A single-chain and fast-responding light-inducible Cre recombinase as a novel optogenetic switch
Figures
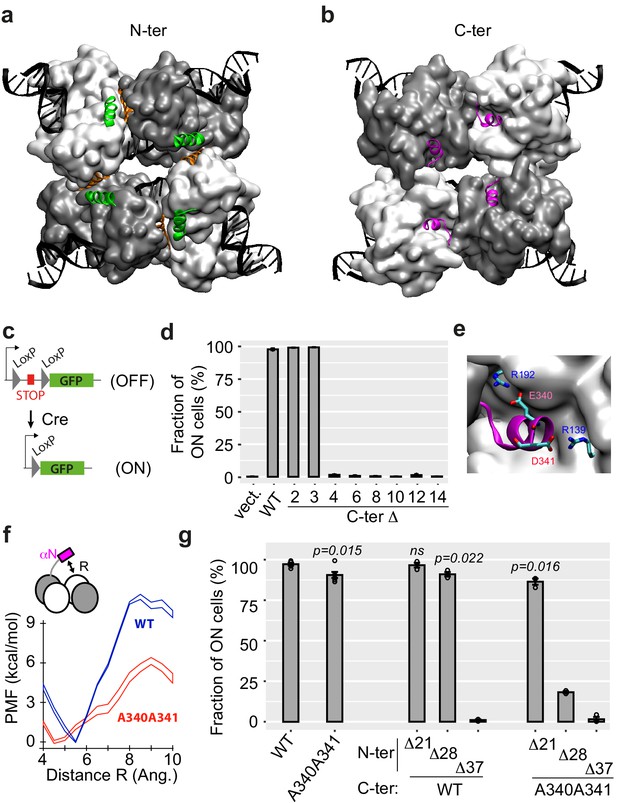
N-ter and C-ter α-helices of Cre are critical for activity.
(a, b) Structure of the Cre tetramer complexed with DNA (PDB: 1NZB). The four N-ter domains (a) interact via contacts between α-helices A (green) and E (orange), and the four C-ter domains (b) interact via α-helices N (magenta). (c) Yeast reporter system to quantify Cre efficiency. The STOP element includes a selectable marker and a terminator sequence, which prevents expression of the downstream green fluorescent protein (GFP) sequence. (d) Activity of wild-type (WT) and C-ter mutants of Cre measured as the fraction of cells expressing GFP (mean ± s.e.m., n = 3 independent transformants). Numbers denote the number of residues deleted from the C-ter extremity. 'Vect': expression plasmid with no insert. (e) Blow-up of αN helix. (f) Energetics of αN displacing (see Materials and methods). PMF: potential mean force (± error defined as in Materials and methods). (g) Activity of Cre mutants lacking N-terminal residues 2 to X, combined or not with the A340 A341 C-terminal mutation (mean ± s.e.m., n = 3 independent transformants). X was 21, 28, or 37 as indicated. P-values: t-test of the difference from WT. ns: not significantly different from WT.
-
Figure 1—source data 1
Data used to produce Figure 1d.
- https://cdn.elifesciences.org/articles/61268/elife-61268-fig1-data1-v2.txt
-
Figure 1—source data 2
Data used to produce Figure 1f.
- https://cdn.elifesciences.org/articles/61268/elife-61268-fig1-data2-v2.txt
-
Figure 1—source data 3
Data used to produce Figure 1g.
- https://cdn.elifesciences.org/articles/61268/elife-61268-fig1-data3-v2.txt
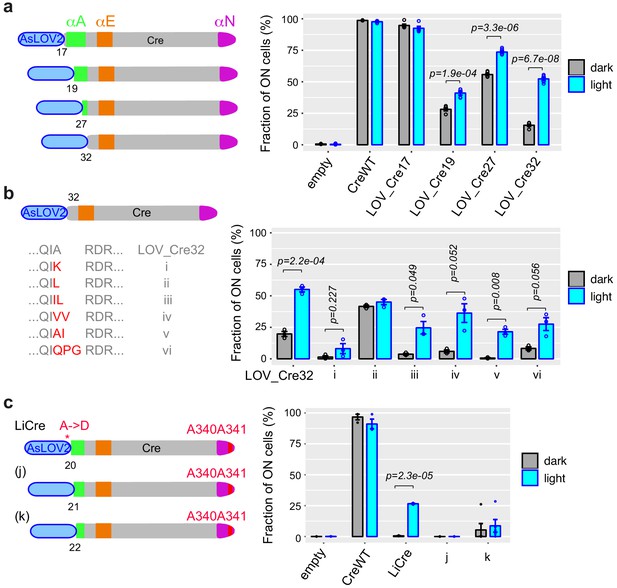
Specific single-chain AsLOV2-Cre fusions display photoactivatable recombinase activity.
(a) Fusions with wild-type Cre. (b) Variants of LOV_Cre32 carrying the indicated mutations at the peptide junction. (c) Fusions with CreE340A,D341A mutant. (a–c) Numbers indicate the positions on the Cre peptidic sequence where AsLOV2 was fused. All bar plots show recombinase activity measured by flow cytometry (mean ± s.e.m. of the proportion of switched cells, n = 5 independent transformants) after galactose-induced expression of the fusion protein, followed (cyan) or not (gray) by illumination at 460 nm, 36.3 mW/cm2, for 30 min (PAUL LED box). Statistical significance of the differences between dark and light conditions is indicated (t-tests). Peptide sequences of all constructs are provided in Supplementary file 1 – Supplementary Text S2. AsLOV2: A. sativa phototropin 1 LOV2; LOV: light, oxygen, or voltage.
-
Figure 2—source data 1
Data used to produce Figure 2a.
- https://cdn.elifesciences.org/articles/61268/elife-61268-fig2-data1-v2.txt
-
Figure 2—source data 2
Data used to produce Figure 2b.
- https://cdn.elifesciences.org/articles/61268/elife-61268-fig2-data2-v2.txt
-
Figure 2—source data 3
Data used to produce Figure 2c.
- https://cdn.elifesciences.org/articles/61268/elife-61268-fig2-data3-v2.txt
-
Figure 2—source data 4
Data used to produce Figure 2—figure supplement 1.
- https://cdn.elifesciences.org/articles/61268/elife-61268-fig2-data4-v2.txt
-
Figure 2—source data 5
Data used to produce Figure 2—figure supplement 2.
- https://cdn.elifesciences.org/articles/61268/elife-61268-fig2-data5-v2.txt
-
Figure 2—source data 6
Data used to produce Figure 2—figure supplement 3.
- https://cdn.elifesciences.org/articles/61268/elife-61268-fig2-data6-v2.txt
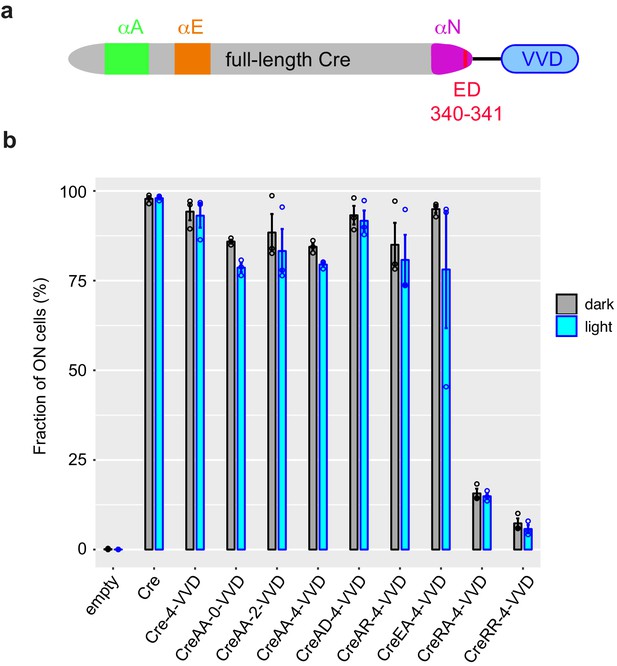
Cre-VVD fusions do not display photoactivatable recombinase activity.
(a) Scheme of chimeric proteins tested. (b) Activity (fraction of switched cells) was measured after galactose-induced expression of the fusion protein, followed (cyan) or not (gray) by illumination at 460 nm, 36.3 mW/cm2, for 30 min, followed by 90 min cultivation in non-dividing medium and flow cytometry. Error bars, s.e.m. (n = 3 colonies of the same transformant). Notation indicates residues at positions 340 and 341, ED being the wild-type sequence. Numbers 0, 2, and 4 indicate the amino acid length of the spacer. Peptide sequences are provided in Supplementary file 1 – Supplementary Text S2. VVD: Vivid.
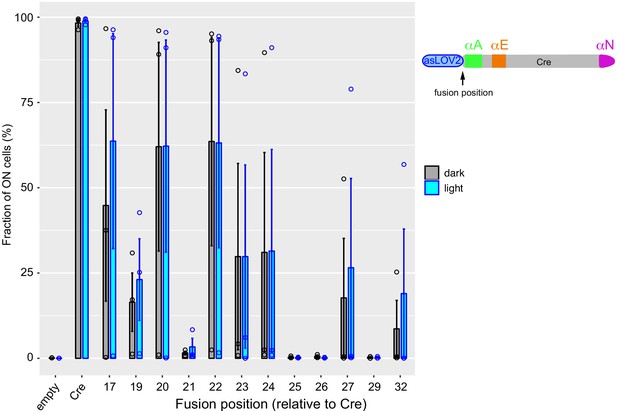
Systematic analysis of AsLOV2-Cre fusions reveals candidate cases of photoactivatable recombinase activity.
Numbers indicate the position of the fusion on the peptide sequence of Cre. Recombinase activity was measured as in Figure 2—figure supplement 1. Error bars: s.e.m., n = 3 independent transformants. AsLOV2: A. sativa phototropin 1 LOV2.
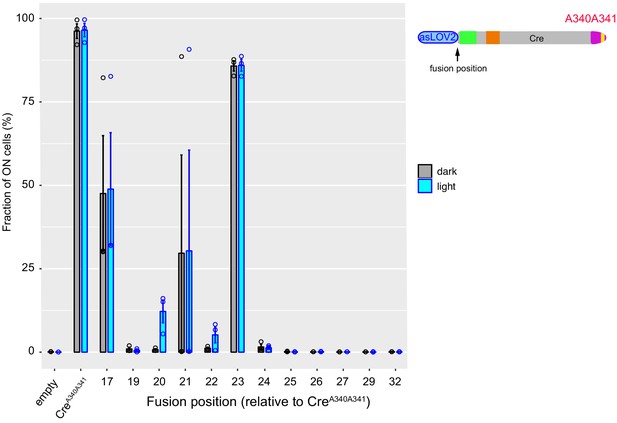
Systematic analysis of AsLOV2-CreA340A341 fusions reveals additional cases of photoactivatable recombinase activity.
Residues at positions 340 and 341 of Cre were replaced by alanines. Numbers indicate the position of the fusion on the peptide sequence of Cre. Recombinase activity was measured as in Figure 2—figure supplement 1. Error bars: s.e.m., n = 3 independent transformants. AsLOV2: A. sativa phototropin 1 LOV2.
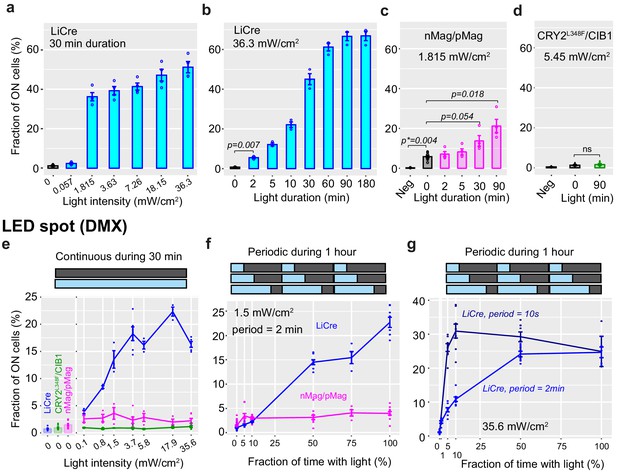
Calibration of light induction.
(a, b) Yeast cells carrying the reporter system of Figure 1c and expressing light-inducible Cre (LiCre) were grown to stationary phase and illuminated with blue light (460 nm, PAUL LED box, see Materials and methods) at indicated intensities, then incubated in non-dividing conditions and processed by flow cytometry (mean ± s.e.m., strain GY1761 transformed with pGY466); n = 4 and 3 colonies in (a) and (b), respectively. Illumination conditions varied either in intensity (a) or duration (b). p: significance from t-test (n = 3). The fraction of ON cells observed at 0 min was not significantly higher than the fraction of ON GY1761 cells transformed with empty vector pRS314 (p=0.19). (c) Yeast strain GY1761 was transformed with plasmids pGY491 and pGY501 to express the two proteins of the nMag/pMag split Cre system (Kawano et al., 2016). Cells were processed as in (b) with a light intensity that matched the authors' recommendations (1.815 mW/cm2, PAUL LED box). Neg: no illumination, cells containing empty vectors only. p: significance from t-tests (n = 4); p*: significance from Wilcoxon rank-sum test (n = 4). (d) Yeast strain GY1761 was transformed with plasmids pGY531 and pGY532 to express the two proteins of the CRY2L348F/CIB1 split Cre system (Taslimi et al., 2016). Cells were processed as in (b) with or without illumination for 90 min at an intensity matching the authors' recommendations (5.45 mW/cm2, PAUL LED box). Neg: no illumination, cells containing empty vectors only. (e–g) Yeast strain GY2214 carrying two reporters (green fluorescent protein and mCherry) of Cre-mediated excision was transformed with expression plasmids for LiCre (blue), the nMag/pMag (magenta), or CRY2L348F/CIB1 (green) split Cre and illuminated at 30°C under a DMX-controlled LED spot. The fraction of ON cells was computed as the fraction of cells that had activated the mCherry reporter. Illumination was either continuous (e) or periodic (f, g) at indicated intensity, duration, period, and duty cycle (fraction of period when light is ON).
-
Figure 3—source data 1
Data used to produce Figure 3a.
- https://cdn.elifesciences.org/articles/61268/elife-61268-fig3-data1-v2.txt
-
Figure 3—source data 2
Data used to produce Figure 3b.
- https://cdn.elifesciences.org/articles/61268/elife-61268-fig3-data2-v2.txt
-
Figure 3—source data 3
Data used to produce Figure 3c.
- https://cdn.elifesciences.org/articles/61268/elife-61268-fig3-data3-v2.txt
-
Figure 3—source data 4
Data used to produce Figure 3d.
- https://cdn.elifesciences.org/articles/61268/elife-61268-fig3-data4-v2.txt
-
Figure 3—source data 5
Data used to produce Figure 3e.
- https://cdn.elifesciences.org/articles/61268/elife-61268-fig3-data5-v2.txt
-
Figure 3—source data 6
Data used to produce Figure 3f.
- https://cdn.elifesciences.org/articles/61268/elife-61268-fig3-data6-v2.txt
-
Figure 3—source data 7
Data used to produce Figure 3g.
- https://cdn.elifesciences.org/articles/61268/elife-61268-fig3-data7-v2.txt
-
Figure 3—source data 8
Data used to produce Figure 3—figure supplement 1.
- https://cdn.elifesciences.org/articles/61268/elife-61268-fig3-data8-v2.txt
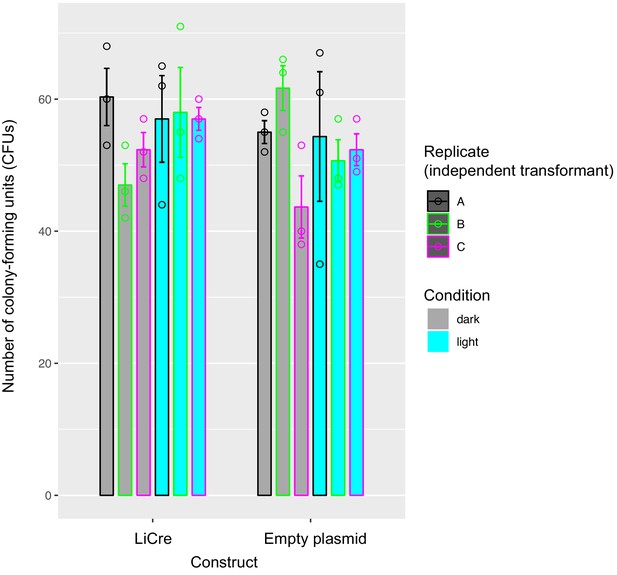
Yeast viability after exposure to blue light.
Yeast strain GY2214 carrying light-inducible Cre-expressing plasmid pGY466 or empty plasmid pRS314 was processed as for recombination scoring: an overnight culture was illuminated (light) or not (dark) with blue light (450 nm) at 35.6 mW/cm2 using a DMX-controlled spot during 60 min at 30°C. Cells were then diluted by serial dilutions and plated on non-selective rich medium. Two days after, colonies were counted. Each data point represents the number of colonies on one plate.
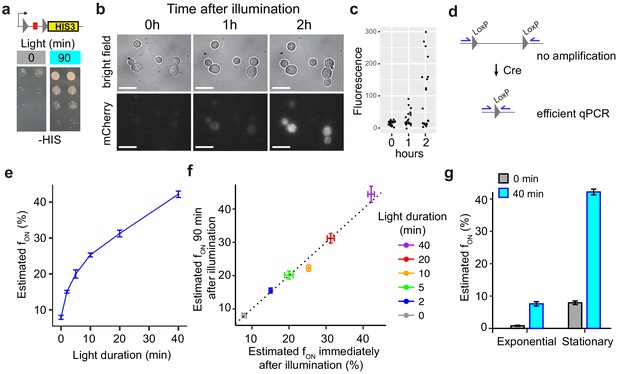
Additional functional properties of light-inducible Cre (LiCre).
(a) Yeast cells expressing LiCre from plasmid pGY466 and carrying an integrated reporter conferring prototrophy to histidine were spotted on two His- plates at decreasing densities. Prior to incubation at 30°C, one plate (right) was illuminated for 90 min at 3.63 mW/cm2 intensity (PAUL LED box). (b) Time-lapse imaging of yeast cells expressing LiCre and carrying an mCherry reporter of Cre-mediated recombination (strain GY2033 with plasmid pGY466). Cells were grown to stationary phase, illuminated for 90 min at 3.63 mW/cm2 intensity, immobilized on bottom-glass wells in dividing condition, and imaged at the indicated time. Bar: 10 μm. (c) Quantification of intracellular mCherry fluorescence from (b), n = 22 cells. (d) Design of qPCR assay allowing to quantify recombination efficiency. (e) Quantification of excision by qPCR immediately after illumination at 3.63 mW/cm2 intensity (stationary phase, strain GY1761 transformed with pGY466, PAUL LED box). ‘Estimated fON’ values are the estimated fraction of DNA molecules that underwent recombination, based on a standard curve (see Materials and methods). (f) DNA excision does not occur after illumination. X-axis: same data as shown on Y-axis in (e). Y-axis: same experiment but after illumination, cells were incubated for 90 min in dark and non-dividing condition prior to harvest and qPCR. (g) Quantification of DNA excision by qPCR on exponentially growing or stationary-phase cells (strain GY1761 transformed with pGY466) illuminated at 3.63 mW/cm2 intensity (PAUL LED box). Gray: no illumination. Bars in (e–g): s.e.m. (n = 10 colonies).
-
Figure 4—source data 1
Data used to produce Figure 4c.
- https://cdn.elifesciences.org/articles/61268/elife-61268-fig4-data1-v2.txt
-
Figure 4—source data 2
Data used to produce Figure 4e–g.
- https://cdn.elifesciences.org/articles/61268/elife-61268-fig4-data2-v2.txt
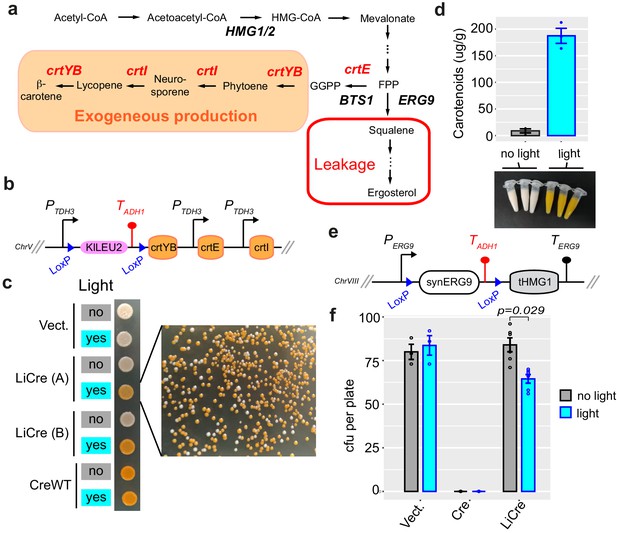
Switching ON carotenoid production with light.
(a) β-Carotene biosynthetic pathway. Exogeneous genes from X. dendrorhous are shown in red. FPP: farnesyl pyrophosphate; GGPP: geranylgeranyl pyrophosphate. (b) Scheme of the switchable locus of yeast GY2247. (c) Photoswitchable bioproduction. Strain GY2247 was transformed with either pRS314 (Vect.), pGY466 (light-inducible Cre [LiCre]), or pGY502 (CreWT). Cells were cultured overnight in SD-M-W, and the cultures were illuminated (460 nm, 90 min, 36.3 mW/cm2) or not and then spotted on agar plates. A and B correspond to two independent transformants of the LiCre plasmid. Colonies on the right originate from the illuminated LiCre (A) culture. (d) Quantification of carotenoids production. Three colonies of strain GY2247 transformed with LiCre plasmid pGY466 were cultured overnight in SD-W-M. The following day, 10 ml of each culture were illuminated as in (c) while another 10 ml was kept in the dark. These cultures were then incubated for 72 hr at 30°C. Cells were pelleted (colors of the cell pellets are shown on picture) and processed for quantification (see Materials and methods). Units are micrograms of total carotenoids per gram of biomass dry weight. Bars: mean ± s.e.m., n = 3. (e) Scheme of the switchable locus of yeast GY2236. (f) Light-induced deletion of squalene synthase gene. Strain GY2236 was transformed with either pRS314 (Vect), pGY502 (Cre), or pGY466 (LiCre). Cells were cultured overnight in 4 ml of SD-M-W. A 100-μl aliquot of each culture was illuminated (as in c) while another 100-μl aliquot was kept in the dark. A dilution at ~1 cell/μl was then plated on SD-W. Colonies were counted after 3 days. cfu: colony-forming units (mean ± s.e.m., n ≥ 3 plates); p: significance from t-test.
-
Figure 5—source data 1
Data used to produce Figure 5d.
- https://cdn.elifesciences.org/articles/61268/elife-61268-fig5-data1-v2.txt
-
Figure 5—source data 2
Data used to produce Figure 5e.
- https://cdn.elifesciences.org/articles/61268/elife-61268-fig5-data2-v2.txt
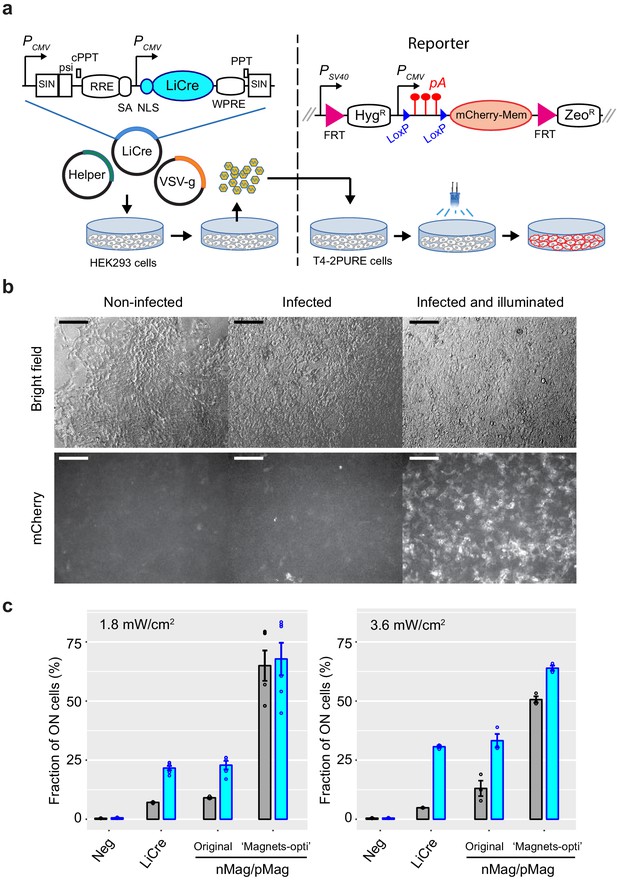
Light-inducible Cre (LiCre) photoactivation in human cells.
(a) Left: lentiviral SIN vector for LiCre expression (plasmid pGY577). PCMV: early cytomegalovirus promoter; SIN: LTR regions of simian immunodeficiency virus comprising a partially deleted 3' U3 region, followed by the R and U5 regions; psi: retroviral psi RNA packaging element; cPPT and PPT: central and 3' polypurine tracks, respectively; RRE: Rev/Rev-responsive element; SA: SIV Rev/Tat splice acceptor; NLS: nuclear localization signal; WPRE: woodchuck hepatitis virus regulatory element; Helper: plasmid coding for gag, pol, tat, and rev; VSV-g: plasmid encoding the envelope of the vesicular stomatitis virus. Co-transfection in HEK293T cells produces pseudotyped particles. These particles are deposited on T4-2PURE reporter cells, which are then illuminated and imaged. Right: genomic reporter locus of T4-2PURE cells. PSV40: promoter from SV40; FRT: FLP recognition targets; HygR: hygromycin resistance; pA: poly-adenylation signal from SV40; ZeoR: zeomycin resistance. Recombination between LoxP sites switches ON the expression of mCherry by removing three pA terminators. (b) Microscopy images of T4-2PURE cells following the assay. Bars, 150 μm. All three fluorescent frames were acquired at the same intensity and exposure time. Illumination corresponded to two 20 min exposures at 3.63 mW/cm2 in the PAUL LED box, separated by 20 min without illumination. (c) Flow-cytometry quantification of recombination efficiencies following the assay (mean fraction of mCherry-positive cells ± s.e.m., n = 3 or 6). T4-2 PURE cells were infected with lentiviral particles coding for either LiCre (produced from pGY577), nMag/pMag split Cre (produced from pGY625), or the 'Magnets-opti' version of nMag/pMag split Cre (produced from pGY626). Illumination was conducted as in (b) at the indicated light intensities. Neg: non-infected. We controlled by qPCR that the lower residual activity of LiCre was not due to a lower particle load of the lentiviral preparation (see Materials and methods).
-
Figure 6—source data 1
Data used to produce Figure 6c, left.
- https://cdn.elifesciences.org/articles/61268/elife-61268-fig6-data1-v2.txt
-
Figure 6—source data 2
Data used to produce Figure 6c, right.
- https://cdn.elifesciences.org/articles/61268/elife-61268-fig6-data2-v2.txt
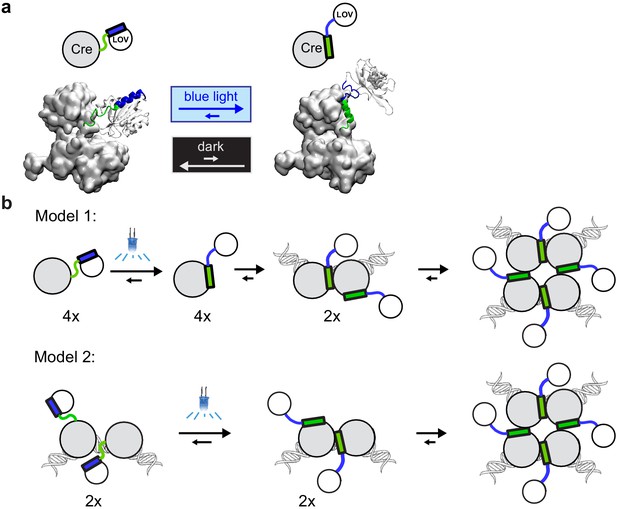
Models of light-inducible Cre (LiCre) activation.
(a) Structural model built using PDB structures 1NZB (Cre) and 4WF0 (AsLOV2). Green: residues of αA helix from Cre. Blue: residues of Jα helix from AsLOV2. (b) Proposed models for the photo-induced formation of the LiCre:DNA recombination synapse. Models differ by the affinity of uninduced LiCre for DNA, which may (Model 2) or may not (Model 1) allow their association prior to illumination.
Additional files
-
Source code 1
R/Sweave code for analysis of flow-cytometry data.
- https://cdn.elifesciences.org/articles/61268/elife-61268-code1-v2.zip
-
Supplementary file 1
File containing supplementary tables and supplementary text.
Table S1: List of plasmids used in this study. Table S2: List of strains used in this study. Table S3: List of DNA oligonucleotides used in this study. Supplementary Text S1: Synthetic nucleotidic sequences. Supplementary Text S2: Peptide sequences.
- https://cdn.elifesciences.org/articles/61268/elife-61268-supp1-v2.odt
-
Transparent reporting form
- https://cdn.elifesciences.org/articles/61268/elife-61268-transrepform-v2.docx





