Sequential perturbations to mouse corticogenesis following in utero maternal immune activation
Figures
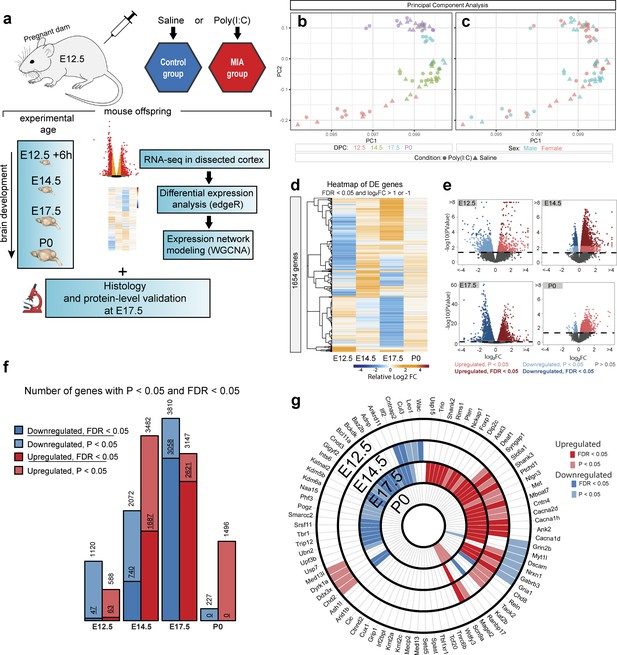
Differential expression across embryonic brain development following maternal immune activation via poly(I:C) injection at E12.5.
(a) Schematic representation of MIA model and study design. E: embryonic day, P: postnatal day, IHC: immunohistochemistry, WB: western blot. Poly(I:C) was injected at E12.5. Samples for RNA-seq were collected at E12.5 + 6 hr, E14.5, E17.5, and at birth (P0). IHC and WB analysis were conducted on a separate animal cohort at E17.5 and P0. (b, c) Principal component analysis (PCA) of RNA-seq data indicates that developmental age accounts for the majority of variance across samples. Age (DPC) or sex are represented as colored symbols in (b) and (c) respectively, and poly(I:C) or saline treatment indicated by circles or triangles, respectively (d) Heatmap representing relative gene expression changes between control and MIA samples across time-points. Hierarchical clustering by relative fold changes shows stage-specific differential expression signatures (DE genes shown have FDR < 0.05 and log2 fold change (log2FC)>1 or < −1). (e) Volcano plots of DE effect size and significance show stage-specific differences in number of DE genes and DE effect size, with the strongest dysregulation at E17.5. Colors represent directionality and statistical significance. (f) Numbers of upregulated and downregulated DE genes at FDR < 0.05 and p<0.05 thresholds again show varying DE genes numbers with a peak of dysregulated genes at E17.5. (g) Intersection of stage-specific DE genes with the 82 SFARI autism-associated mouse gene orthologs that were expressed at measurable levels in the RNA-seq data. E17.5 DE genes (FDR < 0.05) were enriched for SFARI genes (p=3.9e-04, hypergeometric test). Concentric circles represent developmental time-points. Light red, upregulated DE genes (p<0.05); dark red, upregulated DE genes (FDR < 0.05); light blue, downregulated DE genes (p<0.05); dark blue, downregulated DE genes (FDR < 0.05).
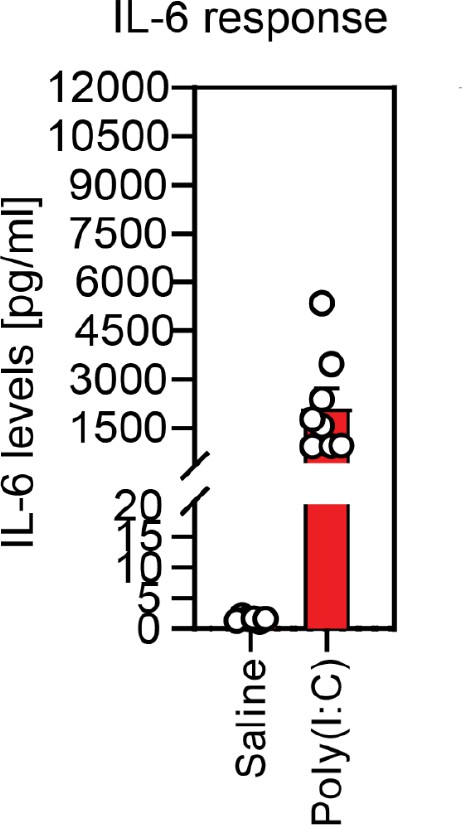
Validation of responsiveness to poly(I:C) based on dam serum IL-6 levels.
Injection of poly(I:C) at E12.5 consistently elevates serum IL-6 in dams 4 hr following injection, compared with little response in dams injected with saline.
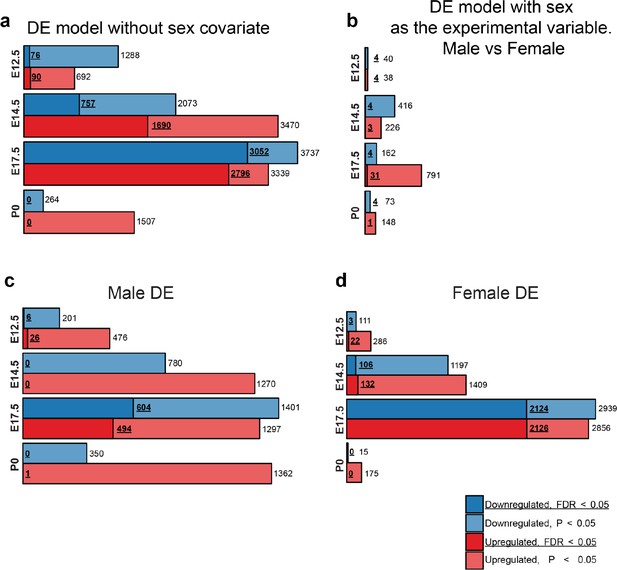
Males and females show largely concordant DE signatures following MIA.
(a) Numbers of upregulated and downregulated DE genes with p < 0.05 and FDR < 0.05, in the DE analysis lacking the sex covariate, are similar to the DE model including the sex covariate (Figure 2c). (b) Numbers of upregulated and downregulated DE genes with p < 0.05 and FDR < 0.05, in a DE analysis testing for differential expression between sexes identifies few DE genes, suggesting limited sex dimorphism between samples. MIA conditions and sequencing lanes were set as covariates for the DE model. (c, d) Numbers of upregulated and downregulated DE genes with p < 0.05 and FDR < 0.05, for the DE analysis performed on (c) males and (b) females demonstrate robust DE at E17.5 in both sexes.
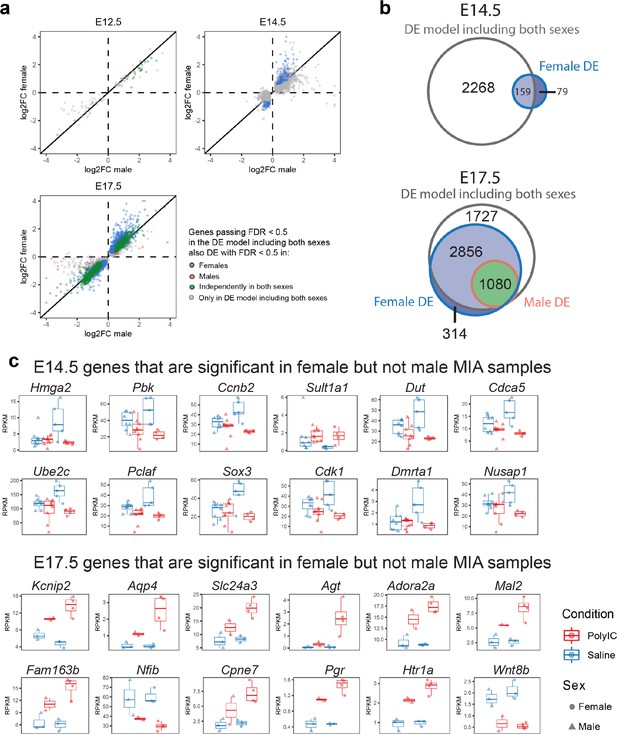
Sex-stratified comparison of DE genes and effect sizes suggest increased DE effect sizes in females.
(a) Correlation of the relative effect sizes (log2FC) for genes passing FDR < 0.05 identifies high degree of concordance between MIA responses between sexes. Genes passing FDR threshold in either or both sexes are color coded. (b) Venn diagrams illustrating overlap between DE gene sets passing FDR < 0.05 at E14.5 and E17.5, in DE models containing samples from both sexes, males, or females. The DE model comprising individuals of both sexes includes most of the DE signature of each sex. Female DE analysis identifies limited and unique DE signature. (c) Examples of E14.5 and E17.5 genes significant in females but not males. At E14.5 genes were selected to pass FDR < 0.05 and logFC > 0.5 or logFC < −0.5 in females, but not logFC > 0.5 or logFC < −0.5 in males. The gene-set selection process was analogous for E17.5 with the addition of FDR < 0.05 condition in males. After gene set filtering, twelve genes with the lowest FDR in females were plotted.
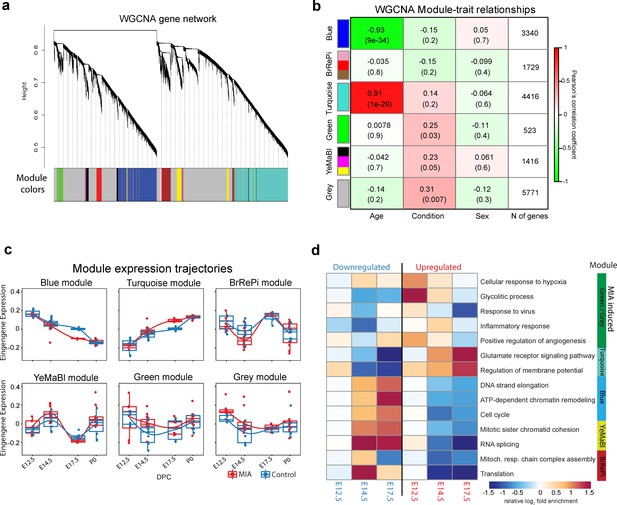
Weighted gene co-expression network analysis (WGCNA) reveals discrete gene networks perturbed by MIA across embryonic cortical neurodevelopment.
(a) WGCNA cluster dendrogram of time series samples identifies genes that were assigned into co-expression modules. Based on similarities in module gene expression, six of the original 10 modules were grouped into two larger modules, BrRePi: Brown, Red and Pink; YeMaBl: Yellow, Magenta and Black. (b) Heatmap of correlation between gene expression modules and experimental traits; age, condition (saline vs poly(I:C)), and sex. Blue and Turquoise modules are strongly associated with age; Green and Grey modules are significantly associated with MIA condition. Numerical values represent signed Pearson’s correlation coefficients, with Student asymptotic p values in brackets. Green represents negative and red represents positive correlation. Color intensity signifies the strength of the correlation. (c) Module eigengene expression for MIA and control groups plotted by time-point illustrates expression trajectories across developmental stages, capturing module- and stage-specific differences between MIA and control groups. (d) Heatmap of enrichment of DE genes for representative gene ontology biological processes (GO BP) by developmental time-point shows stage- and module-specific transcriptional pathology. Y-axis rows show enrichment for GO BP terms among module-specific DE genes (FDR < 0.05). Heatmap color scale represents relative fold enrichment for the GO terms among DE genes. P0 not shown due to insufficient DE gene numbers and absence of GO BP terms passing enrichment criteria.
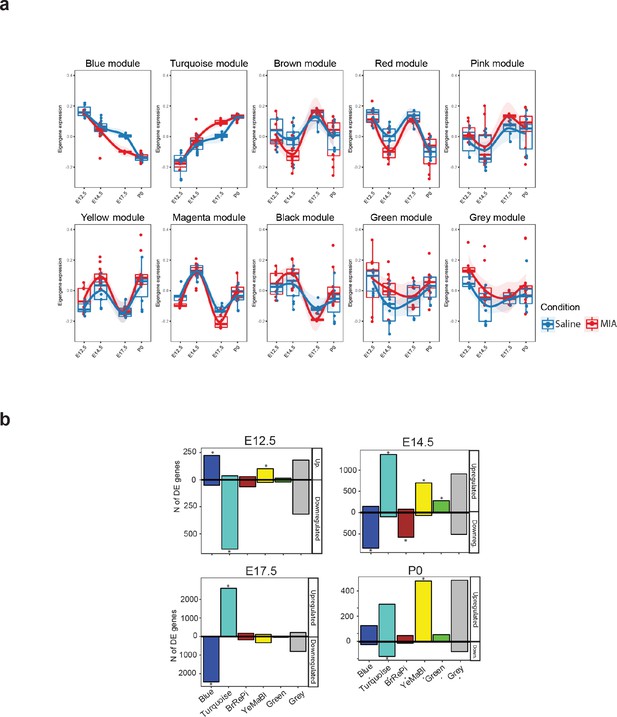
Summary of WGCNA module eigengene expression, association with experimental variables, and overlap with DE genes.
(a) Eigengene trajectories across neurodevelopment of the ten original co-expression modules. (b) DE (p < 0.05) gene set enrichment analysis in WGCNA modules across the developmental time-points of the study. Values representing the number of upregulated and downregulated genes are plotted above and below 0, respectively. Asterisks indicate statistically significant enrichment of DE genes (p < 0.05) in a module (p < 0.05; hypergeometric test).
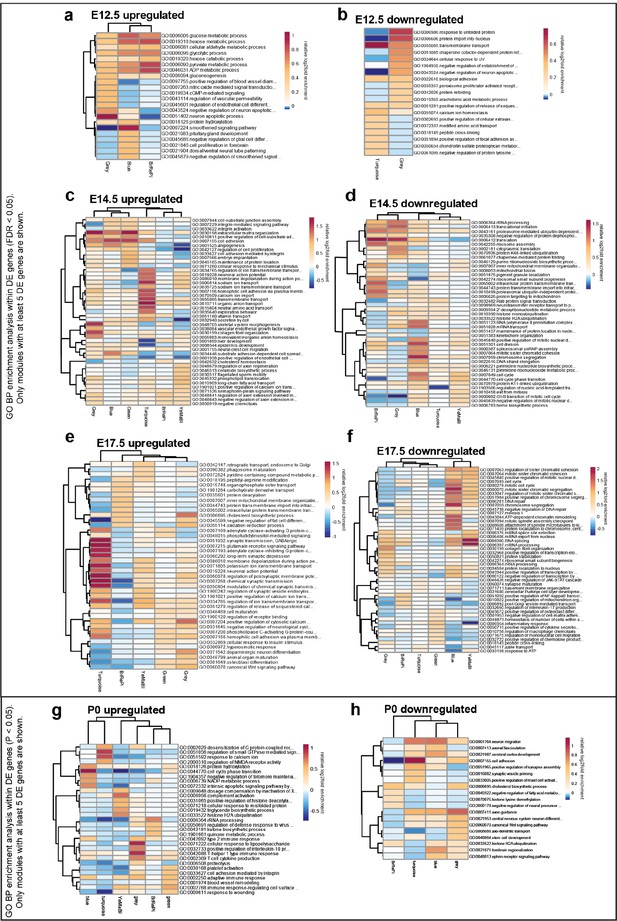
Stage-specific heatmaps of GO BP enrichment analysis of upregulated and downregulated differentially expressed genes in WGCNA modules.
(a) E12.5 upregulated DE genes (FDR < 0.05). (b) E12.5 downregulated DE genes (FDR < 0.05). (c) E14.5 upregulated DE genes (FDR < 0.05). (d) E14.5 downregulated DE genes (FDR < 0.05). (e) E17.5 upregulated DE genes (FDR < 0.05). (f) E17.5 downregulated DE genes (FDR < 0.05). (g) P0 upregulated DE genes (FDR < 0.05). (h) P0 downregulated DE genes (FDR < 0.05). Only modules with more than five DE genes (FDR < 0.05) are shown in the heatmaps.
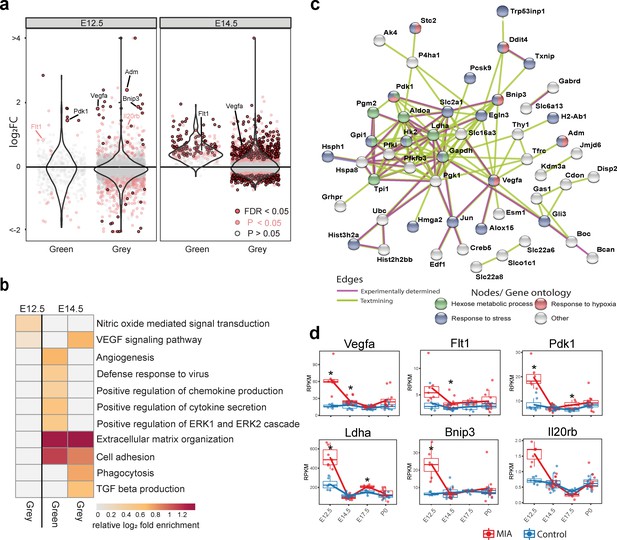
Green and Grey co-expression modules capture acute transcriptional response at 6 hr and 2 days following MIA exposure.
(a) Violin plots visualize distribution of log2 fold changes of Green and Grey module gene expression between control and MIA animals. At E12.5, an initial set of genes in the Green and Grey modules are induced and DE in MIA samples. By E14.5, generalized module expression exhibits induction in the MIA samples, with particularly strong change for the Green module, where nearly all genes are upregulated. Genes with expression trajectories shown in (d) are labeled. (b) Gene set enrichment analysis of GO BP terms significantly enriched among DE genes (FDR < 0.05) at E12.5 and E14.5 in Green and Grey modules showing upregulation of angiogenesis and immune pathways. Representative enriched GO BP terms with p < 0.05 colored by enrichment; gray represents enrichment p > 0.05 (Fisher’s exact test). (c) STRING protein–protein interaction network of E12.5 DE genes (FDR < 0.05) colored by annotation to GO BP terms. There were significantly more interactions than expected by chance among these genes. (d) RPKM expression plots of genes that are associated with this network show acute DE at E12.5. Stars represent FDR < 0.05.
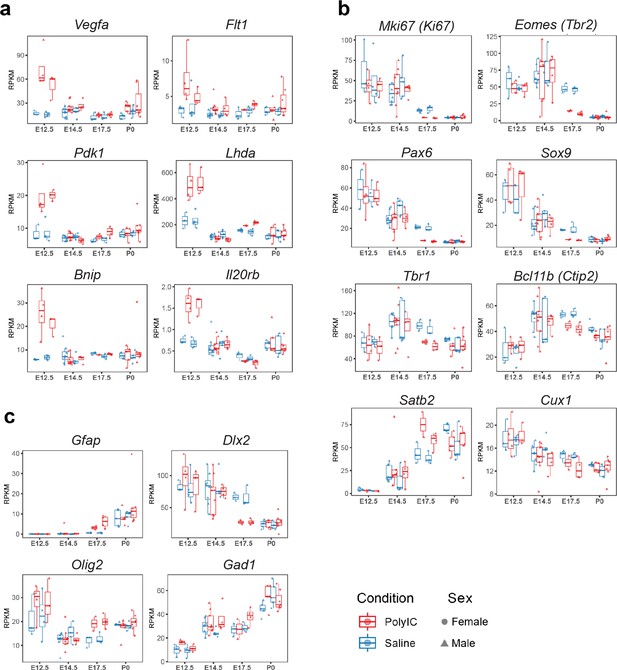
Sex-stratified RPKM gene trajectories of genes presented in the manuscript showing concordant effects between males and females.
(a) Vegfa, Flt1, Pdk1, Ldha, Bnip3, Il20rb. (b) Mki67, Eomes, Pax6, Sox9, Tbr1, Bcl11b, Satb2, Cux1. (c) Gfap, Dlx2, Olig2, Gad1.
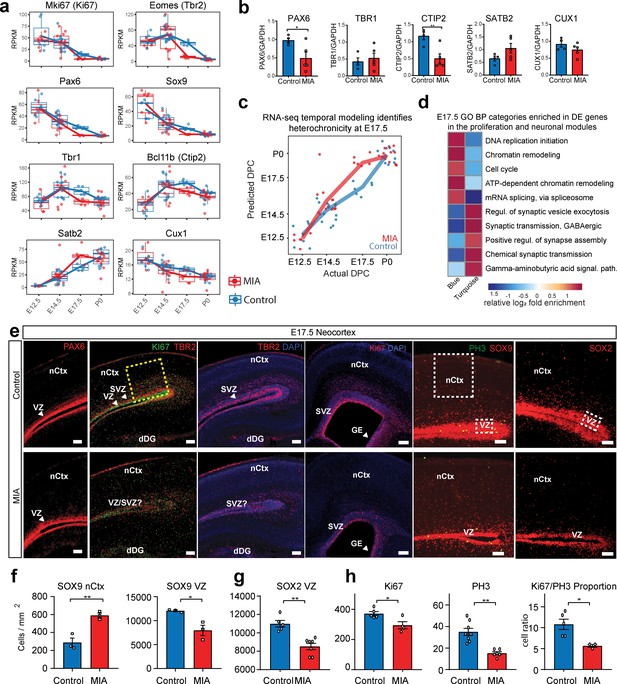
Altered cortical proliferation and lamination dynamics in fetal offspring five days after induction of MIA.
(a) RPKM trajectories of proliferative and cortical lamination markers that were DE and validated at protein level. (b) Quantified protein levels from WB analysis validated DE changes at E17.5. Protein expression data are relative to GAPDH expression (n = 4 control, n = 5 MIA; PAX6 p=0.039, TBR1 p=0.615, CTIP2 p=0.0090, SATB2 p=0.1160, CUX1 p=0.2112; two tailed Student’s t-test). Individual blots are in supplementary data (Figure 4—figure supplement 1a–c,h–i). (c) Temporal modeling of the RNA-seq data suggests acceleration of the neurodevelopmental program in MIA animals at E17.5 (p =4.5e-06, Student’s t-test) and similar trend at E14.5 (p = 0.07, Student’s t-test). Actual age (X-axis) vs predicted age (Y-axis) calculated by the linear model. Control samples were used for training the model. Lines connect average values, points depict individual samples and are jittered along the X-axis. (d) GO BP categories highlighting module-specific enrichment of E17.5 DE genes in the proliferation (Blue) and neuronal (Turquoise) WGCNA modules. (e) Representative images of coronal brain sections from E17.5 saline and MIA animals show reduced progenitor populations (neural stem cells: PAX6, SOX9, SOX2, intermediate progenitors: TBR2, and general proliferative markers: Ki67, PH3) and reduced active proliferation (Ki67:PH3 ratio). Staining for all cell populations is qualitatively decreased in the cortices from MIA offspring. Yellow box indicates the region of the neocortex utilized for Ki67 and Ph3 cell counts, and white boxes indicate regions of the VZ and upper layers of the nCtx utilized for SOX9 and SOX2 cell counts reported in (f-h). Scale bars = 100 µm, (labels: nCtx: neocortex, VZ: ventricular zone, SVZ: sub-ventricular zone, dDG: developing dentate gyrus, GE: ganglionic eminence). (f) SOX9 positive cell density in the VZ as well as in the upper layers of the nCtx shows reduced and ectopic SOX9+ cells in the MIA group (n = 3 per condition, SOX9 VZ *p=0.0198, SOX9 nCtx **p=0.0058, two tailed Student’s t-test) (g) SOX2 positive cell density counts show a specific reduction in the VZ zone in the MIA group (n = 5 control group, n = 7 MIA group, SOX2 VZ **p=0.0006). (h) Ki67 and PH3 cell density quantification along the entire length of the VZ confirms this reduction in proliferative cells in MIA samples (Ki67 n = 5 control group, n = 4 MIA group, Ki67 *p=0.0198; PH3 n = 8 control group, n = 6 MIA group, PH3 **p=0.0003, two tailed Student’s t-test) as well as a reduction in the Ki67/PH3 ratio (n = 5 control group, n = 3 MIA group, *p=0.0005 Chi-Squared).
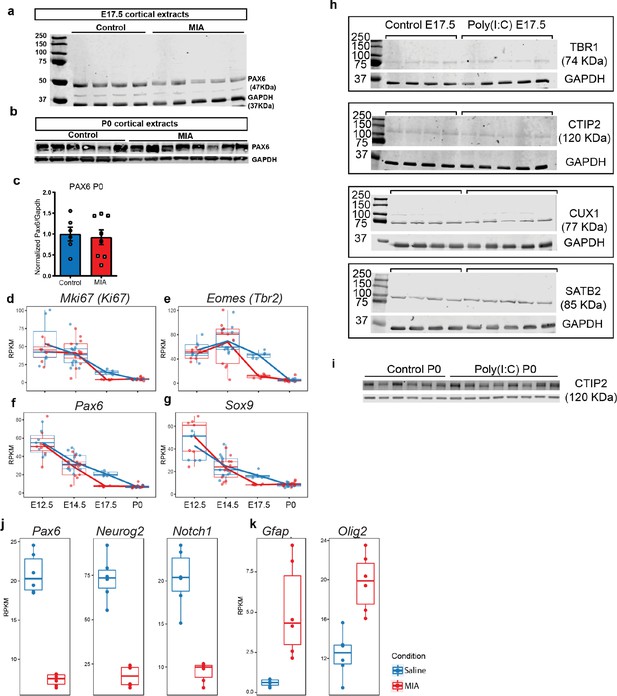
Raw western blot and RNA-seq supporting data to proliferation, lamination, and cell-specificity analyses.
(a) Western blot demonstrates reduced PAX6 protein level in tissue extracts from poly(I:C) treated animals at E17.5, (b) but not at P0. (c) Quantified PAX6 protein levels from western blot analysis at P0 (n = 6 for each control and poly(I:C) groups; p=0.766, two tailed Student’s t-test). (d–g) RNA-seq RPKM expression trajectories throughout development of (d) Ki67, (e) Tbr2, (f) Pax6, and (g) Sox9. (h) Individual western blots detecting TBR1, CTIP2, SATB2 and CUX1 from whole forebrain extracts at E17.5 and (i) CTIP2 at P0. (j) E17.5 RNA-seq RPKM expression levels of Pax6, Neurog2, Notch1, and (k) Gfap and Olig2.
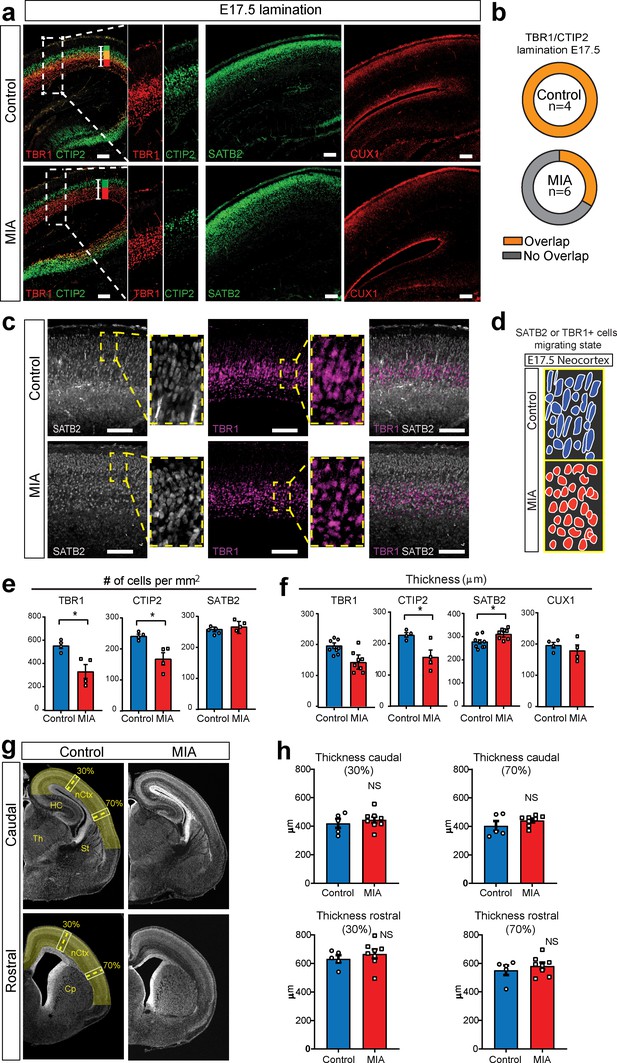
MIA impacts cortical lamination during development.
(a) Coronal fetal brain sections from E17.5 saline (control) and MIA offspring show altered lamination pattern. Immunostaining using antibodies to identify deep cortical lamination layers using CTIP2 (green), and TBR1 (red) markers as well as markers of postmitotic neurons, SATB2 and CUX1, in more superficial cortical layers in brains derived from saline (control) or MIA injected dams. Representative images are shown. CTIP2/TBR1 overlap, represented by yellow color, was generally present in control brains, compared to fully laminated deep layers in MIA brains (no overlap illustrated by adjacent green and red rectangles). Boxes indicate magnified areas used for quantification and CTIP2/TBR1 overlap analyses. Scale bars = 100 µm. (b) TBR1/CTIP2 lamination overlap occurrence is present in all control brains but absent in the majority of MIA samples at E17.5. Brains from three MIA litters and two control litters were co-stained with TBR1 and CTIP2 cortical markers (as shown in a-b) and analyzed for lamination overlap occurrence (n = 4 control, n = 6 MIA). While all control brains showed lamination overlap between TBR1/CTIP2 layers, absence of TBR1/CTIP2 lamination overlap was observed in four out of six analyzed MIA brains. (c) Representative images of SATB2 and TBR1 staining at higher resolution show a change in cell morphology consistent with a difference in migration between MIA and control groups at E17.5. (d) Schematic representation illustrates the oval-radially migrating cell body shapes in control brains and the more rounded cell body shape in the MIA brains typical of static cells after migration. (e) MIA-induced reduction in TBR1+, CTIP2+, but not in SATB2+ cell counts (n = 4–5 brains per condition, TBR1 p=0.0138; CTIP2 p=0.0119; SATB2 p=0.306). (f) Significant decrease in CTIP2 and increase in SATB2 thickness indicates relative expansion of more superficial layers in MIA brains at E17.5 (Layer-specific thickness analysis: n = 4–8 per condition, TBR1 p=0.081; CTIP2 p=0.024; SATB2 p=0.039; CUX1 p=0.430, two tailed Student’s t-test). (g) Representative caudal and rostral coronal sections of E17.5 control and poly(I:C) brains stained with DAPI show no obvious cortical dysplasia or gross anatomy alterations. Yellow shading represents neocortical areas considered for thickness measurements. nCtx: neocortex, Th: thalamus, St: striatum, HC: hippocampus, Cp: caudate-putamen. (h) Quantification of cortical thickness measured at 30% and 70% distance from the dorsal midline and cortical hemispheric circumference indicates no differences in overall thickness of these neocortical regions (n = 5 control, n = 8 poly(I:C); caudal 30% p=0.5252; caudal 70% p=0.2481; rostral 30% p=0.4459; rostral 70% p=0.4583, two tailed Student’s t-test).
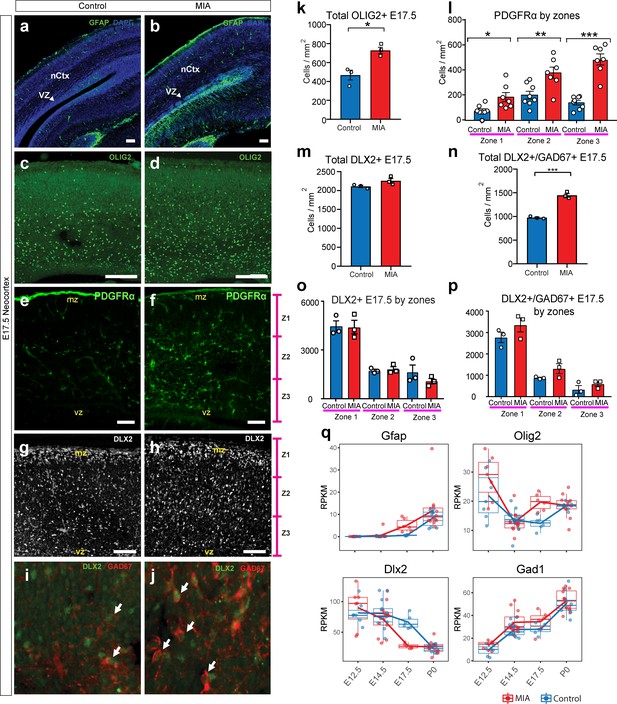
MIA alters development of neuronal and glial cell types in fetal offspring at E17.5.
(a–j) Coronal fetal brain sections from three independent E17.5 saline litters (n = 4 control) and four independent MIA litters (n = 6 MIA) were immunostained for cell-type markers. (a,b) Altered distribution and numbers of GFAP+ cells (radial glia and astrocytes) in MIA samples. (c-j) Reduced sub-population of interneurons and oligodendrocytes using OLIG2, PDGFRα, DLX2, and GAD67. (k-p) Quantification of the cell density in (c-j). Overall MIA-associated increase in OLIG2+ cells (k), with further specific increased PDGFRα+ cells representing oligodendrocytes (l), and no change in overall DLX2+ interneurons (m,o), but increased overall number of DLX2+/GAD67+ interneurons (n,p) are observed in MIA neocortex. Student’s t-test: OLIG2 p=0.0134; DLX2+/GAD67+ cells p=0.0004. Bin quantification analyses of PDGFRα+, DLX2+, and GAD67+ immunoreactive cells in zones 1–3 (Z1 to Z3) indicate that changes are not restricted to a particular zones of the neocortex. Arrows denote co-labeled cells. Scale bars in (a-h) = 100 µm and in (i-j) = 30 µm. Abbreviations: neocortex (nCtx); marginal zone (mz); ventricular zone (VZ). (q) RPKM trajectories of the cell identity markers tested in (a-k) across the developmental time-points of the study show parallel differences in mRNA expression of cell-type markers following MIA induction. Gfap and Olig2 mRNA was significantly upregulated in MIA samples at E17.5. Dlx2 was significantly downregulated at E17.5, with no significant changes observed for Gad1 (encodes GAD67) or for Pdgfra (not shown).
Tables
| Reagent type (species) or resource | Designation | Source or reference | Identifiers | Additional information |
|---|---|---|---|---|
| Chemical compound, drug | poly(I:C) dsRNA | Sigma Aldrich | P0913 | Lot # 016M1451V |
| Antibody | Rabbit polyclonal anti PAX-6 | Covance | PRB-278P-100 | Dilution: (1:250) for IHC; (1:3000) for WB |
| Antibody | Rabbit polyclonal anti-TBR1 | Abcam | ab31940 | Dilution: (1:500) for IHC; (1:2000) WB |
| Antibody | Rat monoclonal anti-CTIP2 | Abcam | ab18465 | Dilution (1:250) for IHC; (1:1000) for WB |
| Antibody | Rabbit polyclonal anti-CUX1 | Abclonal | A2213 | Dilution (1:200) for IHC; (1:1000) for WB |
| Antibody | Mouse monoclonal anti-SATB2 | Abcam | Ab51502 | Dilution (1:500) for IHC; (1:2000) for WB |
| Antibody | Rabbit monoclonal anti-KI67 | Cell Signaling | 12202 | Dilution (1:500) for IHC |
| Antibody | Goat polyclonal anti-SOX9 | R and D Systems | AF3075 | Dilution (1:500) for IHC |
| Antibody | Rabbit polyclonal anti-PH3 | Cell Signaling | 9701 | Dilution (1:500) for IHC |
| Antibody | Rat monoclonal anti-TBR2 | Thermo Fisher Scientific | 14-4875-82 | Dilution (1:500) for IHC |
| Antibody | anti-DLX2 | John Rubenstein Lab | N/A | Dilution (1:200) for IHC |
| Antibody | Rabbit polyclonal anti-GFAP | Agilent Dako | Z0334 | Dilution (1:250) for IHC |
| Antibody | Rabbit polyclonal anti-OLIG2 | Millipore Sigma | AB9610 | Dilution (1:500) for IHC |
Additional files
-
Supplementary file 1
Differential gene expression data of control vs MIA samples, and WGCNA gene module assignment at E12.5.
Columns indicate gene symbols (gene_name), log2 fold change (LogFC), log2 counts per million (LogCPM), likelihood ratio statistics (LR), p value (PValue), false discovery rate adjusted p value (FDR), and WGCNA module assignment (moduleColors and moduleColors_merged). A total of 16,396 genes were tested.
- https://cdn.elifesciences.org/articles/60100/elife-60100-supp1-v2.zip
-
Supplementary file 2
Differential gene expression data of control vs MIA samples, and WGCNA gene module assignment at E14.5.
Columns indicate gene symbols (gene_name), log2 fold change (LogFC), log2 counts per million (LogCPM), likelihood ratio statistics (LR), p value (PValue), false discovery rate adjusted p value (FDR), and WGCNA module assignment (moduleColors and moduleColors_merged). A total of 16,658 genes were tested.
- https://cdn.elifesciences.org/articles/60100/elife-60100-supp2-v2.zip
-
Supplementary file 3
Differential gene expression data of control vs MIA samples, and WGCNA gene module assignment at E17.5.
Columns indicate gene symbols (gene_name), log2 fold change (LogFC), log2 counts per million (LogCPM), likelihood ratio statistics (LR), p value (PValue), false discovery rate adjusted p value (FDR), and WGCNA module assignment (moduleColors and moduleColors_merged). A total of 16,229 genes were tested.
- https://cdn.elifesciences.org/articles/60100/elife-60100-supp3-v2.zip
-
Supplementary file 4
Differential gene expression data of control vs MIA samples, and WGCNA gene module assignment at E12.5.
Columns indicate gene symbols (gene_name), log2 fold change (LogFC), log2 counts per million (LogCPM), likelihood ratio statistics (LR), p value (PValue), false discovery rate adjusted p value (FDR), and WGCNA module assignment (moduleColors and moduleColors_merged). A total of 16,613 genes were tested.
- https://cdn.elifesciences.org/articles/60100/elife-60100-supp4-v2.zip
-
Supplementary file 5
RNA-seq sample metadata including Sample ID, age, treatment (Condition), classification as per their IL-6 prenatal levels (Response), sequencer lane batch parameters (HiSeq Lane, Library barcodes), animal and litter parameters (Animal ID, Litter ID, Liter Size), animal sex inferred from Xist expression (Sex by Xist counts), fastq file name, and fastq file md5sum hash value (md5sum).
Out of 74 samples, 73 were tracked for the Litter ID (explained in the Comments).
- https://cdn.elifesciences.org/articles/60100/elife-60100-supp5-v2.zip
-
Supplementary file 6
Differential gene expression data of control vs MIA male samples at E12.5.
Columns indicate gene symbols (gene_name), log2 fold change (LogFC), log2 counts per million (LogCPM), likelihood ratio statistics (LR), p value (PValue), and false discovery rate adjusted p value (FDR). A total of 16,171 genes were tested.
- https://cdn.elifesciences.org/articles/60100/elife-60100-supp6-v2.zip
-
Supplementary file 7
Differential gene expression data of control vs MIA male samples at E14.5.
Columns indicate gene symbols (gene_name), log2 fold change (LogFC), log2 counts per million (LogCPM), likelihood ratio statistics (LR), p value (PValue), and false discovery rate adjusted p value (FDR). A total of 16,568 genes were tested.
- https://cdn.elifesciences.org/articles/60100/elife-60100-supp7-v2.zip
-
Supplementary file 8
Differential gene expression data of control vs MIA male samples at E17.5.
Columns indicate gene symbols (gene_name), log2 fold change (LogFC), log2 counts per million (LogCPM), likelihood ratio statistics (LR), p value (PValue), and false discovery rate adjusted p value (FDR). A total of 15,883 genes were tested.
- https://cdn.elifesciences.org/articles/60100/elife-60100-supp8-v2.zip
-
Supplementary file 9
Differential gene expression data of control vs MIA male samples at P0.
Columns indicate gene symbols (gene_name), log2 fold change (LogFC), log2 counts per million (LogCPM), likelihood ratio statistics (LR), p value (PValue), and false discovery rate adjusted p value (FDR). A total of 16,394 genes were tested.
- https://cdn.elifesciences.org/articles/60100/elife-60100-supp9-v2.zip
-
Supplementary file 10
Differential gene expression data of control vs MIA female samples at E12.5.
Columns indicate gene symbols (gene_name), log2 fold change (LogFC), log2 counts per million (LogCPM), likelihood ratio statistics (LR), p value (PValue), and false discovery rate adjusted p value (FDR). A total of 16,161 genes were tested.
- https://cdn.elifesciences.org/articles/60100/elife-60100-supp10-v2.zip
-
Supplementary file 11
Differential gene expression data of control vs MIA female samples at E14.5.
Columns indicate gene symbols (gene_name), log2 fold change (LogFC), log2 counts per million (LogCPM), likelihood ratio statistics (LR), p value (PValue), and false discovery rate adjusted p value (FDR). A total of 16,350 genes were tested.
- https://cdn.elifesciences.org/articles/60100/elife-60100-supp11-v2.zip
-
Supplementary file 12
Differential gene expression data of control vs MIA female samples at E17.5.
Columns indicate gene symbols (gene_name), log2 fold change (LogFC), log2 counts per million (LogCPM), likelihood ratio statistics (LR), p value (PValue), and false discovery rate adjusted p value (FDR). A total of 16,093 genes were tested.
- https://cdn.elifesciences.org/articles/60100/elife-60100-supp12-v2.zip
-
Supplementary file 13
Differential gene expression data of control vs MIA female samples at P0.
Columns indicate gene symbols (gene_name), log2 fold change (LogFC), log2 counts per million (LogCPM), likelihood ratio statistics (LR), p value (PValue), and false discovery rate adjusted p value (FDR). A total of 16,487 genes were tested.
- https://cdn.elifesciences.org/articles/60100/elife-60100-supp13-v2.zip
-
Supplementary file 14
Differential gene expression data for all four developmental ages (Age) analyzed without sex covariate.
Columns indicate gene symbols (gene_name), log2 fold change (LogFC), log2 counts per million (LogCPM), likelihood ratio statistics (LR), p value (PValue), and false discovery rate adjusted p value (FDR).
- https://cdn.elifesciences.org/articles/60100/elife-60100-supp14-v2.zip
-
Supplementary file 15
Differential gene expression data contrasting male vs female samples at each developmental timepoint (Age).
Columns indicate gene symbols (gene_name), log2 fold change (LogFC), log2 counts per million (LogCPM), likelihood ratio statistics (LR), p value (PValue), and false discovery rate adjusted p value (FDR).
- https://cdn.elifesciences.org/articles/60100/elife-60100-supp15-v2.zip
-
Supplementary file 16
High confidence SFARI mouse orthologs genes used in the analysis.
Columns indicate gene symbols and names, chromosomal location, genetic category, gene and syndromic scores, number of reports and Mouse Genome Informatics (MGI) symbols for mouse orthologs. 89 mouse orthologs with high confidence SFARI gene scores of one or two were included in the analysis.
- https://cdn.elifesciences.org/articles/60100/elife-60100-supp16-v2.zip
-
Supplementary file 17
Complete SFARI gene database available at the time of conducting the analysis.
- https://cdn.elifesciences.org/articles/60100/elife-60100-supp17-v2.zip
-
Supplementary file 18
Gene ontology biological process enrichment analysis for genes passing FDR < 0.05, for each developmental timepoint and direction of differential expression, summarized in Figure 2d heatmap.
Columns signify gene ontology ID number (GO.ID), gene ontology term (Term), the number of annotated genes in the term (Annotated), the number of genes passing FDR < 0.05 for up- or downregulated genes (Significant), the expected number of genes under the null hypothesis, Fisher’s p value (classicFisher), developmental age (Age), direction of differential expression relative to saline (DE_direction), enrichment.
- https://cdn.elifesciences.org/articles/60100/elife-60100-supp18-v2.zip
-
Supplementary file 19
Gene ontology biological process enrichment analysis for genes passing FDR < 0.05, for each developmental timepoint, direction of differential expression, and WGCNA module membership.
Columns signify gene ontology ID number (GO.ID), gene ontology term (Term), the number of annotated genes in the term (Annotated), the number of genes passing FDR < 0.05 for up- or downregulated genes and WGCNA module (Significant), the expected number of genes under the null hypothesis, Fisher’s p value (classicFisher), WGCNA module membership (module_color), enrichment, direction of differential expression (Direction), and developmental age (Age).
- https://cdn.elifesciences.org/articles/60100/elife-60100-supp19-v2.zip
-
Supplementary file 20
Numbers of RNA-seq samples divided by sex and treatment condition.
- https://cdn.elifesciences.org/articles/60100/elife-60100-supp20-v2.zip
-
Supplementary file 21
Summary of sample numbers (N) used for neuroanatomical IHC validation at E17.5.
Numbers represent samples analyzed for at least one of the individual listed markers or co-stains. Details of specific use of individual samples can be found in supplementary file 22.
- https://cdn.elifesciences.org/articles/60100/elife-60100-supp21-v2.xlsx
-
Supplementary file 22
List of all MIA and control samples analyzed through neuroanatomical IHC validation at E17.5, and its specific use for individual experiments.
- https://cdn.elifesciences.org/articles/60100/elife-60100-supp22-v2.xlsx
-
Transparent reporting form
- https://cdn.elifesciences.org/articles/60100/elife-60100-transrepform-v2.docx





