Wnt/β-catenin signaling is an evolutionarily conserved determinant of chordate dorsal organizer
Figures
Asymmetric activity of the Wnt/β-catenin signaling pathway during the cleavage and early gastrula stages of amphioxus embryo.
(A–D’), (F–F’), (H–H’), (K’) and (M–M’) Immunostaining of β-catenin with specific anti-amphioxus β-catenin antibody produced in the mouse. (J) Bright-field of the embryo shown with the signals in (J’) and (J’’). (J’) Immunostaining with anti-human β-catenin antibody produced in the rabbit. (J’’) Immunostaining of the same embryo as in (J’) with specific anti-amphioxus Goosecoid antibody produced in the mouse. (K) Bright-field of the embryo shown with the signals in (K’). (E), (G), (I), (L) and (N) In-situ hybridization of the Wnt/β-catenin signaling target gene Axin. (A–A’) β-catenin is not present in the nuclei of the embryos at 8-cell stage. Embryos are shown from the vegetal view. (B–C’) Nuclear β-catenin is accumulated in the vegetal half of the embryo at 32-cell stage. Arrowheads show polar body, which demarcates the animal pole. (B–B’) side view, (C) vegetal view, (C’) animal view. Inserts in (C) and (C’) demonstrate individual optical sections from the vegetal and animal poles, respectively. (D–D’) Individual optical section demonstrates that β-catenin is present in the vegetal nuclei of a 64-cell stage embryo. (E) The expression of the Wnt/β-catenin signaling target gene Axin is not ubiquitous in the 64-cell stage embryo. (F–F’) Asymmetrical distribution of β-catenin and asymmetrical expression of Axin (G) in the blastula of amphioxus embryo. (H–H’) Nuclear β-catenin and (I) the expression of Axin are concentrated to the presumptive dorsal endomesoderm and dorsal blastopore lip at the onset of gastrulation. (J) The dorsal part of blastopore of Branchiostoma laceoltum has a typical morphology and is different from the ventral part of blastopore at early gastrula stage. Dashed lines demarcate presumptive dorsal endomesoderm. (J’) β-catenin is located in the dorsal part of blastopore at early gastrula stage and co-expressed with Goosecoid protein (J’’). (K–K’) The signal of β-catenin, which is labeled with specific anti-amphioxus β-catenin antibody, is located in the dorsal endomesoderm. (L) Axin is expressed at a higher level in the dorsal endomesoderm at early gastrula stage. (M–M’) Enrichment of nuclear β-catenin and (N) Axin mRNA in the dorsal endomesoderm of the embryos at early cap-shaped mid-gastrula stage. (H–H’), (I), (M–N) Embryos are shown in the lateral view. (J–L) Blastopore view of the embryos. An, animal; Veg, vegetal; Dbl, dorsal blastopore lip; Bp, blastopore. Scale bar is 100 µM.
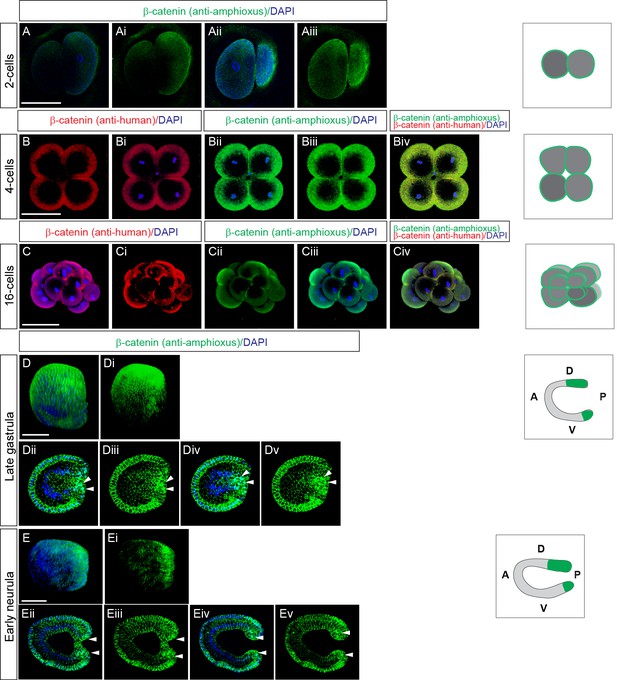
Immunodetection of nuclear β-catenin at the cleavage stage, late gastrula and late neurula stages Immunostaining of amphioxus embryos with antibodies against amphioxus.
(A-Aiii, B–Bi, C–Ci, D–Ev) or human ß-catenin (Bii-Biv). Overlay of double staining in Biv and Civ. Embryos are at the two-cell stage in A-Aiii, four-cell stage in B-Biv, 16-cell stage in C-Civ, late gastrula stage in D-Dv, and early neurula stage in E-Ev. 3D-view of the whole embryo in C-Civ, D-Di and E-Ei. Individual z-stacks in (A-Biv, Dii-Dv and Eii-Ev). Anterior to the left in D-Ev. Panels in the column on the right side represent diagrams of the embryos for a given developmental stage summarizing the expression of β-catenin. Arrowheads denote the nuclear localization of ß-catenin. D, dorsal; V, ventral; A, anterior; P, posterior. Scale bar is 100 µM.
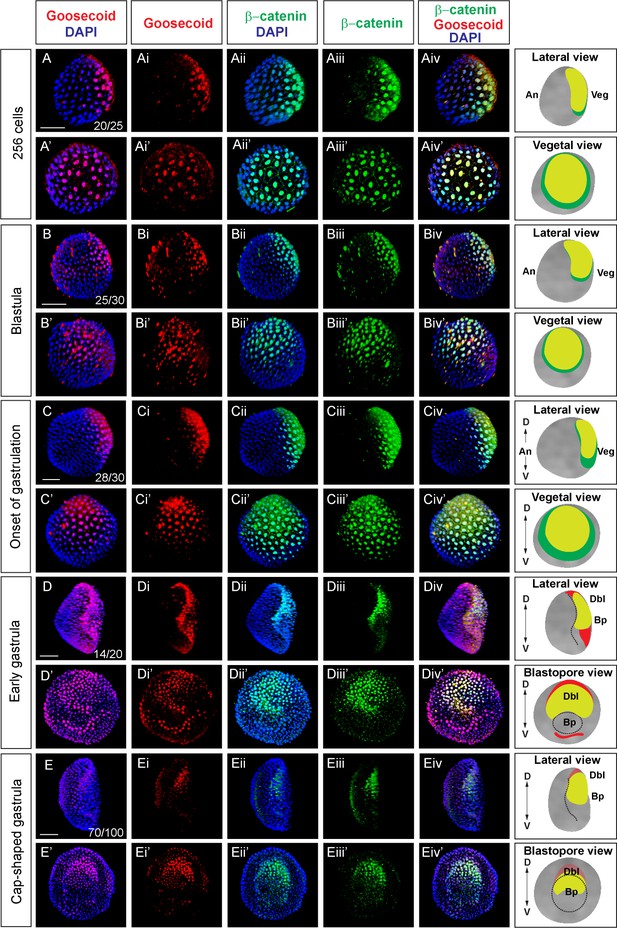
Nuclear β-catenin is colocalized with dorsal-specific protein Goosecoid during early development in amphioxus (A-Eiv’) Double immunostaining of Gooosecoid and β-catenin in Branchiostoma lanceolatum.
Lateral view of the embryos in (A-Aiv), (B-Biv), (C-Civ), (D-Div), (E-Eeiv). The embryos are shown from the vegetal pole view in (A’-Aiv’), (B’-Biv’), (C’-Civ’). Blastopore view of the embryos in (D’-Div’), (E’-Eiv’). Panels in the column on the right side represent diagrams of the embryos for a given developmental stage summarizing the expression of Goosecoid and β-catenin. D, dorsal; V, ventral; An, animal; Veg, vegetal; Dbl, dorsal blastopore lip; Bp, blastopore. Scale bar is 50 µM.
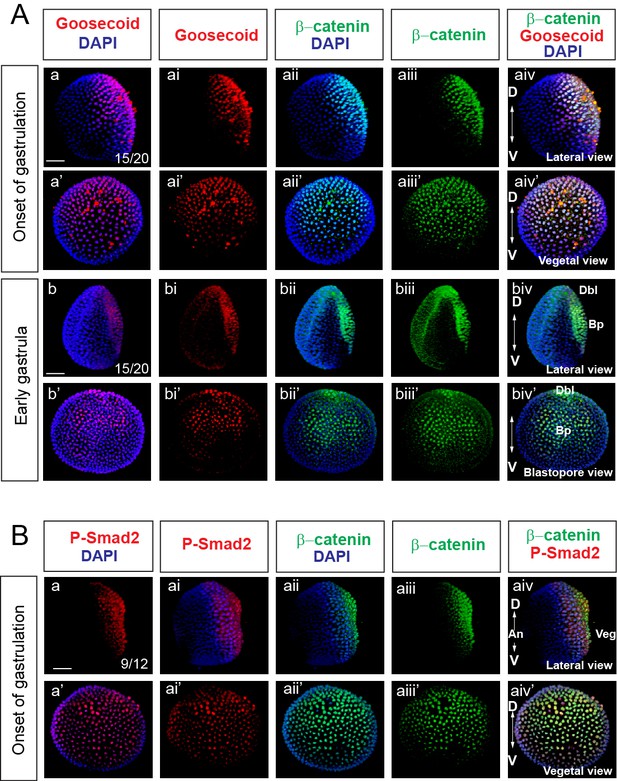
Nuclear β-catenin is colocalized with dorsal-specific protein Goosecoid and P-Smad2 during early development of Branchiostoma floridae.
(Aa-Abiv’) Double immunostaining of Gooosecoid and β-catenin. (Ba-Baiv’) Double immunostaining of P-Smad2 and β-catenin. Lateral view of embryos in (Aa-Aaiv), (Ab-Abiv), and (Ba-Baiv). The embryos are shown from the vegetal pole view in (Aa’-Aaiv’) and (Ba’-Baiv’). Blastopore view of the embryos in (Ab’-Abiv’). D, dorsal; V, ventral; An, animal; Veg, vegetal; Dbl, dorsal blastopore lip; Bp, blastopore. Scale bar is 50 µM.
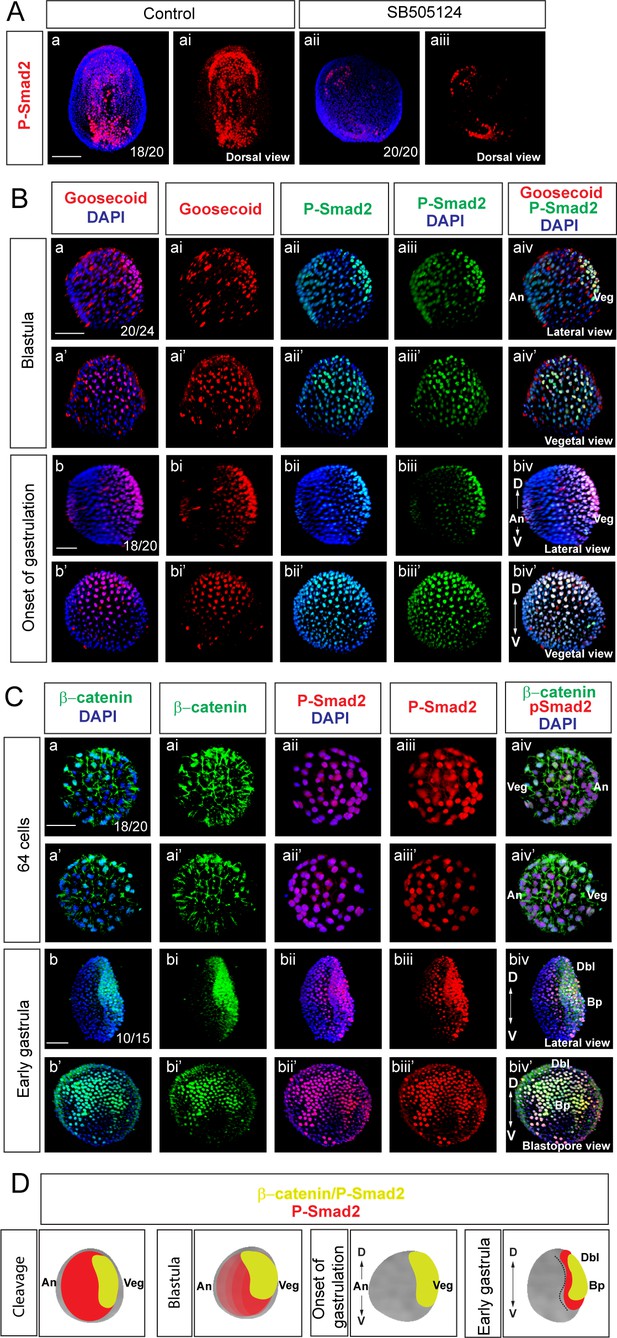
Wnt/β-catenin and Nodal signaling activities overlap during early amphioxus development.
(A) Commercial anti-phosphoSmad2 antibodies specifically label phospho-Smad2 (P-Smad2) in amphioxus. (B) Double immunostaining of Gooosecoid and P-Smad2. (C) Double immunostaining of P-Smad2 and β-catenin during early development of Branchiostoma lanceolatum. (D) Schematic diagrams of the embryos summarizing the expression of P-Smad2 and β-catenin. (Aa-Aai) Dorsal view of early amphioxus neurula demonstrates stronger signal of P-Smad2 in the left endoderm and mesoderm. (Aa-Aaiii) Treatment with P-Smad2-mediated signaling pathway inhibitor SB505124 downregulates the expression of P-Smad2. (B) Goosecoid and P-Smad2 are co-expressed at the blastula (Ba-Baiv’) and onset of gastrulation (Bb-Bbiv’) stages. Red dots that do not correspond to nuclei represent nonspecific random sticking of antibodies on the surface of the embryos. (C) The expression of nuclear β-catenin and P-Smad2 at 64-cell stage (Ca-Caiv’) and early gastrula stage (Cb-Cbiv’). (A) Dorsal view of the embryo. (Ba-Baiv), (Bb-Bbiv), (Cb-Cbiv) and (Da-Daiv) Lateral view of the embryos. The embryos are shown from the view of the vegetal pole in (Ba’-Baiv’), (Bb’-Bbiv’) and (Da’-Daiv’). Blastopore view in (Cb’-Cbiv’). (Ca-Caiv) Two sides of the view with animal pole to the right in (Ca-Caiv) and to the left in (Ca’-Caiv’). D, dorsal; V, ventral; An, animal; Veg, vegetal; Dbl, dorsal blastopore lip; Bp, blastopore. Scale bar is 50 µM.
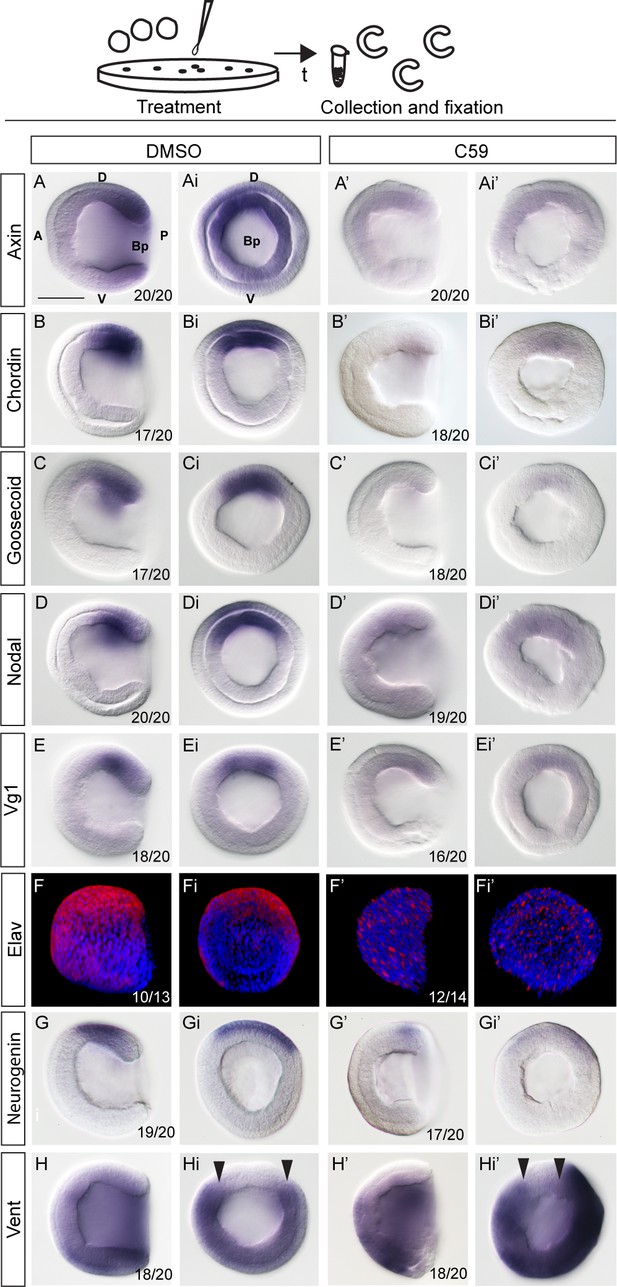
Treatment of amphioxus embryos with the inhibitors of Wnt/β-catenin signaling at one-cell stage results in downregulation of the dorsal-specific genes at the gastrula stage.
The expression of Wnt/β-catenin target Axin, dorsal-specific genes Chordin and Goosecoid, dorsal-specific genes encoding signaling molecules Nodal and Vg1, dorsal-specific neural markers Neurogenin and Elav, ventral-specific gene Vent1 in the control DMSO-treated embryos and embryos treated continuously with Wnt/β-catenin signaling inhibitor C59 after fertilization at one-cell stage. The embryos are at mid-gastrula stage. Lateral view in (A–H and A’–H’) and blastopore view in (Ai-Hi and Ai’-Hi’). Arrowheads in Hi and Hi’ mark the extent of dorsal Vent expression. D, dorsal; V, ventral; A, anterior; P, posterior; Bp, blastopore. Scale bar is 50 µM.
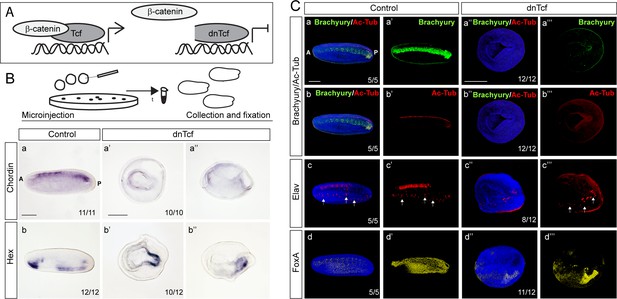
Inhibition of Wnt/ß-catenin signaling by injection of dominant-negative Tcf (dnTcf) mRNA leads to impairment of the axis development in amphioxus neurula.
(A) The scheme illustrates the mechanism of Wnt/ß-catenin signaling inhibition by dnTcf. (B) mRNA expression of Chordin and Hex genes in the control embryos and embryos injected with dnTcf. (C) Protein expression of Brachyury, acetylated tubulin (Ac-Tub), Elav and FoxA in the control embryos and embryos injected with dnTcf. In (Ca), (Cb) and (Ca’’), (Cb’’) the same embryos with double expression of Brachyury and Ac-Tub are shown. White arrows demarcate isolated sensory cells. In (Ba), (Bb), (Ca-a’), (Cb-b’), (Cc-c’) and (Cd-d’) the embryos are shown in lateral view. In (Ba’), (Bb’), (Ca’’-a’’’), (Cb’’-b’’’), (Cc’’-c’’’) and (Cd’’-d’’’) the embryos are shown in blastopore view. Anterior to the left. A, anterior; P, posterior. Scale bar is 100 µm.
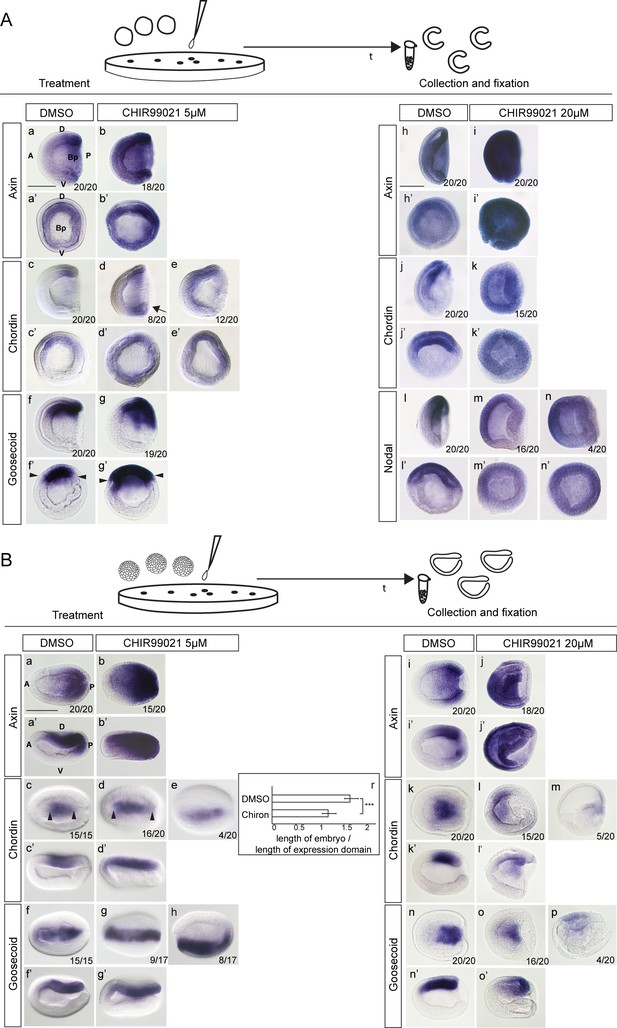
Ectopic activation of Wnt/β-catenin signaling at one-cell stage but not at the blastula stage results in ectopic expression of dorsal-specific genes (Aa-g’).
The expression of Axin, Chordin and Goosecoid in the control embryos and embryos treated with 5 µM of CHIR99021 at one-cell stage. Arrow in Ad marks ventral expansion of Chordin expression. Arrowheads in Af’ and Ag’ mark the extent of Goosecoid expression. (Ah-n’) The expression of Axin, Chordin and Nodal in the control embryos and embryos treated with 20 µM of CHIR99021 at one-cell stage. (Ba-g’) The expression of Axin, Chordin and Goosecoid in the control embryos and embryos treated with 5 µM of CHIR99021 at the blastula stage. Arrowheads in Bc and Bd mark the extent of Chordin expression. (Bi-o’) The expression of Axin, Chordin and Goosecoid in the control embryos and embryos treated with 20 µM of CHIR99021 at the blastula stage. (Br) Chart illustrating significant posterior and anterior extension of the Chordin expression domain in the treated embryos (p<0.01, paired t-test). (Aa, Ab, Ac, Ad, Af, Ag, Ah, Ai, Aj, Ak, Al, Am, An, Ba’, Bb’, Bc’, Bd’, Bf’, Bg’, Bi’, Bj’, Bk’, Bl’, Bn’, Bo’) The embryos are shown in lateral view. (Aa’, Ab’, Ac’, Ad’, Ae’, Af’, Ag’, Ah’, Ai’, Aj’, Ak’, Al’, Am’, An’) The embryos are shown in blastopore view. (Ba, Bb, Bc, bd, Be, Bf, Bg, Bh, Bi, Bj, Bk, Bl, Bm, Bn, Bo, Bp) The embryos are shown in dorsal view. D, dorsal; V, ventral; A, anterior; P, posterior; Bp, blastopore. Scale bar is 100 µM.
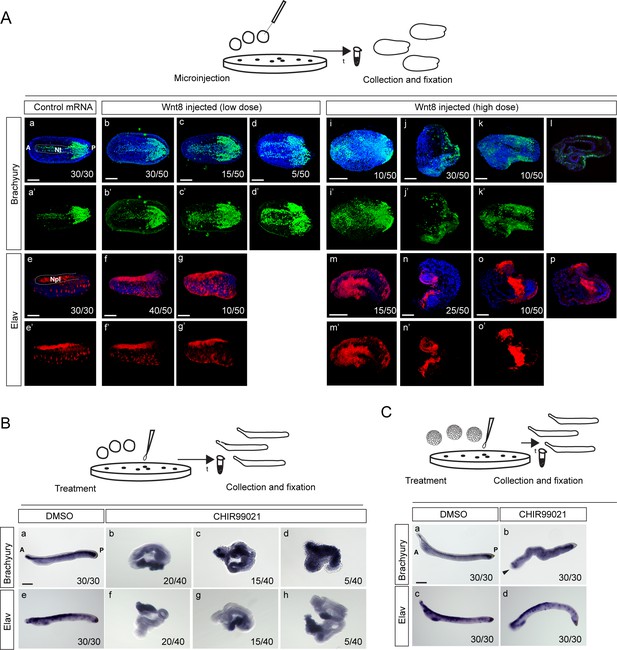
Ectopic activation of Wnt/β-catenin signaling pathway by microinjection of Wnt8 ligand mRNA or treatment with CHIR99021 induces ectopic expression of dorsal axis-specific markers in the amphioxus embryos.
(A) Expression of Brachyury and Elav proteins at the mid-neurula of amphioxus embryos injected with Wnt8 mRNA at a low dose (3 ng/µl) or high dose (7 ng/µl). (B–C) Expression of Brachyury and Elav mRNA at the larval stage of amphioxus embryos treated with 10 µM of CHIR99021 at one-cell stage (B) or at blastula stage (C). Arrowhead in Cb marks the truncation of the head. The embryos are shown in dorsal view in (Aa-Ad’), (Ai-i’) and (Am-m’). Lateral view in (Ae-g’), (Ba), (Be) and (Ca-d). (Al) and (Ap) represent the individual z-stacks of (Ak) and (Ao), respectively. A, anterior; P, posterior; Nt, notochord; Npl, neural plate. Scale bar is 100 µM.
Wide-field image of amphioxus embryos injected with tdTomato mRNA or tdTomato with Wnt8 mRNA.
(A) The embryos injected with mRNA of tdTomato alone. (B) and (C) The embryos injected with mRNA of tdTomato and Wnt8.
Ectopic activation of Wnt/β-catenin signaling pathway by microinjection of Wnt11 ligand mRNA induces formation of a double axis in the amphioxus embryos.
Double immunostaining detecting the protein expression of Brachyury and Ac-Tub in the embryos injected with a low (A) or high (B) dose of Wnt11 mRNA (50 ng/µl or 250 ng/µl, respectively). The insert in (Be) demonstrates the enlarged region from the ventral part of the embryo. Embryos are at early larval (Aa-d’’) or the mid-neurula stage (Ba-e’’). The embryos are shown in lateral view in (Aa-a’’), (Ad-d’’), (Ba-a’’) and (Be-e’’). The embryos are shown in dorsal view in (Ab-c’’) and (Bb-d’’). Arrowheads mark the twinning of notochord and neural tube following the injection of Wnt11 mRNA. A, anterior; P, posterior. Scale bar is 100 µM.
Wnt/β-catenin and Nodal signaling pathways co-act during early development to promote dorsal cell fate in the amphioxus embryo.
(A) Expression of Nodal (Aa-av’’) and Goosecoid (Ab-bv’’) in the control embryos, which were treated with DMSO (Aa-a’’ and Ab-b’’), and the embryo treated with Wnt signaling inhibitor C59 (Aai-ai’’ and Abi-bi’’), Nodal signaling inhibitor SB505124 (Aaii-aii’’ and Abii-bii’’) separately or simultaneously (Aaiii-aiii’’ and Abiii-biii’’), Wnt/β-catenin signaling activator CHIR99021 (Aaiv-aiv’’ and Abiv-biv’’) separately or simultaneously with SB505124 (Aav-av’’ and Abv-bv’’) at one-cell stage. The embryos are at the mid-gastrula stage. (B) Expression of Brachyury (Ba-av’) and Elav (Bb-bv’) in the control embryos, which were treated with DMSO (Ba-a’ and Bb-b’), and the embryo treated with Wnt signaling inhibitor C59 (Bai-ai’ and Bbi-bi’), Nodal signaling inhibitor SB505124 (Baii-aii’ and Bbii-bii’) separately or simultaneously (Baiii-aiii’ and Bbiii-Biii’), Wnt/β-catenin signaling activator CHIR99021 (Baiv-aiv’ and Bbiv-biv’) separately or simultaneously with SB505124 (Bav-av’ and Bbv-bv’) at one-cell stage. The embryos are at the late neurula stage. Arrowheads in Baiv’ and Bbiv’ mark weak ectopic expression of Brachyury and Elav, respectively. (Aa-av, Ab-bv) The embryos are shown from blastopore view. (Aa’-av’, Ab’-bv’, Ba-av, Bb-bv) The embryos are shown in lateral view. (Ba’-av’, Bb’-bv’) The embryos are shown in dorsal view. D, dorsal; V, ventral; A, anterior; P, posterior; Bp, blastopore. Scale bar is 50 µM.
Assaying the responsiveness of cis-regulatory elements of amphioxus Goosecoid, Nodal and Vg1 to Wnt/β-catenin.
Luciferase reporter plasmids containing cis-regulatory elements of amphioxus Goosecoid (A), Nodal and Vg1(B) were cotransfected with expression plasmid encoding a stabilized form of β-catenin (β-cateninΔN) into 293 T cells. Graph values represent the average of triplicates+/-standard deviation. p=0.03 (Student’s t-test).
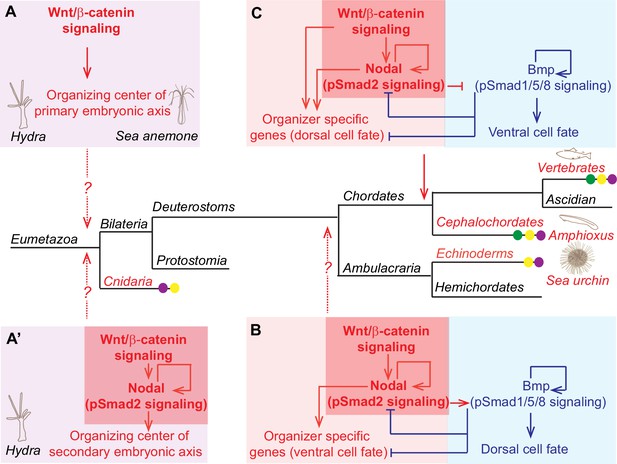
The role of Wnt/β-catenin signaling in the establishment of axial patterning in Metazoa.
(A) Wnt/β-catenin signaling determines the organizing center of the primary body axis formation in cnidarian Hydra and Nematostella. (A’) Wnt/β-catenin upstream of autoactivating Nodal signaling acts to induce a secondary axis formation in cnidarian Hydra. (B) Wnt/β-catenin, Nodal and Bmp signaling specify the secondary ventral/dorsal (also called oral/aboral axis) in sea urchin. Note that according to the hypothesis of dorso-ventral inversion occurred in chordate lineage (Arendt and Nübler-Jung, 1994) the establishment of ventral/dorsal axis in sea urchin corresponds to the establishment DV axis in chordates. (C) Wnt/β-catenin, Nodal and Bmp signaling specify the secondary DV axis in chordates. (A’), (B) and (C) Core-signaling cassette involving Wnt/β-catenin upstream of autoactivating Nodal signaling (highlighted by the red boxes) acts to induce a secondary axis formation in cnidarian Hydra (A’), echinoderms (B) and chordates (C). Cooption of Wnt/β-catenin signaling (violet dot), nodal signaling (yellow dot), and opposing regulatory mechanism between Nodal and Bmp signaling (green dot) for the establishment of organizing centers of embryonic axes is indicated. Mechanisms involved in left-right axial patterning are not shown. For references see text.
Tables
| Reagent type (species) or resource | Designation | Source or reference | Identifiers | Additional information |
|---|---|---|---|---|
| Strain, strain background (Branchiostoma lanceolatum) | Wild type | collected in Argeles-sur-mer, France | NCBITaxon: 7740 | |
| Strain, strain background (Branchiostoma floridae) | Wild type | Laboratory cultures, Institute of Molecular Genetics | NCBITaxon: 7739 | |
| Chemical compound, drug | CHIR99021 | SelleckChem | S1263 | |
| Chemical compound, drug | C59 | Xcess Bioscience Inc | M-60005–2 s | |
| Chemical compound, drug | SB505124 | Sigma | S4696 | |
| Antibody | Anti-β-Catenin, produced in rabbit | Sigma | C2206 | (1:500) |
| Antibody | Anti-Smad2(phospho S255), produced in rabbit | Abcam | ab188334 | (1:20,000) |
| Antibody | Anti-β-Catenin, produced in mouse | Bozzo et al., 2017 | (1:200) | |
| Antibody | Anti-FoxA, produced in mouse | Bozzo et al., 2017 | (1:200) | |
| Antibody | Anti-Goosecoid, produced in mouse | This paper | (1:200) | |
| antibody | Anti-Brachyury, produced in mouse | This paper | (1:200) | |
| Antibody | Anti-Elav, produced in mouse | This paper | (1:200) | |
| Sequence-based reagent | Goosecoid_fw | This paper | GCTTCGGATCCGAAGTCACCGAGCGCACGACC | |
| Sequence-based reagent | Goosecoid_rev | This paper | GGTCTAAGCTTACTGTCCGTCGCTAGGCGT | |
| Sequence-based reagent | Brachyury_fw | This paper | GCTGTGGATCCGGAATGGAAGATTTGCAAGAT | |
| Sequence-based reagent | Brachyury_rev | This paper | CCTGTAAGCTTAGAGCGATGGTGGAGTCAT | |
| Sequence-based reagent | Elav_fw | This paper | GTGCTGGATCCGGCTCGCCCGACGGACGCAC | |
| Sequence-based reagent | Elav_rev | This paper | GTGCTAAGCTTACAGTCCACTCACGTACAAGTT | |
| Sequence-based reagent | Vg1_fw | This paper | GCGGCCACTGCTTTTACTTT | |
| Sequence-based reagent | Vg1_rev | This paper | GTGTAGTAGTGGACAAGACTGAGGG | |
| Sequence-based reagent | Nodal_fw | This paper | TAACGTGAGGCCAGGTGATG | |
| Sequence-based reagent | Nodal_rev | This paper | CGGAGTTTGCGTGTTCGACT | |
| Sequence-based reagent | Wnt8_fw | This paper | GGAATTCGCACGATGCTCGCACAGCTC | |
| Sequence-based reagent | Wnt8_rev | This paper | GGAATTCCTAGTTGTTTCCCCTGTTTCTTC | |
| Sequence-based reagent | Wnt11_fw | This paper | CTCTTCTTCAACCTGCGACTG | |
| Sequence-based reagent | Wnt11_rev | This paper | TTTTCTCTGGCTTTCCCTTGA | |
| Sequence-based reagent | dnTcf_fw | This paper | GCGATCTAGAGGTTCCAAGCCACCGTTGCAA | |
| Sequence-based reagent | dnTcf_rev | This paper | GCTCAAGCTTTCACGTGTTGGGCGTTGACTT | |
| Software, algorithm | Fiji | Schindelin et al., 2012 | ||
| Software, algorithm | Leica Application Suite X (LAS X) | Leica |
Additional files
-
Supplementary file 1
Chemicals used in this study to affect the signaling pathways.
- https://cdn.elifesciences.org/articles/56817/elife-56817-supp1-v3.docx
-
Transparent reporting form
- https://cdn.elifesciences.org/articles/56817/elife-56817-transrepform-v3.docx