Purinergic signaling in cochlear supporting cells reduces hair cell excitability by increasing the extracellular space
Figures
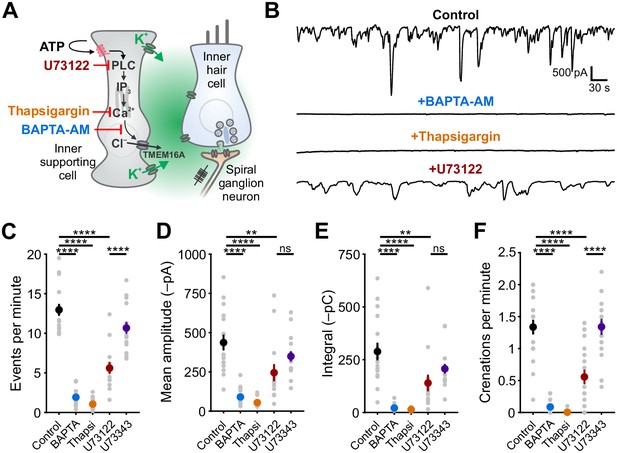
Ca2+ release from intracellular stores is required for spontaneous currents and crenation in inner supporting cells.
(A) Model of ATP-mediated depolarization of inner hair cells. ATP: adenosine triphosphate, PLC: phospholipase C, IP3: inositol triphosphate, TMEM16A: transmembrane member 16A (Ca2+-activated Cl– channel). Inhibitors of key steps in this pathway are indicated. (B) Whole-cell voltage-clamp recordings from inner supporting cells after pre-incubating with indicated inhibitors. (C) Quantification of ISC spontaneous current frequency in the presence of the indicated inhibitors. Data shown as mean ± SEM. n = 16 cells, 11 cochleae from six postnatal day (P) 6–8 rats (control), 18 cells, 10 cochleae from five rats (BAPTA-AM; 100 μM), 20 cells, 11 cochleae from six rats (Thapsigargin; 2 μM), 14 cells, 11 cochleae from seven rats, (U73122; 10 μM), and 16 cells, 10 cochleae from five rats (U73343; 10 μM). ****p<5e-5, one-way ANOVA. (D) Quantification of ISC spontaneous current amplitude in the presence of indicated inhibitors. Data shown as mean ± SEM. n values are reported in (C) (one-way ANOVA; ****p<5e-5, **p<0.005, ns: not significant). (E) Quantification of ISC spontaneous current charge transfer (integral) in the presence of indicated inhibitors. Data shown as mean ± SEM. n values are reported in (C) (one-way ANOVA; ****p<5e-5, **p<0.005, ns: not significant). (F) Quantification of ISC crenation (cell shrinkage) frequency in the presence of indicated inhibitors. Data shown as mean ± SEM. n = 19 videos, 11 cochleae from six rats (control), 15 videos, 8 cochleae from four rats (BAPTA-AM), 22 videos, 12 cochleae from six rats (Thapsigargin), 23 videos, 17 cochleae from 10 rats (U73122), and 20 videos, 10 cochleae from five rats (U73343) (one-way ANOVA; ****p<5e-5, **p<0.005, ns: not significant). See Figure 1—source data 1 for plotted values and statistics.
-
Figure 1—source data 1
Plotted values and statistics for Figure 1.
- https://cdn.elifesciences.org/articles/52160/elife-52160-fig1-data1-v2.xlsx
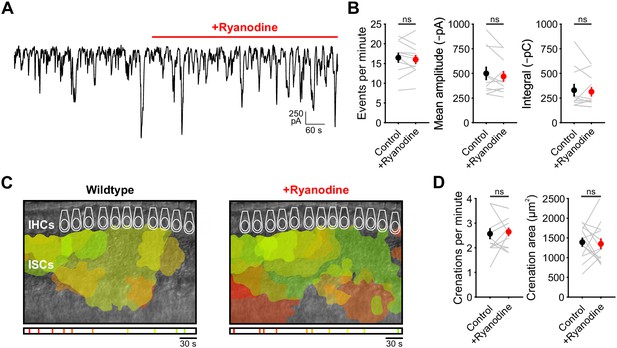
Inhibition of calcium-induced calcium release does not alter spontaneous currents or crenations.
(A) Spontaneous inward currents recorded from an ISC before and after application of ryanodine (10 μM). Recordings were performed near physiological temperature 32–34°C. (B) Quantification of event frequency, amplitude, and charge transfer (integral). Each window measured was 5 min in length. n = 11 ISCs, 11 cochleae from 8 P6-8 mice (two-tailed paired Student's t test with Bonferroni correction; ns, not significant). (C) Intrinsic optical imaging performed before and after application of ryanodine (10 μM). Detected crenations are outlined in colors based on time of occurrence as indicated by timeline below image. Imaging was performed near physiological temperature 32–34°C. (D) Quantification of crenation frequency and area before and after application of ryanodine. n = 13 cochleae from 8 P6-8 mice (two-tailed paired Student's t test with Bonferroni correction; ns, not significant). See Figure 1—figure supplement 1—source data 1 for plotted values and statistics.
-
Figure 1—figure supplement 1—source data 1
Plotted values and statistics for Figure 1—figure supplement 1.
- https://cdn.elifesciences.org/articles/52160/elife-52160-fig1-figsupp1-data1-v2.xlsx
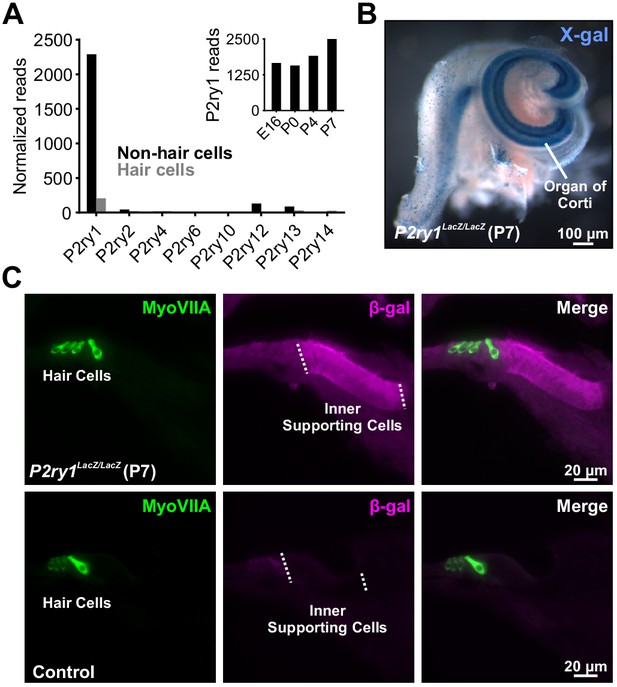
The metabotropic P2Y1 receptor is highly expressed by ISCs.
(A) Expression levels of metabotropic purinergic receptors in hair cells (gray) and non-sensory cells (black) of the developing cochlea (postnatal day 7, P7). (inset) P2ry1 expression in non-sensory cells over development. Data adapted from Scheffer et al. (2015). (B) Image of a cochlea following X-gal reaction in P2ry1 LacZ reporter mice. (C) Immunostaining for B-galactosidase in cochleae from P7 P2ry1 LacZ (top) and control (bottom) cochlea.
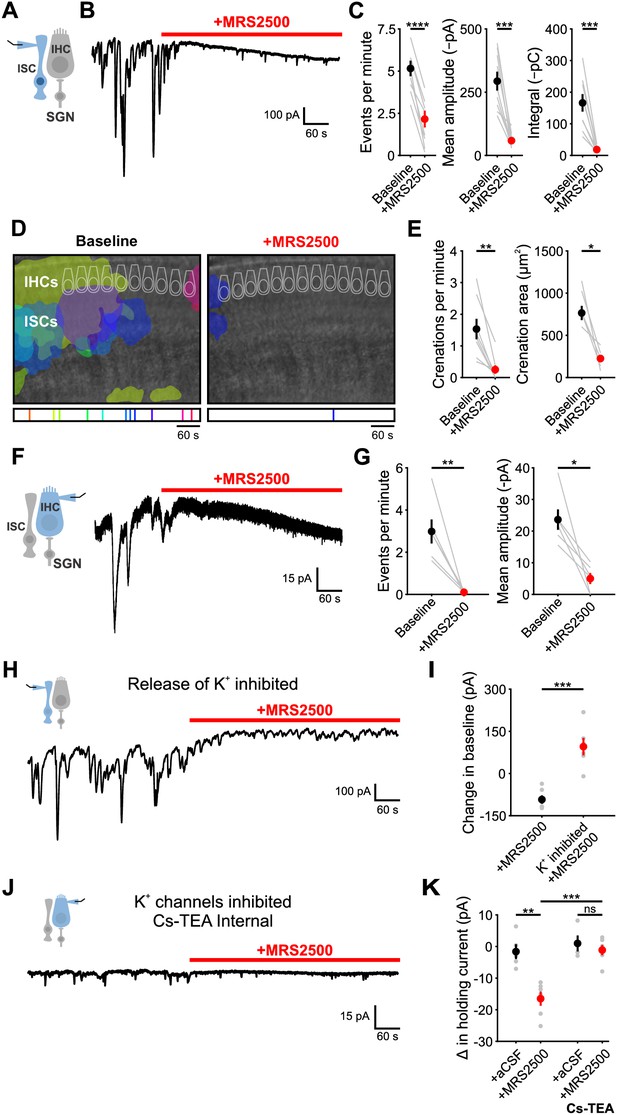
P2Y1 inhibition abolishes spontaneous currents in inner supporting cells and inner hair cells.
(A) Schematic of whole-cell recording configuration from ISCs. (B) Spontaneous inward currents recorded from an inner supporting cell before and during application of MRS2500 (1 μM). Recordings were performed at room temperature (~25°C). (C) Plot of event frequency, amplitude, and integral (charge transfer) before and after application of MRS2500. Measurement periods were five minutes long. n = 9 ISCs, 9 cochleae from 8 P6-8 mice (two-tailed paired Student’s t test; ****p<5e-5, ***p<0.0005) (D) Intrinsic optical imaging performed before and after application of the P2RY1 antagonist, MRS2500 (1 μM). Detected crenations are outlined in colors based on time of occurrence as indicated by timeline below image. Imaging was performed at room temperature (~25°C). (E) Plot of crenation frequency and area before and after application of MRS2500. n = 8 videos, 8 cochleae from 8 P6-8 mice (two-tailed paired Student's t test; **p<0.005) for frequency calculation and n = 5 cochleae (two-tailed paired Student's t test; *p<0.05) for area calculation. Cochleae that did not crenate after MRS2500 were excluded from the area calculation. (F) Schematic of whole-cell recording configuration from IHCs. (right) Whole-cell voltage clamp recording from an IHC before and during application of MRS2500. (G) Plots of event frequency and amplitude before and after application of MRS2500. n = 6 IHCs, 6 cochleae from 6 P6-8 mice (two-tailed paired Student's t test with Bonferroni correction; **p<0.005, *p<0.05). (H) Whole-cell voltage clamp recording of an ISC with application of MRS2500 following pre-incubation in aCSF containing CdCl2 (100 μM), TTX (1 μM), ouabain (10 μM), and bumetanide (50 μM) to limit potassium release into the extracellular space. (I) Plot of the change in holding current, defined as the 95% percentile current value for each period. n = 6 ISCs, 6 cochleae from 6 P6-8 mice for each condition (two-tailed Student’s t test; ***p<0.0005). (J) Whole-cell voltage clamp recording of an IHC with a Cs-TEA internal solution (to inhibit K+ channels) before and after MRS2500 application. (K) Plot of the change in IHC holding current following control (superfusion of aCSF only) and MRS2500 with K-MeS and Cs-TEA internal. n = 5 IHCs, 4 cochleae from 4 P6-8 mice for aCSF, n = 6 IHCs, 6 cochleae from six mice for MRS2500, n = 4 IHCs, 3 cochleae from three mice for aCSF with Cs-TEA internal, and n = 6 IHCs, 6 cochleae from six mice for MRS2500 with Cs-TEA internal (one-way ANOVA; ***p<0.005, **p<0.005, ns, not significant). See Figure 3—source data 1 for plotted values and statistics.
-
Figure 3—source data 1
Plotted values and statistics for Figure 3.
- https://cdn.elifesciences.org/articles/52160/elife-52160-fig3-data1-v2.xlsx
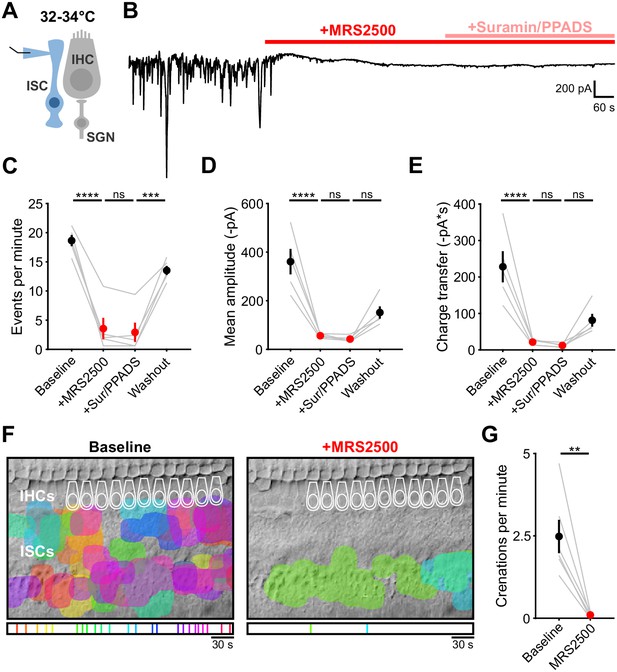
P2ry1 inhibition abolishes spontaneous inward currents near physiological temperature.
(A) Schematic of whole-cell recording configuration from ISCs. (B) Spontaneous inward currents recorded from an ISC before and after application of MRS2500 (1 μM) and subsequent broad spectrum purinergic antagonists suramin (100 μM) and PPADS (50 μM). Recordings were performed near physiological temperature 32–34°C. (C) Quantification of event frequency. Each window measured was 5 min in length, washout was taken 20 min after superfusion of aCSF. n = 5 ISCs, 5 cochleae from 5 P7-8 mice (one-way ANOVA; ****p<5e-5, ***p<0.0005, ns, not significant). (D) Quantification of event amplitude. n = 5 ISCs, 5 cochleae from 5 P7-8 mice (one-way ANOVA; ****p<5e-5, ns, not significant). (E) Quantification of average charge transfer (integral). n = 5 ISCs, 5 cochleae from 5 P7-8 mice (one-way ANOVA; ****p<5e-5, ns, not significant). (F) Intrinsic optical imaging performed before and after application of the P2Y1 antagonist, MRS2500 (1 μM). Detected crenations are outlined in colors based on time of occurrence as indicated by timeline below image. Imaging was performed near physiological temperature 32–34°C. (E) Plot of crenation frequency before and after application of MRS2500. n = 6 videos, 6 cochleae from 3 P6-8 mice (two-tailed paired Student's t test; **p<0.005). See Figure 3—figure supplement 1—source data 1 for plotted values and statistics. (G).
-
Figure 3—figure supplement 1—source data 1
Plotted values and statistics for Figure 3—figure supplement 1.
- https://cdn.elifesciences.org/articles/52160/elife-52160-fig3-figsupp1-data1-v2.xlsx
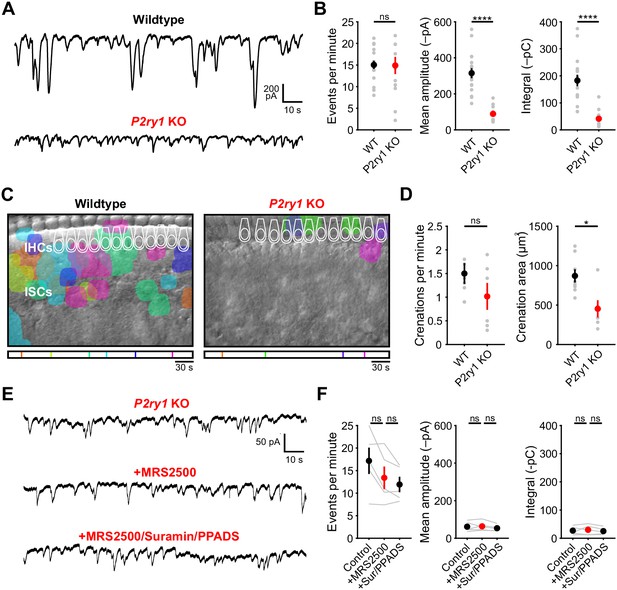
Spontaneous inward currents and crenations are dramatically reduced in P2ry1 KO mice.
(A) Spontaneous inward currents recorded from ISCs in wildtype and P2ry1 KO mice. Recordings were performed near physiological temperature 32–34°C. (B) Plots of event frequency, amplitude, and integral (charge transfer). n = 17 ISCs, 17 cochleae from 17 P6-8 wildtype mice and 14 ISCs, 14 cochleae from 9 P2ry1 KO mice (two-tailed Student's t-test with Bonferroni correction; ****p<0.0005, ns, not significant). (C) Intrinsic optical imaging performed in wildtype and P2ry1 KO mice. Detected crenations are outlined in colors based on time of occurrence as indicated by the timeline below image. Imaging was performed at room temperature. (D) Plots of crenation frequency and area in wildtype and P2ry1 KO mice. n = 8 videos, 8 cochleae from 6 P6-8 wildtype mice and six videos, 6 cochleae from 5 P2ry1 KO mice (two-tailed paired Student's t test with Bonferroni correction; *p<0.05, ns, not significant). (E) Spontaneous inward currents recorded from an inner supporting cell in P2ry1 KO mice before and during application of MRS2500 (1 μM) and subsequent broad spectrum purinergic antagonists suramin (100 μM) and PPADS (50 μM). (F) Plots of event frequency, amplitude, and charge transfer. n = 5 ISCs, 5 cochleae from 4 P2ry1 KO mice (one-way ANOVA; ns, not significant). See Figure 3—figure supplement 2—source data 1 for plotted values and statistics.
-
Figure 3—figure supplement 2—source data 1
Plotted values and statistics for Figure 3—figure supplement 2.
- https://cdn.elifesciences.org/articles/52160/elife-52160-fig3-figsupp2-data1-v2.xlsx
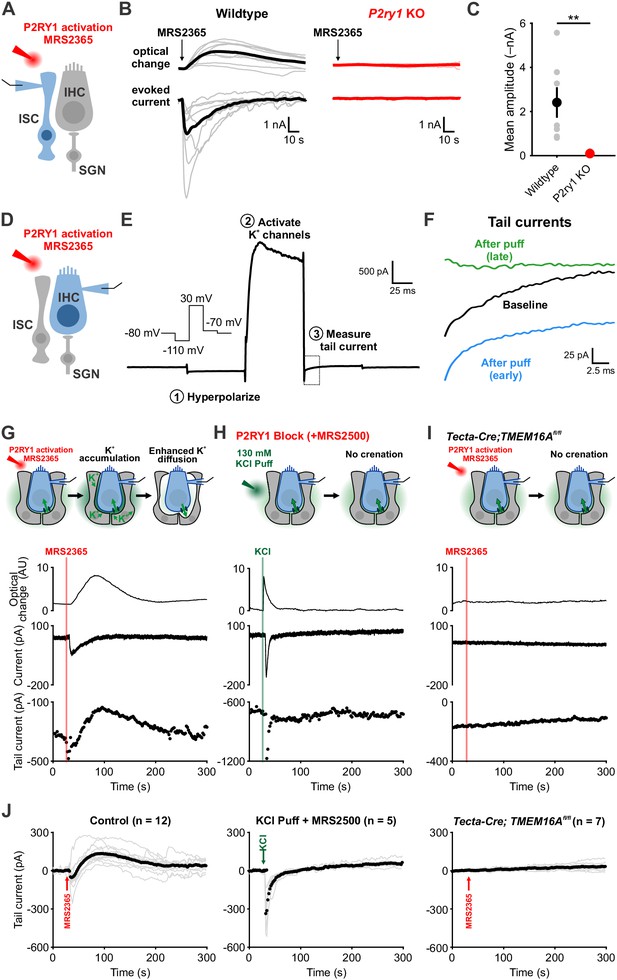
Activation of P2RY1 results in an initial accumulation of extracellular K+, followed by crenation and enhanced K+ clearance.
(A) Schematic of whole-cell recording configuration from ISCs with puffs of MRS2365 (10 μM), a P2RY1 agonist. (B) Optical change (crenation) and current elicited with MRS2365 puffs in wildtype and P2ry1 KO mice (C) Plot of mean current amplitude with MRS2365 puffs. n = 8 ISCs, 8 cochleae from 4 P6-8 wildtype mice and n = 7 ISCs, 7 cochleae from four from P2ry1 KO mice (two-tailed Student’s t test; **p<0.005). (D) Schematic of whole-cell recording configuration from IHCs with puffs of MRS2365 (10 μM). (E) Example current trace and voltage-protocol designed to measure potassium accumulation. This protocol consisted of: (1) a hyperpolarizing step to −110 mV to relieve K+ channel inactivation, (2) a depolarizing step to +30 mV to activate outward K+ currents, and (3) a step to −70 mV to obtain a ‘tail’ current. Dashed box indicated tail current measurement period indicated in (F). (F) Tail currents observed during baseline, immediately following the MRS2365 puff (with 2 s), and after the puff (30 s). (G) Model of K+ dynamics following MRS2365 stimulation. Initially, extracellular K+ rapidly increases following stimulation, but ISCs crenate, increasing the amount of extracellular space and K+ buffering. (bottom) Exemplar optical change (crenation), holding current, and tail current as a function of time with respect to MRS2365 puff. (H) Similar to G, but with KCl puffs (130 μM) in cochleae treated with MRS2500. (I) Similar to G, but in Tecta-Cre;TMEM16Afl/fl mice where TMEM16A has been conditionally removed from the sensory epithelium (see Figure 4—figure supplement 1). No crenations were observed with MRS2365 stimulation. (J) Group tail currents across conditions from G-I. Gray lines indicate individual recordings; black points indicate the mean. Baseline was normalized to 0 pA for all traces. n = 12 IHCs, 11 cochleae from 9 P6-8 wildtype mice with MRS2365 stimulation, n = 5 IHCs, 5 cochleae from five wildtype mice with KCl stimulation, and n = 8 IHCs, 8 cochleae from 5 Tecta-Cre;TMEM16Afl/fl mice with MRS2365 stimulation. See Figure 4—source data 1 for plotted values and statistics.
-
Figure 4—source data 1
Plotted values and statistics for Figure 4.
- https://cdn.elifesciences.org/articles/52160/elife-52160-fig4-data1-v2.xlsx
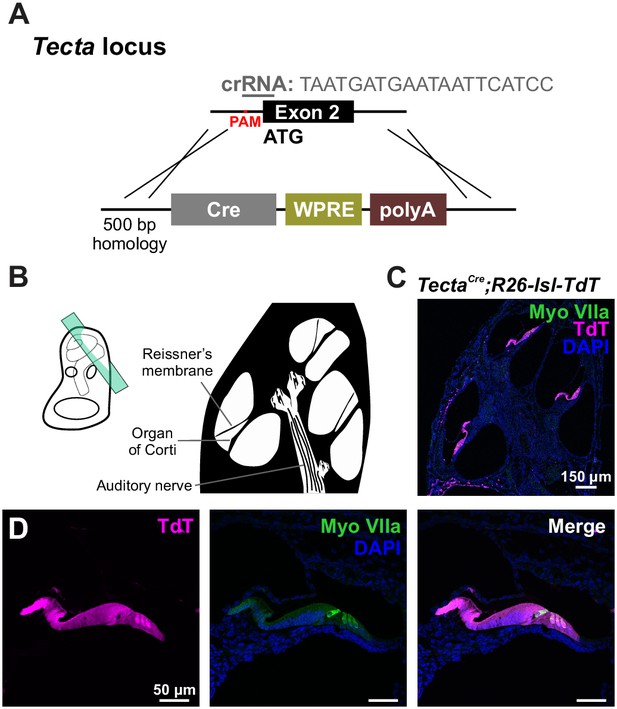
Crispr-Cas9 mediated generation of the supporting cell specific Tecta-Cre mouse line.
(A) Targeting strategy for introducing an iCre coding sequence into the endogenous Tecta locus. Note: start ATG is located in exon 2 of the Tecta locus. (B) Schematic of temporal bone with sectioning orientation indicated with green plane. (C) TdTomato reporter expression observed along the entire length of a P7 cochlea. Expression was absent in stria vascularis and very sparse in SGNs. (D) TdTomato reporter expression observed in nearly all cells throughout the sensory epithelium, including hair cells (MyoVIIa, green).
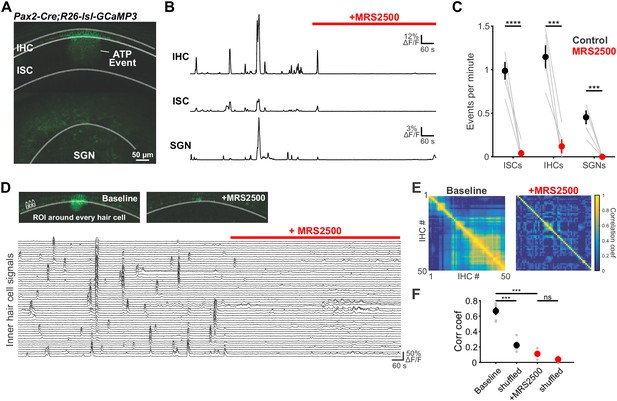
Large-scale coordinated activity in the cochlea requires P2RY1.
(A) Exemplar Ca2+ transient in excised cochlea from Pax2-Cre;R26-lsl-GCaMP3 mice. Note the coordinated activation of ISCs, IHCs, and SGNs. (B) Traces of fluorescence intensity over time taken from ROIs that span the entire IHC, ISC, and SGN regions indicated in (A). (C) Plot of event frequency before and during application of MRS2500 (1 μM). n = 5 cochleae from 3 P5+1DIV (day in vitro) Pax2-Cre;R26-lsl-GCaMP3 mice (two-tailed paired Student's t test with Bonferroni correction; ****p<5e-5, ***p<0.0005). (D) Exemplar images of IHC Ca2+ transients. ROIs were drawn around every IHC for subsequent analysis (bottom). (E) Correlation matrices generated by calculating the linear correlation coefficient for all IHC pairs before and after MRS2500 application. (F) Plot of average correlation coefficient calculated between the four nearest IHCs. n = 5 P5+1DIV (day in vitro) cochleae from 3 Pax2-Cre;R26-lsl-GCaMP3 mice (two-tailed paired Student's t test with Bonferroni correction; ***p<0.0005, ns, not significant). See Figure 5—source data 1 for plotted values and statistics.
-
Figure 5—source data 1
Plotted values and statistics for Figure 5.
- https://cdn.elifesciences.org/articles/52160/elife-52160-fig5-data1-v2.xlsx
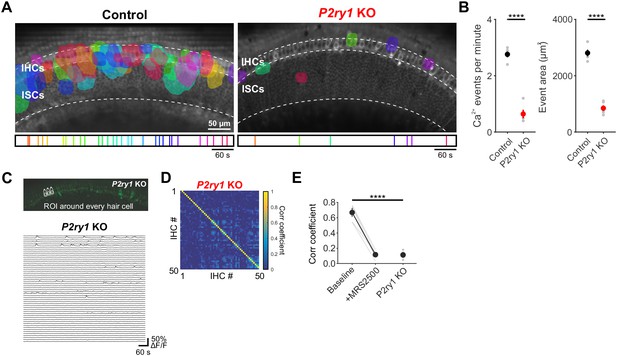
P2ry1 KO mice exhibit reduced Ca2+ transients in ISCs.
(A) Maps showing maximum area of spontaneous Ca2+ transients in control (Pax2-Cre;R26-lsl-GCaMP3) and P2ry1 KO (Pax2-Cre;R26-lsl-GCaMP3; P2ry1–/–) mice. Ca2+ transients in the ISC and IHC regions are color-coded based on time of occurrence as indicated in timeline below image. Imaging was performed at room temperature. (B) Plots of Ca2+ event frequency and area in control and P2ry1 KO mice. n = 5 P5+1DIV cochleae from three control mice and 5 P5+1DIV cochleae from 4 P2ry1 KO mice (two-tailed Student's t test with Bonferroni correction, ****p<0.05). (C) Exemplar images of IHC Ca2+ transients. ROIs were drawn around every IHC for subsequent analysis (bottom). (D) Correlation matrices generated by calculating the linear correlation coefficient for all IHC pairs in P2ry1 KO mice. (E) Plot of average correlation coefficient among the four nearest neighboring hair cells. Data from MRS2500 experiment (Figure 4) is reproduced here for comparison. n = 5 cochleae from 3 control and 4 P2ry1 KO mice (two-tailed paired Student's t test with Bonferroni correction; ****p<5e-5). See Figure 5—figure supplement 1—source data 1 for plotted values and statistics.
-
Figure 5—figure supplement 1—source data 1
Plotted values and statistics for Figure 5—figure supplement 1.
- https://cdn.elifesciences.org/articles/52160/elife-52160-fig5-figsupp1-data1-v2.xlsx
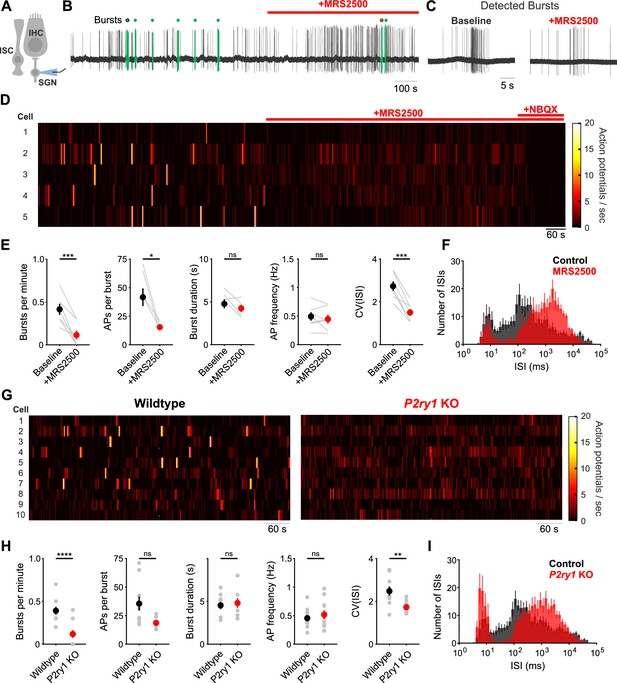
Inhibition of P2ry1 disrupts burst firing in spiral ganglion neurons.
(A) Schematic of the juxtacellular recording configuration used to assess spiral ganglion neurons (SGNs). All SGN recordings were performed at room temperature. (B) Action potentials recorded before and during application of MRS2500 (1 μM). Detected bursts are indicated in green (see Materials and methods and Materials for parameters used for burst detection). Circle with black and red outlines are expanded in (C). (C) Action potentials within a detected burst before and after MRS2500 application. (D) Raster plots indicating the average firing rate of SGNs (bin: 1 s) before and during application of MRS2500 (1 μM) and subsequent NBQX (50 μM) (E) Plots of average burst frequency, burst duration, action potentials (AP) per burst, average AP frequency, and coefficient of variation for all interspike intervals (ISIs) measured. n = 7 SGNs, 7 P5+2DIV cochleae from six wildtype mice (two-tailed paired Student's t-test with Bonferroni correction; ***p<0.0005, *p<0.05, ns, not significant). (F) Average log-binned interspike interval histograms before and after MRS2500 application. (G) Raster plots indicating the average firing rate of SGNs (bin: 1 s) in wildtype and P2ry1 KO mice. (H) Plots of average burst frequency, burst duration, action potentials (AP) per burst, average AP frequency, and coefficient of variation for all ISIs measured. n = 10 SGNs, 10 P5+2DIV cochleae from wildtype mice and 11 SGNs, 10 cochleae from 9 P2ry1 KO mice (two-tailed Student's t-test with Bonferroni correction; ****p<5e-5, **p<0.005, ns, not significant). (I) Average log-binned interspike interval histograms from wildtype and P2ry1 KO SGN recordings. See Figure 6—source data 1 for plotted values and statistics.
-
Figure 6—source data 1
Plotted values and statistics for Figure 6.
- https://cdn.elifesciences.org/articles/52160/elife-52160-fig6-data1-v2.xlsx
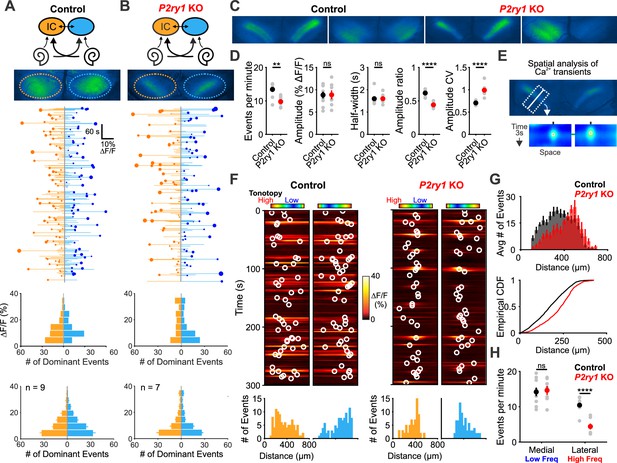
P2ry1 KO mice exhibit reduced and spatially restricted spontaneous activity in the inferior colliculus.
(A) Diagram illustrating flow of information through the auditory system and average intensity image over the 10 min imaging session. (middle) Activity over time in left and right IC in an individual where each line indicates the fluorescence intensity of each detected event; the circle indicates the dominant lobe, and the size of the circle indicated the difference in fluorescence. (bottom) Histograms showing the frequency of dominant events of a given amplitude for this experiment and for all experiments. Imaging was performed in Snap25-T2A-GCaMP6s mice (n = 9 mice). (B) Similar to (A), but in Snap25-T2A-GCaMP6s;P2ry1–/– (P2ry1 KO) mice (n = 7 P6-8 mice). (C) Images of spontaneous events in the IC of in control (Snap25-T2A-GCaMP6s) and P2ry1 KO mice (Snap25-T2A-GCaMP6s;P2ry1–/–). (D) Comparisons of average frequency, amplitude, half-width, and event ratio from control and P2ry1 KO mice. Bilateral amplitude ratio was calculated for events simultaneous across both lobes of the IC and defined as the ratio of the weak to the strong side amplitude. A ratio of 1 would indicate complete synchrony between lobes; a ratio of 0 would indicate complete asymmetry. n = 9 control and n = 7 P2ry1 KO P6-8 mice (two-tailed Student’s t test with Bonferroni correction; ****p<5e-4, **p<0.005, ns: not significant). (E) Exemplars of a single-banded event. Rectangular ROIs were placed as shown and averaged to create a `line-scan' across the tonotopic axis. (bottom) Heat maps of activity as a function of time and distance; circles indicate detected peaks. (F) Activity over a five-minute time frame in the left and right IC of control and P2ry1 KO mice. Circles indicate detected peaks. (bottom) Histograms of peak locations. (G) Histogram of average number of events across all control (black) and P2ry1 KO (red) mice. (bottom) Cumulative distribution function of event locations across the tonotopic axis pooled from all animals. Events from left and right IC were combined for each experiment. (H) Quantification of event frequency in the medial (low frequency) and lateral (high frequency) regions of the IC. n = 9 control and 7 P2ry1 KO P6-8 mice (two-tailed Student's t test with Bonferroni correction; ****p<5e-5, ns, not significant). See Figure 7—source data 1 for plotted values and statistics.
-
Figure 7—source data 1
Plotted values and statistics for Figure 7.
- https://cdn.elifesciences.org/articles/52160/elife-52160-fig7-data1-v2.xlsx
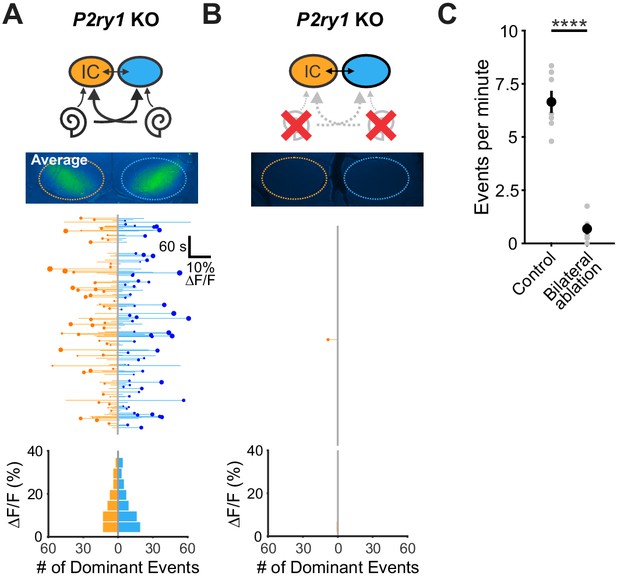
Spontaneous activity in P2ry1 KO mice originates in the cochlea.
(A) Top: Diagram illustrating flow of information through the auditory system and average intensity image over the 10 min imaging session. Middle: Activity over time in left and right IC in an individual where each line indicates the fluorescence intensity of each detected event; the circle indicates the dominant lobe, and the size of the circle indicated the difference in fluorescence. Bottom: Histograms showing the frequency of dominant events of a given amplitude. (B) Similar to A, but with bilateral ablation of the cochleae. (C) Plot of IC event frequency in control (P2ry1 KO) and bilaterally ablated (P2ry1 KO) mice. n = 7 control and n = 5 bilaterally ablated P2ry1 KO mice (two-tailed Student’s t test; ****p<5e-5). See Figure 7—figure supplement 1—source data 1 for plotted values and statistics.
-
Figure 7—figure supplement 1—source data 1
Plotted values and statistics for Figure 7—figure supplement 1.
- https://cdn.elifesciences.org/articles/52160/elife-52160-fig7-figsupp1-data1-v2.xlsx
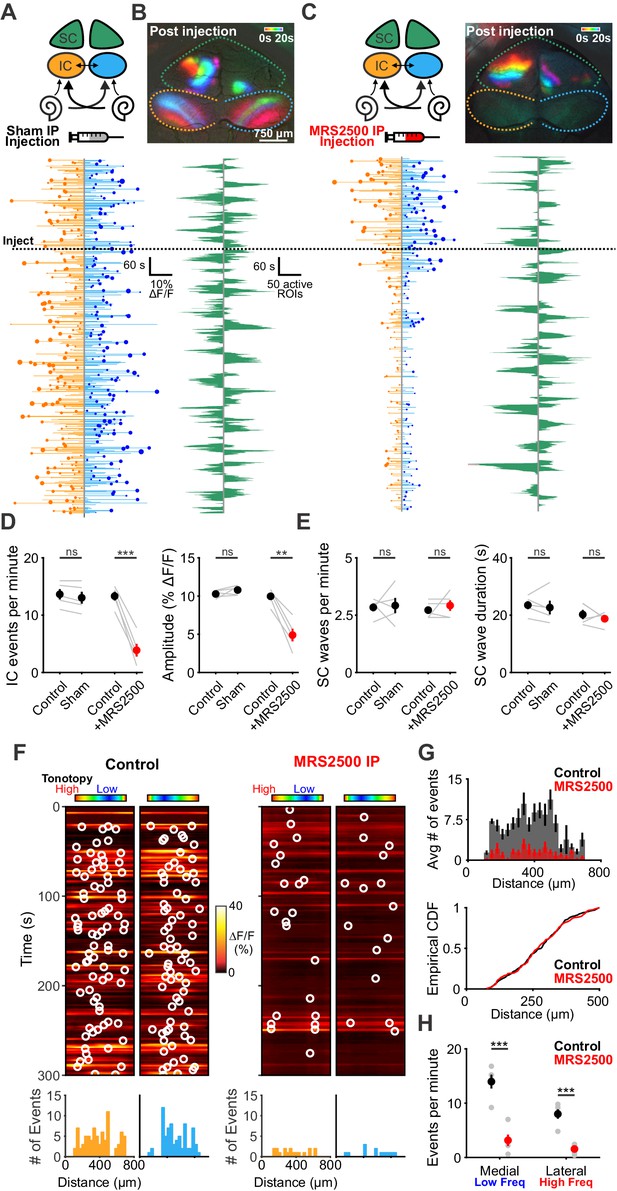
Delivery of MRS2500 in vivo dramatically reduces spontaneous activity in the developing auditory system.
(A) Diagram illustrating flow of information to the midbrain and the visual superior colliculus. Sham solution (5% mannitol) was injected via IP catheter during imaging. (bottom) Activity over time in left and right IC in an individual where each line indicates the fluorescence intensity of each detected event; the circle indicates the dominant lobe, and the size of the circle indicated the difference in fluorescence. Dashed line indicates time of injection. (B) Calcium transients in the midbrain, color-coded based on time of occurrence following sham injection. (bottom) Calcium transients observed in the left and right SC. (C) Similar to (A) and (B), but with injection of MRS2500 (50 μL of 500 μM MRS2500 in 5% mannitol solution). (D) Plot of IC event frequency and amplitude in sham and MRS2500 injected animals. n = 5 mice for each condition (two-tailed paired Student’s t test with Bonferroni correction; ***p<0.005, **p<0.005, ns: not significant). (E) Plot of SC wave frequency and duration in sham and MRS2500 injected animals. n = 5 P6-8 mice for each condition (two-tailed paired Student’s t test with Bonferroni correction; ns: not significant). (F) Activity along the tonotopic axis over a five-minute time frame in the left and right IC before (left) and after (right) MRS2500 injection. Circles indicate detected peaks. (bottom) Histograms of peak locations. (G) Histogram of average number of events before (black) and after (red) MRS2500 injection. (bottom) Cumulative distribution function of event locations across the tonotopic axis pooled from all animals. Events from left and right IC were combined for each experiment. (H) Quantification of event frequency in the medial (low frequency) and lateral (high frequency) regions of the IC. n = 5 P6-8 mice (two-tailed Student's t test with Bonferroni correction; ***p<0.005). See Figure 8—source data 1 for plotted values and statistics.
-
Figure 8—source data 1
Plotted values and statistics for Figure 8.
- https://cdn.elifesciences.org/articles/52160/elife-52160-fig8-data1-v2.xlsx
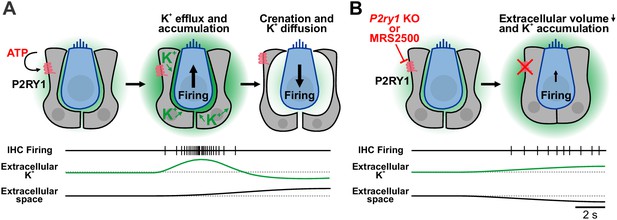
Summary of the dualistic action of P2Y1 signaling.
(A) Spontaneous release of ATP and P2RY1 activation of ISCs initiates a cascade of intracellular signaling events that culiminates in a transient increase in extracellular K+ levels and depolarization of IHCs, resulting in burst firing. The large efflux of ions during each ATP event draws water out of ISCs through osmosis, leading to pronounced shrinkage (crenation) of ISCs and enhanced diffusion of K+ through the increased extracellular volume. (B) Inhibition or genetic removal of P2RY1 induces cellular swelling and loss of extracellular space, leading to extracellular K+ accumulation and tonic firing of IHCs.
Tables
| Reagent type (species) or resource | Designation | Source or reference | Identifiers | Additional information |
|---|---|---|---|---|
| Strain, strain background (Rattus norvegicus) | Sprague Dawley | Charles River | RRID:MGI:5651135 | |
| Strain, strain background (Mus musculus) | FVB mice | Charles River | RRID:IMSR_CRL:207 | |
| Genetic reagent (Mus musculus) | P2ry1tm1Bh;P2ry1 KO | (Fabre et al., 1999) | RRID:MGI:3623279 | |
| Genetic reagent (Mus musculus) | Pax2-Cre | (Ohyama and Groves, 2004) | RRID:IMSR_RBRC09434 | |
| Genetic reagent (Mus musculus) | R26-lsl-GCaMP3 | (Paukert et al., 2014) | ||
| Genetic reagent (Mus musculus) | B6.Cg-Snap25tm3.1Hze/J; Snap25-T2A-GCaMP6s | Jackson Laboratory | RRID:IMSR_JAX:025111 | |
| Genetic reagent (Mus musculus) | TMEM16Afl/fl | (Schreiber et al., 2015) | ||
| Genetic reagent (Mus musculus) | Tecta-Cre | this paper | A mouseline with Cre expression limited primarily to the sensory epithelium. Cross to TdTomato line reveals sparse labeling of cells within the temporal bone and spiral ganglion neurons. | |
| Genetic reagent (Mus musculus) | P2ry1tm1(KOMP)Vlcg;P2ry1 LacZ | KOMP | RRID:IMSR_KOMP:VG12793-1-Vlcg | |
| Genetic reagent (Mus musculus) | B6.Cg-Gt(ROSA)26Sortm14(CAG-tdTomato)Hze/J; Ai14; TdTomato | Jackson Laboratory | RRID:IMSR_JAX:007909 | |
| Antibody | Chicken polyclonal anti-beta-gal | Aves | Cat:BGL-1010;RRID:AB_2313508 | (1:4000) |
| Antibody | Rabbit polyclonal anti-Myosin-VIIa | Proteus Biosciences | Cat:25–6790; RRID:AB_2314838 | (1:500) |
| Antibody | Donkey anti-chicken Alexa Fluor 488 | Life Technologies | Cat:ab150073; RRID:AB_2636877 | (1:2000) |
| Antibody | Donkey anit-rabbit Alexa Fluor 546 | Jackson ImmunoResearch | Cat:711-165-152; RRID:AB_2307443 | (1:2000) |
| Sequence-based reagent | Primer: cccagttgagattggaaagtg (Snap25GC6s-com-s) | Jackson Laboratory | ||
| Sequence-based reagent | Primer: acttcgcacaggatccaaga (Snap25GC6s-mut-as) | Jackson Laboratory | ||
| Sequence-based reagent | Primer: ctggttttgttggaatcagc (Snap25GC6s-wt-as) | Jackson Laboratory | ||
| Sequence-based reagent | Primer: ccgtcaggacaattatcacc (P2ry1-com-as) | this paper | ||
| Sequence-based reagent | Primer: cctaccagccctcatcttct (P2ry1-wt-s) | this paper | ||
| Sequence-based reagent | Primer: cttctatcgccttcttgacg (P2ry1-KO-s) | this paper | ||
| Sequence-based reagent | Primer: gatggttgtggtgtgtctcg (Tecta-com-s) | this paper | ||
| Sequence-based reagent | Primer: cagtgatgagggaggaggtg (Tecta-wt-as) | this paper | ||
| Sequence-based reagent | Primer: cctgtccctgaacatgtcca (Tecta-Cre-as) | this paper | ||
| Sequence-based reagent | Primer: gctgcctgagttggaaagaa (P2ry1-LacZ-com-s) | this paper | ||
| Sequence-based reagent | Primer: ggcttcatgtggaaaacgaa (P2ry1-LacZ-wt-as) | this paper | ||
| Sequence-based reagent | Primer: ctctgctgcctcctggcttct (Rosa26-s) | Paukert et al., 2014 | ||
| Sequence-based reagent | Primer: cgaggcggatcacaagcaata (Rosa26-as) | Paukert et al., 2014 | ||
| Sequence-based reagent | Primer: tcaatgggcgggggtcgtt (CMV-E-as) | Paukert et al., 2014 | ||
| Sequence-based reagent | Primer: attcagacggcaaacgactg (P2ry1-LacZ-LacZ-as) | this paper | ||
| Sequence-based reagent | crRNA: TAATGATGAATAATTCATCC (Tecta exon two targeting) | this paper | ||
| Chemical compound, drug | BAPTA-AM | Sigma | Cat:A1076; CAS:126150-97-8 | (100 μM) |
| Chemical compound, drug | Thapsigargin | Sigma | Cat:T9033; CAS:67526-95-8 | (2 μM) |
| Chemical compound, drug | U73122 | Tocris | Cat:1268; CAS:112648-68-7 | (10 μM) |
| Chemical compound, drug | U73343 | Tocris | Cat:4133; CAS:142878-12-4 | (10 μM) |
| Chemical compound, drug | Ryanodine | Tocris | Cat: 1329; CAS:15662-33-6 | (10 μM) |
| Chemical compound, drug | MRS2500 | Tocris | Cat:2159; CAS:630103-23-0 | (1 μM) |
| Chemical compound, drug | PPADS | Sigma | Cat:P178; CAS:192575-19-2 (anhydrous) | (100 μM) |
| Chemical compound, drug | Suramin | Sigma | Cat:S2671; CAS:129-46-4 | (50 μM) |
| Chemical compound, drug | TTX | Abcam | Cat: ab120055; CAS: 18660-81-6 | (1 μM) |
| Chemical compound, drug | Ouabain | Tocris | Cat: 1076; CAS: 630-60-4 | (10 μM) |
| Chemical compound, drug | Bumetanide | Tocris | Cat: 3108; CAS: 28395-03-1 | (50 μM) |
| Chemical compound, drug | CsCl2 | Sigma | Cat: C4036; CAS: 7647-17-8 | (100 μM) |
| Software, algorithm | ZEN Blue/Black | Zeiss | RRID:SCR_013672 | |
| Software, algorithm | ImageJ | https://imagej.nih.gov/ij/ | RRID:SCR_003070 | |
| Software, algorithm | MultiStackReg | http://bradbusse.net/sciencedownloads.html | RRID:SCR_016098 | |
| Software, algorithm | MATLAB 2017b | Mathworks | RRID:SCR_001622 | |
| Software, algorithm | CorelDRAW Graphics Suite | Corel | RRID:SCR_014235 | |
| Strain, strain background (Rattus norvegicus) | Sprague Dawley | Charles River | RRID:MGI:5651135 |





