Residue-by-residue analysis of cotranslational membrane protein integration in vivo
Figures
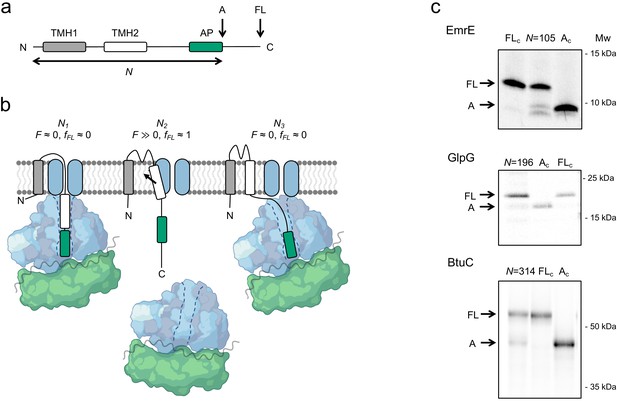
The force profile assay.
(a) Basic construct. Arrested (A) and full-length (FL) products are indicated. (b) At construct length N1, TMH2 has not yet entered the SecYEG channel and no pulling force F is generated. At N2, TMH2 is integrating into the membrane and F ≫0. At N3, TMH2 is already integrated and F ≈ 0. (c) SDS-PAGE gels showing A and FL products for [35S]-Met labeled and immunoprecipitated EmrE(Cout) (N = 105), GlpG (N = 196), and BtuC (N = 314). Control constructs AC and FLc have, respectively, a stop codon and an inactivating Ala codon replacing the last Pro codon in the arrest peptide (AP). The band just below the A band in the EmrE(Cout) (N = 105) lane most likely represents ribosomes stacked behind the AP-stalled ribosomes (Notari et al., 2018) and is not included in the calculation of fFL. See Figure 1—figure supplement 1 for additional gels.
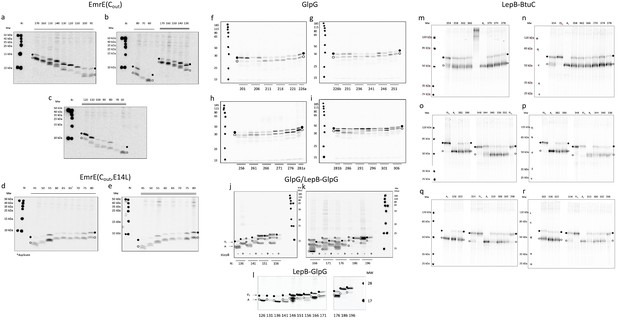
Gel gallery with selected EmrE (a–e), GlpG (f–l), and BtuC (m–r) constructs.
Full-length (FL) and arrested (A) products are indicated by black and white circles, respectively. Repeat experiments are indicated by single and double lines above the lanes for EmrE (a–e), by short lines beneath the lanes for GlpG (f–i), and are on neighboring gels for BtuC. For BtuC, some full-length (FLc) and arrest (Ac) controls are included (c.f., Figure 1c); these are for the construct immediately to the right of the control construct lanes. GlpG (j, k) show GlpG constructs (indicated by –) and the corresponding LepB-GlpG constructs (indicated by +) for N = 136–196. For N = 141–186, GlpG constructs have the expected full-length band (lower gray circles) that runs slightly faster than the corresponding LepB-GlpG construct (black circles), plus an extra band of unknown provenance that runs more slowly than the full-length LepB-GlpG construct (upper gray circles). The GlpG fFL values shown in Figure 3—figure supplement 1a were calculated by assigning only the lower (dashed magenta curve), or both (dashed green curve), of the bands indicated by gray circles as full-length product. Panel l shows a repeat experiment for the LepB-GlpG constructs included in panels j and k.
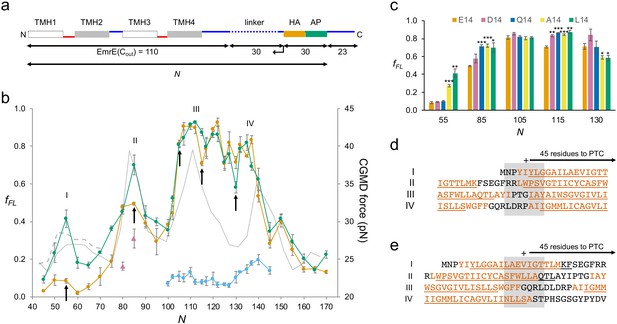
EmrE(Cout).
(a) Construct design. EmrE(Cout) is shortened from the C-terminal end of the LepB-derived linker (dotted), as indicated by the arrow. Cytoplasmic (red) and periplasmic (blue) loops, and lengths of full-length EmrE(Cout), LepB-derived linker, HA tag + arrest peptide (AP), and C-terminal tail, are indicated. Since the 30-residue HA + AP segment is constant in all constructs, the force profile (FP) reflects nascent chain interactions occurring mainly outside the ribosome exit tunnel. (b) FPs for EmrE(Cout) (orange), EmrE(Cout,E14L) (green), EmrE(Cout) with SecM(Ec-sup1) AP (blue), EmrE(Cout, I37I38→NN) (magenta triangles), and coarse-grained molecular dynamics (CGMD-FP) calculated with a −100 mV membrane potential (gray). (c) Effects of mutations in E14 on fFL values for the N values are indicated by arrows in (b). p-values (two-sided t-test): *p < 0.05; **p < 0.01; ***p < 0.001. (d, e) Sequences corresponding to peaks I–IV aligned from their Nstart (d) and Nend (e) values. The + sign indicates 45 residues from the polypeptide transferase center (PTC). Hydrophobic transmembrane helix (TMH) segments are shown in orange and transmembrane α-helices underlined (PDB: 3B5D). Error bars in b and c indicate SEM values.
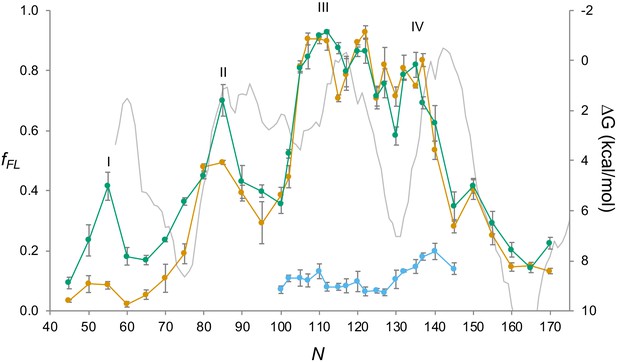
EmrE(Cout).
As in Figure 2b, but with a hydrophobicity plot (HP) (ΔG) calculated by TOPCONS (3, 50) (gray). Since the HP represents the membrane integration energy, and the force profile (FP) the force generated during integration, the two profiles have been aligned such that peaks in the FP approximately align with maxima in the derivative of the HP.
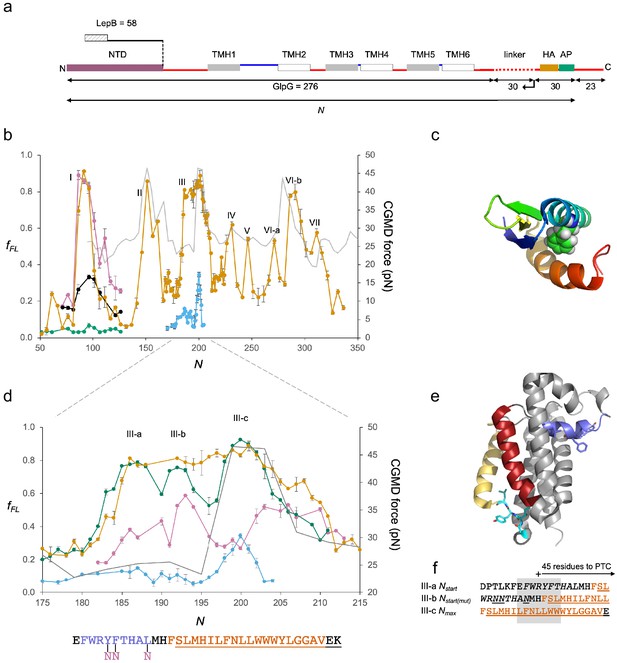
GplG.
(a) Construct design, c.f., Figure 2a. The N-terminal LepB fusion is indicated. (b) Force profiles (FPs) for GlpG and LepB-GlpG (N = 131–224) (orange), NTD(F16E) (green), in vitro translated N-terminal domain (NTD) (magenta), and NTD(F16E) (black), LepB-GlpG with SecM(Ec-Sup1) AP (blue), and coarse-grained molecular dynamics (CGMD)-FP calculated with a −100 mV membrane potential (gray). Error bars indicate SEM values. Note that the LepB-GlpG constructs are two residues shorter than the corresponding GlpG constructs but are plotted with the same N values as the latter to facilitate comparison. (c) NTD (PDB ID: 2LEP), with F16 in spacefill. (d) Enlarged FPs for LepB-GlpG with SecM(Ec) AP (orange), SecM(Ec-Ms) AP (green), SecM(Ec-sup1) AP (blue), and GlpG(Y138F139L143→NNN) with SecM(Ec-Ms) AP (magenta). CGMD-FP in gray. (e) Structure of GlpG with the periplasmic surface helix in blue, TMH2 in red, the membrane-associated cytoplasmic segment in cyan, and TMH5 in yellow. Y138F139L143 and G222I223Y224L225 are shown as sticks. (f) LepB-GlpG peak III-a and III-c sequences aligned, respectively, from their Nstart and Nmax values, and the mutant LepB-GlpG(Y138F139L143→NNN) peak III-c sequence aligned from its Nmax value. Hydrophobic transmembrane helix (TMH) segments are shown in orange and transmembrane α-helices (PDB: 2IC8)underlined. The periplasmic surface helix is italicized. AP: arrest peptide; PTC: polypeptide transferase center.
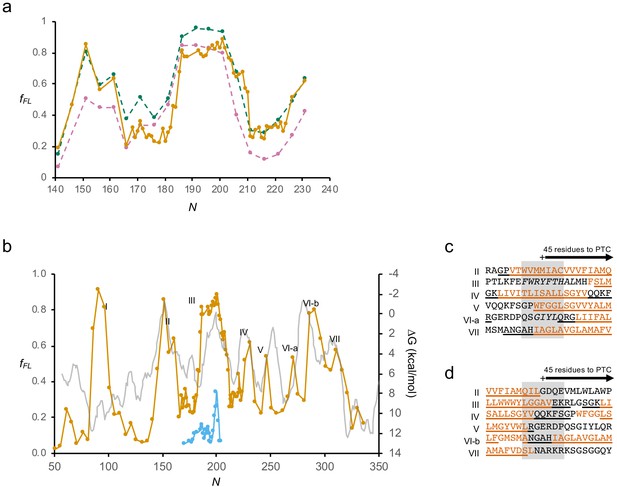
GlpG.
(a) fFL values for peak III obtained for LepB-GlpG fusion constructs (orange) and GlpG constructs calculated either including (dashed green) or excluding (dashed magenta) the slowly migrating band indicated in Figure 1—figure supplement 1j,k in IFL. The two latter are from single measurements. (b) As in Figure 3b, but with a hydrophobicity plot (HP) (ΔG) calculated by TOPCONS (Hessa et al., 2007; Tsirigos et al., 2015) (gray). (c) Sequences corresponding to peaks II–VII aligned based on the Nstart values. The periplasmic surface helix upstream of TMH2 and the hydrophobic patch upstream of TMH5 are in italics. Hydrophobic transmembrane helix (TMH) segments are shown in orange and membrane-embedded α-helices underlined. (d) Sequences corresponding to peaks II–VII aligned based on the Nend values. Hydrophobic TMH segments are shown in orange and membrane-embedded α-helices underlined. PTC: polypeptide transferase center.
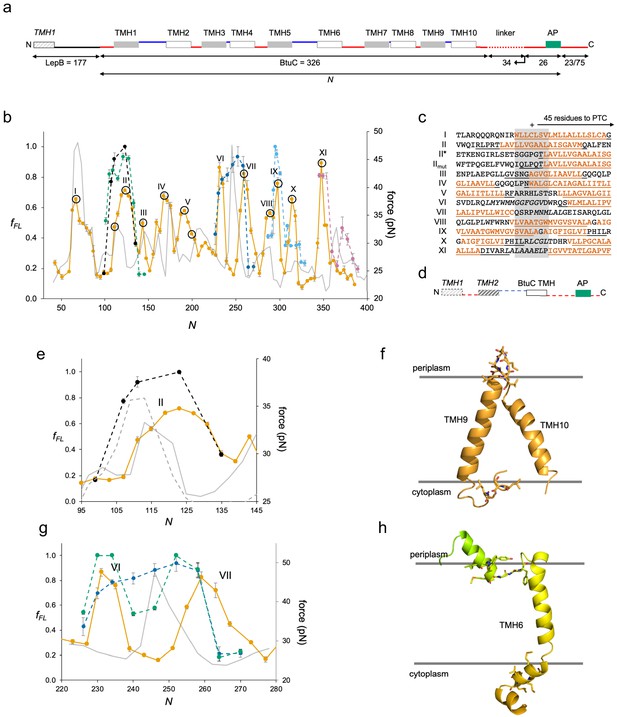
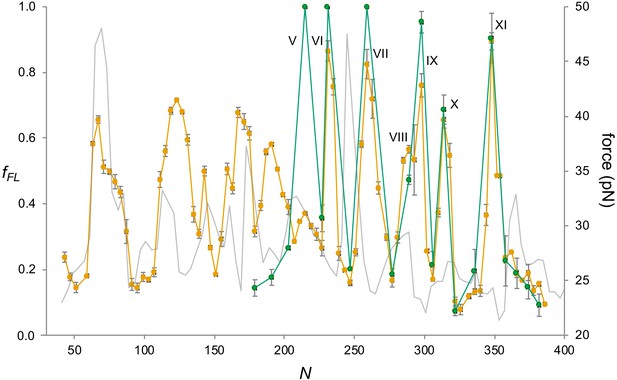
BtuC.
(a) Construct design, cf. Figure 2a. The N-terminal LepB fusion is indicated. N values are calculated from the N-terminus of BtuC. For constructs with N ≥ 298, the C-terminal tail is 75 residues long. Circles indicate constructs for which mutations were made in the corresponding transmembrane helix (TMH) (see Figure 4—figure supplement 2. (b) Force profiles (FPs) for BtuC (orange), BtuC-TMH2 (green), BtuC(R47R56R59→QQQ) (black), BtuC-TMH6 (dark blue), BtuC-TMH8 (blue), BtuC-TMH10 (pink), and CGMD-FP calculated with a −100 mV membrane potential (gray). Error bars indicate SEM values. Note that the BtuC-TMH2, BtuC-TMH6, BtuC-TMH8, and BtuC-TMH10 constructs are plotted with the same N values as the corresponding BtuC constructs to facilitate comparison (i.e., the number of residues between the TMH in question and the last residue of the AP is the same in both types of constructs, see Supplementary file 1). (c) Sequences corresponding to peaks I–XI aligned from their Nstart values. Hydrophobic TMH segments are shown in orange and membrane-embedded α-helices according to the OPM database (Lomize et al., 2012) underlined. Re-entrant loops and surface helices discussed in the text are italicized. (d) Construct design for obtaining FPs of isolated Nout-oriented BtuC TMHs. Dashed segments are derived from LepB. (e) Enlarged FPs for BtuC (orange) and (R47R56R59→QQQ) (black), together with coarse-grained molecular dynamics (CGMD)-FPs calculated with (gray) and without (dashed gray) a −100 mV potential. (f) BtuC TMH9-TMH10, with hydrophobic flanking residues in stick representation (PDB ID: 2QI9). (g) Enlarged FPs for BtuC (orange), isolated TMH6 (residues 187–206; blue), and isolated TMH5-6 (residues 138–206; green). In the latter construct, LepB TMH2 was not included in order to maintain the correct membrane topology of the BtuC TMH5-TMH6 part. The CGMD-FP is in gray. (h) Structure of TMH6 including the upstream periplasmic re-entrant helix and the downstream cytoplasmic surface helix, with hydrophobic flanking residues in stick representation. AP: arrest peptide; PTC: polypeptide transferase center.
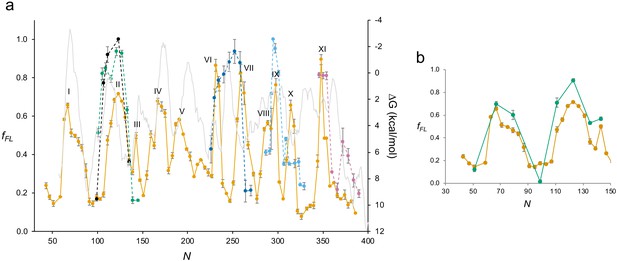
BtuC.
(a) As in Figure 4b, but with a hydrophobicity plot (ΔG) calculated by TOPCONS (3, 50) (gray). (b) Close-up view of the BtuC force profile (FP) (N = 30–150; orange), and the corresponding FP obtained with BtuC constructs lacking the N-terminal LepB fusion (green). For the latter, an HA tag was included just upstream of the arrest peptide, and cells were radiolabeled with [35S]-Met for 1 min before trichloroacetic acid precipitation.
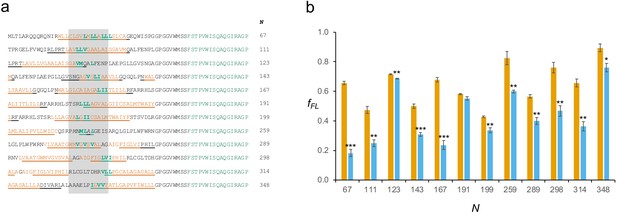
Mutations in constructs representing peaks in the BtuC force profile (FP).
(a) Sequences of the 67 residues leading up to the end of the arrest peptide (AP) for constructs with the indicated N-values. The constructs are identified by black circles on the BtuC FP in Figure 4b. For each construct, the residues indicated in bold green were simultaneously mutated to Ala. The shaded area encompasses residues located 40–50 residues away from the C-terminal end of the AP in the respective constructs. Hydrophobic transmembrane helix segments are shown in orange and membrane-embedded α-helices underlined. (b) fFL values for the unmutated constructs (orange) and the Ala-replacement mutants (blue). Error bars indicate SEM values, and stars indicate p-values calculated using a two-sided t-test (*p < 0.05; **p < 0.01; ***p < 0.001).
BtuC.
As in Figure 4b, with the force profile (FP) for construct BtuC(ΔTMH1-TMH4) in green.
Tables
| Reagent type (species) or resource | Designation | Source or reference | Identifiers | Additional information |
|---|---|---|---|---|
| Strain, strain background (Escherichia coli) | BL21(DE3) | Sigma-Aldrich | CMC0016 | Electrocompetent cells |
| Strain, strain background (Escherichia coli) | MC1061 | J Biol Chem. 261:13844–9. PMID:3531212 | NA | Electrocompetent cells |
| Other | Protein-G-agarose | Roche | 11243233001 | Resin used for immunoprecipitation |
| Antibody | Anti-HA.11 epitope tag antibody (mouse monoclonal) IgG | BioLegend | Cat# 901533 | Used for immunoprecipitation (1 μl of 1 mg/ml, diluted 1:820) |
| Antibody | LepB antibody (rabbit polyclonal) IgG | Generated in-house | NA | Used for immunoprecipitation (dilution 1:820) |
| Recombinant DNA reagent | pET Duet-1 (plasmid) | Novagen | Cat# 71146 | Expression plasmid |
| Recombinant DNA reagent | pING1 (plasmid) | Gene 34:137–45. PMID:4007491 | NA | Expression plasmid |
| Commercial assay, kit | GeneJET Plasmid miniprep kit | Thermo Fisher Scientific RRID: SCR_008452 | Cat# 0502 | Used to purify plasmids |
| Commercial assay, kit | GeneJET PCR Purification Kit | Thermo Fisher Scientific | Cat# K0701 | Used to purify linear fragments for in vitro expression |
| Commercial assay, kit | PURExpress | New England Biolabs | Cat# E6800L | Used for in vitro expression |
| Chemical compound, drug | 35S methionine | PerkinElmer | Cat# NEG009T001MC | 35S Methionine is incorporated into the protein during in vitro and in vivo translation and aids detection by phosphor imaging |
| Software, algorithm | EasyQuant | Developed in-house Nat Struct Mol Biol. 19:1018–22. PMID: 23001004 | Used to quantify relative fraction full length of translated protein from SDS-PAGE |
Additional files
-
Source data 1
Measured fFL values for all EmrE, GlpG, and BtuC constucts reported in Figures 2, 3 and 4 (and the corresponding Supplementary Figures and Supplementary file 1).
- https://cdn.elifesciences.org/articles/64302/elife-64302-data1-v2.xlsx
-
Supplementary file 1
Amino acid sequences of EmrE(Cout), GlpG, and BtuC constructs.
- https://cdn.elifesciences.org/articles/64302/elife-64302-supp1-v2.docx
-
Transparent reporting form
- https://cdn.elifesciences.org/articles/64302/elife-64302-transrepform-v2.docx





