Mushroom body evolution demonstrates homology and divergence across Pancrustacea
Figures
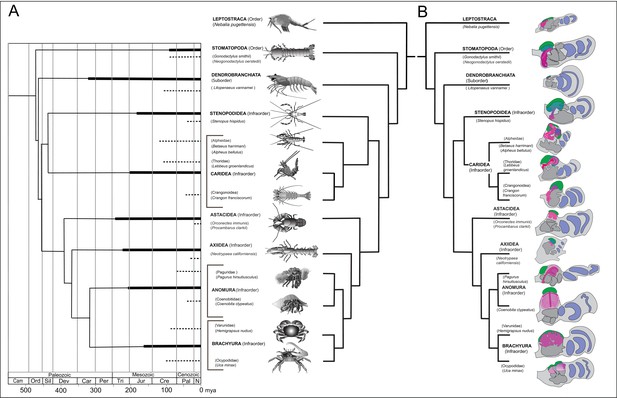
Time lines, phylogeny and protocerebral morphology of malacostracan lineage representatives.
Time lines and lineage relationships are based on the molecular phylogeny of Wolfe et al. (2019). (A) Geological time scale shown as millions of years ago (mya). Solid lines indicate estimated occurrence of lineages sampled for this study; dashed lines indicate estimated age of representative taxa (see citations in text). Images depict species used for this study. (B) Schematics showing proportions of anti-DC0-immunoreactive centers (shades of magenta) in the right lateral protocerebrum of species described in this account. Rostral is up, distal to the right. Nested optic lobe neuropils shown blue; rostrally disposed globuli cell clusters, green; generalized neuropil domains of the lateral protocerebrum, mid-gray.
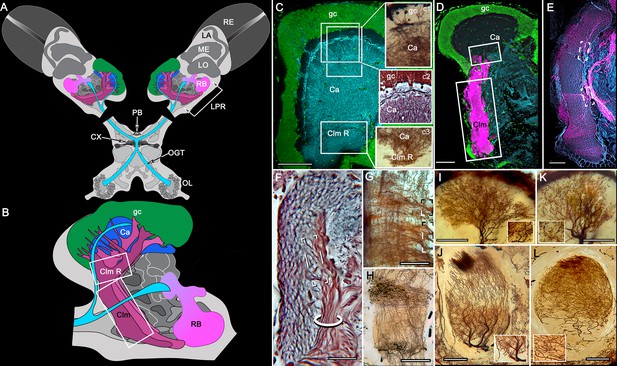
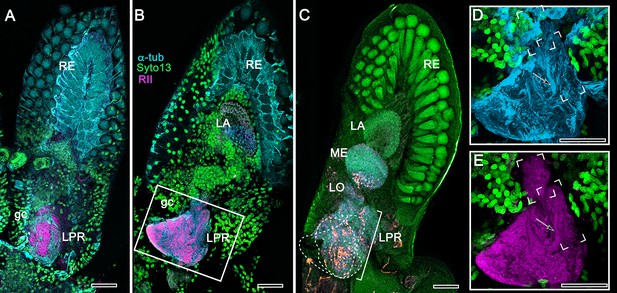
Disposition and cardinal features of the stomatopod mushroom body.
(A) Schematic showing the relative proportions of the lateral protocerebra (LPR) and medial protocerebrum. The latter is defined by the central body (CX), protocerebral bridge (PB), and associated neuropils located anterior to the deutocerebrum, which is denoted by its olfactory lobes (OL). Axons of olfactory relay neurons leave the olfactory lobes to provide the olfactory globular tract (OGT, cyan) that terminates in the mushroom body calyces and more rostral neuropils of the lateral protocerebrum (LPR). (B) Enlargement of the stomatopod mushroom body showing the globuli cell layer (gc) supplying intrinsic neurons that branch in the calyces (Ca). Their axon-like fibers converge at the columnar root (Clm R) and extend through the lengths of the columnar lobes (Clm). In this species (Neogonodactylus oerstedii), four lobes (dark magenta) extend in parallel. A second prominent neuropil is the reniform body (RB), which like the columnar lobes is immunoreactive to anti-DC0. Here and in other species, the RB resides between the mushroom body and optic lobes beneath the retina (RE; lamina, LA; medulla, ME; lobula, LO). (C-L) Histological features that define the mushroom body. Globuli cells (gc; green Syto13) provide thousands of overlapping dendritic trees that populate the calyx (Ca; cyan: anti-α-tubulin). Axons of intrinsic cells converge deep in the calyx to provide the roots of the columnar lobes (Clm R). The boxed areas in C refer to panels (c1-3). Panel c1 shows Golgi-impregnated globuli cells (gc), with neurites extending into the dense layered meshwork of intrinsic neuron dendrites that comprise the calyx; Bodian staining (c2) resolves the packed globuli cells overlying their stratified dendrites in calycal layers. Golgi impregnation (c3) shows axon-like processes converging at the base of the Ca where they form the root of the columnar lobe (Clm R). (D) Confocal laser scan showing anti-DC0 labelling along the length of a columnar lobe (Clm; magenta, anti-DC0; cyan: anti-α-tubulin labelled neuropil). The small rectangle denotes the origin of the column from the calyx, as shown in panel B. The larger rectangle corresponds to comparable lengths of columnar neuropil in panels B, E and F. (E) Anti-tyrosine hydroxylase (magenta) and anti-α-tubulin (cyan) immunolabelling reveals discrete synaptic domains of mushroom body output neurons (MBON) along the length of a column. Their exit points are indicated by open boxes. (F) Bodian-stained section showing braid-like bundles of intrinsic neuron parallel fibers. The open box indicates the exit points of MBONs, the axons of which converge to form a prominent bundle (ringed) destined for volumes of the rostral midbrain protocerebrum (not shown). (G) Golgi-impregnated afferent and efferent processes enter at discrete domains (open boxes) along the stomatopod’s mushroom body column. (H) For comparison: organization of homologous neurons in the mushroom body column of the cockroach Periplanata americana . (I, J) Cross sections of mushroom body column in a stomatopod (panel I) and cockroach (panel J) demonstrate spine-like specializations (insets) of dendrites typifying MBONs. (K, L) Cross sections of afferent terminals in a stomatopod columnar lobe (panel K) and cockroach (panel L). Insets show corresponding beaded specializations. Scale bars in A, 50 μm; B, 100 μm; C-G, 50 μm; H, 100 μm; I, 50 μm; J, 100 μm.
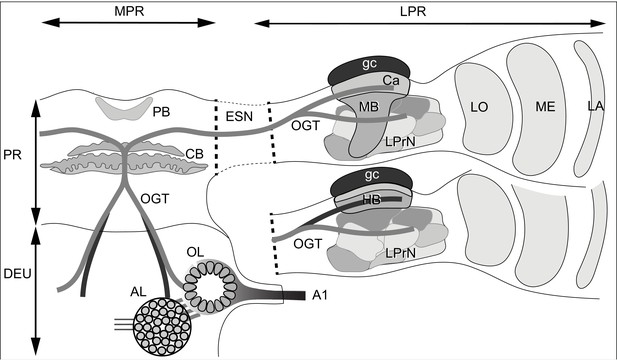
Ground pattern of the pancrustacean olfactory pathway and its lateral protocerebral termini.
In malacostracans, olfactory receptor neurons supply the olfactory lobe (OL) through the antennular nerve A1. Relays from the olfactory lobe (or, in certain Reptantia, from the accessory lobe (AL) which receives inputs from the olfactory lobe) are carried by the olfactory globular tract (OGT). This branches at the midline immediately dorsal to the central body (CB). Its ipsi- and contralateral axon branches extend to the lateral protocerebra (LPR) via the eyestalk nerve (ESN). Branches of the OGT supplying a mushroom body (MB), innervate its calyx (Ca) and several discrete lateral protocerebral neuropils beneath it (LPrN). The mushroom body transformation to a hemiellipsoid body morphology (HB), is shown for the lower lateral protocerebrum. Corresponding levels of organization are shown with identical gray shading. In certain reptantians, the OGT carries axons from the AL to supply the hemiellipsoid body and from the OL to supply LPrN. Other abbreviations: MPR medial protocerebrum; PR protocerebrum; DEU deutocerebrum; PB protocerebral bridge; LA, ME, LO: first, second and third optic neuropils.
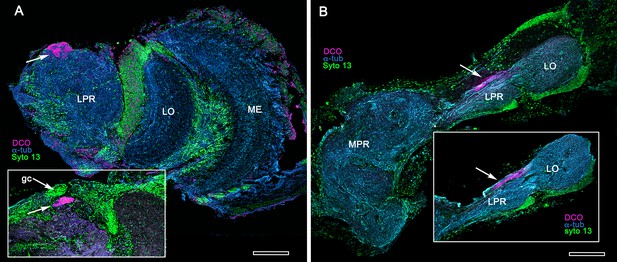
Extreme reduction of DC0-immunoreactive neuropils in decapods and isopods.
(A) The pelagic white leg shrimp Penaeus vannamei, showing substantial lateral protocerebral neuropils (LPR) and optic neuropils (ME, medulla; LO, lobula), but a minute anti-DC0-immunoreactive center, the location of which corresponds to that of the ancestral mushroom body of Stomatopoda and Caridea, and the derived hemiellipsoid body of Reptantia. Inset: Another example showing also the clustered globuli cells (gc) overlying the anti-DC0-immunoreactive center. (B) Preparations from two individual isopods Ligia pallasii, showing the medial protocerebrum (MPR) connected to a greatly reduced lateral protocerebrum (LPR). A narrow layer of anti-DC0-immunoreactive neuropil resides at a position corresponding to that occupied by the mushroom body of a stomatopod or the hemiellipsoid body of a reptantian. Magenta, anti-DC0; cyan, α-tubulin; green, Syto13. Scale bars,100 μm.
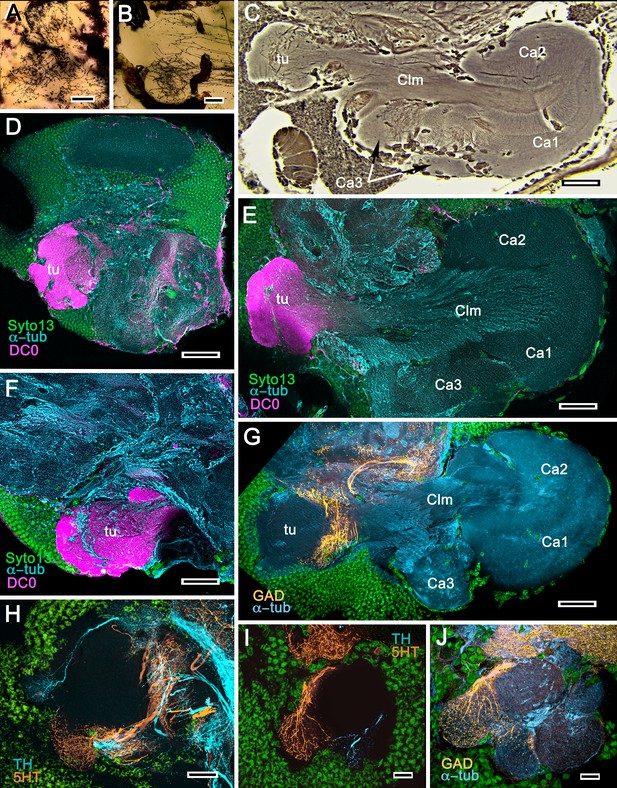
Mushroom bodies of Stenopus hispidus: DC0 and neuromodulatory delineation of tubercular domains.
(A,B) Golgi-impregnated neurites arborizing in the mushroom body columns’ tubercles. (C) Bodian silver-stained overview of the stenopid mushroom body. Axon-like extensions from three distinct calycal regions (Ca1, Ca2, Ca3) provide the column (Clm) that terminates in discrete tubercular domains (tu). (D–F) Immunolabelled sections showing regions containing elevated levels of anti-DC0 (magenta). Anti-DC0 immunoreactivity dominates the entirety of the column’s tubercular domain (cyan, α-tubulin; green, Syto13). (G) Calycal domains and the column show sparse, if any, anti-GAD immunoreactivity (yellow), with the exception of processes that innervate a zone spanning the width of the column immediately distal to its tubercles (tu, cyan: anti-α-tubulin). Neuron cell bodies are shown stained green with Syto13. (H,I) Anti-serotonin (5HT, orange) and anti-tyrosine hydroxylase (TH, cyan) immunoreactive fibers similarly occupy discrete territories in the column’s tubercle. Neural and glial cell bodies are shown stained green with Syto13. (J) Tubercular domains showing discrete territories, each a subset of intrinsic neuron processes, occupied by anti-GAD-immunoreactive processes (yellow; cyan, α-tubulin; green, Syto13). Scale bars in A, B, 20 μm; C, 50 μm; D-H, 50 μm; I, J, 20 μm.
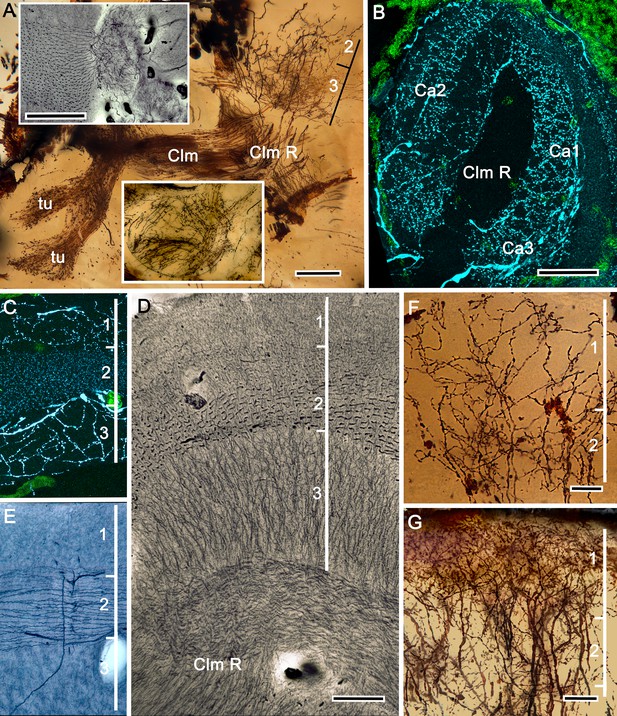
The stenopid mushroom body: cytoarchitecture of the calyx.
(A) Mushroom body intrinsic neurons extend their axon-like processes from the calyx into the column (Clm) terminating in its tubercles (tu). Levels 2, 3 indicate the two inner layers of the calyx. Upper inset (Bodian reduced silver) and lower inset (Golgi- impregnated processes) show axon-like extensions from intrinsic neurons undergoing elaborate sorting to enter the columnar lobe; thus comprising three fused lobes, one from each calyx. (B) Anti-tyrosine hydroxylase immunolabelling (TH, cyan; cell bodies, green) reveals the supply by anti-TH-immunoreactive processes to all three calyces surrounding the root of the mushroom body column (Clm R). (C) Enlargement of the calyx showing anti-TH-positive neuronal arborizations in calycal layers 1 and 3, while anti-TH labelling in layer 2 shows very fine, yet densely populated neuron varicosities. (D) Bodian stain showing the stratified stenopid calyx. Layer 1 contains the apical dendrites of mushroom body intrinsic neurons, as well the terminals of afferent neurons from other lateral protocerebral centers. Terminals of the olfactory globular tract (OGT) arborize in layer 2. Layer 3 is occupied by the extensions of the inner dendrites of intrinsic neurons. Intrinsic neurons send their axon-like extensions into the root of the columnar lobe (Clm R; also see A), top inset) after extending laterally beneath layer 3. (E) Bodian stain of layer 2, which is further defined by numerous parallel projecting terminals from the olfactory lobes carried by the OGT. (F, G) Golgi impregnations of slender, varicose afferent neurons (F) and apical dendrites of intrinsic neurons (G) in layer 1. Scale bars In A, 20 μm; B, 50 μm; C-E, 20 μm; F, 100 μm; G, 50 μm.
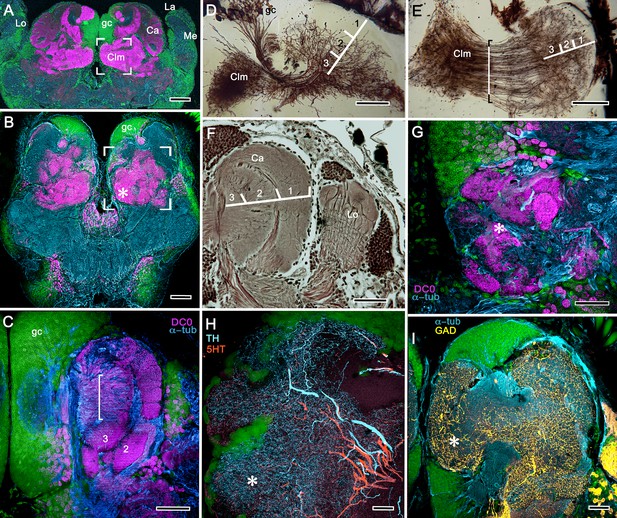
Neuroarchitecture of the alpheid mushroom body.
(A) Anti-DC0-immunoreactivity (magenta) in the brain of Alpheus bellulus. The mushroom bodies (Ca calyx; Clm columnar lobe) are flanked on both sides of the brain by the optic lobes (cyan, anti-α-tubulin; green, Syto13; La, lamina; Me, medulla; Lo, lobula). (B) Anti-DC0 (magenta) immunolabelling in the brain of Betaeus harrimani. Asterisk marks a columnar lobe ending in tubercles (cyan, anti-α-tubulin; green, Syto13). (C) The mushroom body calyx of A. bellulus. Throughout its depth, and in each layer (layers 2 and 3 are shown here), the calyx shows a high affinity for antibodies raised against DC0. Striations (bracket) correspond to bundles of intrinsic neuron processes supplying the columnar lobe, correspondingly bracketed in panel E. (D,E) Golgi impregnations of mushroom body intrinsic cell clusters, originating from globuli cells (gc) and giving rise to dendrites in the calyces (Ca, layers numbered 1–3) and column (Clm). The distinct calycal layers are most clearly revealed in Bodian-stained sections (F). (G) Anti-DC0 is expressed in distinct territories within the A. bellulus tubercles. (H, I) Aminergic processes in the lateral protocerebrum of B. harrimani. Asterisks in G, H, I denote corresponding regions. (H) Anti-5HT (orange) and anti-TH (cyan) show these neural arborizations invading most regions of the lateral protocerebrum coincident with volumes denoted by anti-DC0-labelled mushroom body-associated structures. (I) Anti-GAD (yellow) immunoreactivity shows an expression pattern throughout the anti-DC0-positive domains shown in B (cyan, anti-α-tubulin). Scale bars in A (A. bellulus), 100 μm; B (B. harrimani), 100 μm; C-F (A. bellulus), 100 μm; G (A. bellulus), 40 μm; H (B. harrimani), 20 μm; I (B. harrimani) 50 μm.
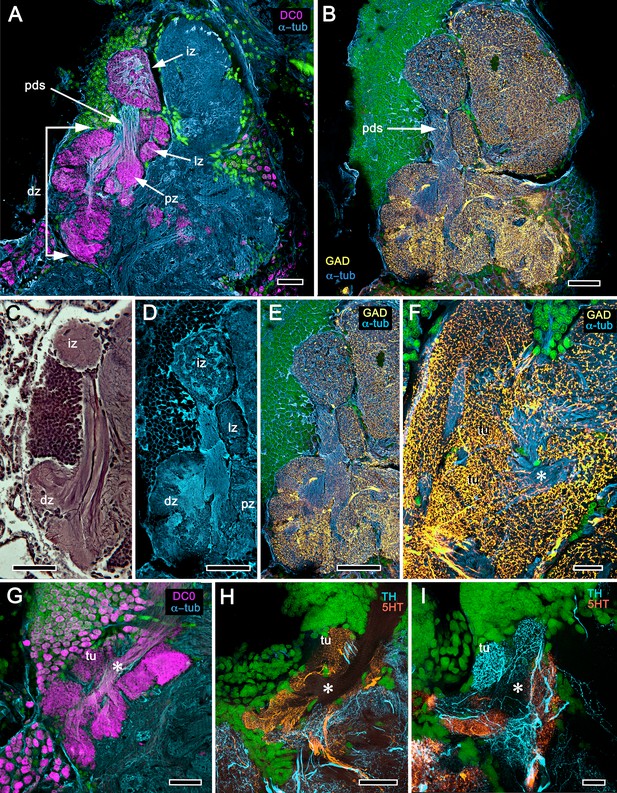
Reniform body (A–E) and mushroom body lobes (F–I) in alpheid shrimps.
(A-E) Neuroarchitecture of the Alpheus bellulus reniform body (RB; cyan, anti-α-tubulin; green, Syto13). (A) Components of the alpheid reniform body are clearly distinguished by their high affinity for anti-DC0 (magenta). (A) pedestal-like (pds) bundle of neurites gives rise to four distinct zones: the initial zone (iz), the proximal zone (pz), the distal zone (dz) and the lateral zone (lz). (B,E) Anti-GAD immunolabelling (yellow) reveals putative inhibitory processes occupying major zones of the reniform body, other than the pedestal. (C) Bodian silver-stained Betaeus harrimani and (D) anti-α-tubulin-labelled A. bellulus sections reveal the nearly identical layout of the reniform body across the two alpheid species. (F–I) Anti-DC0 and aminergic innervation of discrete domains within tubercles branching off the mushroom body’s columnar lobe. Asterisks in panels F–I indicate the MB column with its processes defasciculate into discrete tubercular domains. One corresponding tubercle (tu) is indicated in panels G-I. (F) Innervation of tubercular domains by anti-GAD-immunoreactive processes (yellow). (G) Anti-DC0 labelling expressed within tubercles. (H, I) Tubercular domains delineated by anti-TH- and anti-5HT-immunoreactive fibers. In all panels green indicates Syto13-stained neuronal cell bodies. Scale bars in A-C,E, 50 μm (B. harrimani); D,G-I, 50 μm; F, 10 μm (A. bellulus).
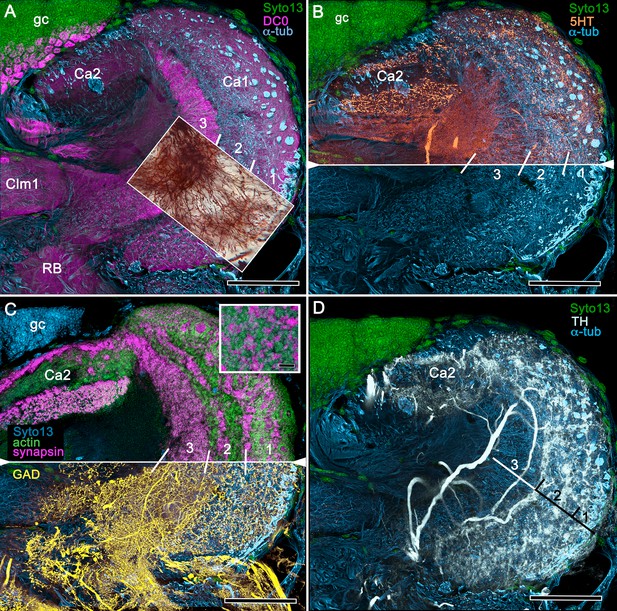
Neuroanatomy of mushroom body calyces in the thorid shrimp, Lebbeus groenlandicus.
(A-D) Confocal laser scans of immunohistochemically labelled sections (anti-α-tubulin is cyan in all panels except C). (A) Anti-DC0 (magenta) immunoreactivity in both calycal regions (Ca1, calyx 1; Ca2, calyx 2), as well as in the columnar extension of intrinsic cells (Clm1) and the reniform body (RB). The inset demonstrates a palisade of Golgi-impregnated intrinsic cells, the dendritic organization of which reflects the presence of three distinct layers in Ca1 (1–3 against inset). (B) Anti-5HT immunolabelling (orange) showing afferent neuron terminals from various regions in the lateral protocerebrum ending in both calyces, and the three layers in Ca2 (upper half of panel). Calycal cytoarchitecture is distinguishable by anti-α-tubulin labelling alone (lower half of panel). (C) Upper half. Double labelling with anti-synapsin (magenta) and F-actin (green) further resolves layering (1-3) of synaptic sites in both Ca1 and Ca2. Globuli cells (gc) are shown in cyan. Inset. High-resolution scans resolve synaptic microglomeruli. Lower half. Anti-GAD-immunopositive fibers (yellow) extending from the lateral protocerebrum to provide dense innervation into all calycal levels. (D) Large-diameter anti-TH-positive terminals (white) spread their tributaries throughout calycal layers 1 and 2. Scale bars in A-D, 100 μm.
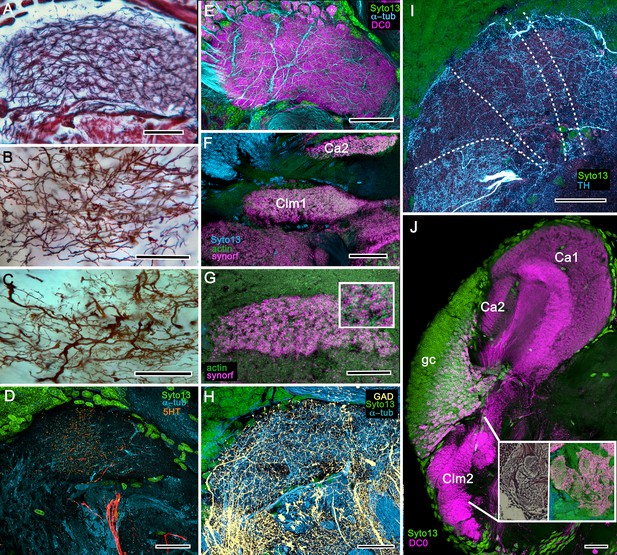
Columnar projections of Lebbeus mushroom body intrinsic neurons.
(A) Bodian-stained axon-like extensions of intrinsic neurons are not parallel but interweave throughout the length of the first column (Clm1). (B). Golgi impregnations resolve these decorated by bouton- and spine-like specializations denoting, respectively, afferent pre- and postsynaptic specializations. (C) Golgi impregnations resolve afferent and efferent (MBON) processes intersecting serial domains within Clm1. (D) Sparse anti-5HT-immunoreactive neurons (orange) send terminals into Clm1 from regions in the lateral protocerebrum. (E) Anti-DC0 (magenta) immunoreactivity reveals a high affinity for this antibody in Clm1 where it outlines afferent and α-tubulin-positive efferent fibers (cyan). (F) Anti-synapsin (magenta) expression in Clm1 and Ca2., (G) Double labelling with anti-synapsin (SYNORF1) and F-actin (green) reveals microglomeruli in Clm1 (inset). (H) Anti-GAD-positive processes (yellow) project along the full length of Clm1. (I) Discrete serial domains along Clm1 are defined by processes of putative MBONs immunoreactive to anti-TH (cyan). (J) Anti-DC0 immunoreactivity (magenta) defines Clm2 showing its characteristic terminal tubercles (compare with Figure 10A). The two insets resolve tubercles in Bodian silver-stained material (left) and anti-synapsin/F-actin (magenta/green) labelling of their synaptic zones (right). Scale bars in A-J, 50 μm.
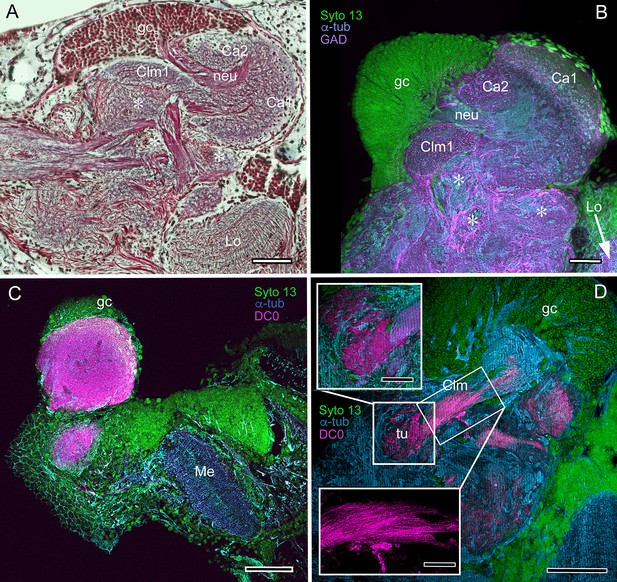
Mushroom body neuroarchitecture: Spirontocaris lamellicornis and Paracrangon echinata.
(A) Bodian-stained section of Lebbeus groenlandicus demonstrates that the organization of the mushroom body calyces and columns of this thorid shrimp is almost identical to that of species sharing the same ecological niche, exemplified by the thorid S. lamellicornis and the crangonidid Paracrangon echinata. (B) Anti-α-tubulin (cyan) and anti-GAD immunoreactivity (magenta) in S. lamellicornis resolve column 1 (Clm1), the two calyces Ca1, Ca2, and their neurites (neu) leading from globuli cells (gc; green, Syto13). More caudal regions of the corresponding lateral protocerebrum are indicated by asterisks in panels A and B. (C) Anti-DC0 immunostaining (magenta) of P. echinata reveals high levels of the antigen in a substantial rounded calyx with a narrowly differentiated outer stratum covered by a dense layer of globuli cells (gc, green). (D) A second specimen showing evidence of a diminutive column (Clm), resolved by anti-DC0 (magenta), comprising parallel fibers (inset, lower left) ending in a small tubercular (tu) domain (inset, upper left). gc globuli cells (green, Syto13); Me medulla; Lo lobula. Scale bars in A-D, 100 μm; insets in D, 50 μm.
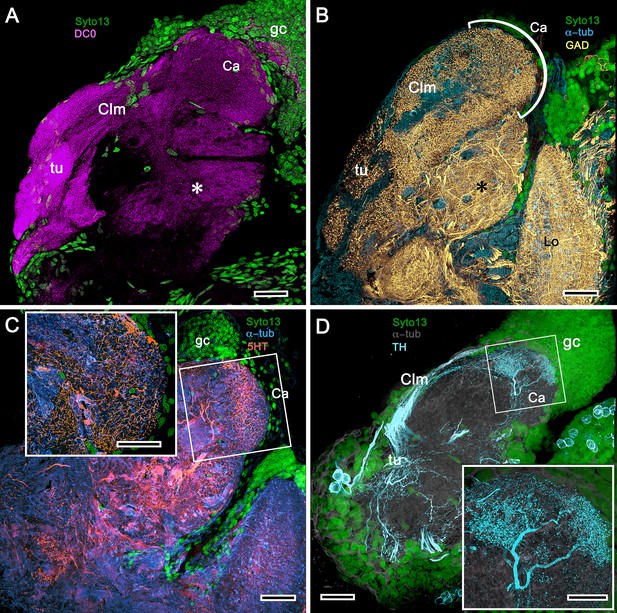
Mushroom body of Crangon franciscorum.
(A) Immunohistochemically labelled sections reveal moderate anti-DC0 expression (magenta) in the mushroom body calyx (Ca) of C. franciscorum but a system of intensely labelled columnar lobes (Clm) ending as tubercular swellings (tu). As in carideans and reptantians, this species reveals lower levels of anti-DC0 immunoreactivity (asterisk) in the more distal parts of the lateral protocerebrum, beneath the calyx. (B) Anti-GAD immunoreactivity (yellow) is abundant in the calyx and the tubercles, but is sparser in the columnar lobes. Anti-GAD immunoreactivity also extends throughout the lateral protocerebrum and lobula (Lo). (C) Anti-5HT labelling (orange) reveals putative serotoninergic processes in the calyx (inset: details shown in single scan) and regions within the lateral protocerebrum. (D) Putative dopaminergic neurons, labelled with anti-TH (cyan), branch in two distinct territories of the calyx (Ca), enlarged in the inset lower right (gray, α-tubulin). gc globuli cells (green, Syto13). Scale bars in A-D, 50 μm, both insets, 20 μm.
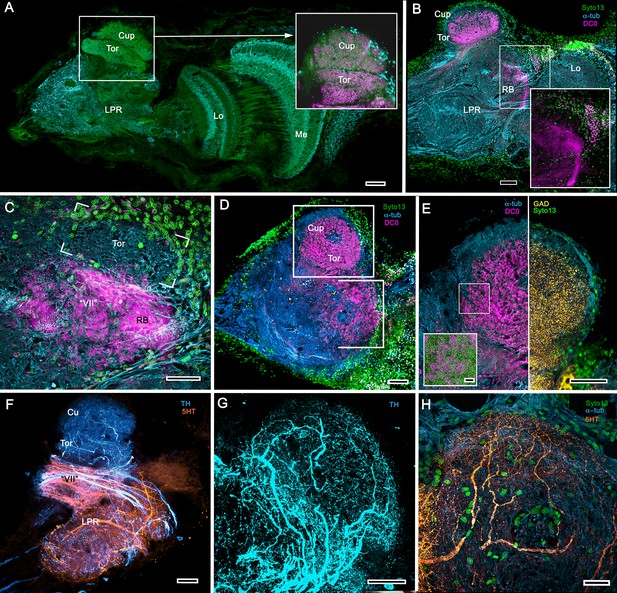
Transformed mushroom bodies in Astacidea.
(A) An overview of the crayfish lateral protocerebrum showing its distinctive hemiellipsoid body composed of two components, the rostral cupola (Cup) overlying the torus (Tor). All panels show the right lateral protocerebrum, where rostral is upwards and distal is to the right (F-actin, green; anti-allatostatin, cyan). Inset to A. anti-synapsin and F-actin staining (magenta/green) shows the torus equipped with denser synaptic clusters than the cupola above it. However, it is the neuropil beneath the torus, receiving a massive input from the olfactory lobes, that shows the greatest synaptic density. (B) Anti-DC0 immunolabelling (magenta) here shows the torus as more densely labelled. Neuropils ascribed to the reniform body (RB) are further distal, at the border between the lateral protocerebrum (LPR) and the optic lobe’s lobula (Lo). The inset shows a cluster of anti-DC0-positive cell bodies at the rostral surface associated with the reniform body like those identified in Brachyura (see Figure 15E, (F). (C) A section just glancing the torus, here showing almost no anti-DC0 immunoreactivity in the torus, but substantial anti-DC0 labelling in sub-calycal neuropils corresponding to neuropil (VII) described by Blaustein et al. (1988) and the probable location of the reniform body (RB). (D) Intense anti-DC0 labelling of both levels of the hemiellipsoid body and neuropils beneath and distal to this center (bracketed). (E) Alignment of the anti-DC0-positive hemiellipsoid body in D to the left with that of another specimen immunolabelled with anti-GAD (yellow) to the right. Discrete small glomerulus-like aggregates resolved by anti-DC0 (corresponding area enlarged in inset) contrast with the uniform distribution of GAD-immunoreactive profiles. The inset in E (left) demonstrates structures corresponding to the anti-DC0-immunolabelled aggregates comprise anti-synapsin/F-actin labelled (magenta-green) synaptic microglomeruli comparable to those identified in the stomatopod mushroom body calyces (Wolff et al., 2017). (F–H) Antibodies against TH (cyan) and 5HT (orange) demonstrate distributions of these efferent neurons, which correspond to multimodal parasol cells (efferent neurons equivalent to MBONs) described by Mellon (2003) and McKinzie et al. (2003). Scale bars in A, B, D, F, 100 μm; E, G, H, 50 μm; inset to E, 2 μm.
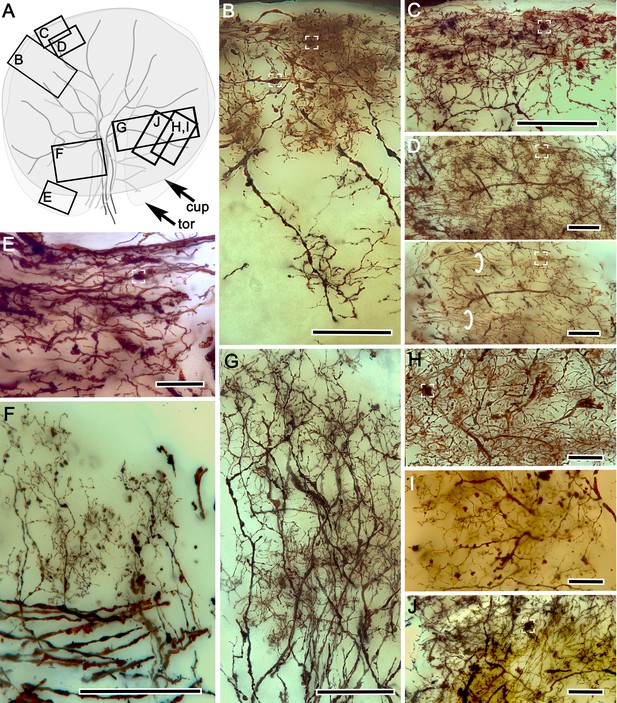
Neuronal arrangements in the hemiellipsoid derivative.
(A) Schematic of crayfish hemiellipsoid body viewed from above, showing the outlines of the torus (tor) overlaid by the cupola (cup). Large branching elements correspond to TH- and 5HT-immunoreactive efferent neurons. Boxes B-J denote locations of neurons illustrated in panels (B-J). Neurons contributing to the cupola’s palisade of intrinsic neurons. Presynaptic boutons of terminals from the olfactory globular tract (OGT) indicated by white brackets (B–E). (C) Mass impregnation of presynaptic OGT endings in the outer layer of the cupola. (D) Two images, the upper showing a top-down view through 40 microns of the cupola outer layer, the lower showing an optical slice in which bundled parallel fibers (circled) can be more clearly isolated from other elements. (E) Terminal OGT boutons and parallel fibers at the margin of the torus. (F) Palisades of collateral dendrites originating from efferent neurons. (G) Bistratified palisades of intrinsic neurons in the torus. (H–J) Dorsal view of systems of local interneurons intersecting the inner intrinsic neuron layer. Scale bars for B, C, F and G = 50 μm; for C, D, E, H-J = 20 μm.
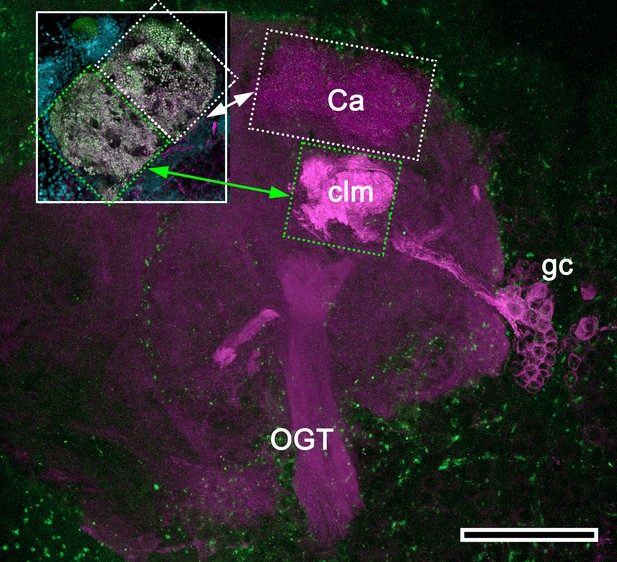
Mushroom body hypotrophy of the ghost shrimp Neotrypaea californiensis.
Modified from Wolff et al. (2012). Immunolabelled sections stained with antisera against DC0 (magenta) and phalloidin conjugated to Alexa 488 (green). Intrinsic cell bodies of the calyx (Ca) were not stained with Syto13 in this preparation; however, a cluster of anti-DC0-immunoreactive intrinsic neurons (gc) gives rise to a greatly condensed column (clm) extending from beneath the calyx (Ca). The inset shows the calyx consisting of a dense population of microglomerular synaptic sites (inset; synapsin, magenta; F-actin, green; Syto13, cyan). Very low anti-DC0 immunoreactivity typically shows the surrounding protocerebral neuropils and olfactory globular tract (OGT). Scale bar, 100 μm.
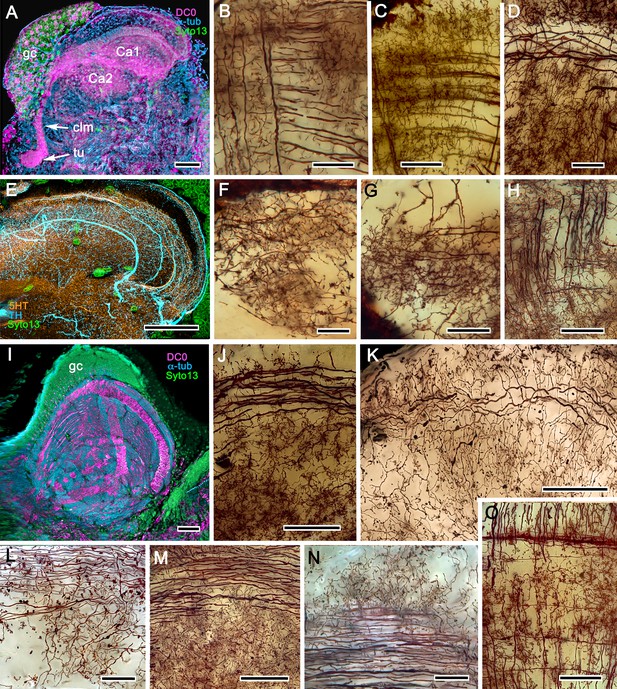
The reptantian mushroom body with and without its columnar lobe.
Golgi impregnations demonstrate corresponding orthogonal arrangements in the two calyces of Pagurus (Ca1, Ca2 in panel A), which extend as a columnar lobe (clm) ending in tubercular swellings (tu), and in the multistratified hemiellipsoid body of Coenobita (I), which lacks a lobe. (B-D), (F-H) Golgi impregnations of Pagurus calyces show characteristic orthogonal arrangement of intrinsic neuron dendrites (B,F,G) originating from parallel arrangements of their initial processes (C). These neurons are invested by beaded fibers from the olfactory globular tract (OGT; F) and provide axon-like elongations that extend to the lobe (D; see also, Strausfeld and Sayre, 2020). Anti-5HT (orange) and anti-TH (cyan) immunostaining resolves corresponding efferent dendritic trees aligned with orthogonal networks (E). These are shown for the multistratified Coenobita hemiellipsoid body (panel I). (J–O) Golgi impregnations of the Coenobita hemiellipsoid body demonstrate orthogonal and rectilinear organization of its intrinsic neurons. Scale bars in A, 50 μm; B-D, 25 μm; E, 50μ; F, 10 μm; G, 25 μm; H,J,K, 50 μm; I, 100 μm; L-O, 25 μm.
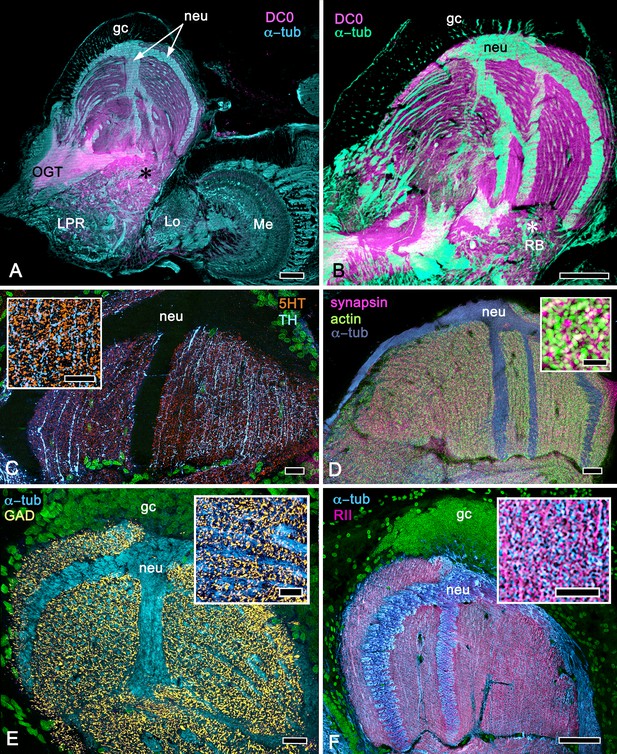
Calycal hypertrophy in the Coenobita clypeatus mushroom body.
(A,B) DC0 and other immunological markers reveal a characteristic system of nested strata comprising orthogonal fibers that originate from the intrinsic neurons (neu; cyan, anti-α-tubulin). Anti-DC0 (magenta) defines these strata as well as the entrance of the olfactory globular tract (OGT) into the calyx and several regions of the lateral protocerebrum beneath the calyx, including the reniform body (RB, asterisk in A,B). Other regions of the lateral protocerebrum (LPR,) and optic lobe (Lo, lobula; Me, medulla) show little or no anti-DC0 affinity. (C) Anti-5HT- (orange) and anti-TH-immunoreactive (cyan) fibers in the calyx extend within each of the anti-DC0-positive strata but are notably absent from the neurite bundles of intrinsic neurons. The distribution of anti-TH is not uniform (inset), but patchy indicating discrete domains of TH-immunopositive processes within the stratified intrinsic neuron networks. (D) Anti-synapsin (magenta) and F-actin (green) demonstrate regions of dense synaptic connectivity indicated by microglomerular configurations (inset). In this panel, intrinsic neuron bundles (neu) are labelled with anti-α-tubulin (cyan). (E) Distributed processes labelled with antisera against GAD (yellow) extend across strata, revealing neuropil occupied by afferents to the calyces (inset). (F) RII, an antiserum developed in Drosophila melanogaster against the regulatory subunit of PKA, confirms an expression pattern (magenta) corresponding to that of DC0 (A,B) and is shown comingling with anti-α-tubulin labelled neuropil in and between stratifications (inset). Scale bars in A,B, 100 μm; C-E, 20 μm; F, 50 μm; inset C, 5 μm; inset D, 2 μm; insets E-F, 5 μm.
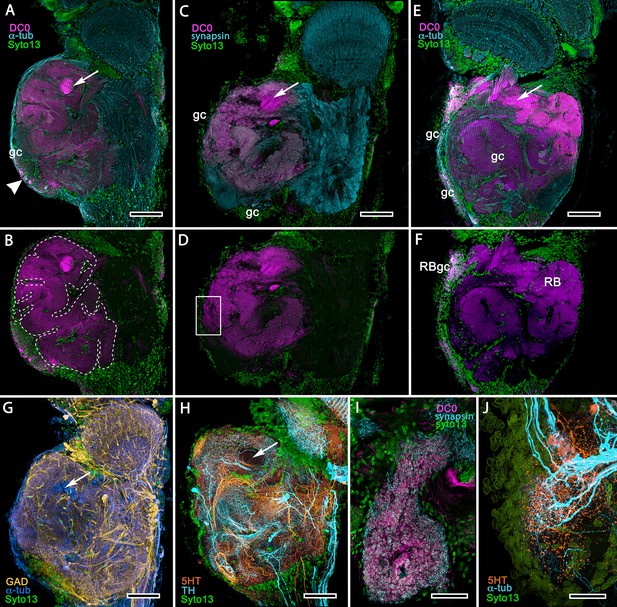
Mushroom body homologues of the shore crab Hemigrapsus nudus.
(A-H) Depictions of sections of the right lateral protocerebrum: rostral is to the left; distal (towards the optic lobes) is the top of each panel. (A–F) Anti-DC0-immunoreactive territories (magenta) are interpreted as modified mushroom bodies, lacking columnar lobes and enormously expanded in the rostral lateral protocerebrum. Distally, each of these centers is penetrated by the pedestal (bright magenta in A-F, arrow in panels A,C,E,G,H) belonging to the large reniform body typifying varunid crabs. The entire territory occupied by the reniform body (RB) is shown in E against the dimmed magenta surround. Anti-DC0 labelling is shown at normal intensities in panels A-D, F. The maximum extent of the mushroom body neuropil is indicated by the dotted outline in B. Cell bodies of intrinsic neurons (gc, green: Syto13) occur scattered over the rostral and dorsal surface of the lateral protocerebrum (A–F), some also showing elevated anti-DC0 immunoreactivity (e.g., arrowhead in A). Globuli cells providing the reniform body (RBgc) also show anti-DC0 immunoreactivity, as indicated in panel F. (G) Distribution of anti-GAD-immunoreactive (yellow) processes mainly in caudal volumes of the lateral protocerebrum and the lobula. (H) Distribution of anti-5HT- (orange) and anti-TH-immunoreactive (cyan) processes. (I,J) Labelling with anti-DC0 (magenta) and the synaptic marker SYNORF1 (cyan; I) and with anti-5HT (orange) and anti-TH (J) reveal discrete regions (one boxed in D) in anti-DC0-immunoreactive territories that are suggestive of mushroom body-like circuitry. Scale bars in A-H, 100 μm; I, J, 25 μm.
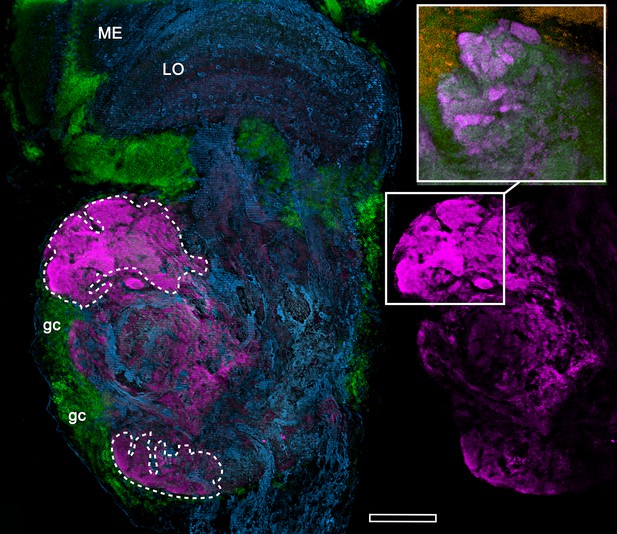
Anti-DC0-immunoreactive neuropils (magenta) of the fiddler crab Uca minax (rostral to the left, distal upwards).
The left panel delineates the boundaries (dashed lines) of two DC0-positive territories (also shown alone, to the right), of which the most distal has dense immunoreactive folds and fissures (boxed in lower right image). The inset upper right, shows the folds and fissures in another specimen, which has been labelled with anti-synapsin (magenta) and F-actin (green; Syto13-labelled cell bodies shown orange) to resolve a particularly dense population of synaptic sites. ME, medulla; LO, lobula; gc, globuli cells. Scale bar is 100 μm.
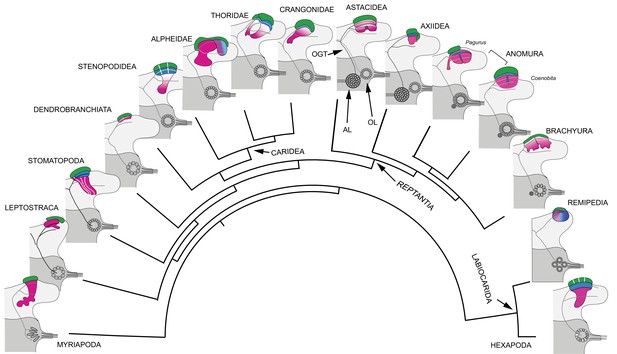
Retention and divergence of the mushroom body ground pattern in Mandibulata.
Schematized shapes of mushroom bodies described in this study (for actual profiles see Figure 1B). Their evolved derivatives are mapped onto a pancrustacean molecular phylogeny (after: Oakley et al., 2013; Wolfe et al., 2019; Schwentner et al., 2017), here extended to include the mandibulate outgroup Myriapoda (represented by the chilopod mushroom body; Wolff and Strausfeld, 2015). Each schematic depicts the right lateral protocerebrum (light gray) without its optic lobes and the right deutocerebrum (dark gray). The deutocerebrum is shown with its olfactory lobe (OL); or, in Reptantia, the olfactory and adjoining accessory lobe (AL). The antennal globular tracts (OGT) are shown with their contralateral extension indicated in all examples except Myriapoda and Hexapoda, where the OGT is exclusively homolateral. Magenta indicates anti-DC0/RII identification of corresponding neuropils; green indicates globuli cell clusters; blue indicates distinct calycal organization. Evolved diminution is here shown for Dendrobranchiata. Despite miniaturization in Leptostraca, the RII-immunoreactive center reveals reduced columnar lobes. A comparable arrangement is resolved in Remipedia (see Fig. 3 in Stemme et al., 2016).
Mushroom body of Nebalia pugettensis.
(A,B) Two successive sections reveal an ensemble of neuropils strongly immunoreactive to antibodies raised against RII (magenta, see Methods). An anti-RII-immunoreactive region of neuropil (A) expands into a planar calyx (B) and gives rise to two small but well-defined columnar lobes extending forwards. These regions (dotted lines) are superimposed in panel (C) beneath which are deeper levels of the lateral protocerebrum (LPR) comprising several discrete neuropils central to the nested optic lobe neuropils (LA, lamina; ME, medulla; LO, lobula). The boxed area in panel B is enlarged in (D, E). (D) Neuroarchitecture of the calyx resolved with anti-α-tubulin (cyan); the columnar lobes are bracketed, one showing bundled parallel fibers (arrowed in D,E). (E) Anti-RII-labeled (magenta) processes (arrowed) extending from the triangular calyx. RE, retina; gc, globuli cells. Orange in panel C is FMRFamide immunoreactivity. Scale bars indicate 50 μm.
Tables
Comparison of main morphological characters defining the crustacean and insect olfactory pathways.
| Character | Hexapoda (Dicondylia) | Eumalacostraca and Remipedia | Correspondence |
|---|---|---|---|
| Ligand-gated odorant receptors (ORs) | Likely ubiquitous | None reported | – |
| Ionotropic olfactory receptors (IRs) | Present | Likely ubiquitous | + |
| Olfactory neurons in each sensillum | 1-5 (in Dicondylia) | Numerous, >10 and can exceed 100s | – |
| Axons of olfactory receptor neurons | Target-defined glomeruli | Non-targeting: can innervate multiple glomeruli | – |
| Olfactory lobe subunits | Unique glomeruli, relating to genetic receptor identity | Discrete subunits, isomorphic, number unrelated to receptor identities | – |
| Accessory lobes | None | Reptantian lineages (possible remipede homologue) | – |
| OL projection neurons (PNs) | Most uniglomerular | All multiglomerular | – |
| Accessory lobeprojection neurons (via OGT) | N/A | In Reptantia | – |
| Number of PN axons from olfactory lobe | 2–5 times the number of glomeruli | Some thousands, unrelated to glomerulus number | – |
| Projection neuron axon tract (OGT or AGT) | AGT stays ipsilateral | OGT bifurcates, ipsi- and contralateral. (Exception is OGT ipsilateral in Cephalocarida) | – |
| Protocerebral PN terminals | In MB calyx + other lateral protocerebraltargets | In MB calyx + other lateral protocerebral targets | + |
| Reagent type (species) or resource | Designation | Source or reference | Identifiers | Additional information |
|---|---|---|---|---|
| Biological sample | Stenopus hispidus | Salty Bottom Reef Co. | SBRC-I-CBS | n = 25 |
| Biological sample | Uca minax | Salty Bottom Reef Co. | N/A | n = 12 |
| Biological sample | Coenobita clypeatus | Carolina Biological Supply Co. | 142415 | n = 13 |
| Biological sample | Orconectes immunis | Carolina Biological Supply Co. | N/A | n = 8 |
| Biological sample | Procambarus clarkii | Carolina Biological Supply Co. | 142512 | n = 10 |
| Biological sample | Periplaneta americana | Carolina Biological Supply Co. | 143642 | n = 20 |
| Biological sample | Alpheus bellulus | LiveAquaria | CN-90061 | n = 20 |
| Biological sample | Stenopus hispidus | LIveAquaria | CN-78439 | n = 3 |
| Biological sample | Penaeus vannamei | Gulf Specimen Marine Laboratories | N/A | n = 24 |
| Biological sample | Neogonodactylus oerstedii | Gulf Specimen Marine Laboratories | Ar-1600 | n = 25 |
| Biological sample | Lebbeus groenlandicus | Friday Harbor Laboratories | N/A | n = 37 |
| Biological sample | Munida quadrispina | Friday Harbor Laboratories | N/A | n = 2 |
| Biological sample | Crangon franciscorum | Friday Harbor Laboratories | N/A | n = 10 |
| Biological sample | Spirontocaris lamellicornis | Friday Harbor Laboratories | N/A | n = 3 |
| Biological sample | Paracrangon echinata | Friday Harbor Laboratories | N/A | n = 8 |
| Biological sample | Neotrypaea californiensis | Friday Harbor Laboratories | N/A | n = 11 |
| Biological sample | Betaeus harrimani | Friday Harbor Laboratories | N/A | n = 3 |
| Biological sample | Hemigrapsus nudus | Friday Harbor Laboratories | N/A | n = 40 |
| Biological sample | Nebalia pugettensis | Friday Harbor Laboratories | N/A | n = 4 |
| Biological sample | Ligia pallasii | Friday Harbor Laboratories | N/A | n = 3 |
| Biological sample | Pagurus hirsutiusculus | Friday Harbor Laboratories | N/A | n = 35 |
| Antibody | α-Tubulin (Mouse, monoclonal) | Developmental Studies Hybridoma Bank (DHSB) | CAT#: 12G10; RRID: AB_1157911 | 1:100 |
| Antibody | α-Tubulin (Rabbit, polyclonal) | Abcam | CAT#: ab15246; RRID: AB_301787 | 1:250 |
| Antibody | Synapsin (SYNORF1; Mouse, monoclonal) | Developmental Studies Hybridoma Bank (DHSB) | CAT#: 3C11; RRID: AB_528479 | 1:100 |
| Antibody | Serotonin (5HT; Rabbit, polyclonal) | ImmunoStar | CAT#: 20080; RRID: AB_572263 | 1:1000 |
| Antibody | Glutamic acid decarboxylase (GAD; Rabbit, polyclonal) | Sigma-Aldrich | CAT#: G5163; RRID: AB_477019 | 1:500 |
| Antibody | Tyrosine hydroxylase (TH; Mouse, monoclonal) | ImmunoStar | CAT#: 22941; RRID: AB_572268 | 1:250 |
| Antibody | DC0 (Rabbit, polyclonal) | Generous gift from Dr. Daniel Kalderon (Skoulakis et al., 1993) | RRID: AB_2314293 | 1:400 |
| Antibody | RII (Rabbit, polyclonal) | Generous gift from Dr. Daniel Kalderon (Li et al., 1999) | N/A | 1:400 |
| Antibody | AffiniPure Donkey Anti-Mouse IgG (H+L) Cy3 (polyclonal) | Jackson ImmunoResearch | CAT#: 715-165-150; RRID: AB_2340813 | 1:400 |
| Antibody | AffiniPure Donkey Anti-Rabbit IgG (H+L) Cy5 (polyclonal) | Jackson ImmunoResearch | CAT#: 711-175-152; RRID: AB_2340607 | 1:400 |
| Antibody | AffiniPure Donkey Anti-Rabbit IgG (H+L) Alexa Flour 647 (polyclonal) | Jackson ImmunoResearch | CAT#: 711-605-152; RRID: AB_2492288 | 1:400 |
| Other (serum) | Normal donkey serum | Jackson ImmunoResearch | RRID: AB_2337258 | N/A |
| Other (DNA stain) | SYTO 13 Green Fluorescent Nucleic Acid Stain | Thermo Fisher Scientific | CAT#: S7575 | 1:2000 |
| Other (Phalloidin stain) | Alexa Fluor 488 Phalloidin | Thermo Fisher Scientific | CAT#: 12379; RRID: AB_2315147 | 1:40 |
| Other (Chemical) | α-terpineol | Sigma-Aldrich | CAT#: 432628 | N/A |
| Other (histology chemical) | Protargol (silver proteinate) | Winthrop Chemical Co. | Product discontinued | N/A |
| Other (histology chemical) | Silver proteinate | Generated in-house Followed recipe described in Pan et al. (2013) | N/A | |
| Other (histology chemical) | Potassium dichromate | Sigma-Aldrich | CAT#: 207802 | N/A |
| Other (HPLC purified water) | HPLC Water | Sigma-Aldrich | CAT#: 270733 | N/A |
| Other (histology chemical) | Osmium tetroxide | Electron Microscopy Sciences | CAT#: 19150 | N/A |
| Other (histology chemical) | Siler nitrate | Electron Microscopy Sciences | CAT#: 21050 | N/A |
| Other (histology chemical) | Propylene oxide | Electron Microscopy Sciences | CAT#: 20401 | N/A |
| Other (embedding resin) | Durcupan ACM resin (4-part component kit) | Sigma-Aldrich | CAT#: 44610 | N/A |
| Other (mounting medium) | Permount mounting medium | Fisher Scientific | CAT#: SP15-100 | N/A |
| Software, algorithm | Photoshop CC | Adobe Inc | N/A | N/A |
| Software, algorithm | Fiji | Open-source software Schindelin et al. (2012) | z-project plugin | |
| Software, algorithm | Helicon Focus | Helicon Soft | N/A | N/A |
| Other (microscope) | LSM 5 Pascal confocal microscope | Zeiss | N/A | N/A |
| Other (microscope) | Orthoplan light microscope | Leitz | N/A | N/A |





