-blocking antibody; red column, conditional Sox9 expression construct, no integrin-blocking antibody; red checkered column, conditional Sox9 expression construct plus integrin-blocking antibody (Ab).
Additional files
-
Supplementary file 1
Lists of genes whose expression is up- or downregulated 48 hr after conditional Sox9 expression.
Tab-1 (Sox9_up_PC): List of genes that are upregulated upon conditional Sox9 expression. Tab-2 (Sox9_down_PC): List of genes that are downregulated upon conditional Sox9 expression. Columns in each tab denote the following information consecutively: A: Ensemble gene ID, B: Gene name, C: Description, D: Average count for Sox9 sample, E: Average count for control sample, G: Logarithmic fold change (FC) of counts between Sox9 and control samples, J: p-value, K: adjusted p-value, W: Average FPKM value for control sample, X: Average FPKM value for Sox9 sample, Y: Fold change (FC) between average FPKM values of Sox9 and control samples, Z: Logarithmic fold change (FC) between average FPKM values of Sox9 and control samples.
- https://cdn.elifesciences.org/articles/49808/elife-49808-supp1-v2.xlsx
-
Supplementary file 2
Datasets of RNA-sequencing and gene ontology (GO) term enrichment analysis of Sox9 and control samples.
Tab-1 (FPKM): FPKM values for each replicate of Sox9 and control samples. Tab-2 (GO_BP): Gene ontology (GO) term enrichment analysis for biological process (BP). Tab-3 (GO_CC): Gene ontology (GO) term enrichment analysis for cellular component. Tab-4 (KEGG): KEGG pathway analysis.
- https://cdn.elifesciences.org/articles/49808/elife-49808-supp2-v2.xlsx
-
Supplementary file 3
List of ECM-related genes that were identified by GO term and KEGG pathway analyses.
Tab-1 (ECM-related genes): List of all genes that were enriched for ECM related terms (Figure 5C,D and E) in GO term and KEGG pathway analyses.
Columns in each tab denote the following information consecutively: A: Ensemble gene ID, B: Gene name, C: Description, D: Average count for Sox9 sample, E: Average count for control sample, G: Logarithmic fold change (FC) of counts between Sox9 and control samples, J: p-value, K: adjusted p-value, W: Average FPKM value for control sample, X: Average FPKM value for Sox9 sample, Y: Fold change (FC) between average FPKM values of Sox9 and control samples, Z: Logarithmic fold change (FC) between average FPKM values of Sox9 and control samples.
- https://cdn.elifesciences.org/articles/49808/elife-49808-supp3-v2.xlsx
-
Supplementary file 4
Key Resources Table.
- https://cdn.elifesciences.org/articles/49808/elife-49808-supp4-v2.docx
-
Transparent reporting form
- https://cdn.elifesciences.org/articles/49808/elife-49808-transrepform-v2.docx





