AirID, a novel proximity biotinylation enzyme, for analysis of protein–protein interactions
Figures
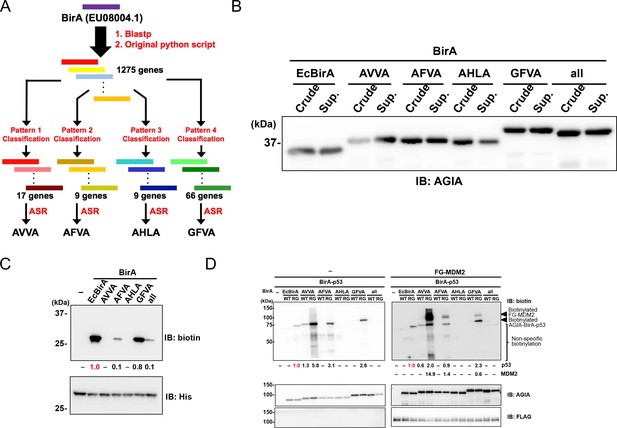
Characterization of novel BirA enzymes designed using metagenome data.
(A) A homolog library of BirA from E. coli (EcBirA) was generated using blastp and curated using an original python script. The curated library was multiple aligned using INTMSAlign and sequences were classified into four groups. Each group was phylogenetically analyzed, and ancestral sequences were designed. (B) AGIA-tagged AncBirAs were synthesized using the wheat cell-free system. Their expressions were confirmed using anti-AGIA antibody immunoblotting. (C) EcBirA or each AncBirA was added to the reaction mixture when His-bls-FLAG-GST was synthesized. Biotinylation of bls by each BirA was examined using anti-biotin antibody immunoblotting. As a control, the expression of each BirA was detected using His antibody. The band intensity of biotinylated His-bls-FLAG-GST was quantified with image J software, with the index intensity (value 1.0) shown in in red characters. (D) The WT or RG mutant of each BirA was fused to p53 (BirA-p53). They analyzed biotinylation activity with or without FLAG-GST-MDM2. As a control, the expression of each BirA-p53 and MDM2 was detected using an anti-AGIA antibody and an anti-FLAG antibody, respectively. The band intensities of biotinylated p53 and MDM2 were quantified with image J software. The index intensity (value 1.0) is shown in red characters.
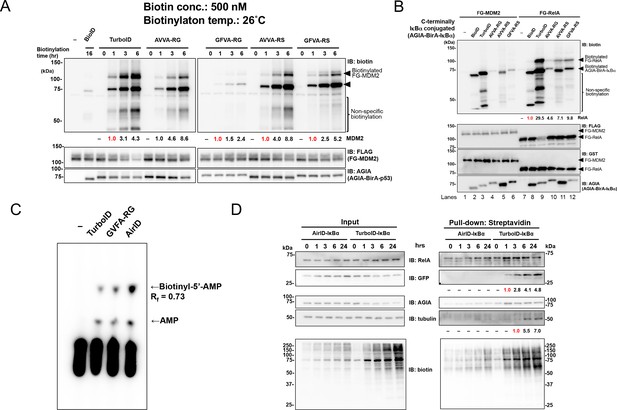
Validation of PPI dependency of novel designed BirA enzymes.
(A) RS mutants of AVVA and GFVA were cloned, and biotinylations of FLAG-GST-MDM2 (FG-MDM2) by BirA-p53 including RS mutants were analyzed. The reaction was performed at 500 nM of biotin at 26 °C for the described time. As a control, the expression levels of both BirA-p53 and MDM2 were detected using anti-AGIA antibody and anti-FLAG antibody, respectively. The band intensity of biotinylated MDM2 was quantified with image J software. The index intensity (value 1.0) is shown in red characters. (B) FG-RelA biotinylation by BirA-IκBα was examined. FG-MDM2 was used as the negative control. Biotinylations were performed at 500 nM of biotin at 26 °C for 1 hr (TurboID), 3 hr (AVVA-RG, AVVA-RS, and GFVA-RS), or 16 hr (BioID). As a control, the expression levels of BirA-p53 and MDM2 were detected using anti-AGIA antibody, anti-FLAG antibody and anti-GST antibody. The band intensity of biotinylated RelA was quantified with image J software. The index intensity (value 1.0) is shown in red characters. (C) GFVA-RG and GFVA-RS expressed using E. coli were purified using Ni beads and mixed with His-bls-FLAG-GST, which was synthesized using a wheat cell-free system and purified using glutathione beads. The mixtures were incubated a solution including [α-32P]ATP and biotin for 30 min at 37 °C. The resultant biotinyl-5′-AMP, AMP, or unreacted ATP was separated using cellulose thin-layer chromatography. (D) GFP and either AirID-IκBα or TurboID-IκBα were transfected in HEK293T, and biotin was added to 5 µM of this mixture for the described time period. After transfecting for 24 hr, cells were lysed by RIPA buffer including protease inhibitors, and biotinylated proteins were pulled down with streptavidin beads. As a control, the expression levels of enzyme-fused protein and target proteins were detected using each protein-specific antibody (left panel). The band intensity of pulled-down GFP and tubulin was quantified with image J software. The index intensity (value 1.0) is shown in red characters.
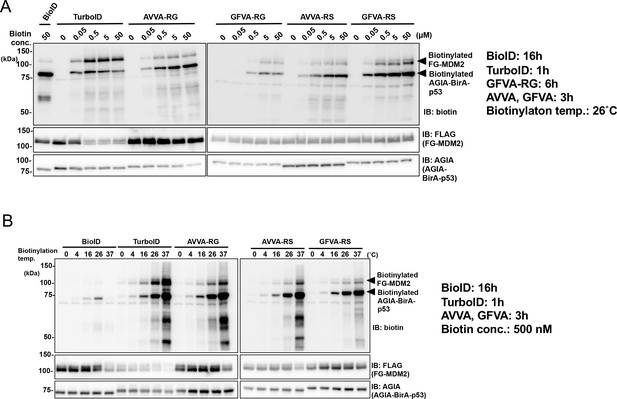
Condition for in vitro biotinylation.
Biotinylation of FG-MDM2 was performed using BioID, TurboID, AVVA-RG, GFVA-RG, AVVA-RS, or GFVA-RS fused to p53. (A) Biotin concentration and (B) reaction temperature were examined for in vitro proximal biotinylation. As a control, expression of each BirA-p53 and MDM2 was detected using anti-AGIA antibody and anti-FLAG antibody, respectively.
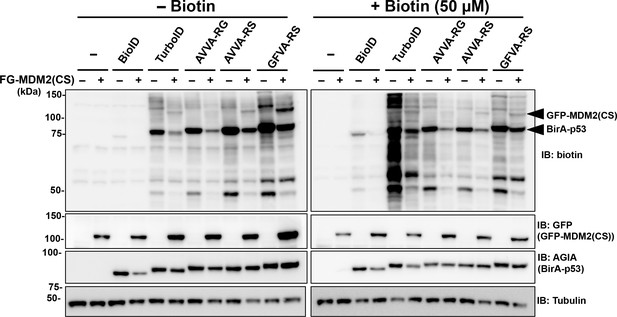
Comparison of MDM2 biotinylation by BirA-p53 enzymes.
BirA-p53-overexpressing HEK293T cells were cultured with or without GFP-MDM2 (CS mutant) and with or without 50 µM of biotin. Cells were lysed after incubating for 24 hr, and biotinylated p53 and MDM2 were detected using a specific antibody. As a control, the expression levels of BirA-p53 and MDM2 (CS) were detected using anti-AGIA antibody and anti-GFP antibody, respectively.
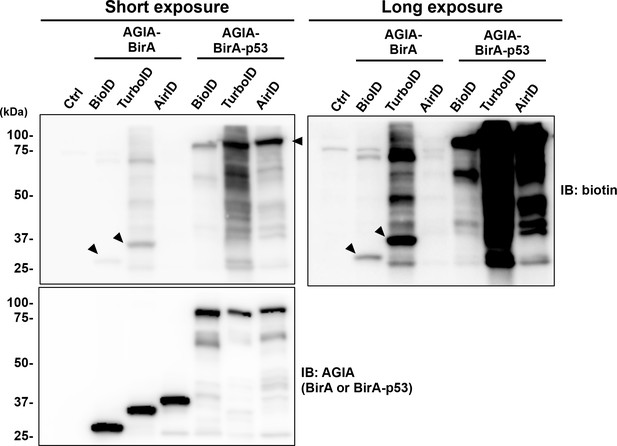
Self-biotinylation activity of novel designed BirA enzymes.
Each BirA or BirA-p53 was synthesized with the wheat cell-free system. Biotin (500 nM) was added and incubated without substrate at 26 °C in overnight. After the reaction, SDS-sample buffer was added to the mixtures, and the degree of self-biotinylation was analyzed. Arrow heads denote self biotinylation. As a control, the expression of each BirA-p53 was detected using anti-AGIA antibody.
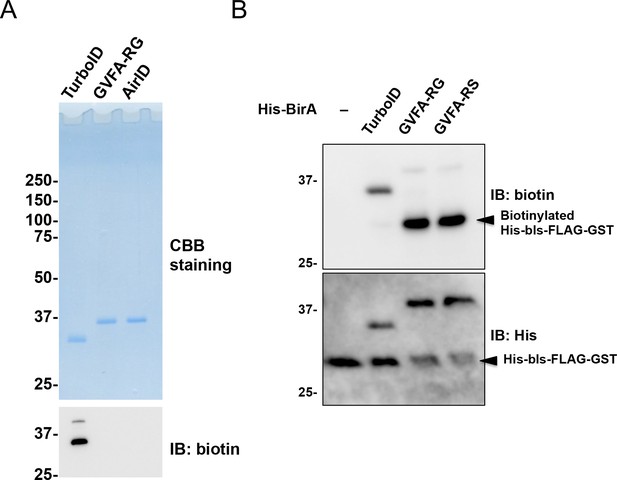
Comparison of enzymatic activity among GFVA-RG, AirID (GFVA-RS), and TurboID.
(A) TurboID, GFVA-RG, and AirID (GFVA-RS) were synthesized using E. coli cells. Synthesized proteins were purified, and (panel A, top) purifications were determined using CBB staining. In addition, (panel A, bottom) purified proteins were immunoblotted using an anti-biotin antibody. (B) His-bls-FLAG-GST was biotinylated using purified TurboID, GFVA-RG, and GFVA-RS. Biotinylation was analyzed using immunoblotting. As a control, the expression of each BirA-p53 and His-bls-FLAG-GST was detected using anti-His antibody. (C) Analysis of biotinyl-5′-AMP. RI-labelled ATP and biotin were added to purified proteins. The resultant biotinyl-5′-AMP was analyzed using thin layer chromatography.
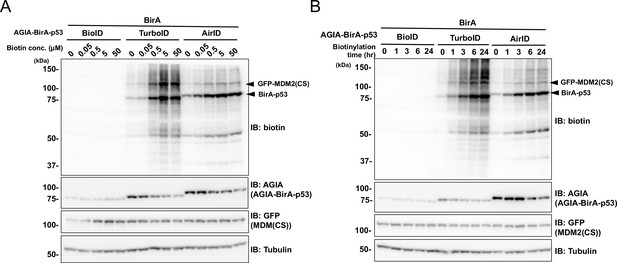
Optimization of AirID-dependent biotinylation in cells.
Conditions for in vivo biotinylation. Biotinylation of FG-MDM2 was performed using BioID-, TurboID-, or AirID-fused p53. Conditions of (A) biotin concentration and (B) biotinylation time were examined for in vitro proximal biotinylation. As a control, the expression levels of BirA-p53 and MDM2 (CS) were detected using anti-AGIA antibody and anti-GFP antibody, respectively.
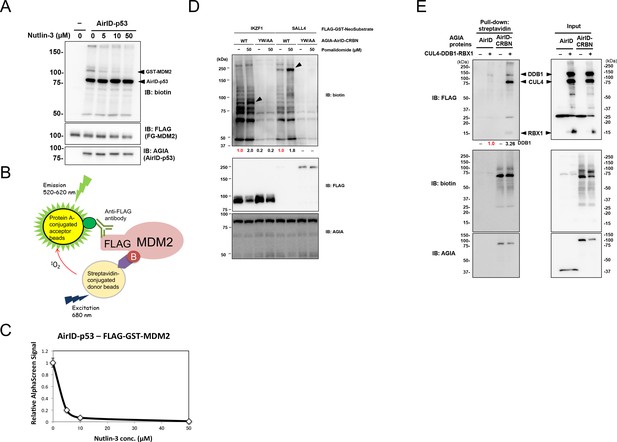
Biochemical applications of AirID-dependent biotinylation on PPI.
(A) Biotinylations of FG-MDM2 by AirID-p53 were carried out with or without Nutlin-3, which inhibits the interaction between p53 and MDM2, at 500 nM of biotin at 26 °C for 3 hr. Biotinylated MDM2 was detected using immunoblotting. As a control, expression levels of BirA-p53 and MDM2 were detected using anti-AGIA antibody and anti-FLAG antibody, respectively. (B) MDM2 biotinylation was detected using AlphaScreen with the reaction mixtures described for panel (A). Biotinylated FG-MDM2 interacts with both streptavidin donor beads and protein A acceptor beads to which the anti-FLAG antibody binds. The AlphaScreen results are shown in panel (C). (D) Pomalidomide-dependent biotinylations of FG-IKZF1 and FG-SALL4 by AirID-CRBN were analyzed. FG-IKZF1 or FG-SALL4 was biotinylated with or without pomalidomide at 500 nM of biotin at 26 °C for 3 hr. As the negative control, YW/AA mutant of AirID-CRBN, which does not bind to pomalidomide, was used. As a control, expression of AirID-CRBN and IKZF1 or SALL4 was detected using anti-AGIA antibody and anti-FLAG antibody, respectively. The band intensity of biotinylated IKZF1 or SALL4 was quantified with image J software. The index intensity (value 1.0) is shown in red characters. (E) CRL4CRBN complex proteins were biotinylated using AirID or AirID-CRBN. Biotinylated proteins were pulled down with streptavidin beads. As a control, the expression levels of AirID-CRBN and the complex component proteins was detected using anti-AGIA antibody and anti-FLAG antibody, respectively (right panel). The band intensity of biotinylated DDB1 was quantified with image J software. The index intensity (value 1.0) is shown in red characters.
-
Figure 3—source data 1
AlphaScreen data used to generate Figure 3C.
- https://cdn.elifesciences.org/articles/54983/elife-54983-fig3-data1-v2.xlsx
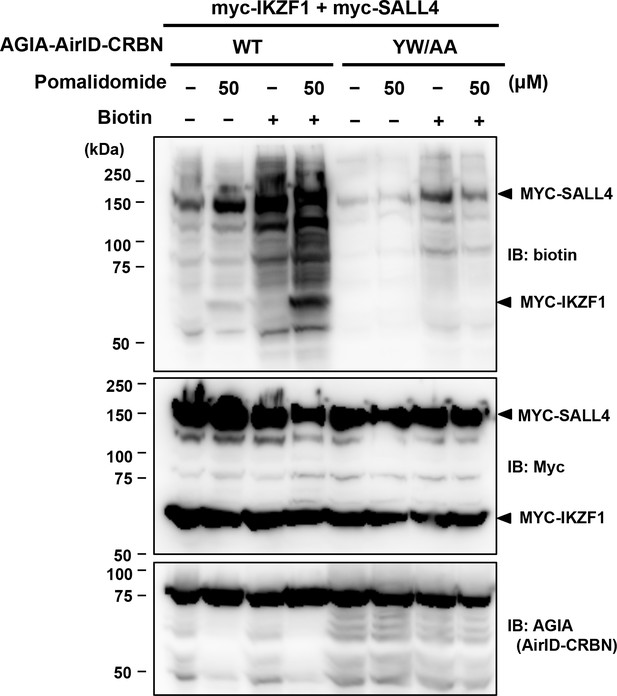
CRBN-dependent biotinylation of IKZF1 and SALL4 with pomalidomide.
AGIA-AirID-CRBN, myc-IKZF1, and myc-SALL4 were co-expressed in HEK293T cells. After treating with pomalidomide and MG132 for 6 hr, biotin was added and biotinylations were analyzed using immunoblotting. As a control, expression of AirID-CRBN and IKZF1 or SALL4 was detected using anti-AGIA antibody and anti-myc antibody, respectively.
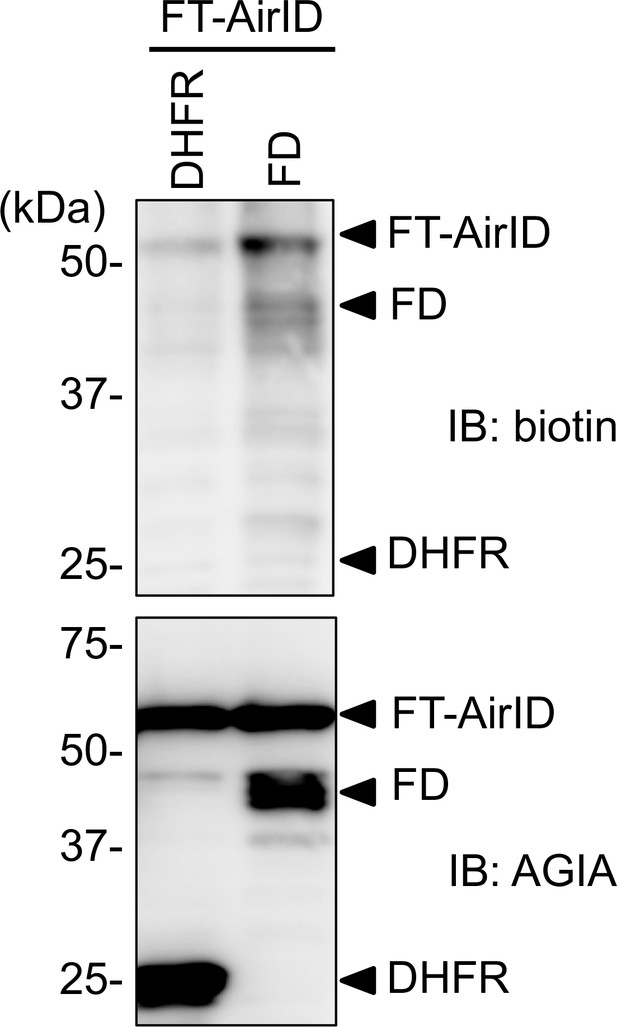
FT-dependent biotinylation of FD in the co-translational condition, based on the cell-free system.
FT-AGIA-AirID was synthesized using the wheat cell-free system with AGIA-FD or AGIA-DHFR in the presence of biotin. After the synthesis reaction, biotinylated proteins (upper panel) and synthesized proteins (lower panel) were identified using immunoblot analysis with the indicated antibodies. As a control, the expression levels of AirID-FT and FD were detected using anti-AGIA antibody.
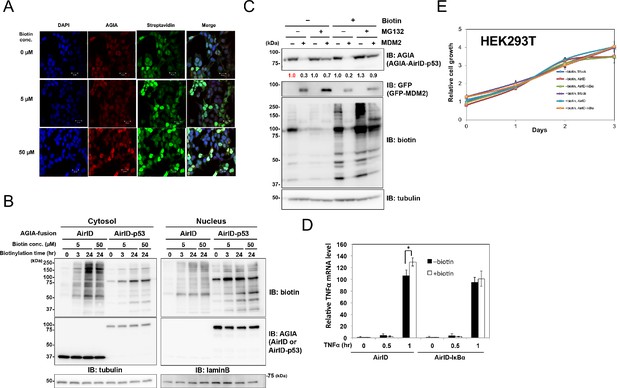
Dynamics and effects of AirID- and AirID-fusion proteins in cells.
Localization analysis was carried out for AirID using (A) immunostaining and (B) a fractionation assay. For immunostaining, HEK293 cells overexpressing AGIA-AirID-p53 were supplemented with the described biotin concentration for 3 hr. The cells were immobilized using anti-AGIA antibody and visualized using anti-rabbit IgG antibody-AlexaFluor555 and streptavidin-AlexaFluor488. AGIA-tagged AirID or AirID-p53 was transfected in HEK293T for the fractionation assay. The next day, biotin was added to 5 µM or 50 µM for the described time. Cytoplasmic and nuclear proteins were fractionated using a ProteoExtract Subcellular Proteome Extraction kit (Merck). As a control, expression of AirID or AirID-p53 was detected using anti-AGIA antibody. (C) AGIA tagged AirID-p53 was co-transfected with or without GFP-MDM2 in HEK293T. Biotin was added to a concentration of 50 µM at the same time. After 6 hr, DMSO or MG132 was added to a concentration of 10 µM. As a control, expression of MDM2 was detected using anti-GFP antibody. The band intensity of AirID-p53 was quantified with image J software. The index intensity (value 1.0) is shown in red characters. (D) qRT-PCR using AirID-IκBα. AGIA-tagged AirID or AirID-IκBα was stably expressed using renti-virus in HEK293T. Cells were seeded in a 96-well plate, and biotin was added at the same time. Next day, cells were stimulated using TNFα (20 ng/mL) for 0, 0.5, or 1 hr. In the cells, the mRNA level of TNFα was analyzed by qRT-PCR. Mean ± S.D. (n = 3). *, p<0.05. (E) Viability of AirID-expressing cells. AGIA-tagged AirID or AirID-IκBα was stably expressed using renti-virus in HEK293T. Cells were seeded in 96-well plates, and biotin was added the next day. The MTS assay was performed 0, 1, 2, or 3 days after adding biotin to measure cell viability.
-
Figure 4—source data 1
Cell growth analysis data relating to Figure 4E.
- https://cdn.elifesciences.org/articles/54983/elife-54983-fig4-data1-v2.xlsx
-
Figure 4—source data 2
qRT-PCR data related to Figure 4D.
- https://cdn.elifesciences.org/articles/54983/elife-54983-fig4-data2-v2.xlsx
Biotinylation conditions of cells stably expressing AirID.
AGIA-AirID or AGIA-AirID-IκBα was stably expressed in HEK293T. Expressions were determined using immunoblotting. AirID-dependent biotinylation of endogenous RelA was confirmed under different conditions of biotin concentration (A) or biotinylation time (B). As a control, expression of AirID or AirID-IκBα and RelA was detected using anti-AGIA antibody and anti-RelA antibody (right panel).
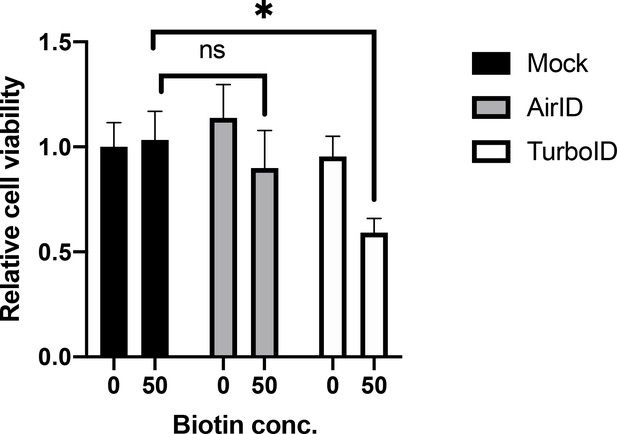
Comparison of the effect on cell growth of TurboID and AirID.
AirID or TurboID was transiently expressed in HEK293T cells. Cell viability was measured using the CellTiter-Glo luminescent cell viability assay system 2 days after the addition of biotin. Mean ± S.D. (n = 3). *, p<0.05.
-
Figure 4—figure supplement 2—source data 1
Viability analysis data related to Figure 4—figure supplement 2.
This excel file contains viability analysis data obtained using a CellTiter-Glo Luminescent Cell Viability Assay (Promega) shown in Figure 4—figure supplement 2. The graph in Figure 4—figure supplement 2 was generated using a ratio calculated on the basis of value subtracted background signal value.
- https://cdn.elifesciences.org/articles/54983/elife-54983-fig4-figsupp2-data1-v2.xlsx
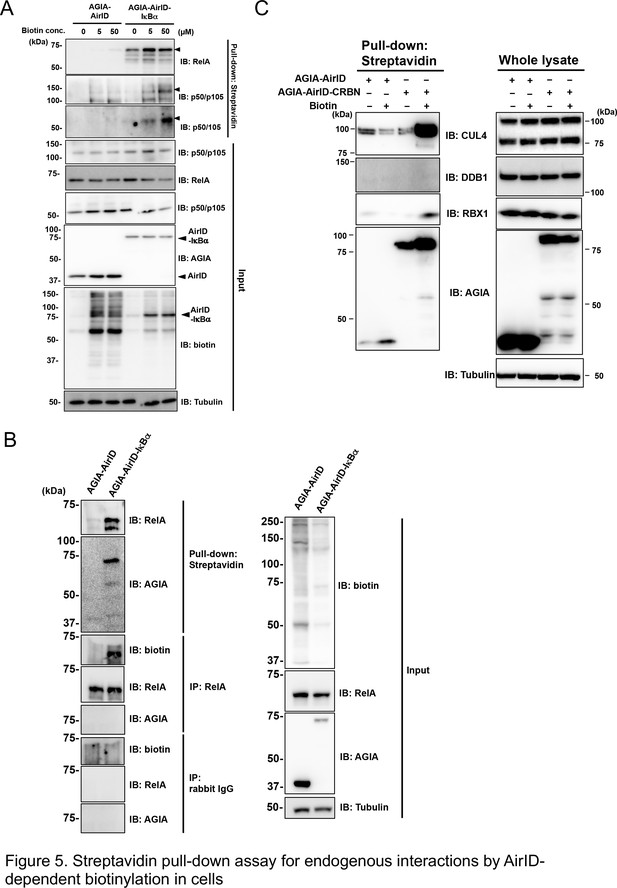
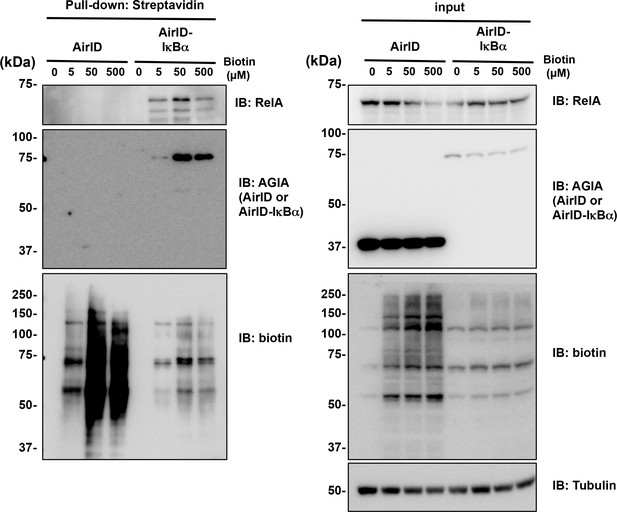
Streptavidin pull-down assay for endogenous interactions using AirID-dependent biotinylation in cells.
(A) Biotin was added to HEK293T cells expressing AirID or AirID-IκBα to concentrations of 0, 5, or 50 µM before incubating for 3 hr. Cells were lysed before immunoprecipitating with streptavidin beads. Pulled-down proteins were detected using immunoblotting with the described antibody. (B) AirID- or AirID-IκBα-expressing HEK293T cells were supplemented with 5 µM biotin and incubated for 3 hr. Cells were lysed and pulled down with streptavidin beads or immunoprecipitated with anti-RelA. Normal rabbit-IgG was also used as a negative immunoprecipitation control. Pulled-down or immunoprecipitated proteins were detected using immunoblotting with the described antibody. (C) Biotinylation of the CRL4CRBN complex was performed using AirID-CRBN. AirID- or AirID-CRBN-expressing HEK293T cells were incubated with or without 5 µM of biotin for 3 hr. Cells were lysed and pulled down with streptavidin. CUL4, DDB1, and RBX1 were detected using immunoblotting with each antibody.
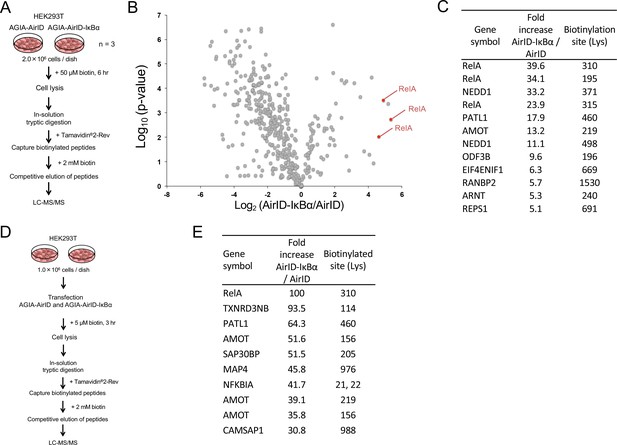
Mass spectrometry analysis of biotinylated proteins in AirID-IκBα expressing cells.
(A) Schematic figure for detecting biotinylated proteins using cells stably expressing AirID. HEK293T cells that stably expressed AGIA-AirID or AGIA-AirID-IκBα were cultured in DMEM containing 50 µM for 6 hr before collecting (n = 3). Collected cells were lysed, and proteins were digested in solution using trypsin. Biotinylated peptides were captured from digested peptides using Tamavidin2-Rev beads (Wako), which can elute biotinylated samples using 2 mM biotin. Eluted peptides were detected using LC-MS/MS. (B) A volcano plot showing AirID-IκBα versus AirID against p-value of triplicate experiments. (C) A list of peptides increased by more than 5-fold. (D) Schematic figure for detecting biotinylated proteins using cells transiently expressing AirID. HEK293T cells that transiently expressed AGIA-AirID or AGIA-AirID-IκBα were cultured in DMEM containing 5 µM for 3 hr before collecting (n = 1). Biotinylated proteins were detected using a similar method. (E) A list of the top ten peptides increased by AirID-IκBα.
-
Figure 6—source data 1
Mass spectrometry data related to Figure 6B.
This excel file contains data from mass spectrometry analyses using AirID-expressing cells. The graph in Figure 6B was generated using a logarithmic value of the abundance ratio and the p-value.
- https://cdn.elifesciences.org/articles/54983/elife-54983-fig6-data1-v2.xlsx
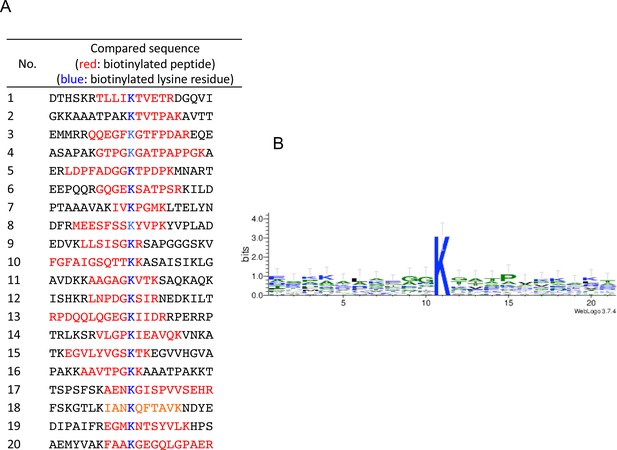
Biotinylation site characteristics.
Twenty sequences around the biotinylation site were compared on the basis of the results of analysis by LC-MS/MS. The results were (A) listed, and (B) the sequence logo was generated using WebLogo (http://weblogo.berkeley.edu/).
Tables
| Reagent type (species) or resource | Designation | Source or reference | Identifiers | Additional information |
|---|---|---|---|---|
| Gene (E. coli-modified) | BioID | Kim et al., 2014; DOI: 10.1073/pnas.1406459111 | Obtained by mutating 118Arg of BirA to Gly | |
| Gene (E. coli-modified) | TurboID | Branon et al., 2018 DOI: 10.1038/nbt.4201 | Synthetic gene fragment was purchased using Invitrogen Gene Art | |
| Gene (artificially designed) | AVVA | This paper | Synthetic gene fragment was purchased using Invitrogen Gene Art | |
| Gene (artificially designed) | AFVA | This paper | Synthetic gene fragment was purchased using Invitrogen Gene Art | |
| Gene (artificially designed) | AHLA | This paper | Synthetic gene fragment was purchased using Invitrogen Gene Art | |
| Gene (artificially designed) | GFVA | This paper | Synthetic gene fragment was purchased using Invitrogen Gene Art | |
| Gene (artificially designed) | All | This paper | Synthetic gene fragment was purchased using Invitrogen Gene Art | |
| Gene (Homo sapiens) | IκBα | Mammalian Gene Collection | ||
| Gene (H. sapiens) | RelA | Mammalian Gene Collection | ||
| Gene (H. sapiens) | p53 | Mammalian Gene Collection | ||
| Gene (H. sapiens) | Mdm2 | Mammalian Gene Collection | ||
| Gene (H. sapiens) | CRBN | Mammalian Gene Collection | ||
| Gene (H. sapiens) | RBX1 | Mammalian Gene Collection | ||
| Gene (H. sapiens) | DDB1 | Mammalian Gene Collection | ||
| Gene (H. sapiens) | CUL4 | Mammalian Gene Collection | ||
| Gene (H. sapiens) | SALL4 | Mammalian Gene Collection | ||
| Gene (H. sapiens) | IKZF1 | Mammalian Gene Collection | ||
| Recombinant DNA reagent | pEU (plasmid) | Cell-Free Science | For protein expression using wheat germ cell-free system | |
| Recombinant DNA reagent | pcDNA3.1 (plasmid) | Modified Invitrogen | For protein expression using human cell | |
| Recombinant DNA reagent | pET30a (plasmid) | Modified Merck | For protein expression using E. coli | |
| Antibody | Anti-AGIA HRP | Yano et al., 2016 DOI: 10.1371/journal.pone.0156716 | Rabbit mAb | WB (1:10,000) |
| Antibody | Anti-biotin, HRP-linked antibody (from goat) | Cell Signaling | #7075S RRID:AB_10696897 | WB (1:10,000) |
| Antibody | His-probe antibody (H-3) (mouse mAb) | SantaCruz | sc-8036 RRID:AB_627727 | WB (1:1000) |
| Antibody | Anti-FLAG M2-HRP (mouse mAb) | Sigma | A8592 RRID:AB_439702 | WB (1:10,000) |
| Antibody | Anti-GST-tag pAb-HRP-DirecT (rabbit pAb) | MBL | PM013-7 RRID:AB_10598029 | WB (1:1000) |
| Antibody | Anti-p65 (D14E12) (rabbit mAb) | Cell Signaling | #8242S RRID:AB_10859369 | WB (1:1000) |
| Antibody | Anti-GFP (1E4) (mouse mAb) | MBL | M048-3 RRID:AB_591823 | WB (1:5000) |
| Antibody | Anti-α-tubulin pAb-HRP-DirecT (rabbit pAb) | MBL | PM054-7 RRID:AB_10695326 | WB (1:10,000) |
| Antibody | Anti-laminB (goat pAb) | SantaCruz | sc-6217 RRID:AB_648158 | WB (1:1000) |
| Antibody | Anti-Myc (4A6) (mouse mAb) | Merck | 05–724 RRID:AB_11211891 | WB (1:2000) |
| Antibody | Anti-p50/p105 (D7H5M) (rabbit mAb) | Cell Signaling | #12540S RRID:AB_2687614 | WB (1:1000) |
| Antibody | Anti-CUL4 (H-11) | SantaCruz | sc-377188 | 1:1000; 5% skimmed milk in TBST |
| Antibody | Anti-DDB1 (E-11) | SantaCruz | sc-376860 | 1:1000; 5% skimmed milk in TBST |
| Antibody | Anti-RBX1 (E-11) | SantaCruz | sc-393640 RRID:AB_2722527 | 1:1000; 5% skimmed milk in TBST |
| Antibody | Anti-Rabbit IgG, HRP-Linked F (ab’)2Fragment Donkey | GE Healthcare | NA9340V RRID:AB_772191 | WB (1:10,000) |
| Antibody | Anti-Mouse IgG, HRP-Linked F (ab’)2Fragment Sheep | GE Healthcare | NA9310V RRID:AB_772193 | WB (1:10,000) |
| Antibody | Normal rabbit IgG | MBL | PM035 | |
| Antibody | Streptavidin, Alexa Fluor 488 | Thermo | S32354 RRID:AB_2315383 | |
| Antibody | F(ab')2-Goat anti-Rabbit IgG (H+L) Cross-Adsorbed Secondary Antibody, Alexa Fluor 555 | Thermo | A21431 RRID:AB_1500601 | |
| Cell line (H. sapiens) | HEK293T | RIKEN BRC | RCB2202 | Maintained in DMEM supplementedwith 10% FBS |
| Beads | Dynabeads proteinG | Invitrogen | DB10004 | |
| Beads | Ni Sepharose High Performance | GE Healthcare | 17526801 | |
| Beads | Glutathione Sepharose 4B | GE Healthcare | 17526801 | |
| Beads | Streptavidin Sepharose High Performance | GE Healthcare | 17511301 | |
| Beads | Tamavidin 2-Rev magnetic beads | Fujifilm | 133–18611 | |
| Commercial assay kit | Alphascreen ProteinA detection kit | Perkin Elmer | 6760617R | |
| Commercial assay kit | CellTiter 96 AQueous One Solution Cell Proliferation Assay | Promega | G5430 | |
| Commercial assay kit | SuperPrep II Cell Lysis and RT kit for qPCR | TOYOBO | SCQ401 | |
| Commercial assay kit | KOD SYBR qPCR Mix | TOYOBO | QKD-201 | |
| Commercial assay kit | Wheat germ cell-free protein synthesis kit | Cell-Free Science | ||
| Chemical compound and drug | Fetal Bovine Serum | Wako | ||
| Chemical compound and drug | DMEM, low glucose | Wako | 041–29775 | |
| Chemical compound and drug | Nutlin-3 | Sigma | N6287-1MG | |
| Chemical compound and drug | Pomalidomide | TCI | P2074 | |
| Chemical compound and drug | MG-132 | Peptide Institute | 3175 v | |
| Chemical compound and drug | D-Biotin | Nacalai tesque | 04822–91 | |
| Chemical compound and drug | Protease inhibitor cocktail | Sigma | P8340-5ML | |
| Chemical compound and drug | Penicillin-Streptomycin | Thermo | 15140122 | |
| Chemical compound and drug | Isobutyric acid | Nacalai tesque | 06429–85 | |
| Chemical compound and drug | Ammonia solution | Nacalai tesque | 025–12 | |
| Other | cellulose TLC plate | Merck | 1.05552.0001 | Toll for chromatography |
Additional files
-
Source code 1
Library-curation toolkit.
- https://cdn.elifesciences.org/articles/54983/elife-54983-code1-v2.tar.gz
-
Supplementary file 1
Amino acid and nucleic acid sequences of ancestral BirAs.
Amino acid and nucleic acid sequences for the ancestral BirAs designed (AVVA, AFVA, AHLA, GFVA, and all) in this report.
- https://cdn.elifesciences.org/articles/54983/elife-54983-supp1-v2.xlsx





