Short and long sleeping mutants reveal links between sleep and macroautophagy
Figures
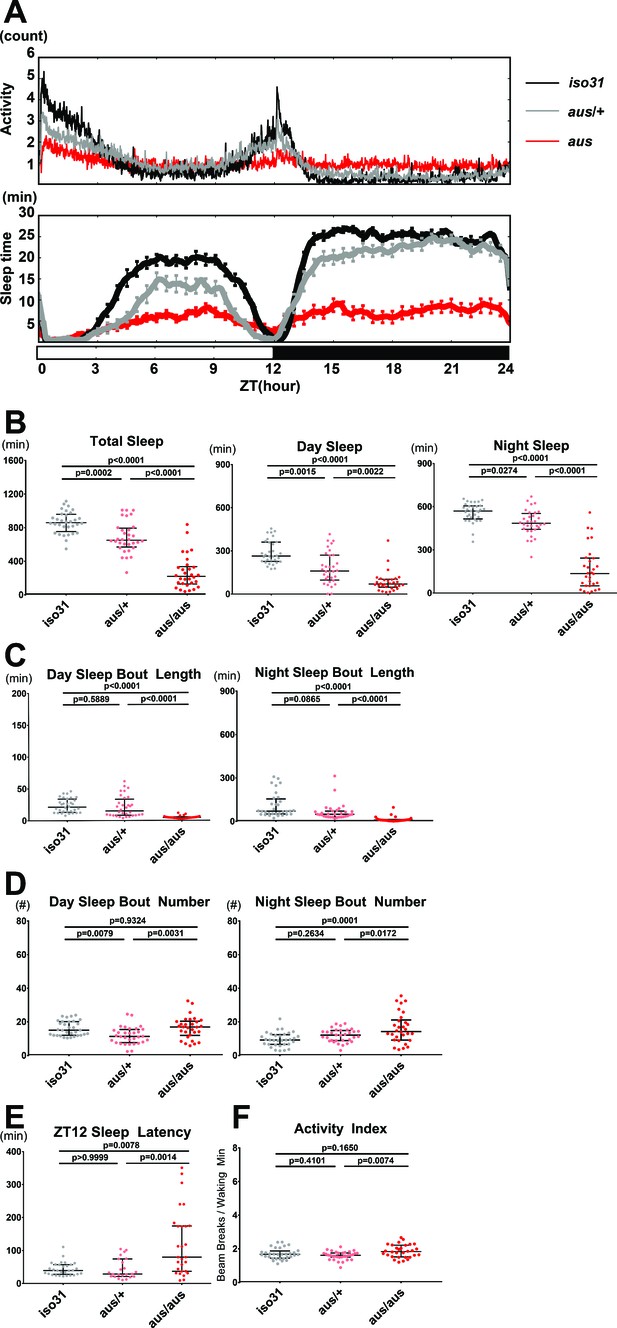
Sleep phenotype of argus mutants.
All sleep metrics were measured under a 12 hr:12 hr light:dark cycle in female iso31 (gray), aus/+ (pink) and aus/aus (red) flies. (A) Mean activity (top panel) and sleep (bottom panel) over time during the 24-hr cycle. (B) Total sleep amount during the whole 24-hr cycle (left), day (middle), and night (right). (C) Mean sleep bout duration during the day (left) and night (right). (D) Sleep bout number during the day (left) and night (right). (E) Latency to first sleep bout after ZT12 lights off. (F) Activity index of beam breaks per waking minute over the 24-hr cycle. n = 30–32 (A–D,F) or n = 23–30 (E); individual flies overlaid with median±interquartiles (B–F); Tukey test (B-total+ night,C,F) or Dunn test (B-day,D-E). (A).
-
Figure 1—source data 1
Sleep Phenotype of Argus Mutants.
- https://cdn.elifesciences.org/articles/64140/elife-64140-fig1-data1-v1.xlsx

Circadian rhythms are intact in the aus mutant.
(A) Sample actograms from iso31 control and aus mutant flies. (B) Activity data from flies assayed during constant darkness were assessed for circadian rhythmicity. Average circadian period length (tau) of iso31 controls and aus mutants was based on Clocklab analysis. Fast Fourier Transform (FFT) was used to establish a cutoff for rhythmicity, such that FFT values > 0.01 were considered indicative of a rhythm. The average FFT value is calculated from rhythmic flies.
-
Figure 1—figure supplement 1—source data 1
Circadian rhythms are intact in the aus mutant.
- https://cdn.elifesciences.org/articles/64140/elife-64140-fig2-data2-v1.xlsx
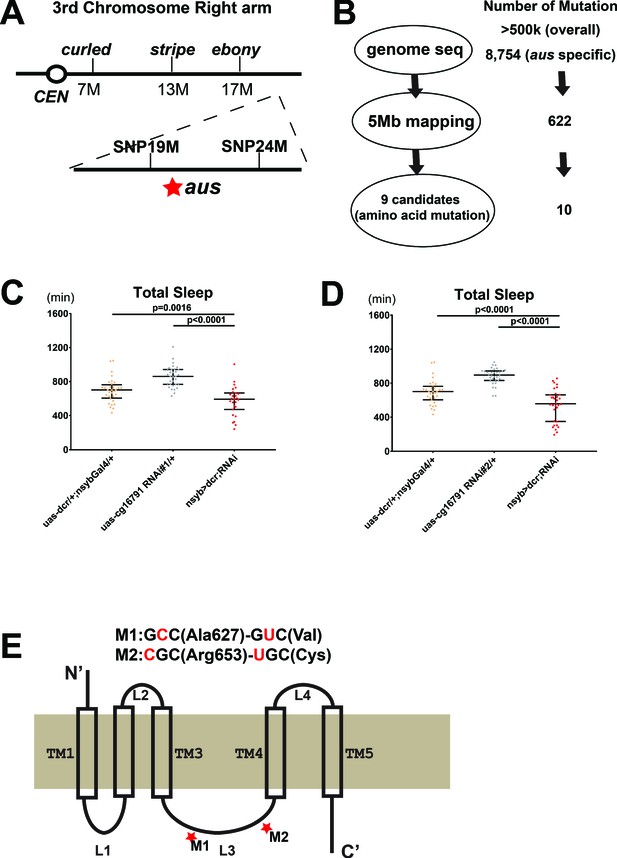
Mapping the argus sleep phenotype to a single gene: cg16791.
(A) The genomic location of argus is indicated as a star within a 5 Mb region on the right arm of the third chromosome, following genetic mapping with visible mutations and SNP markers. (B) Schematic of the genome sequencing procedure of argus homozygotes with the number of mutations identified in each step listed on the right. The initial alignment revealed more than half a million mutations relative to the published Drosophila genome. More than eight thousand mutations remained after removing mutations also found in the iso31 control strain. Factoring in the mapping data (shown in A) and focusing on missense mutations narrowed the number of candidate genes to nine. (C–D) Total sleep with cg16791 RNAi knockdown in females, using pan-neuronal driver nsyb-Gal4, uas-dicer, and either of two independent RNAi lines, compared to RNAi-alone and nsyb-Gal4+ Dcr alone controls. n = 27–32; Fischer’s LSD; individual flies overlaid with median±interquartiles. (E) Predicted protein of CG16791. Two GC-AT transitions that cause missense mutations in the loop3 region were identified by Sanger-sequencing in aus mutants.
-
Figure 2—source data 1
Mapping the argus sleep phenotype to a single gene: cg16791.
- https://cdn.elifesciences.org/articles/64140/elife-64140-fig2-data1-v1.xlsx
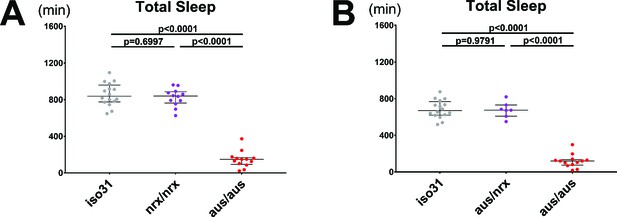
A mutation in Nrx1 does not underlie the aus reduced sleep phenotype.
(A) Total sleep in nrx/nrx mutants is comparable to iso31 control and greater than aus/aus mutants. n = 12–16; individual brain values overlaid with population median±interquartiles; Tukey test. (B) Total sleep in Nrx/aus transheterozygotes is comparable to iso31 control and greater than aus/aus mutants. n = 7–16; individual brain values overlaid with population median±interquartiles; Tukey test.
-
Figure 2—figure supplement 1—source data 1
A mutation in Nrx1 does not underlie the aus reduced sleep phenotype.
- https://cdn.elifesciences.org/articles/64140/elife-64140-fig4-data4-v1.xlsx
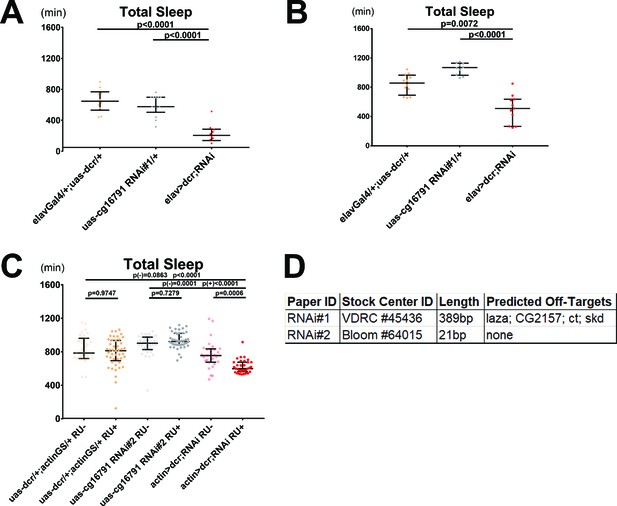
Knockdown of aus in adult neurons via RNAi reduces sleep.
(A–B) Pan-neuronal knockdown of cg16791 with elav-Gal4> dcr,cg16791-RNAi#1 reduced total sleep length in both female (A) and male (B) flies. n = 10–13 (females) and 10–12 (males); individual fly values overlaid with population median±interquartiles; Fischer’s LSD (females) or uncorrected Dunn’s test (males). (C) Total sleep is RU-inducibly reduced by whole-fly knockdown of cg16791 with actinGS> dcr,cg16791-RNAi#2, compared to controls. n = 28–45; individual fly values overlaid with median±interquartiles; Steel-Dwass test; p(-) indicates RU- p-values and p(+) indicates RU+ p-values. (D) UP-TORR bioinformatic off-target search (15 bp, mismatches allowed) for cg16791 RNAi’s. Includes stock center ID, length of inserted RNAi sequence, and predicted off-targets for each RNAi line.
-
Figure 2—figure supplement 2—source data 1
Knockdown of aus in adult neurons via RNAi reduces sleep.
- https://cdn.elifesciences.org/articles/64140/elife-64140-fig5-data5-v1.xlsx
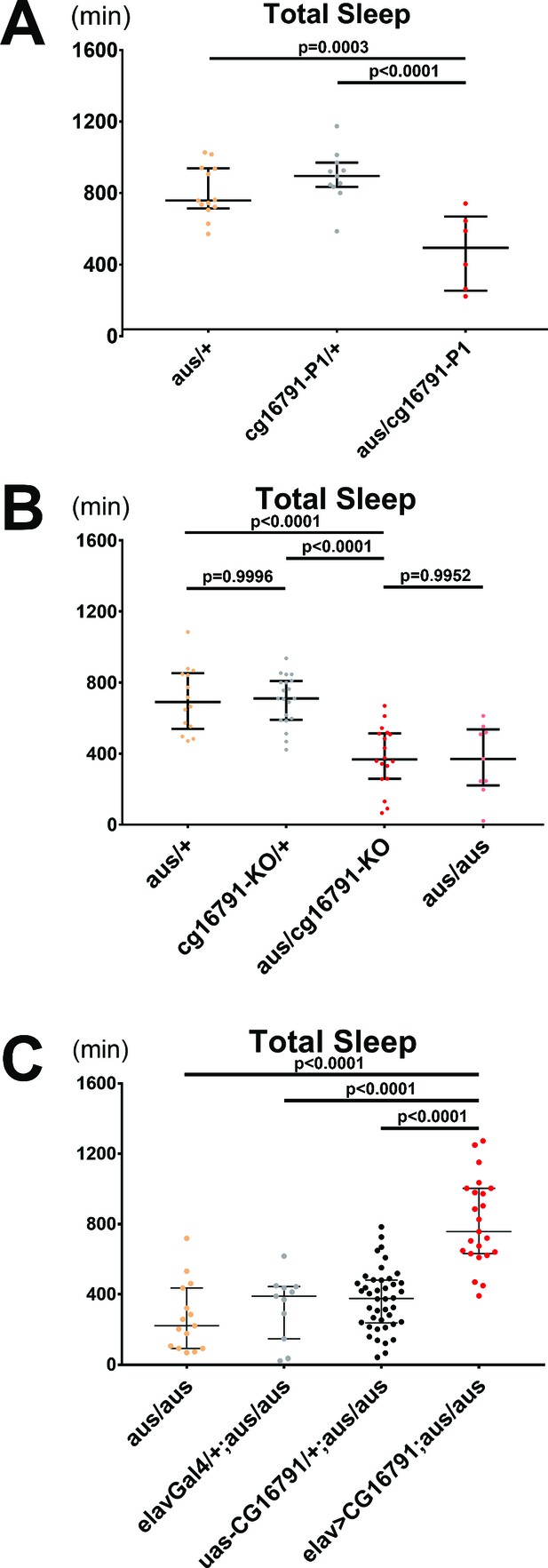
CG16791 underlies the argus sleep phenotype.
(A) Transheterozygotes of male aus and cg16791 insertional mutant (P1) have reduced total sleep compared to aus/+ and cg16791-P1/+ controls. n = 6–13; individual flies overlaid with median±interquartiles; Fischer’s LSD. (B) Female cg16791-KO and aus (EMS) transheterozygotes have reduced total sleep compared to aus (EMS) or cg16791-KO heterozygotes. Transheterozygote total sleep is comparable to aus homozygotes. n = 9–20; individual flies overlaid with median±interquartiles; Tukey test. (C) Pan-neuronal expression of uas-cg16791 with elav-Gal4 partially rescues female aus homozygote short-sleep, to significantly above aus-homozygous Gal4 and UAS controls. n = 11–42; individual flies overlaid with median±interquartiles; Fischer’s LSD.
-
Figure 3—source data 1
CG16791 underlies the argus sleep phenotype.
- https://cdn.elifesciences.org/articles/64140/elife-64140-fig3-data1-v1.xlsx

CRISPR-targeting of argus to generate a null mutant; supplemental Crispr-KO and full-length rescue data.
(A) CRISPR-targeting of argus exon1 to replace it with a selectable marker, Dsred. (B) Southern blot analysis of CG16791 (KO) using part of the Dsred gene as DIGI-probe. A single ~3 kb DIGI-positive band is expected for correct integration of Dsred at the argus locus. A small amount of DIGI label ( < 1 ng) was loaded in lane1 as a control. The Life Science 1 kb plus ladder was loaded in lane 2, and non-specific binding with DIGI probe was observed. iso31 gDNA digested by EcoRI in lane three or CG16791 KO/iso31 gDNA digested by EcoRI in lane 4. (C) Male cg16791-KO and aus (EMS) transheterozygotes have reduced total sleep compared to aus (EMS) or cg16791-KO heterozygotes. Transheterozygote total sleep is comparable to aus homozygotes. n = 9–16; individual flies overlaid with median±interquartiles; Tukey test. (D) Pan-neuronal expression of uas-cg16791(full-length) with elav-Gal4 partially rescues female aus homozygote short-sleep, to significantly above aus-homozygous Gal4 and UAS controls. n = 15–25; individual flies overlaid with median±interquartiles; Fischer’s LSD.
-
Figure 3—figure supplement 1—source data 1
CRISPR-targeting of argus to generate a null mutant; supplemental Crispr-KO and full-length rescue data.
- https://cdn.elifesciences.org/articles/64140/elife-64140-fig7-data7-v1.xlsx
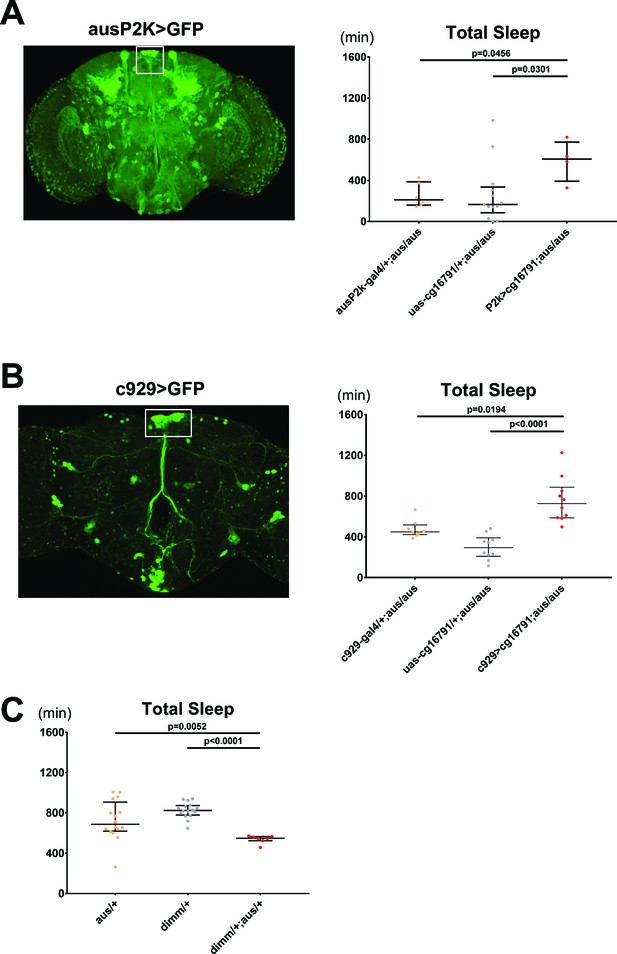
Argus functions in dimmed positive neurons to regulate sleep.
(A) The aus promoter region was subcloned, and a ~ 2000 bp sequence was inserted upstream of Gal4 and used to drive GFP (left). aus2kGal4 driving uas-cg16791 partially rescues the short sleep phenotype in female fruit flies (ausP2K, UAS-cg16791, or ausP2K > UAS-cg16791 in aus/aus mutant background). n = 4–13; individual flies overlaid with median±interquartiles; Fischer’s LSD. (B) C929-Gal4 (a peptidergic Gal4 line representing Dimmed expression) driving GFP (left). C929 driving uas-cg16791 expression rescues the short sleep phenotype in male aus flies. (c929, UAS-cg16791, or c929> UAS-cg16791 in aus/aus mutant background). n = 8–10; individual flies overlaid with median±interquartiles; uncorrected Dunn’s test. (C) aus and dimm interact genetically in female transheterozygotes to reduce sleep (aus/+, dimm/+, and aus dim transheterozygotes). n = 7–16; individual flies overlaid with median±interquartiles; uncorrected Dunn’s test.
-
Figure 4—source data 1
Argus Functions in Dimmed Positive Neurons to Regulate Sleep.
- https://cdn.elifesciences.org/articles/64140/elife-64140-fig4-data1-v1.xlsx
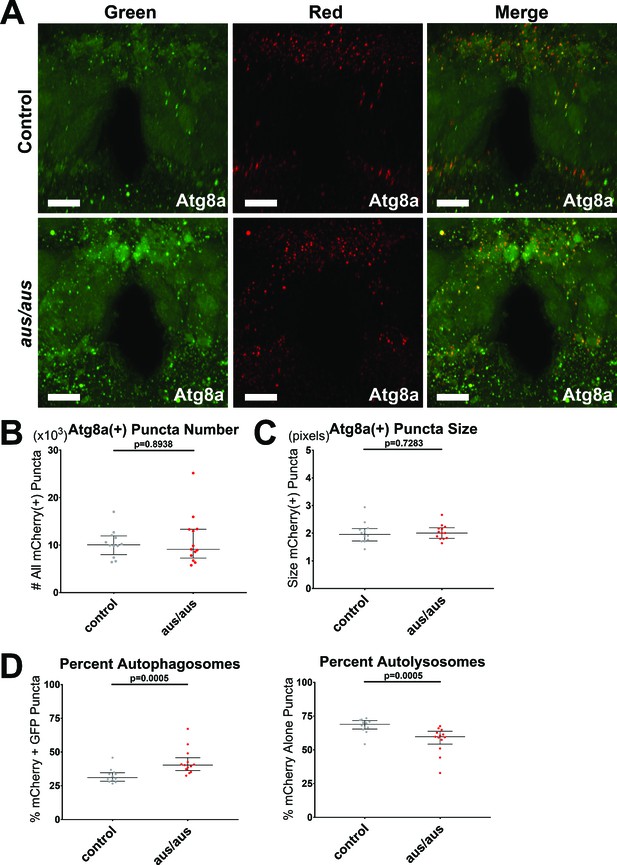
The argus mutant displays accumulation of autophagosomes.
Female iso31 control and aus/aus brains with elav-Gal4> UAS-GFP-mCherry-Atg8a driving pan-neuronal autophagy sensor were live imaged from ZT0-2. mCherry fluoresces in all Atg8a(+) puncta, while GFP fluoresces in autophagosomes and is quenched in autolysosomes. (A) Max-projected z-stacks of representative brains showing GFP (left), mCherry (middle), and merged (right) fluorescence. Scale bar = 25 um. (B) The number of all neuronal mCherry(+) puncta was similar in both genotypes. (C) The size of all neuronal mCherry(+) puncta was similar in both genotypes. (D) aus neuronal mCherry(+) puncta were significantly skewed toward % mCherry+ GFP(+) autophagosomes (left) and away from % mCherry-only(+) autolysosomes (right) compared to control. n = 12–13; individual brains overlaid with median±interquartiles; Mann-Whitney tests.
-
Figure 5—source data 1
The argus mutant displays accumulation of autophagosomes.
- https://cdn.elifesciences.org/articles/64140/elife-64140-fig5-data1-v1.xlsx
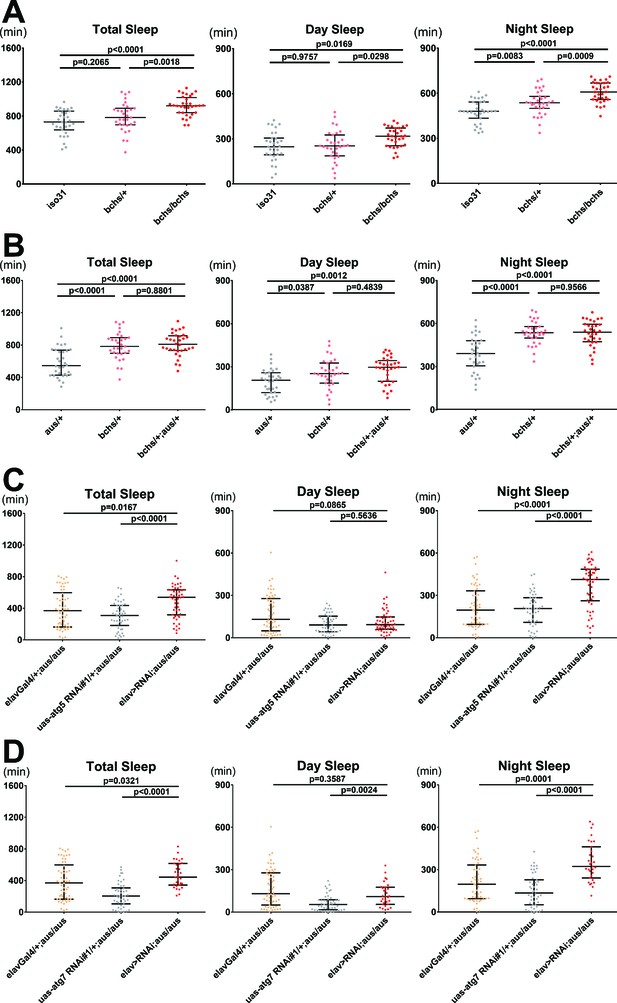
Blocking autophagosome production rescues the short sleep phenotype of the argus mutant.
(A) Total, day, and night sleep were measured under 12 hr:12 hr light:dark in iso31 control, bchs/+, and bchs/bchs female flies. n = 31–32; individual flies overlaid with median±interquartiles; Tukey tests. (B) Total, day, and night sleep were measured under 12 hr:12 hr light:dark in aus/+, bchs/+, and bchs/+; aus/+ transheterozygous female flies. n = 31–32; individual flies overlaid with median±interquartiles; Tukey tests. (C–D) Total, day, and night sleep were measured under 12 hr:12 hr light:dark in elav-Gal4/+, UAS-RNAi/+ and elav-Gal4> UAS RNAi female flies in aus/aus mutant background. RNAi’s used were atg5 RNAi#1 (C) and atg7 RNAi#1 (D). n = 46–54 (C) or n = 31–54 (D); individual flies overlaid with median±interquartiles; uncorrected Dunn’s tests.
-
Figure 6—source data 1
Blocking autophagosome production rescues the short sleep phenotype of the argus mutant.
- https://cdn.elifesciences.org/articles/64140/elife-64140-fig6-data1-v1.xlsx
Effects of aus and bchs on sleep consolidation, latency, and activity index.
(A–E) All sleep metrics were measured under a 12 hr:12 hr light:dark cycle in iso31 (gray), bchs/+ (pink) or bchs/bchs (red) flies. (A) Male total sleep amount during the whole 24-hr cycle (left), day (middle), and night (right). (B) Female (top) and male (bottom) mean sleep bout duration during the whole 24-hr cycle (left), day (middle) and night (right). (C) Female (top) and male (bottom) sleep bout number during the whole 24-hr cycle (left), day (middle) and night (right). (D) Female (left) and male (right) latency to first sleep bout after ZT12 lights off. (E) Female (left) and male (right) activity index of beam breaks per waking minute over the 24-hr cycle. (F) Activity index of beam breaks per waking minute over the 24-hr cycle in female aus/+ (gray), bchs/+ (pink) and transheterozygote (red) flies. n = 31–32; individual flies overlaid with median±interquartiles; Dunn tests (A–E) or Tukey test (F).
-
Figure 6—figure supplement 1—source data 1
Effects of aus and bchs on Sleep Consolidation, Latency, and Activity Index.
- https://cdn.elifesciences.org/articles/64140/elife-64140-fig11-data11-v1.xlsx

Rescue of aus mutants by atg5/atg7 RNAi.
(A–B) Total sleep amount with pan-neuronal atg5 (A) or atg7 (B) knockdown in female flies. elav+ Dicer2/+, UAS-atg RNAi/+ or elav+ Dicer2> UAS atg RNAi. n = 16–25 (atg5) or n = 16 (atg7); individual flies overlaid with median±interquartiles; Fischer’s LSD. (C–D) Activity index of beam breaks per waking minute over the 24-hr cycle in female elav-Gal4/+, UAS-atg RNAi/+, or elav-Gal4/UAS-atg RNAi on aus/aus background. atg5 RNAi (C) or atg7 RNAi (D). n = 46–54 (C) or n = 31–54 (D); individual flies overlaid with median±interquartiles; uncorrected Dunn’s test. (E–F) Total sleep amount in female elav-Gal4/UAS-atg RNAi, elav-Gal4/+, elav-Gal4/+; aus/+ or elav-Gal4/UAS-atg RNAi, aus/+. atg5 RNAi (E) or atg7 RNAi (F). n = 65–92 (E) or n = 35–95; individual flies overlaid with median±interquartiles; uncorrected Dunn’s test.
-
Figure 6—figure supplement 2—source data 1
Rescue of aus mutants by atg5/atg7 RNAi.
- https://cdn.elifesciences.org/articles/64140/elife-64140-fig12-data12-v1.xlsx
Blocking neuronal or whole-fly autophagosome formation increases sleep.
(A) Difference in first-pass population median sleep on RU+ food for a range of female nsybGS> dcr;autophagy-RNAi crosses compared with nsybGS> dcr control (x-axis) and RNAi control (y-axis). Red, numbered dots indicate significant hits that passed all validation steps: (1) bip RNAi#3; (2) atg1 RNAi#4; (3) daor RNAi#1; (4) atg1 RNAi#1; (5) atg10 RNAi#3. N = 133 viable crosses shown; n = 3–16 flies per group for each first-pass experiment. (B) Difference in first-pass population median sleep on RU+ food for a range of female actinGS> dcr;autophagy-RNAi crosses compared with actinGS> dcr control (x-axis) and RNAi control (y-axis). Red, numbered dots indicate significant hits that passed all validation steps: (1) dor RNAi#2; (2) atf6 RNAi#1; (3) atg8b RNAi#2; (4) wacky RNAi#2; (5) atg8b RNAi#1; (6) atg7 RNAi#1; (7) daor RNAi#1; (8) atg14 RNAi#3; (9) dram RNAi#2; (10) aduk RNAi#3; (11) atg12 RNAi#2. N = 106 viable crosses shown; n = 3–16 flies per group for each first-pass experiment. See Supplementary file 3 for details on first-pass screen and Figure 7—figure supplement 1 for combined first/second pass sleep data for significant hits, for the screens shown in both 7A and 7B. (C–F) Total (left), day (middle), and night (right) sleep in GS> dcr;RNAi, GS> dcr control, and RNAi control female flies on both RU+ and RU- food. All data shown as individual flies overlaid with median±interquartiles; p(-) indicates RU- p-values and p(+) indicates RU+ p-values. (C) nsybGS> dcr;atg1-RNAi#1: n = 31–32; Steel-Dwass test (total,night) and Tukey test. (day). (D) nsybGS> dcr;atg1-RNAi#4: n = 21–32; Steel-Dwass tests. (E) actinGS> dcr;atg8b-RNAi#1: n = 25–47; Steel-Dwass test (total,night) and Tukey test. (day). (F) actinGS> dcr;atg8b-RNAi#2: n = 17–32; Steel-Dwass tests.
-
Figure 7—source data 1
Blocking Neuronal or Whole-Fly Autophagosome Formation Increases Sleep.
- https://cdn.elifesciences.org/articles/64140/elife-64140-fig7-data1-v1.xlsx

Validated hits from autophagy RNAi screens.
(A–E) Total sleep amount in female UAS-dcr/+;nsybGS/+, UAS-RNAi/+, or nsybGS> dcr,RNAi flies on RU+ food. (F–P) Total sleep amount in female UAS-dcr/+;actinGS/+, UAS-RNAi/+, or actinGS> dcr,RNAi flies on RU+ food. All data shown is individual flies overlaid with median±interquartiles. (A) nsybGS> dcr;atg1-RNAi#1: n = 20–31; Student’s t-tests. (B) nsybGS> dcr;atg1-RNAi#4: n = 21–32; Student’s t-tests. (C) nsybGS> dcr;atg10-RNAi#3: n = 30–32; Student’s t-tests. (D) nsybGS> dcr;bip-RNAi#3: n = 18–31; Student’s t-tests. (E) nsybGS> dcr;daor-RNAi#1: n = 22–31; Student’s t-tests. (F) actinGS> dcr;aduk-RNAi#3: n = 31–32; Student’s t-tests. (G) actinGS> dcr;atf6-RNAi#1: n = 30–32; Student’s t-tests. (H) actinGS> dcr;atg7-RNAi#1: n = 31; Mann-Whitney tests. (I) actinGS> dcr;atg8b-RNAi#1: n = 29–31; Student’s t-tests. (J) actinGS> dcr;atg8b-RNAi#2: n = 20–32; Student’s t-tests. (K) actinGS> dcr;atg12-RNAi#2: n = 28–31; Student’s t-tests. (L) actinGS> dcr;atg14-RNAi#3: n = 28–32; Student’s t-tests. (M) actinGS> dcr;daor-RNAi#1: n = 9–31; Student’s t-tests. (N) actinGS> dcr;dor-RNAi#2: n = 19–31; Student’s t-tests. (O) actinGS> dcr;dram-RNAi#2: n = 30–31; Mann-Whitney tests. (P) actinGS> dcr;wacky-RNAi#2: n = 20–32; Mann-Whitney tests.
-
Figure 7—figure supplement 1—source data 1
Validated hits from autophagy RNAi screens.
- https://cdn.elifesciences.org/articles/64140/elife-64140-fig14-data14-v1.xlsx
atg1 and atg8b RNAi additional sleep metrics, activity index, and validation of knockdown.
Additional metrics comparing GS+ UAS dcr control, UAS-RNAi control, and GS> dcr,RNAi females back-crossed to iso31, on both RU+ and RU- food. All data shown as individual flies overlaid with median±interquartiles (A–E, G–K, M–Q,S–W) or individual biological replicates (F,L,R,X); p(-) indicates RU- p-values and p(+) indicates RU+ p-values. (A–F) atg1-RNAi#1: experiments with nsybGS; n = 31–32 (A–E) or actinGS; n = 2 (F). (G–L) atg1-RNAi#4: experiments with nsybGS; n = 21–32 (G–K) or actinGS; n = 2 (L). (M–R) atg8b-RNAi#1: experiments with actinGS; n = 25–47 (M–Q) or actinGS; n = 2 (R). (S–X) atg8b-RNAi#2: experiments with actinGS; n = 17–32 (S–W) or actinGS; n = 2 (X). (A,G,M,S) Mean sleep bout duration during the whole 24-hr cycle (left), day (middle) and night (right). All comparisons are Steel-Dwass tests. (B,H,N,T) Sleep bout number during the whole 24-hr cycle (left), day (middle) and night (right). Comparisons are Tukey test (atg1 RNAi#1 day-bouts) or Steel-Dwass tests (all other comparisons). (C,I,O,U) Longest sleep bout during the whole 24-hr cycle. Comparisons are Tukey test (atg1 RNAi#1) or Steel-Dwass tests (all other comparisons). (D,J,P,V) Latency to first sleep bout after ZT12 lights off. All comparisons are Steel-Dwass tests. (E,K,Q,W) Activity index of beam breaks per waking minute over the 24-hr cycle. All comparisons are Steel-Dwass tests. (F,L,R,X) Relative cDNA expression level in whole-fly lysate. All comparisons are one-tailed t-tests.
-
Figure 7—figure supplement 2—source data 1
atg1 and atg8b RNAi additional sleep metrics, activity index, and validation of knockdown.
- https://cdn.elifesciences.org/articles/64140/elife-64140-fig15-data15-v1.xlsx
Sleep regulates autophagosome production.
elav-Gal4> UAS-GFP-mCherry-Atg8a flies expressing pan-neuronal autophagy sensor were live imaged as follows. All quantification shows individual brain values overlaid with population median±interquartiles. (A–D) ZT0-2 or ZT12-14. n = 15; Student’s t-tests. (E–H) ZT0-2 after either a control night of unchallenged sleep or at least 12 hr of mechanical sleep deprivation (SD) beginning at the prior ZT12. n = 13–20; Student’s t-tests. (I–L) ZT12-14 after either a control day of feeding with vehicle or at least 11 hr of feeding with 0.1 mg/mL gaboxadol that verifiably and markedly increased daytime sleep, beginning at the prior ZT0-1. n = 25–26. (A,E,I) Max-projected z-stacks of representative brains showing GFP (left), mCherry (middle), and merged (right) fluorescence for ZT time comparison (A), control vs SD (E), or vehicle vs gaboxadol (I). Scale bars = 25 µm. (B,F,J) The number of all neuronal mCherry(+) puncta was higher at nightfall than daybreak (B), elevated at daybreak by 12 hr overnight SD (F), and depressed at nightfall by 12 hr daytime of gaboxadol-induced sleep (J). (C,G,K) The size of all neuronal mCherry(+) puncta was unaffected by ZT time, SD, and gaboxadol. (D,H,L) The percentage of neuronal mCherry(+) puncta that are mCherry+ GFP(+) autophagosomes (left) and mCherry-only(+) autolysosomes (right) was unaffected by ZT time, SD, and gaboxadol.
-
Figure 8—source data 1
Sleep regulates autophagosome production.
- https://cdn.elifesciences.org/articles/64140/elife-64140-fig8-data1-v1.xlsx
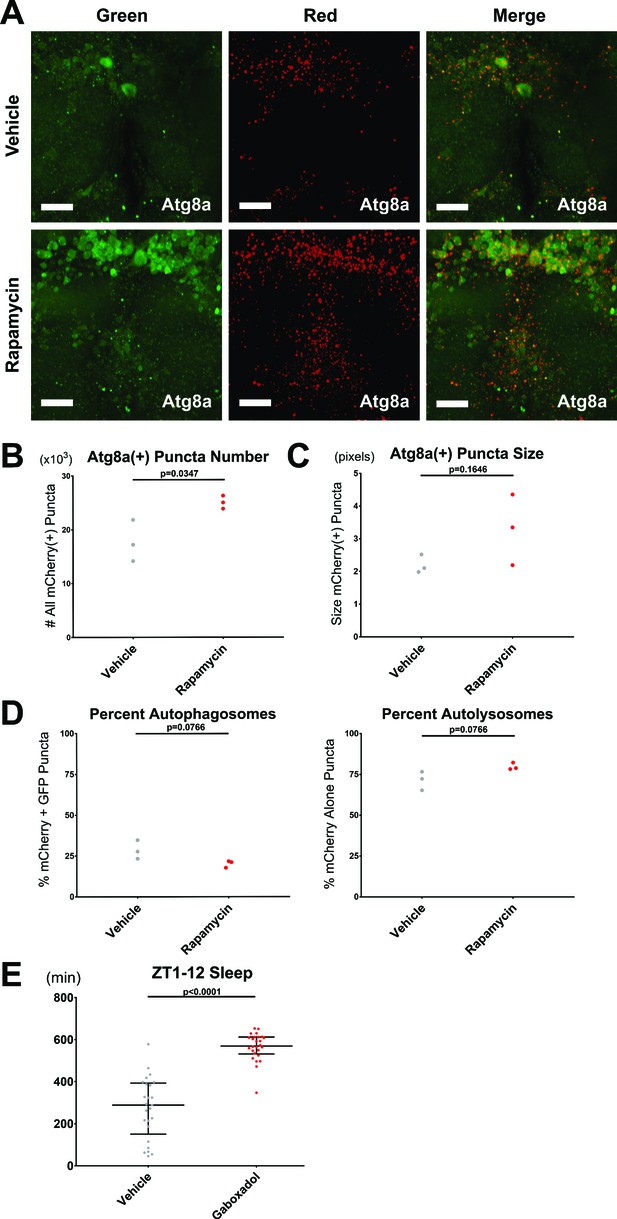
Validation of the Ilastik algorithm for scoring autophagy and the gaboxadol effect on sleep.
(A–D) Female elav-Gal4> UAS-GFP-mCherry-Atg8a brains driving pan-neuronal autophagy sensor were live imaged from ZT2-4, after ~2 hr of pre-incubation in either vehicle or 2 µM rapamycin supplemented AHL. mCherry fluoresces in all Atg8a(+) puncta, while GFP fluoresces in autophagosomes and is quenched in autolysosomes. n = 3; individual brains. (A) Max-projected z-stacks of representative brains showing GFP (left), mCherry (middle), and merged (right) fluorescence. Scale bar = 25 um. (B) The number of all neuronal mCherry(+) puncta was significantly increased by rapamycin treatment. (C) The size of all neuronal mCherry(+) puncta were similar in both groups. (D) The percentages of mCherry+ GFP(+) autophagosomes (left) and mCherry-only(+) autolysosomes (right) were similar in both groups. (E) Total sleep amount was measured in the same flies later dissected for live imaging in Figure 8I–L, from ZT1 (shortly after flip onto either vehicle or gaboxadol-laced food) to ZT12 (when the first fly live imaged was removed from the sleep monitors for dissection). As expected, gaboxadol feeding robustly increased sleep during this window. n = 25–26; individual flies overlaid with median±interquartiles; Student’s t-test.
-
Figure 8—figure supplement 1—source data 1
Validation of the Ilastik algorithm for scoring autophagy and the gaboxadol effect on sleep.
- https://cdn.elifesciences.org/articles/64140/elife-64140-fig17-data17-v1.xlsx
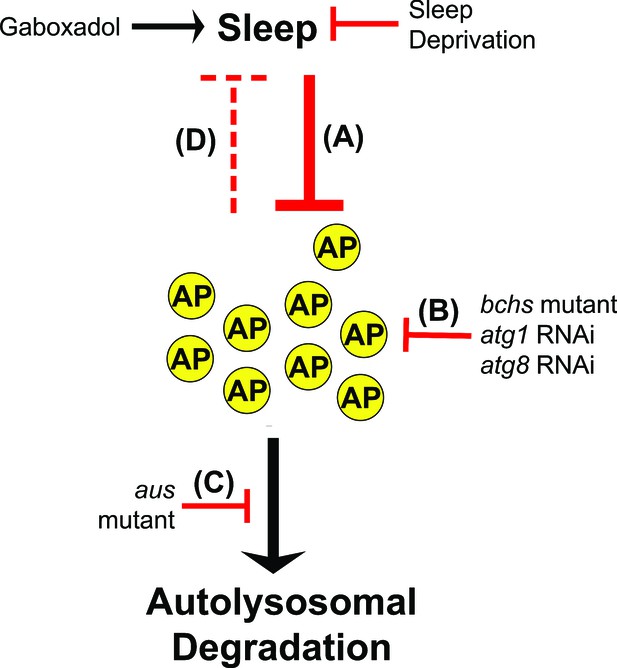
Model for sleep-autophagy interaction.
This schematic details our model for how sleep and macroautophagy interact, based on our results. (A) Sleep decreases autophagosome number under normal conditions, in a manner that is sensitive to both gaboxadol gain or SD loss of sleep lasting between 11 and 14 hr. (B) The mutant blue cheese, pan-neuronal RNAi for atg1, and whole-fly RNAi for atg8b (suppressing both 8a and 8b homologs) are all known to inhibit autophagosome formation, and all increase sleep. (C) Neuronal loss-of-function in the aus mutant inhibits autophagosome degradation, and decreases sleep in a manner that is rescued by blocking autophagosome formation upstream. (D) The wake-promoting / sleep-inhibiting effects of autophagosome number are able to drive sleep behavior when strongly and sustainably adjusted by our genetic manipulations, but are unable to drive sustained waking after a single night of SD, as acute rebound sleep is well established to occur after sleep deprivation on this timescale. Together, this suggests that autophagosome inhibition of sleep is considerably weaker than sleep inhibition of autophagosome accumulation, with autophagosome number only becoming a strong enough signal to control sleep behavioral output with a very strong and/or sustained stimulus.
Additional files
-
Supplementary file 1
Lines and Primers.
Tab 1: A figure-by-figure breakdown of alleles, sources, and backgrounds for each fly line used in most figures of the manuscript. Tab2: A list of all primer sequences used in producing and validating the novel fly lines described in the manuscript.
- https://cdn.elifesciences.org/articles/64140/elife-64140-supp1-v1.xlsx
-
Supplementary file 2
Bioinformatic analysis of the CG16791/ Aus protein product.
An unbiased ProDom analysis of the full-length CG16791, Isoform A protein sequence identified a number of candidate transmembrane domains. Validation with TMPred produced a similar 5-transmembrane best-fit topological prediction for all naturally occurring isoforms of CG16791, as well as our UAS-aus construct protein product. Deep-Loc-1.0 predicted the cell membrane as the most likely initial insertion site for all of these same CG16791 sequences, with the endoplasmic reticulum and Golgi apparatus as possible alternative insertion sites.
- https://cdn.elifesciences.org/articles/64140/elife-64140-supp2-v1.xlsx
-
Supplementary file 3
Autophagy RNAi Screen, First-Pass Sleep for All Crosses.
Tab 1: A list of all RNAi’s used in the screens, including unambiguous stock center IDs. Tab 2: First-pass medians, interquartiles, and n’s for total sleep in females on RU+ food for each nsybGS> dcr,RNAi cross with appropriate controls. Crosses that passed primary criterion are indicated, and annotated with whether they passed subsequent criteria or not and, if not, why. Tab 3: First-pass medians, interquartiles, and n’s for total sleep in females on RU+ food for each actinGS> dcr,RNAi cross with appropriate controls. Crosses that passed primary criterion are indicated, and annotated with whether they passed subsequent criteria or not and, if not, why.
- https://cdn.elifesciences.org/articles/64140/elife-64140-supp3-v1.xlsx
-
Transparent reporting form
- https://cdn.elifesciences.org/articles/64140/elife-64140-transrepform1-v1.docx





