The membrane periodic skeleton is an actomyosin network that regulates axonal diameter and conduction
Figures
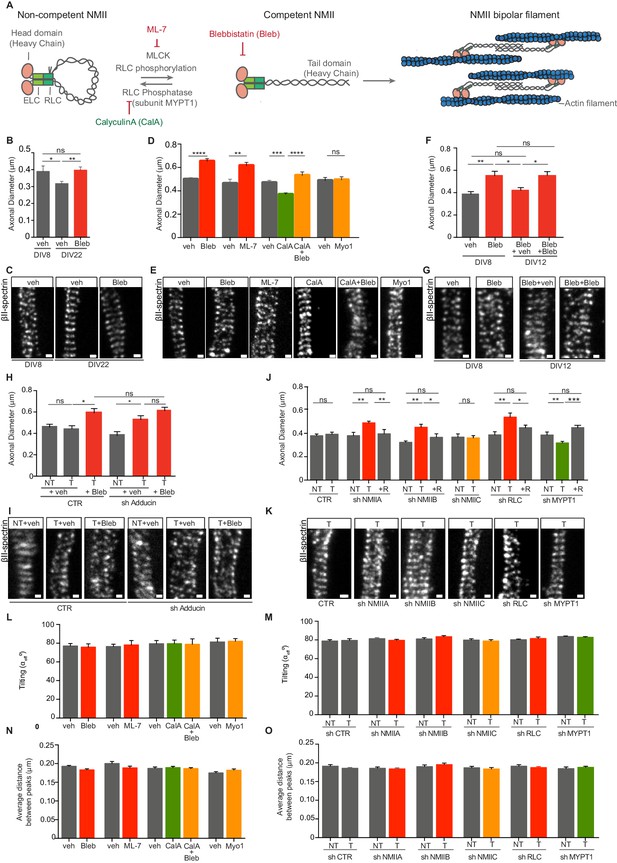
Modulation of NMII activity regulates axon diameter.
(A) Representation of NMII structure and activity regulation. NMII contains two heavy chains (HC), two RLCs and two ELCs. The head domain includes an actin-binding site and an ATPase motor domain. ELCs and RLCs are bound to NMII head domains. In the absence of RLC phosphorylation, NMII is in a non-competent conformation. Upon RLC phosphorylation by MLCK, NMII unfolds to generate a competent form. NMII is then able to assemble into bipolar filaments, which bind to actin. MLC phosphatase that includes a regulatory subunit -MYPT1- can revert this activation. NMII activity can be modulated by drugs including ML-7, a specific MLCK inhibitor; calyculin A (CalA) an inhibitor of RLC phosphatase, and blebbistatin (Bleb) that inhibits NMII-mediated ATP hydrolysis. (B) Axon diameter of DIV8 and DIV22 hippocampal neurons incubated either with vehicle (veh) or 3 μM blebbistatin (Bleb) at DIV3 and DIV8 (1 hr before fixation); n = 6–8 axons/condition and 20–87 rings/axon. (C) Representative STED images of βII-spectrin immunostaining using a STAR 635P secondary antibody related to (B). (D) Axon diameter of DIV8 rat hippocampal neurons incubated with drug modulators of myosin activity: 3 μM blebbistatin (Bleb), 4 μM ML-7, 5 nM calyculin A (CalA), 5 nM calyculin A + 3 μM blebbistation (CalA+Bleb), 4 μM myovin1 (Myo1) and the respective vehicles (veh). Calyculin A was added at DIV8 (25 min before fixation), while the remaining drugs were added at DIV3 and DIV8 1 hr before fixation; n = 8–13 axons/condition and 10–77 rings/axon. (E) Representative STED images of βII-spectrin immunostaining using a STAR 635P secondary antibody related to (D). (F) Axon diameter of DIV8 rat hippocampal neurons incubated with either 3 μM blebbistatin (Bleb) or vehicle (veh) at DIV3 and DIV8 (1 hr before fixation). In cells treated with blebbistatin up to DIV8, drug was replaced by either vehicle (Bleb+veh) or drug treatment was continued (Bleb+Bleb) and axonal diameter was analysed at DIV12; n = 7–10 axons/condition and 12–79 rings/axon. (G) Representative STED images of βII-spectrin immunostaining using a STAR 635P secondary antibody related to (F). (H) Axon diameter of DIV8 rat hippocampal neurons upon shRNA-mediated knockdown of adducin (sh Adducin) or transfection using a control shRNA (CTR) and subsequent incubation with either Bleb or veh; T- transfected; NT- non-transfected; n = 4–13 axons/condition and n = 12–88 rings/axon. (I) Representative STED images of βII-spectrin immunostaining using a STAR 635P secondary antibody related to (H). (J) Axon diameter of DIV8 rat hippocampal neurons upon shRNA-mediated knockdown of different NMII isoforms (NMIIA, NMIIB and NMIIC), RLC or MYPT1; scramble shRNA was used as control (CTR); shRNA resistant constructs (R) for each specific shRNA were used to rescue their effect; T- transfected; NT- non-transfected; n = 5–15 axons/condition and 6–103 rings/axon. (K) Representative STED images of βII-spectrin immunostaining using a STAR 635P secondary antibody related to (J). (L) Tilting (effective angle αeff) of the actin rings in relation to the axonal axis after incubation with NMII chemical modulators; similar concentrations and incubation times as defined in (D); n = 8–14 axons/condition and 14–72 rings/axon and (M) after shRNA-mediated downregulation; n = 5–15 axons/condition and 7–70 rings/axon. Tilting was quantified in STED images after βII-spectrin immunostaining. (N) MPS periodicity after incubation with myosin-targeting drugs; similar concentrations and incubation times as defined in (D). The average distance between peaks was quantified by STED after βII-spectrin immunostaining; n = 8–14 axons/condition and 14–72 rings/axon. (O) MPS periodicity after shRNA-mediated knockdown of different NMII isoforms (NMIIA, NMIIB and NMIIC), RLC or MYPT1; a scramble shRNA was used as control (CTR). The average distance between peaks was quantified by STED after βII-spectrin immunostaining; T- transfected; NT- non-transfected; n = 5–15 axons/condition and 7–70 rings/axon. In all panels: Scale bars, 200 nm. *p<0.05; **p<0.01; ***<0.001; ****p<0.0001; ns-non significant. Graphs show mean ± s.e.m. In every case displayed in this Figure, at least 3 independent experiments have been performed.
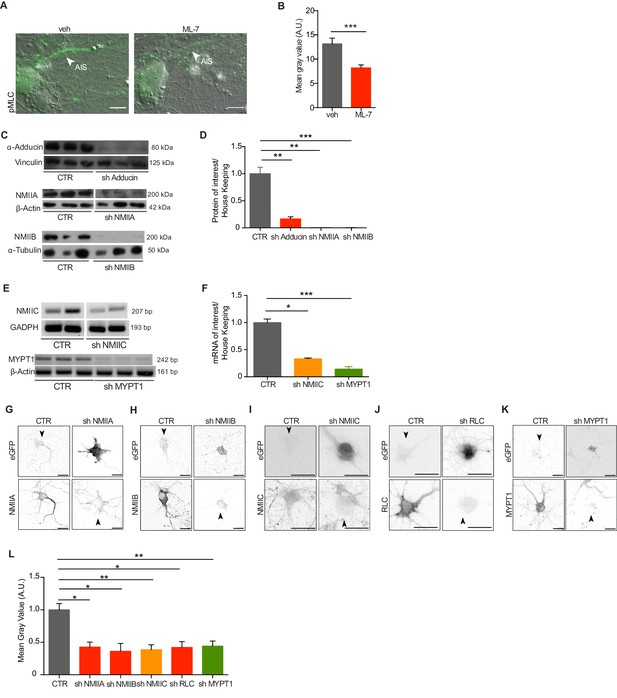
Analysis of ML-7 activity and ShRNA-mediated downregulation.
(A) Representative images of DIC and pMLC immunolabeling of hippocampal neurons treated with either vehicle (veh) or 4 μM ML-7 at DIV3 and DIV8, (1 hr before fixation). Scale bar: 10 μm. (B) Quantification related to (A). The mean gray intensity (arbitrary units- A.U.) corresponding to the raw integrated density/area of the region of interest, was determined using Fiji. (C) Western blot analysis of expression levels following shRNA-mediated knockdown of α-adducin (upper panel), NMIIA (middle panel) and NMIIB (bottom panel) after CAD cell transfection. CAD cells were transfected with control vectors (CTR; empty pLKO-1 vector for the upper panel and sh scramble for the middle and bottom panels) or with plamids encoding specific shRNAs for α-adducin (sh Adducin), NMIIA (sh NMIIA) or NMIIB (sh NMIIB). Vinculin (upper panel), β-actin (middle panel) and α-tubulin (bottom panel) were used as housekeeping proteins. In each case triplicates are shown. (D) Quantification related to (C). (E) RT-PCR following shRNA-mediated knockdown of NMIIC (upper panel) and MYPT1 (bottom panel) after transfection of either PC-12 cells or CAD cells, respectively. Cells were transfected with shRNA scramble control vectors (CTR) or with plasmids encoding specific shRNAs for NMIIC (sh NMIIC) or MYPT1 (sh MYPT1). GAPDH (upper panel) and β-actin (bottom panel) were used as housekeeping genes. Either duplicates or triplicates are shown. (F) Quantification related to (E). (G–K) Representative images of shRNA-mediated down regulation in primary hippocampal neurons of NMIIA (G), NMIIB (H), NMIIC (I), RLC (J) and MYPT1 (K). ShRNA transfected cells are eGFP-positive (upper right panels), whereas non-transfected cells of the same well are shown as eGFP negative (upper left panels- CTR). Immunofluorescence against proteins of interest are shown in the lower panels. (L) Quantification of shRNA-mediated downregulation as assessed by immunofluorescence, related to (G–K). Scale bar: 30 μm. *p<0.05; **p<0.01; ***<0.001; ****p<0.0001; ns-non significant. Graphs show mean ± s.e.m. In each case, three independent experiments were performed.
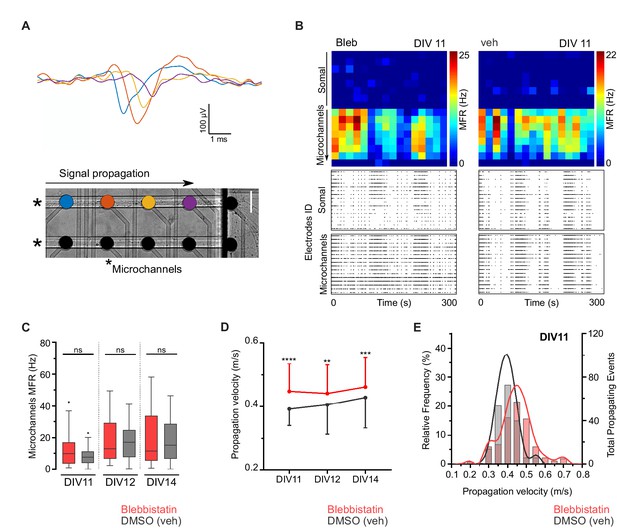
Blebbistatin treatment increases axonal signal propagation velocity.
(A) Single propagating event recorded along four electrodes inside a microchannel. Each colored signal trace corresponds to an electrode from a single microchannel. Two microchannels are indicated by asterisks in the phase-contrast image of hippocampal neurons cultured on a μEF platform at DIV8 (scale bar = 50 μm). (B) Activity maps of vehicle and 3 μM blebbistatin (Bleb) treated cultures at DIV11. Each pixel corresponds to one recording electrode and the mean firing rate (MFR) is color-coded for each electrode. Notice that μEFs allow for the recording of both somal and axonal activity. Below are shown raster plots of 300 s of activity from 16 active electrodes of the somal compartment and from 16 electrodes within the microchannels (one electrode per microchannel). (C) MFR from the electrodes in the microchannels for vehicle and Bleb-treated cultures (DIV11-14). (D) Median propagation velocity (± SD) for vehicle and blebbistatin-treated cultures at DIV11, 12, and 14 (propagating events pooled from 25 to 43 microchannels, from 3 to 4 independent μEFs). Unpaired t-test or Mann-Whitney test; ns = not significant, **p<0.01, ***p<0.001 and ****p<0.0001. (E) Frequency distribution of the propagation velocity domains (0.05 m/s binning) at DIV11 for both conditions.
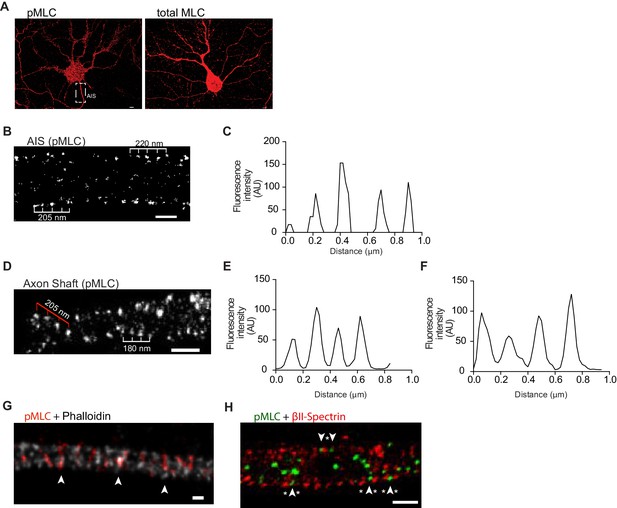
Phosphorylated NMII light chains are organized as circular periodic structures persisting throughout the axon shaft.
(A) Immunolabeling of DIV8 hippocampal neurons with rabbit anti-pMLC (left) and rabbit anti-total MLC (right) using a secondary anti- rabbit Alexa Fluor 647 antibody. Scale bar: 5 μm. (B) Single colour SMLM of pMLC distribution in the AIS using a secondary Alexa Fluor 647 antibody. Scale bar: 500 nm. (C) Analysis of the periodicity related to (B). (D) 2D single colour SMLM of the axon shaft of a DIV8 hippocampal neuron immunostained for pMLC using a secondary Alexa Fluor 647 antibody. Scale bar: 500 nm. Periodic distribution in outer most regions of the axon shaft is highlighted with white ruler; inner distribution consistent with anchoring in different positions of adjacent actin rings is highlighted with red ruler. (E) Analysis of periodicity in outer most regions of the axon shaft (highlighted in white in D). (F) Analysis of inner periodic distribution (highlighted in red in D). (G) Two colour STED of a DIV8 hippocampal neuron immunostained against pMLC (red) and stained for actin (gray), using a STAR 580 secondary antibody and phalloidin 635, respectively. pMLC molecules, highlighted with arrowheads, co-localize with actin. Scale bar: 200 nm. The raw image was deconvolved using the CMLE algorithm (Huygens Professional, Scientific Volume Imaging). (H) Z projection of 3D two-colour SMLM of a DIV8 hippocampal neuron immunostained against βII-spectrin (red) and pMLC (green) using secondary antibodies labeled with Alexa Fluor 532 and 647, respectively. Molecules intercalating at outermost positions are highlighted using arrowheads (pMLC) and asterisks (βII-spectrin). Scale bar: 500 nm.
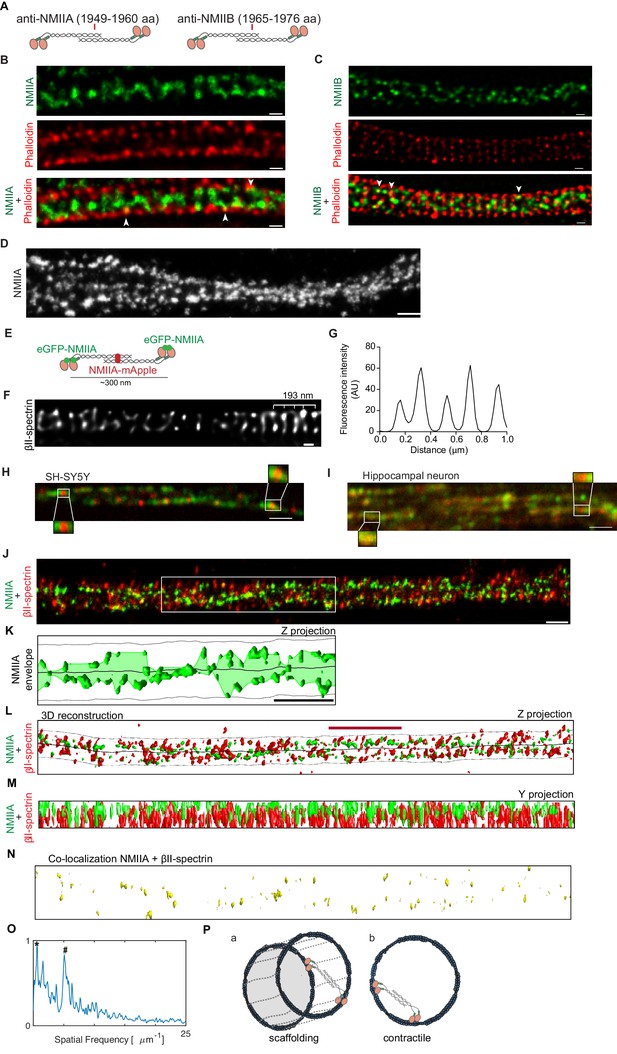
NMII heavy chains organize into filaments distributed in multiple orientations along the axon shaft.
(A) Representation of recognition sites of antibodies against NMIIA and NMIIB (NMIIA: 1949–1960 aa; NMIIB: 1965–1976 aa). (B) Representative STED analysis of a DIV8 hippocampal neuron co-stained with NMIIA using a secondary anti-rabbit STAR 580 (upper) and phalloidin 635 (middle); the merged image is shown in the lower panel. Scale bar: 200 nm. (C) Representative STED analysis of a DIV8 hippocampal neuron co-stained with NMIIB using a secondary anti-rabbit STAR 580 (upper) and phalloidin 635 (middle); the merged image is shown in the lower panel. Scale bar: 200 nm. (D) Z projection of 3D SMLM of an axon single labeled for NMII heavy chain A using an Alexa Fluor 647 labeled secondary antibody. NMIIA labeling following different orientations in relation to the axonal axis is highlighted with arrowheads. Scale bar: 500 nm. (E) Representation of a bipolar NMIIA filament, with the N-terminal eGFP tagged and the C-terminal mApple tagged. (F) SH-SY5Y was immunolabelled with βII-spectrin using a STAR 635P secondary and imaged using STED. Scale bar: 200 nm. The raw image was deconvolved using the CMLE algorithm (Huygens Professional, SVI). (G) Analysis of SH-SY5Y βII-spectrin periodicity related to (F). (H) Representative spinning disk image of SH-SY5Y after co-transfection with eGFP-NMIIA and NMIIA-mApple. Scale bar: 1 µm. Insets highlight bipolar NMIIA filaments of ~300 nm. (I) Representative spinning disk image of a primary hippocampal neuron axon after co-transfection with eGFP-NMIIA and NMIIA-mApple. Scale bar: 1 µm. (J) Z projection of a 3D SMLM double stained for βII-spectrin (red) and NMII heavy chain A (green) using anti-mouse Alexa Fluor 532 and anti-rabbit Alexa Fluor 647, respectively. Scale bar: 500 nm. (K) Analysis of the envelope of NMIIA labeling (Z projection) relative to the region highlighted by the white box in (J); the axonal membrane (dashed line) and centerline (solid line) are depicted. Scale bar: 1 µm. (L) Computer 3D reconstruction of the SMLM double stained for βII-spectrin (red) and NMII heavy chain A (green) of the image shown in (J). Solid line follows the axon centerline, whereas the dashed line marks the cellular membrane. The red scale bar is 1.7 µm, which corresponds to the size of the βII-spectrin secondary structure (see panel O). A Z projection is shown. (M) Y projection related to (J). (N) Co-localization of βII-spectrin and NMII heavy chain A for the reconstruction shown in (L). (Q) Fluorescence intensity spatial frequency of βII-spectrin on the axonal axis, analyzed by Fourier transform. In addition to the 0.20 µm peak (marked with ‘#”), there is also a consistent peak at 1.7 µm (marked with ‘*”). (P) Models for the distribution of the NMII along axons. NMII filaments may crosslink adjacent actin rings (model a) or span individual rings (model b), in both cases with variable angles relative to the axonal axis.
Videos
3D single colour SMLM of pMLC distribution in the AIS of a DIV8 hippocampal neuron.
Scale bar: 500 nm.
3D two-color SMLM of DIV8 hippocampal neurons immunostained against βII-spectrin (red) and NMIIA (green).
Scale bar: 500 nm.
Tables
| Reagent type (species) or resource | Designation | Source or reference | Identifiers | Additional information |
|---|---|---|---|---|
| Antibody | Rabbit polyclonal anti-NMIIA | Sigma-Aldrich | Cat# M8064, RRID:AB_260673 | WB_1:1000; IF_1:200 |
| Antibody | Rabbit polyclonal anti-NMIIB | Sigma-Aldrich | Cat# M7939, RRID:AB_260669 | WB_1:1000; IF_1:200 |
| Antibody | Rabbit polyclonal canti-α-adducin | Abcam | Cat# ab51130, RRID:AB_867519 | WB_1:1000 |
| Antibody | Rabbit monoclonal anti-vinculin | Abcam | Cat# ab129002, RRID:AB_11144129 | WB_1:1000 |
| Antibody | Mouse monoclonal anti-βactin | Sigma-Aldrich | Cat# A5441, RRID:AB_476744 | WB_1:5000 |
| Antibody | Mouse monoclonal anti-αtubulin | Sigma-Aldrich | Cat# T6199, RRID:AB_477583 | WB_1:1000 |
| Antibody | Peroxidase-AffiniPure donkey polyclonal anti-rabbit IgG (H+L) | Jackson Immuno Research Labs | Cat# 711-035-152, RRID:AB_10015282 | WB_1:5000 |
| Antibody | Peroxidase-AffiniPure donkey polyclonal anti-mouse IgG (H+L) | Jackson Immuno Research Labs | Cat# 715-035-151, RRID:AB_2340771 | WB_1:5000 |
| Antibody | Mouse monoclonal anti-βII-spectrin | BD Transduction | Cat# 612563,RRID:AB_399854 | IF_1:200 |
| Antibody | Rabbit polyclonal anti-MAP2 | Synaptic Systems | Cat# 188002, RRID:AB_2138183 | IF_1:20000 |
| Antibody | Rabbit polyclonal anti-NMIIC | Robert Adelstein NHLBI, Bethesda, USA | N/A | IF_1:40 |
| Antibody | Rabbit polyclonal anti-pMLC2 Thr18/Ser19 | Cell Signaling | Cat# 3674, RRID:AB_2147464 | IF_1:50 |
| Antibody | Rabbit polyclonal anti-MYPT1 | Cell Signaling | Cat# 2634, RRID:AB_915965 | IF_1:50 |
| Antibody | Rabbit monoclonal anti-MLC2 | Cell Signaling | Cat# 1678505S | IF_1:40 |
| Antibody | Goat polyclonal anti-mouse STAR 635P | Abberior GmbH | Cat# 2-0002-007-5 | IF_1:200 |
| Antibody | Goat polyclonal anti-rabbit STAR 635P | Abberior GmbH | Cat# 2-0012-007-2 | IF_1:200 |
| Antibody | Goat polyclonal anti-mouse STAR 580 | Abberior GmbH | Cat# 2-0002-005-1, RRID:AB_2620153 | IF_1:200 |
| Antibody | Goat polyclonal anti-mouse STAR 580 | Abberior GmbH | Cat# 2-0012-005-8 | IF_1:200 |
| Antibody | Goat polyclonal anti- mouse Alexa Fluor 532 | Thermo Fischer Scientific | Cat# A-11002, RRID:AB_2534070 | IF_1:200 |
| Antibody | Donkey polyclonal anti- rabbit Alexa Fluor 647 | Jackson ImmunoResearch | Cat# 711-605-152 | IF_1:1000 STORM_ 1:200 |
| Chemical compound, drug | Blebbistatin | Sigma-Aldrich | Cat# B0560 | 3 µM |
| Chemical compound, drug | ML-7 | Sigma-Aldrich | Cat# I2764 | 4 µM |
| Chemical compound, drug | Myovin1 | Calbiochem | Cat# 475984 | 4 µM |
| Chemical compound, drug | Calyculin A | Sigma-Aldrich | Cat# C5552 | 5 nM |
| Chemical compound, drug | Dimethyl sulfoxide (DMSO) | VWR International | Cat# A3672.0050 | N/A |
| Chemical compound, drug | Phalloidin 635P | Abberior GmbH | Cat# 2-0205-002-5 | 0.33 µM |
| Cell line | CAD (mouse) | ECACC through Sigma-Aldrich | Cat# 08100805, RRID:CVCL_0199 | N/A |
| Cell line | PC-12 (rat) | ATCC | Cat# CTL-1721, RRID:CVCL_F659 | N/A |
| Cell line | SH-SY5Y (human) | ATCC | Cat# CRL-2266, RRID:CVCL_0019 | N/A |
| Recombinant DNA reagent | pEGFP-C1 | Addgene | N/A | N/A |
| Recombinant DNA reagent | pLKO.1 | Addgene | N/A | N/A |
| Recombinant DNA reagent | shRNA NMIIA | Rai et al., 2017 | N/A | 5’GCGATACTACTCAGGGCTTAT3’ |
| Recombinant DNA reagent | shRNA NMIIB | This paper | N/A | 5‘GCCAACATTGAAACATACCT3’ |
| Recombinant DNA reagent | shRNA NMIIC | This paper | N/A | 5’CCGGGCTCATTTATACCTACT3’ |
| Recombinant DNA reagent | shRNA RLC | Wang et al., 2008 | N/A | 5′GCACGGAGCGAAAGACAAA3′ |
| Recombinant DNA reagent | shRNA MYPT1 | This paper | N/A | 5′GAGCCTTGATCAGAGTTATAAC3′ |
| Recombinant DNA reagent | shRNA α-adducin | Sigma-Aldrich | Cat# TRCN0000108809 | 5’GCAGAAGAAGAGGGTGTCTAT3’ |
| Recombinant DNA reagent | Human mutated ShRNA-resistant NMIIA | This paper | N/A | Vectorbuilder Page 20_line 374–376 |
| Recombinant DNA reagent | Human mutated ShRNA-resistant NMIIB | This paper | N/A | Vectorbuilder Page 20_line 374–376 |
| Recombinant DNA reagent | Human ShRNA-resistant RLC | Addgene | Cat #35680 | N/A |
| Recombinant DNA reagent | Human mutated ShRNA-resistant MYPT1 | This paper | N/A | Vectorbuilder Page 20_line 374–376 |
| Recombinant DNA reagent | CMV-eGFP-NMIIA | Addgene | Cat #11347 | N/A |
| Recombinant DNA reagent | CMV-NMIIA-mApple | John Hammer, NHLBI, Bethesda, USA | N/A | N/A |
| Commercial assay or kit | NZY Total RNA Isolation Kit | NZY Tech | Cat# MB13402 | N/A |
| Commercial assay or kit | SuperScript First-Strand Synthesis System for RT-PCR | Thermo Fisher Scientific | Cat# 11904018 | N/A |
| Sequence-based reagent | NMIIC sense primer | This paper | N/A | 5’CCTGGCTGAGTTCTCCTCAC3’ |
| Sequence-based reagent | NMIIC antisense primer | This paper | N/A | 5’TGCTTCTGCTCCATCATCTG3’ |
| Sequence-based reagent | RLC sense primer | This paper | N/A | 5’CCTTTGCCTGCTTTGATGAG3’ |
| Sequence-based reagent | RLC antisense primer | This paper | N/A | 5’GTGACTGGGATGGGGTGTAG3’ |
| Sequence-based reagent | MYPT1 sense primer | This paper | N/A | 5’AAGGGAACGAAGAGCTCTAGAAA3' |
| Sequence-based reagent | MYPT1 antisense primer | This paper | N/A | 5’TGACAGTCTCCAGGGGTTCT3’ |
| Sequence-based reagent | β-actin sense primer | This paper | N/A | 5’ACCACACCTTCTACAATGAG3’ |
| Sequence-based reagent | β-actin antisense primer | This paper | N/A | 5’TAGCACAGCCTGGATAGC3’ |
| Sequence-based reagent | GADPH sense primer | This paper | N/A | 5’AGGCACCAAGATACTTACAAAAAC3’ |
| Sequence-based reagent | GADPH antisense primer | This paper | N/A | 5’TGTATTGTAACCAGTCATCAGCA3' |
| Software, algorithm | MATLAB R2018a | MATLAB | RRID:SCR_001622 | N/A |
| Software, algorithm | Fiji | NIH | SRRID:SCR_002285 | N/A |
| Software, algorithm | Leica LAS X software | Leica | RRID:SCR_013673 | N/A |
| Software, algorithm | µSpikeHunter software | Heiney et al., 2019 | N/A | N/A |
| Software, algorithm | GraphPad Prism | GraphPad | RRID:SCR_002798 | N/A |
| Software, algorithm | Huygens Software | Scientific Volume Imaging | RRID:SCR_014237 | N/A |





