A single regulator NrtR controls bacterial NAD+ homeostasis via its acetylation
Figures
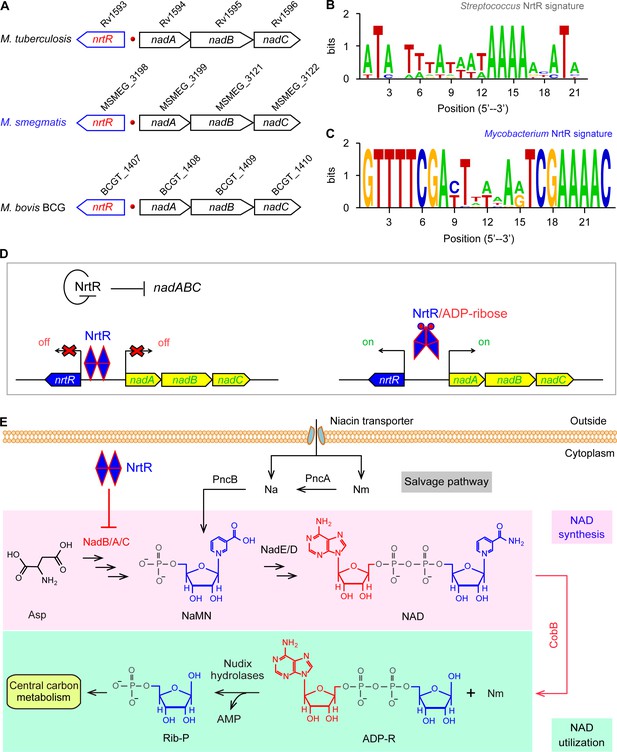
Working model for the regulation of NAD homeostasis by NrtR in Mycobacterium.
(A) The genetic context of nrtR and its signature in Mycobacterium compared with the NrtR-binding sequences in Streptococcus (B) and Mycobacterium (C). (D) NrtR acts as an auto-repressor and represses the transcription of the nadA-nadB-nadC operon that is responsible for the de novo synthesis of the NAD+ cofactor in Mycobacterium. (E) NAD+ homeostasis proceeds through cooperation of a salvage pathway with de novo synthesis in Mycobacterium. Designations: nadA, the gene encoding quinolinate synthase; nadB, gene encoding L-aspartate oxidase; nadC, gene encoding quinolinate phosphoribosyltransferase; PncA, nicotinamide deaminase; PncB, nicotinate phosphoribosyltransferase; NrtR, a bifunctional transcriptional factor involved in the regulation of NAD+ synthesis; ADP-R, ADP-ribose; Na, nicotinic acid; Nm, nicotinamide; Rib-P, ribose-5-phosphate; Asp, aspartate; NaMN, nicotinate mononucleotide; NAD+, nicotinamide adenine dinucleotide; CobB, an NAD+-consuming deacetylase.
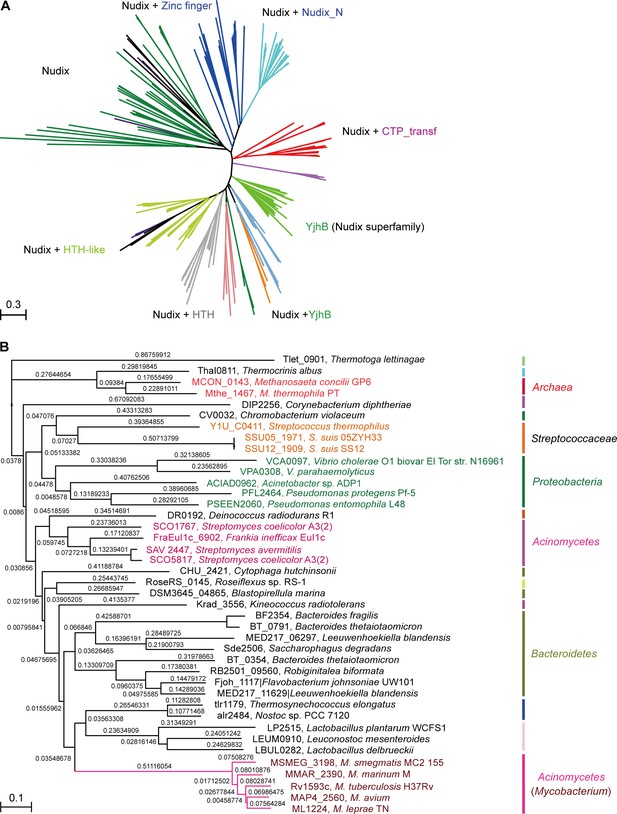
Phylogeny of NrtR proteins.
(A) The unrooted radial phylogeny of Nudix-like proteins. A variety of distinct subclades involve homologs containing a Nudix domain alone, a Nudix domain combined with DNA-binding domains or zinc finger domains, a Nudix domain combined with a CTP-transf (Cytidylyltransferase family) domain, Nudix+Nudix_N (Nudix located at N-terminal), or a Nudix pyrophosphate hydrolase with ADP-ribose substrate preference (YjhB, Nudix+YjhB superfamily). These distinct subclades seem to coincide with known taxonomic groups with few exceptions. NrtR candidates in Mycobacterium, Vibrio and Streptococcus species are indicated with purple, green and orange text, respectively. (B) Hierarchical tree of NrtR homologs. Several distinct sub-clades are clustered in a pattern that is generally consistent with bacterial taxonomic groups. The protein-sequence-based phylogeny of NrtR homologs was inferred using the maximum likelihood method and the WAG substitution model. The evolutionary distance for each node is shown next to the branches. Gene locus tags and strain names corresponding to the protein sequences used are indicated in the figure.
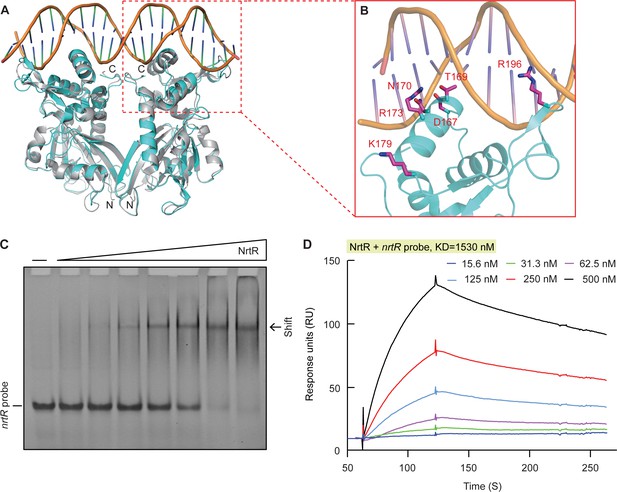
Structural and functional insights into the binding of MsNrtR to its cognate DNA target.
(A) Structural analysis of the predicted DNA-binding motif through structural modeling of M. smegmatis NrtR (http://swissmodel.espasy.org/). The image shows the superposition of M. smegmatis NrtR with the Shewanella oneidensis NrtR-DNA complex (PDB: 3GZ6). MsNrtR is highlighted in cyan and soNrtR is indicated in gray. Double-stranded DNA is denoted by two orange lines. (B) Structural prediction of the critical DNA-binding residues of the M. smegmatis NrtR. The six residues (D167, T169, N170, R173, K179, and R196) that are implicated in direct or indirect contact with cognate DNA are labeled in red. (C) Electrophoretic mobility shift assay (EMSA)-based visualization of the interaction of MsNrtR with the nrtR probe. The amount of NrtR protein incubated with the DNA probe is in each lane is (left to right): 0, 0.5, 1, 2, 5, 10, 20, and 40 pmol. (D) Surface plasmon resonance (SPR) measurements of M. smegmatis NrtR binding to the nrtR promoter. NrtR protein at various concentrations (typically 15.625–500 nM) were injected over the immobilized DNA probe comprising of the NrtR palindrome of nrtR gene. KD, kd/ka, ka, association constant; kd, dissociation constant; RU, response units.
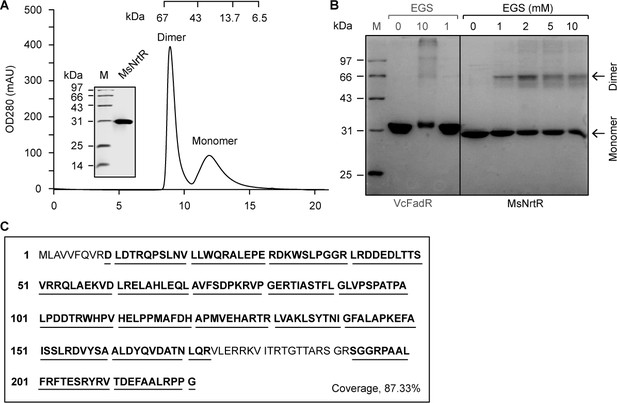
Characterization of the M. smegmatis NrtR.
(A) Fast protein liquid chromatography (FPLC) profile for the M. smegmatis NrtR. The protein sample was analyzed in a Superdex 75 column run by AKTA Purifier. The inset shows the SDS-PAGE gel of the recombinant 6 × His NrtR. The apparent mass of the recombinant NrtR of M. smegmatis is ~30 kDa. Note that M. smegmatis NrtR can easily form dimers in solution. OD280, optical density at 280 nm; AU, absorbance units. (B) Chemical cross-linking analyses of the purified NrtR protein. The samples were separated by 12% SDS-PAGE following the chemical cross-linking assays. The ethylene glycol bissuccinimidylsuccinate (EGS) cross-linking reagent was added at the concentrations shown at the top. M, protein standard marker. FadR is a known dimeric protein that was used as a positive control. (C) Mass spectrometry identification of the recombinant M. smegmatis NrtR. The peptide fragments that were matched are shown in bold and underlined (87.33%).
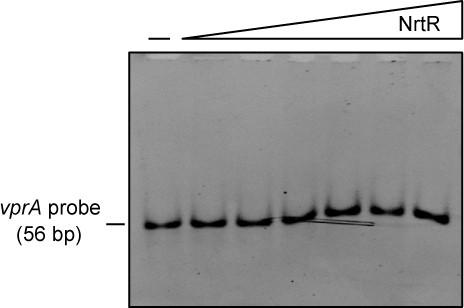
The M. smegmatis NrtR cannot bind to an unrelated DNA, the vprA probe.
The vprA probe (56 bp), an unrelated DNA (Supplementary file 2), acts here as a negative control. Above the gel image, the minus ‘–' sign indicates no addition of NrtR protein, and the triangle on right refers to the addition of increasingly levels of NrtR protein (0.5, 1.0, 2.0, 5.0, 10.0 and 20.0 pmols). Although it binds the nrtR probe (57 bp, Supplementary file 2), the NrtR of M. smegmatis can’t interact with the Vibrio cholerae vprA probe, an unrelated DNA. This verifies that the physical interplay between NrtR and the nadADC operon depends on specific binding to the nrtR probe located in the intergenic region (Figures 1A and 3C).
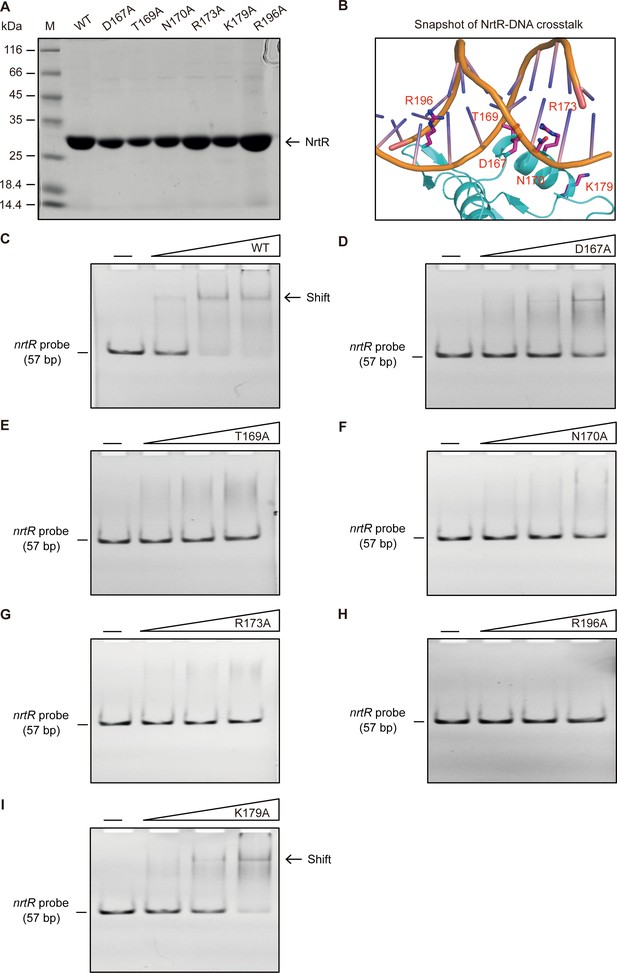
Mapping of NrtR-DNA interactions.
(A) 12% SDS-PAGE profile of the purified NrtR and its mutants. (B) Enlarged view of the predicted DNA-binding residues of M. smegmatis NrtR. (C) Binding ability of wild-type NrtR to the nrtR probe evaluated by EMSA. (D) Binding ability of the D167A point mutant of NrtR to the nrtR probe evaluated by EMSA. (E) Loss of binding of the T169A point mutant of NrtR to the nrtR probe evaluated by EMSA. (F) Loss of binding of the N170A point mutant of NrtR to the nrtR probe evaluated by EMSA. (G) Loss of binding of the R173A point mutant of NrtR to the nrtR probe evaluated by EMSA. (H) Loss of binding of the R196A point mutant of NrtR to the nrtR probe evaluated by EMSA. (I) Impairment of binding of the K179A point mutant of NrtR to the nrtR probe evaluated by EMSA. Minus ‘--' signs denote no addition of NrtR protein, and the triangles to the right of these signs refer to the addition of increasing levels of NrtR protein (2, 5, and 10 pmols).
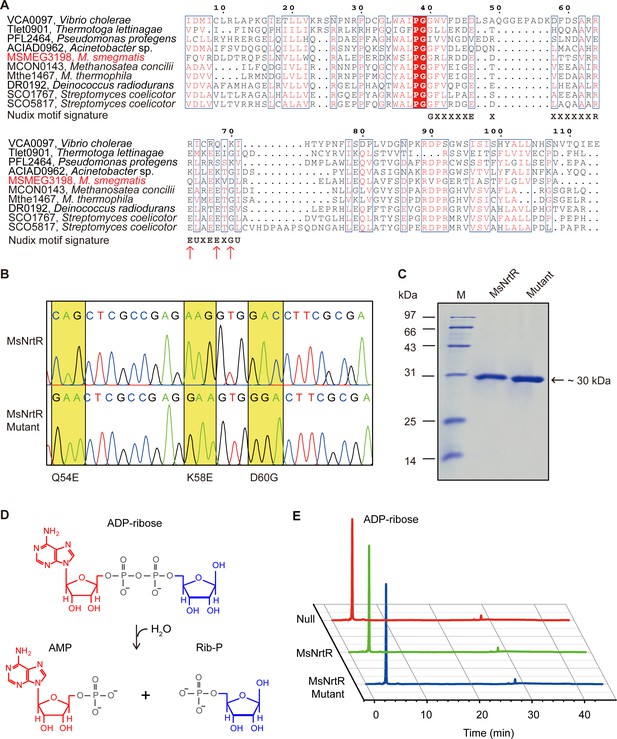
Identification of the ADPR pyrophosphatase activity of the M. smegmatis NrtR and its mutant (Q54E, K58E and D60G).
(A) Multiple sequence alignments of the Nudix domain of M. smegmatis NrtR and its homologous proteins. The multiple alignment was conducted using ClustalW2 (http://www.ebi.ac.uk/Tools/clustalw2/index.html), and the final output was expressed through processing by the ESPript 2.2 program (Robert and Gouet, 2014). Identical residues are in white letters with red background, similar residues are in red letters with white background, varied residues are in black letters, and dots represent gaps. GenBank accession numbers and organisms are as follows: VCA0097, Vibrio cholerae; Tlet0901, Thermotoga lettinagae; ACIAD0962, Acinetobacter sp.; MSMEG_3198, M. smegmatis; MCON0143, Methanosatea concilli; Mthe1467, Methanosatea themophila; DR0192, Deinococcus radiodurans; SCO1767, Streptomyces coelicotor; and SCO5817, Streptomyces coelicotor. The Nudix motif signature is shown below the sequences as GX5EX7REUXEEXGU (U: Ile, Leu, or Val). (B) The mutations (Q54E, K58E and D60G) present in M. smegmatis nrtR revealed by direct DNA sequencing. The mutation of Q54E and K58E and D60G at DNA level denotes the genotype of the mutant protein. (C) The SDS-PAGE gel of the purified NrtR and its mutant (Q54E and K58E and D60G). (D) Schematic diagram of the hydrolyzation of ADP-ribose by ADP-ribose pyrophosphatase which belongs to the Nudix hydrolase superfamily. (E) Identification of the reaction product upon hydrolysis of ADP-ribose by NrtR. Null, before the addition of the recombinant NrtR or its mutant; MsNrtR, after incubation with M. smegmatis NrtR for 30 min; MsNrtR Mutant, after incubation with NrtRQ54E&K58E&D60G.
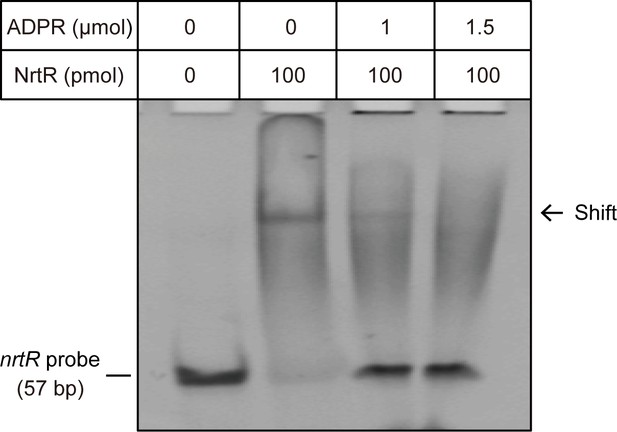
The NAD metabolite ADP-ribose interferes with the binding of MsNrtR to cognate DNA. The image shows the electrophoretic mobility of the nrtR (0.2 μM) probe incubated alone (lane 1) or with purified MsNrtR (5 μM) in the absence (lane 2) and in the presence of 50 (lane 3) and 75 (lane 4) mM of ADP-ribose. The volume of the EMSA reaction system is 20 μl.
https://doi.org/10.7554/eLife.51603.009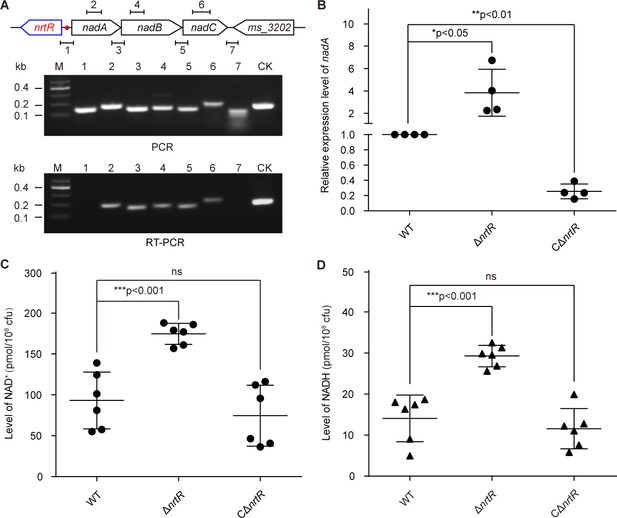
NrtR is a repressor for the nadABC operon that is responsible for NAD+ and NADH concentration in M. smegmatis.
(A) Genetic organization and transcriptional analyses of the nrtR and its neighboring de novo NAD+ synthesis genes. The arrows represent open reading frames, and the numbered short lines (1 to 7) represent the specific PCR amplicons that were observed in the following PCR and RT-PCR assays (in the bottom panels). PCR and RT-PCR were applied to analyze the transcription of the putative NAD+ de novo synthesis loci. The primer numbering was identical to that shown in the top panel. CK (control) denotes the 16S rDNA. (B) RT-qPCR analyses of nad operon expression in the wild-type strain and in the ΔnrtR mutant and nrtR complementary strains. RT-qPCR experiments were performed at least three times and the data were expressed as means ± standard deviations (SD). The p-value was calculated using one-way ANOVA along with Tukey's test. *p<0.05 and **p<0.01. Comparison of the intra-cellular level of NAD+ (C) and NADH (D) among the WT, ΔnrtR and CΔnrtR strains. Each dark circle or triangle represents an independent experiment. The data are shown as means ± SD. The statistical significance of differences among WT, ΔnrtR and CΔnrtR was determined by Student’s t test and by ANOVA with heterogeneous variances. ***p<0.001; ns, no significant difference.
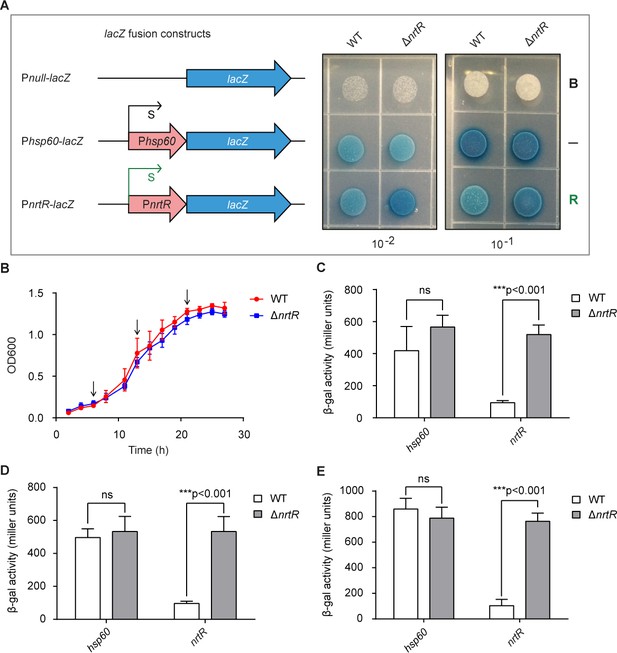
In vivo evidence that MsNrtR is an auto-repressor.
(A) LacZ-based visualization of the auto-regulation of NrtR in M. smegmatis. Left column: schematic representation of promoter-lacZ transcriptional fusions. A promoter-less LacZ refers to the blank control (abbreviated as ‘B’); the hsp60 promoter-fused LacZ acts as the negative control (indicated by ‘–'); and the nrtR promoter-driven LacZ is used to evaluate the regulatory role of the NrtR repressor (highlighted with ‘R’). Right column: the exponentially growing M. smegmatis cultures of the wild-type and ΔnrtR strains were diluted appropriately and spotted onto 7H10 plates containing 50 μg/ml kanamycin and 50 mg/ml X-gal. The plates were incubated at 37°C for 48 hr. (B) Growth curves of WT and nrtR deletion strains carrying a transcriptional fusion plasmid, pMV261-promoter-lacZ. Cultures were grown in LB medium supplemented with 0.5% glycerol, 0.05% tween 80, and 50 mg/ml kan at 37°C, 220 rpm, and absorbance at 600 nm was recorded at 2 hr intervals for 28 hr. (C) Transcriptional levels of nrtR in lag-phase cultures of the wild-type and in the ΔnrtR mutant of M. smegmatis, evaluated using lacZ-transcriptional fusions. A LacZ controlled by the hsp60 promoter acts as the negative control. Results are expressed as an average ± standard deviation (SD) from no less than three independent tests. (D) Transcriptional levels of nrtR in exponential-phase cultures of wild-type and nrtR-deleted strains of M. smegmatis, evaluated using lacZ-transcriptional fusions (E) Transcriptional levels of nrtR in stationary-phase cultures of wild-type M. smegmatis and its nrtR deletion mutant, evaluated using lacZ-transcriptional fusions. Data are presented as mean ± SD. The p-value was measured using one-way ANOVA along with Tukey's test. **, p<0.01; ***, p<0.001.
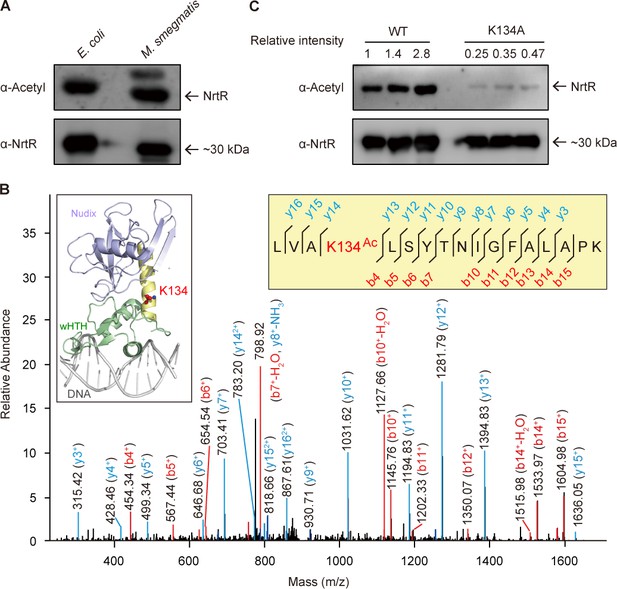
The discovery of acetylation of K134 in MsNrtR.
(A) Use of Western blot to probe the acetylation of recombinant MsNrtR protein in both E. coli and M. smegmatis. The two forms of recombinant NrtR protein were purified from E. coli BL21 and M. smegmatis, and analyzed by western blotting using both anti-acetyl-lysine antibody (α-Acetyl) and poly anti-MsNrtR rabbit serum. The bigger version of MsNrtR is produced by the pET28 expression plasmid in E. coli, whose N-terminus is fused to the 6xHis-containing tag of 23 residues (Supplementary file 1). By contrast, the smaller version of MsNrtR is generated by pMV261 in M. smegmatis, which is only tagged with C-terminal 6xHis. The altered molecular mass (~2 kDa) is the reason why the migration rate of protein electrophoresis differs slightly for the two MsNrtR versions. A representative result is given from three independent trials. (B) The discovery of a unique Lys134 acetylation site in MsNrtR. A LC/MS spectrum reveals that a charged peptide (LVAkLSYTNIGFALAPK) of MsNrtR bears an acetylated lysine (K134Ac). The sequence depicted in the yellow box illustrates the K134 site of acetylation in the context of the modeled structure of MsNrtR-DNA. (C) The mutation of K134A results in reduced acetylation of MsNrtR in M. smegmatis MC2 155 (Magni et al., 2004). A representative result from three independent experiments is given.
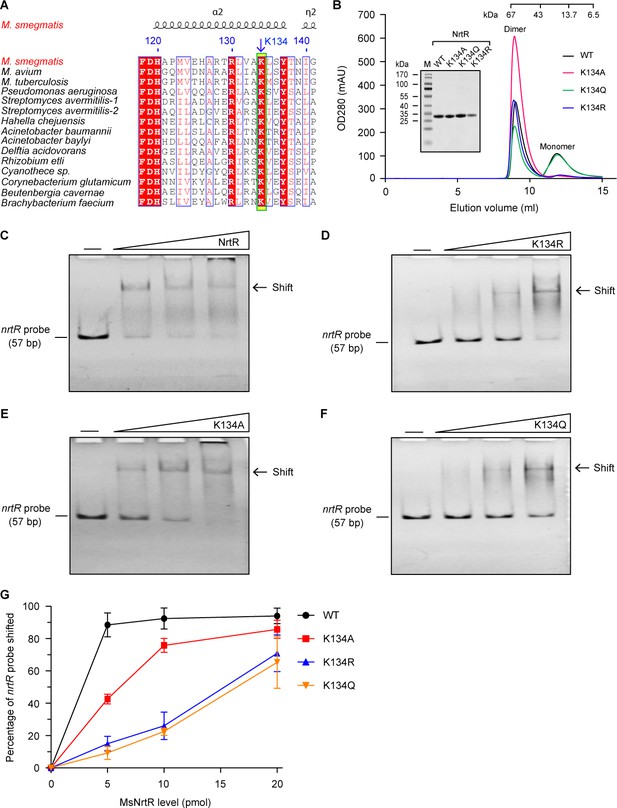
Dependence on K134 acetylation in the binding of MsNrtR to a cognate DNA target.
(A) Conservation analysis of the NrtR K134 residue of M. smegmatis through multiple sequence alignment. An arrow denotes the conserved lysine residue. The amino acid sequences are from M. smegmatis MC2 155 (Magni et al., 2004) (Acc. no.: YP_887512), M. avium (Acc. no.: WP_011725315), M. tuberculosis H37Rv (Acc. no.: NP_216109), Pseudomonas aeruginosa (Acc. no.: PTC34113.1), Streptomyces avermitilis-1 (Acc. no.: WP_010987913), Streptomyces avermitilis-2 (Acc. no.: WP_010983884), Hahella chejuensis (Acc. no.: WP_011400661), Acinetobacter baumannii (Acc. no.: WP_000155308), Acinetobacter baylyi (Acc. no.: WP_011182178), Delftia acidovorans (Acc. no.: WP_012207554), Rhizobium etli (Acc. no.: WP_011428339.1), Cyanothece sp. (Acc. no.: WP_009544558), Corynebacterium glutamicum (Acc. no.: WP_011014097), Beutenbergia cavernae (Acc. no.: YP_002882784), and Brachybacterium faecium (Acc. no.: YP_003153838). (B) Size exclusion chromatographic profile and inset SDS-PAGE gel of M. smegmatis NrtR and its three mutants K134A, K134Q, and K134R. This work was carried out as described in Figure 3—figure supplement 1. Designations: OD280, optical density at 280 nm; mAU, milli-absorbance units. (C) Binding of wild-type NrtR to DNA containing the MsNrtR palindrome evaluated by EMSA. (D) Binding of the NrtR K134R mutant to DNA containing the MsNrtR palindrome evaluated by EMSA. (E) Binding of the NrtR K134A mutant to DNA containing the MsNrtR palindrome evaluated by EMSA. (F) Binding of the NrtR K134Q mutant to DNA containing the MsNrtR palindrome evaluated by EMSA. The amount of NrtR added per lane is increased from left to right (0, 5, 10, and 20 pmol), with the minus sign denoting the absence of NrtR protein. (G) Comparative analyses of the relative DNA-binding affinity of MsNrtR and its three K134 mutants (K134R, K134A and K134Q).
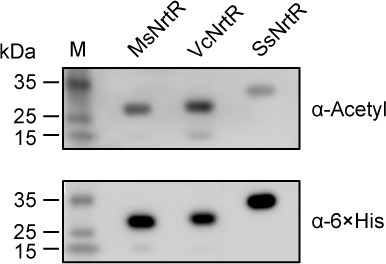
Acetylation is ubiquitous in three bacterial NrtR proteins.
Western blot was routinely applied to detect the acetylation of the NrtR homologs MsNrtR (MSMEG_3198, 27.35 kDa), VcNrtR (VCA0097, 29 kDa) of Vibrio cholerae, and SsNrtR (SSU05_1971, 32 kDa) of Streptococcus suis. These recombinant NrtR proteins were overexpressed, purified from E. coli BL21, and analyzed by Western blot using both anti-acetyl-lysine antibody (α-Acetyl) and anti-6 ×His antibody (α−6 × His). A representative result is given from three independent trials. The molecular weights of the NrtR homologs were predicted by an online server (web.expasy.org).
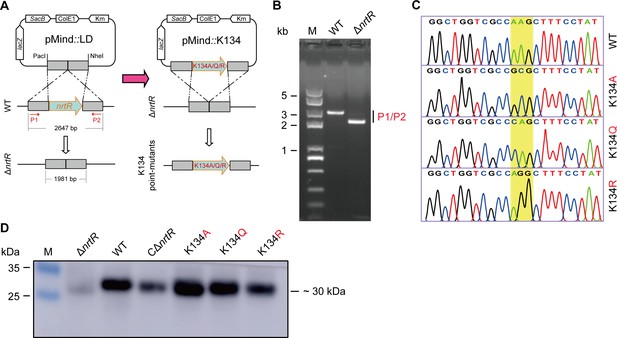
Construction and identification of NrtR K134 point mutants on the M. smegmatis chromosome.
(A) A schematic for the construction of an in-frame deletion of nrtR and its K134 point mutants on the chromosome. The M. smegmatis lines carrying the point-mutants of K134 in NrtR are constructed by re-introducing the mutated version of nrtR after the removal of full-length nrtR-encoding sequence from M. smegmatis. Primers 1 and 2, abbreviated as P1 and P2, represent the primer pair nrtR-U (PacI)-F and nrtR-D-(NheI)-R (Supplementary file 2). (B) PCR confirmation of the in-frame nrtR deletion mutant of M. smegmatis, ΔnrtR. (C) Direct DNA sequencing of the three point-mutants (K134A, K134Q, and K134R) of nrtR on the chromosome of M. smegmatis. (D) Western blot to evaluate the expression of NrtR in wild-type M. smegmatis and its derivatives (ΔnrtR and the three point-mutants [K134A, K134R, and K134Q]). Anti-MsNrtR rabbit serum refers to a primary antibody.
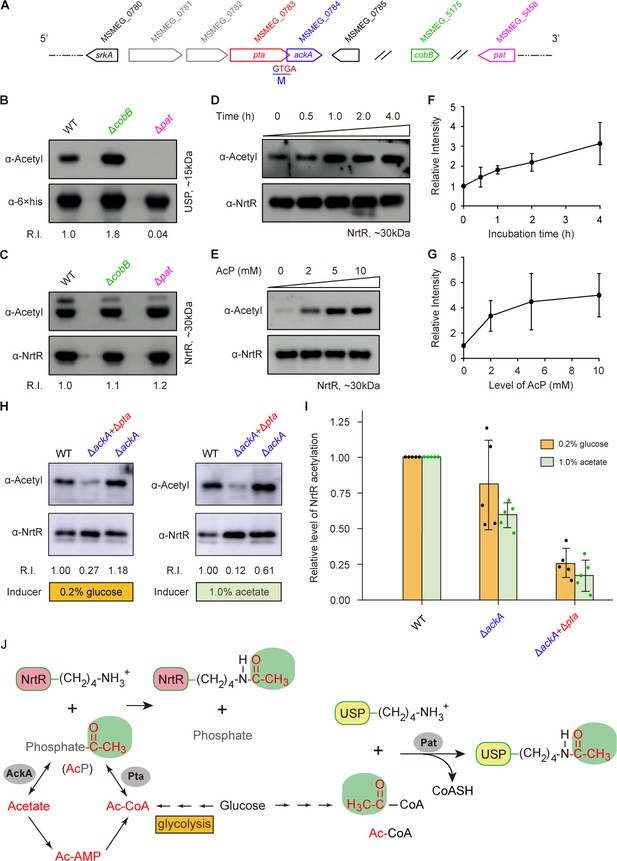
Acetyl phosphate-mediated acetylation of MsNrtR.
(A) Genetic context of the two types of acetylation pathways in M. smegmatis. The two genes pat (MSMEG_5458) and cobB (MSMEG_5175) are responsible for the reversible enzymatic route of acetylation. The two loci ackA (MSMEG_0784) and pta (MSMEG_0783) participate in the non-enzymatic AcP pathway. Of note, ackA and pta are two overlapping loci that appear as an operon. (B) The acetylation levels of USP are dependent on the Pat/CobB-requiring enzymatic route in M. smegmatis. USP denotes the universal stress protein (MSMEG_4207). Using the recombinant plasmid pMV261-usp, the 6 × His tagged USP protein was expressed in wild-type M. smegmatis and its derivatives (ΔcobB and Δpat). As a result, the acetylation levels of the purified USP proteins were detected with the pan anti-acetyl lysine antibody (α-Acetyl) and an anti-6 ×his antibody was used as a loading control. A representative result for three independent experiments displayed. (C) The acetylation levels of NrtR are not distinguishable in the three strains of M. smegmatis (wild-type, ΔcobB and Δpat). 6 × His tagged MsNrtR was expressed in the three strains described for panel (B) using pMV261-nrtR. The acetylation levels of the purified MsNrtR proteins were determined using the α-Acetyl antibody, and anti-NrtR antiserum (α-NrtR) acted as a loading control. Western blots were conducted in triplicates. (D) Western-blot-based detection of the in vitro non-enzymatic acetylation of MsNrtR using AcP as the phosphate donor. Acetylation of MsNrtR by AcP (10 mM) was measured by incubating MsNrtR and AcP for 0, 0.5, 1, 2 and 4 hr at 37°C. The concentration of NrtR was determined by Western blot with anti-NrtR serum as a primary antibody (lower panel). (E) Acetylation of MsNrtR is AcP dose-dependent. MsNrtR was incubated with different levels of AcP (0, 2, 5 and 10 mM) for 2 hr at 37°C. (F) Altered acetylation of MsNrtR as incubation progresses over time with constant AcP. Acetylation was quantified using Image J software and normalized to the signal at 0 hr. (G) MsNrtR acetylation at various levels of AcP. Data were measured with Image J software and normalized to the signal at 0 mM AcP. Data are shown as mean ± standard deviation (SD). (H) In vivo evidence that the AcP pathway is associated with NrtR acetylation. In addition to the parental strain, a single mutant (ΔackA) and double mutant (ΔackA+Δpta) were used to prepare the recombinant MsNrtR proteins with varied levels of acetylation. Of note, the bacterial growth conditions were supplemented with an inducer of 0.2% glucose or 1.0% acetate recommended by Weinert et al. (2013). The Western blot was performed as described for panels (B) and (C). Representative results of three or more independent experiments are shown. (I) Contribution of the AckA and Pta-requiring AcP pathway to NrtR acetylation. The acetylation signal was quantified using Image J software, and the density in the WT was normalized as 1. Each dot denotes a Western blot experiment. (J) Working model for non-enzymatic acetylation of MsNrtR in a metabolic context, and the working model for the enzymatic acetylation of USP. Abbreviations: MsNrtR, M. smegmatis NrtR; AcP, Acetyl-phosphate; AcAMP, Acetyl-AMP; Ac-CoA, acetyl-CoA; USP (MSMEG_4207), universal stress protein (130aa); Pta (MSMEG_0783), phosphate acetyltransferase (692aa); and AckA (MSMEG_0784), acetate kinase (376aa).
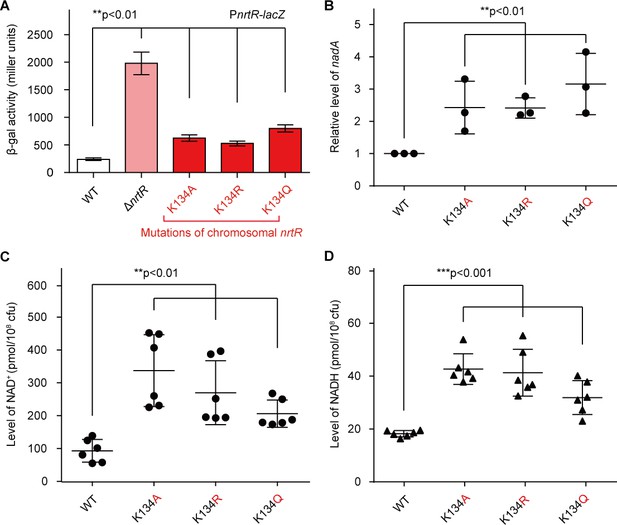
Acetylation of K134 in MsNrtR determines its role in the homeostasis of the intracellular NAD+ pool.
(A) Genetic assays for the auto-repression of nrtR using PnrtR-lacZ transcriptional fusion. These results suggest that functional impairments in K134 acetylation lead to de-repression of nrtR, as does the removal of nrtR. (B) RT-qPCR analyses of the transcription of the nad operon in the mutant carrying a point-mutation of K134 in nrtR (K134A, K134R and K134Q). Levels of intracellular NAD+ (C) and NADH (D) in the WT nrtR strain and its point-mutants (K134A, K134R and K134Q). All of the experiments were performed at least three times, and the data are presented as means ± SD. The p-values were calculated using one-way ANOVA along with Tukey's test.
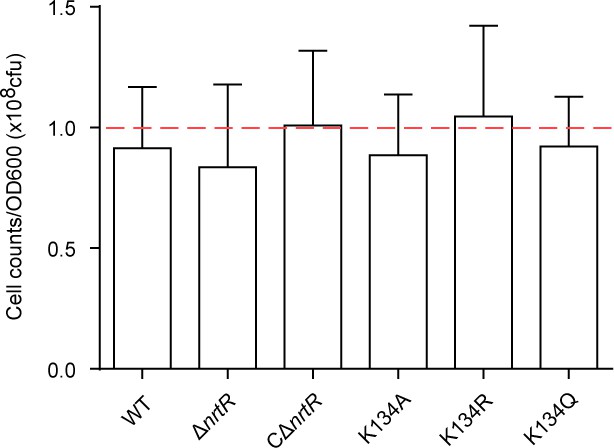
Determining the number of live cells at OD600 during the exponential phase.
The wild-type and mutant strains were cultured to exponential phase, harvested by centrifugation, and then re-suspended with fresh 7H9 broth. The optical density (OD600) was adjusted to ~1.0. Following series of 10-fold dilution (1 × 104, 1 × 105 and 1 × 106 times) with fresh 7H9 broth, 100 μl of diluted bacterial suspension was plated on LB agar plates. Colony counting was performed after incubation at 37°C for three days. It suggested that the OD600 of M. smegmatis represents around 108 cells (indicated with a dashed line).
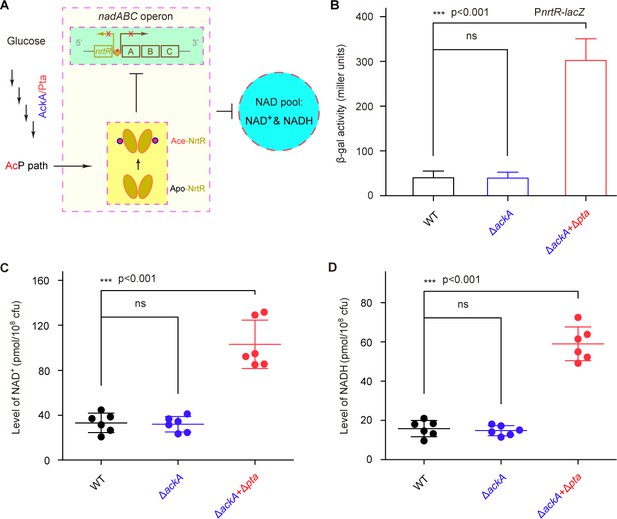
AcP-pathway-dependent repression of NAD+ synthesis by NrtR in M. smegmatis.
(A) Scheme for the maintenance of NAD+ homeostasis by AcP-dependent NrtR acetylation (B) The removal of ackA and pta from M. smegmatis increases the β-gal level of Pnrt-lacZ transcriptional fusion in the presence of 0.2% glucose in the growth medium. (C) The level of the cytosolic NAD+ pool is elevated in the double mutant of M. smegmatis (ΔackA + Δpta) in the growth condition with 0.2% glucose added. (D) The inactivation of the AcP path gives an increase of ~3 fold in the cytosolic NADH pool Three strains of M. smegmatis (WT, ΔackA, and ΔackA + Δpta) were cultivated in 7H10 medium supplemented with 0.2% glucose. No less than three independent measures were carried out, and the values presented here are averages ± SD. *, p<0.001; ns, no significance.
Additional files
-
Supplementary file 1
Strains and plasmids used in this study.
- https://doi.org/10.7554/eLife.51603.020
-
Supplementary file 2
Primers used in this study.
- https://doi.org/10.7554/eLife.51603.021
-
Supplementary file 3
Nudix family protein in M. smegmatis MC2 155 (Magni et al., 2004).
- https://doi.org/10.7554/eLife.51603.022
-
Transparent reporting form
- https://doi.org/10.7554/eLife.51603.023





