Nanoscale binding site localization by molecular distance estimation on native cell surfaces using topological image averaging
Figures
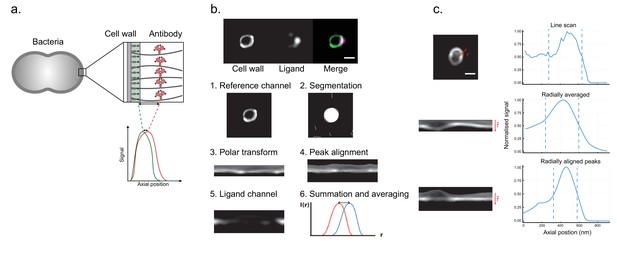
Binding site localization is based on resolving the distance between bound ligands and a reference surface.
(a) Site localization on bacterial surfaces. The illustration exemplifies a measurement with antibody-coated bacteria. Antibodies with red fluorescent dyes are bound to bacterial surface proteins. A bacterial cell wall is labelled with a green fluorescent dye. High-resolution images of single bacteria are acquired in the focal plane, and the fluorescent peak signal is averaged along the bacterial contour. The distance d is determined by resolving the difference between the fluorescent signal peaks. (b) Analysis pipeline for the site localization method. Structured illumination microscopy (SIM) images are shown at the top. S. pyogenes strain SF370 has been fixed, stained with Alexa Fluor 488-conjugated wheat germ agglutinin (WGA), and coated with antibody Fc fragments. The antibody fragments were stained with Fc-specific Alexa Fluor 647-conjugated F(ab’)2 fragments. The scale bar is 500 nm. The aim is to locate the antibody binding site by calculating the average distance between the antibody channel and a reference signal channel. (1) A raw image in the reference channel is shown. (2) Bacteria are identified by fitting circles to an edge-detection-processed image. (3) The data in each circle mask is isolated and transformed to polar coordinates. (4) An alignment of the reference position is performed by identifying peaks at each radial position. (5) The spatially corresponding data in the antibody channel is extracted. (6) The radially aligned peaks are then averaged. The peak distance between these intensity profiles should then correspond to the distance between the binding site and the bacterial peptidoglycan layer. (c) Representation of improved SNR by peak alignment for non-spherical particles Intensity profiles (right) for the reference channel is shown together with their respective image data (left). The signal along a single line for an oval-shaped bacterium is shown at the top. The intensity profile of a radial average is shown in the middle. An improved SNR is seen as a peak alignment is performed (bottom).
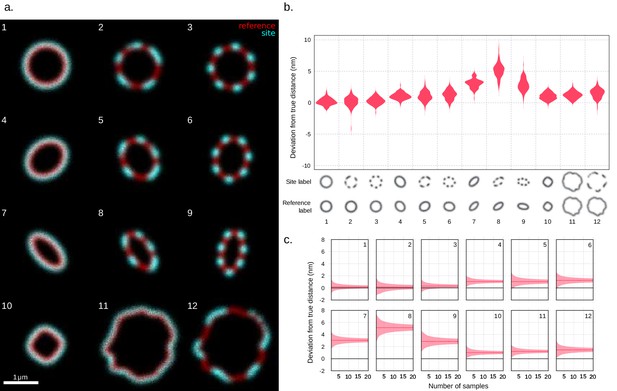
Validation of site localization method through simulated images with various cell morphologies.
(a) Examples of simulated images. The different shapes that the method was tested on are shown. From each of the 12 morphologies, 100 time series with 10 frames each were generated. Each frame consists of a set number of photons sampled from the shape’s spatial distribution. Before creating a time series, a random translation and rotation were applied to the distribution. The chosen shapes emulate various eccentricities and degrees of surface protein patchiness found, for example, in bacteria as well as the surface irregularity of larger cells. The 1 µm scale bar assumes a pixel length of 20.5 nm corresponding to that of the structured illumination microscopy (SIM) images, with this pixel length the simulated distance between the reference channel and the target channel is 41 nm. (b) Site localization results expressed as deviation from simulated distances. The deviations of the measured values from the simulated distances are shown in a violin plot grouped by shape. The mean distance deviations are in order from left to right [mean ± SD, median (IQR)]: 0.2 ± 0.5 nm, 0.1 (0.7) nm; 0.2 (1.0) nm; 0.3 ± 0.6 nm, 0.3 (0.8) nm; 1.1 ± 0.5 nm, 1.0 (0.7) nm; 1.0 ± 0.8 nm, 1.0 (1.1) nm; 1.2 ± 0.8 nm, 1.3 (1.1) nm; 3.0 ± 0.7 nm, 3.1 (0.9) nm; 5.1 ± 1.1 nm, 5.2 (1.3) nm; 2.9 ± 1.0 nm, 2.8 (1.2) nm; 1.0 ± 0.6 nm, 1.0 (0.7) nm; 1.2 ± 0.6 nm, 1.2 (0.8) nm; 1.5 ± 0.8 nm, 1.6 (1.1) nm. (c) Bootstrap of measured mean distances as a function of the number of time series used. The plots show an estimate of the 95% confidence interval of the mean of a number of measurements computed by percentile bootstrap with 50,000 resamples. As the number of measurements used in the calculation of the mean increases, the confidence interval narrows.
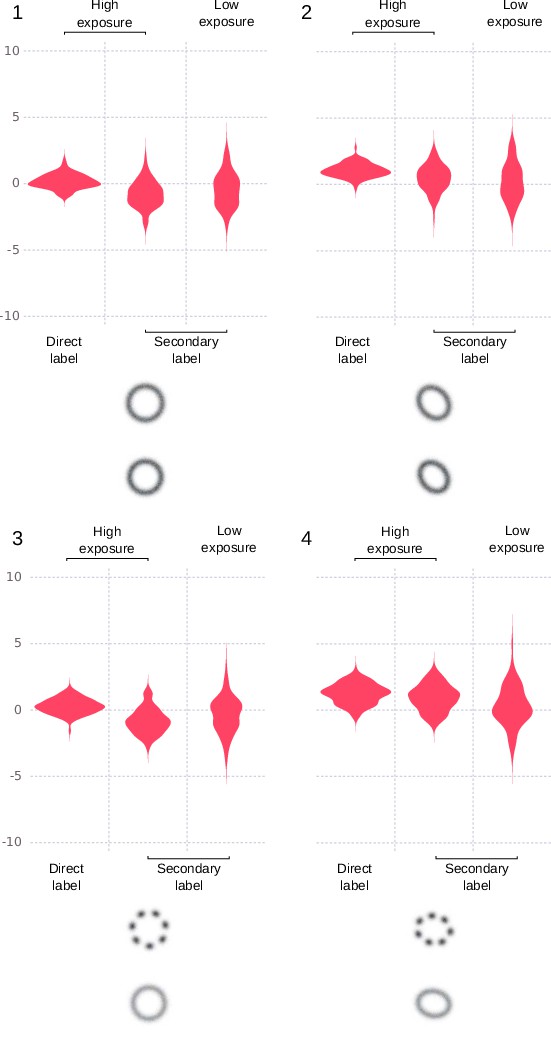
Site localization results on simulated images expressed as deviation from simulated distance for different types of labelling and signal.
The deviations of the measured values from the simulated distance are shown in violin plots grouped by shape. For each shape, the result is shown for parameters corresponding to direct labelling of the binding site, parameters corresponding to labelling with a secondary antibody, and parameters corresponding to labelling with a secondary antibody and low exposure. The median distance deviations are in order from left to right in each subplot [median (IQR)]: 1: 0.1 (0.7) nm, –0.8 (1.3) nm, 0.4 (1.8) nm. 2: 1.0 (0.7) nm, 0.5 (1.3) nm, 0.1 (2.0) nm. 3: 0.3 (0.8) nm, –0.9 (1.1) nm, –0.2 (1.7) nm. 4: 1.3 (1.1) nm, 1.1 (1.2) nm, 0.2 (1.9) nm.
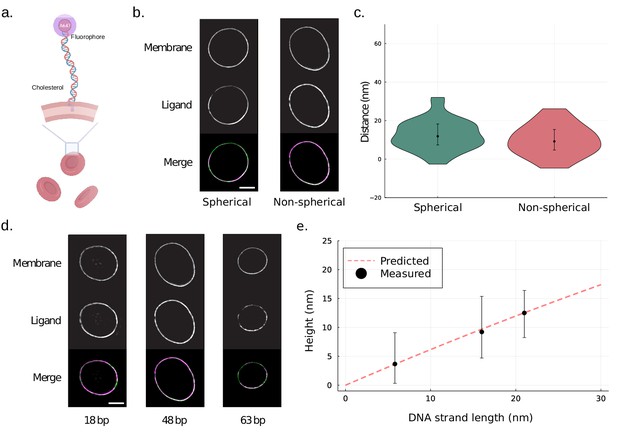
Site localization measurements of DNA probes on human red blood cells.
(a) A schematic illustrating the binding of DNA probe to red blood cells. Human red blood cells were coated with Alexa Fluor 647-conjugated dsDNA of varying lengths and a cholesterol anchor. (b) Representative structured illumination microscopy (SIM) images of spherical and non-spherical erythrocytes. Human erythrocytes were stained with membrane dye CellBrite Fix 488 and Alexa Fluor 647-conjugated dsDNA of length 48 bp. Scale bar is 2 µm. (c) Site localization method performs well for both spherical and non-spherical particles. The measured distances for 48 bp dsDNA probes on erythrocytes are shown in a violin plot. The resolved distance is [median (IQR), mean ± SD] 11.5 (11.6) nm, 11.4 ± 9.3 nm (n = 31) for DNA on the spherical cells and 9.2 (10.7) nm, 9.8 ± 8.1 nm (n = 33) for the non-spherical erythrocytes. Error bars indicate interquartile range. Additionally, the result for spherical cells was confirmed using an existing method for site localization on spherical objects (Figure 3—figure supplement 1). (d) Representative SIM images of DNA-coated erythrocytes. Human erythrocytes were stained with membrane dye CellBrite Fix 488 and Alexa Fluor 647-conjugated dsDNA of length 18 bp, 48 bp, and 63 bp. Scale bar is 2 µm. (e) Site localization measurements of DNA probes show good agreement with predicted worm-like chain (WLC) heights The measured heights of the DNA probes are plotted as a function of the fully extended length of the DNA strands. The measured height is [median (IQR), mean ± SD] 3.7 (8.8) nm, 5.1 ± 8.3 nm (n = 25) for 18 bp DNA, 9.2 (10.7) nm, 9.8 ± 8.1 nm (n = 33) for 48 bp DNA, and 12.5 (8.2)nm, 10.3 ± 10.5 nm (n = 30) for 63 bp DNA. Error bars indicate interquartile range. The dashed line represents predicted height based on modelling the DNA strand as a WLC with a persistence length of 50 nm free to move in a hemisphere above the surface.
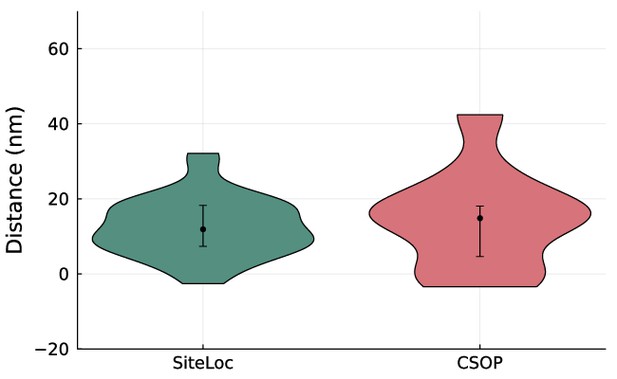
Distance measurements with site localization and cell surface optical profilometry (CSOP) for DNA probes on human red blood cells The measured heights of 48 bp dsDNA probes on erythrocytes are shown in a violin plot.
The resolved distance between the two channels is [median (IQR), mean ± SD] 11.5 (11.6) nm, 11.4 ± 9.3 nm (n = 31) with the site localization method and 14.8 (13.5) nm, 14.6 ± 13.6 nm (n = 10) with CSOP. Error bars indicate interquartile range.
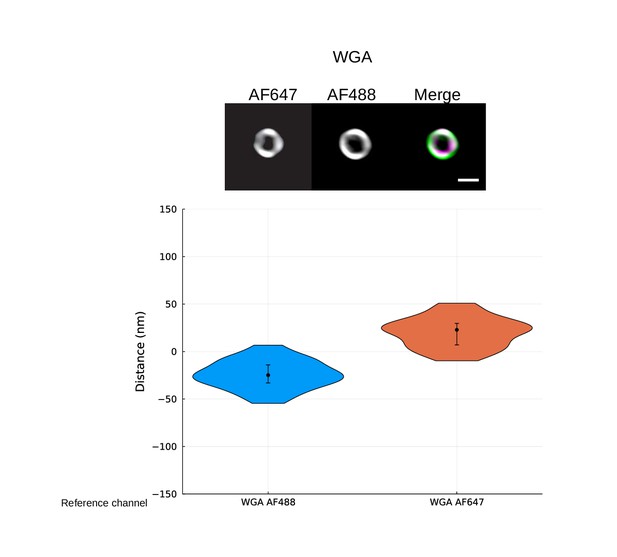
Control of site localization method using double-stain wall control.
Representative images are shown at the top. S. pyogenes strain SF370 has been fixed and stained with Alexa Fluor 488- and Alexa Fluor 647-conjugated wheat germ agglutinin (WGA). Scale bar is 500 nm. The violin plots show the resolved distance between the two cell wall signals. The median resolved distance between the two channels is [median (IQR)] –24.8 (19.0) nm with Alexa Fluor 488 as reference and 22.8 (22.8) nm with Alexa Fluor 647 as reference. A mirroring of the results is expected as the reference and target channels are flipped, and this is evident from the shape of the violin plots. The measured offset is mainly due to chromatic aberration at the imaging plane and provides a correction constant for subsequent site localization measurements on structured illumination microscopy (SIM images).
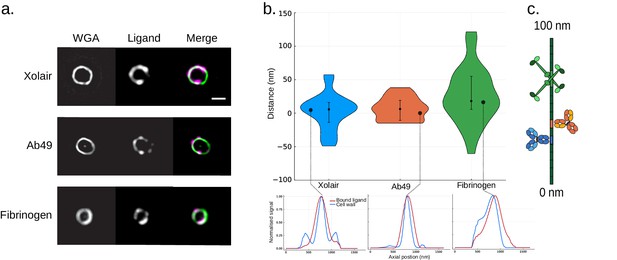
Site localization measurements of bound ligands on bacterial surface protein.
(a) Representative structured illumination microscopy (SIM) images of ligand coated bacteria. S. pyogenes strain SF370 has been fixed, stained with Alexa Fluor 488-conjugated wheat germ agglutinin (WGA), and coated with Alexa Fluor 647-conjugated Xolair (top), Ab49 (middle), or fibrinogen. Scale bar is 500 nm. (b) Binding site measurements of antibodies and fibrinogen on bacterial M protein The measured distances are shown in a violin plot. The resolved distance between the two channels is [median (IQR), mean ± SD] 5.6 (29.7) nm, 2.5 ± 26.7 nm (n = 41) for the Xolair Fc binding site, 6.2 (29.8) nm, 6.5 ± 16.8 nm (n = 28) for Ab49 Fab binding site, and 18.0 (49.3) nm, 26.2 ± 41.6 nm (n = 33) for the two fibrinogen binding sites. Error bars indicate the interquartile range. An intensity profile from a single bacterium is shown for each of the ligands below the violin plot. (c) A schematic illustrating the determined binding sites on M protein.
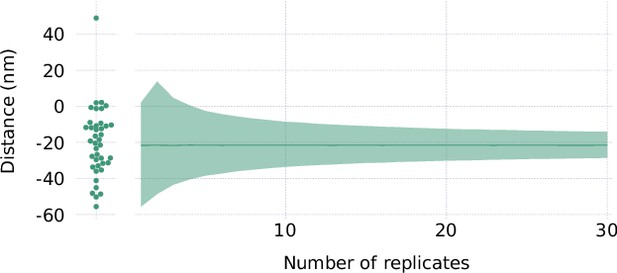
Percentile bootstrap on site localization results for double wall stain control.
The left part of the plot shows the data points for the distance from the Alexa Fluor 488-conjugated wheat germ agglutinin (WGA) (reference) channel to the Alexa Fluor 647-conjugated WGA channel as resolved by the site localization method. The ribbon plot to the right shows an estimate of the 95% confidence interval of the mean of a number of measurements computed by percentile bootstrap with 50,000 resamples for each chosen number of replicates.
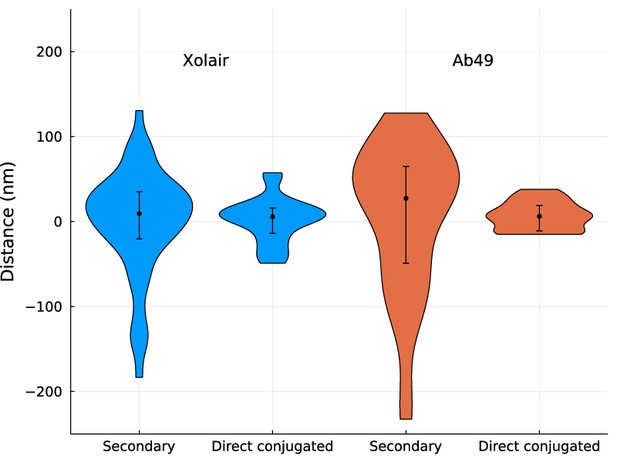
A comparison of site localization measurements of antibody binding sites using secondary antibody fragments and direct conjugation.
SF370 wild type was stained with Alexa Fluor 488-conjugated wheat germ agglutinin (WGA). For secondary antibody labelling, bacteria were incubated with IdeS-cleaved Xolair or Ab49. The antibody samples were stained with fluorescently labelled IgGFab (Ab49) or IgGFc (Xolair)-specific F(ab’)2 fragments (Alexa Fluor 647-conjugated anti-human IgGFc or IgGFab). For direct labelling, Alexa Fluor 647-conjugated Xolair or Ab49 was used. The median measurements are in order from left to right: [median (IQR), mean ± SD] 9.5 (55.5) nm, –0.1 ± 63.8 nm (n = 48), 5.6 (29.7) nm, 2.5 ± 26.7 nm (n = 41), 27.4 (113.7) nm, 6.1 ± 88.6 nm (n = 33), 6.2 (29.8) nm, 6.5 ± 16.8 nm (n = 28). It is evident from these results that site localization precision is increased when using directly conjugated antibodies.
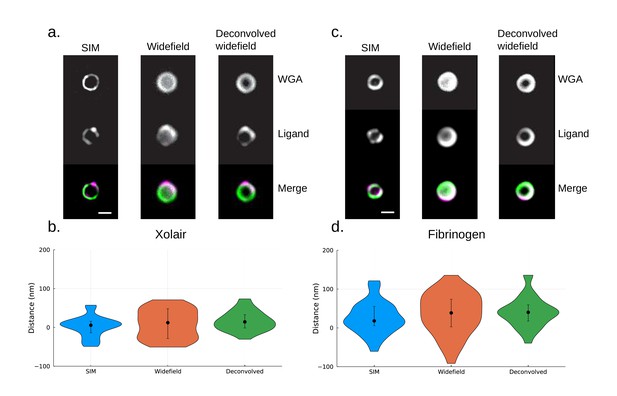
A comparison of site localization measurements using structured illumination microscopy (SIM), widefield, and deconvolved widefield images.
(a) Representative images of Xolair-coated bacteriaare shown at the top. Scale bar is 500 nm. S. pyogenes strain SF370 has been fixed, stained with Alexa Fluor 488-conjugated wheat germ agglutinin (WGA), and coated with Alexa Fluor 647-conjugated Xolair antibodies. (b) Site localization measurement of Fc binding site yields similar results with the widefield dataset. The resolved distance between the two channels is [median (IQR), mean ± SD] 5.6 (29.7) nm, 2.5 ± 26.7 nm (n = 41) with SIM data, 12.3 (76.5) nm, 8.7 ± 36.6 nm (n = 32) with widefield data, and 14.2 (34.5) nm, 17.0 ± 25.4 nm (n = 31) with deconvolved widefield data. Error bars indicate the interquartile range. (c) Representative images of fibrinogen-coated bacteria are shown at the top. S. pyogenes strain SF370 has been fixed, stained with Alexa Fluor 488-conjugated WGA, and coated with antibody Alexa Fluor 647-conjugated fibrinogen. Scale bar is 500 nm. (d) Site localization measurement of fibrinogen binding sites yields similar results with the widefield dataset. The resolved distance between the two channels is [median (IQR), mean ± SD] 18.0 (49.3) nm, 26.2 ± 41.6 nm (n = 33) with SIM data, 38.5 (71.0) nm, 35.9 ± 53.3 nm (n = 37) with widefield data, and 40.1 (42.0) nm, 41.1 ± 40.1 nm (n = 29) with deconvolved widefield data. Error bars indicate the interquartile range.
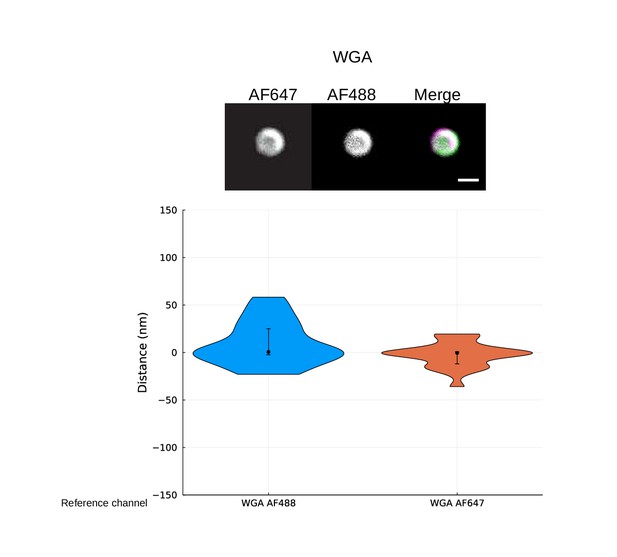
Control of site localization method using double-stain wall control.
Representative images are shown at the top. S. pyogenes strain SF370 has been fixed and stained with Alexa Fluor 488- and Alexa Fluor 647-conjugated wheat germ agglutinin (WGA). Scale bar is 500 nm. The violin plots show the resolved distance between the two cell wall signals. The median resolved distance between the two channels is [median (IQR)] 0.6 (27.4) nm with Alexa Fluor 488 as reference and –0.4 (12.6) nm with Alexa Fluor 647 as reference. A mirroring of the results is expected as the reference and target channels are flipped, and this is evident from the shape of the violin plots. The measured offset is mainly due to chromatic aberration at the imaging plane and provides a correction constant for subsequent site localization measurements for widefield images.
Tables
| Reagent type (species) or resource | Designation | Source or reference | Identifiers | Additional information |
|---|---|---|---|---|
| Strain, strain background (Streptococcus pyogenes) | SF370 | ATCC | Cat# 700294 | |
| Biological sample (Homo sapiens) | Venous blood (normal, adult) | This paper | Freshly taken from Homo sapiens | |
| Antibody | Xolair (humanized from mouse monoclonal) | Novartis | Cat# 028268; RRID#:AB_2459636 | (500 μg/ml) |
| Antibody | Anti-streptococcal M protein; Ab49 (human monoclonal) | Bahnan et al., 2021 | (10–μg/ml) | |
| Antibody | Alexa Fluor 647 AffiniPure F(ab’)2 fragment goat anti-human IgG (Fab specific) (goat polyclonal) | Jackson ImmunoResearch Laboratories | Cat# 109-606-097; RRID:AB_2337898 | (3 μg/ml) |
| Antibody | Alexa Fluor 647 AffiniPure F(ab’)2 fragment goat anti-human IgG (Fc specific) (goat polyclonal) | Jackson ImmunoResearch Laboratories | Cat# 109-606-170; RRID:AB_2337902 | (3 μg/ml) |
| Other | Alexa Fluor 647 (Invitrogen) | Thermo Fisher | Cat# 820006 | |
| Other | CellBrite Fix Membrane stain 488 | Cat# 30090 | ||
| Sequence-based reagent | Conjugated DNA_F 18 base pair | This paper, Son et al., 2020 | Oligonucleotide | 5′-Alexa647-N-AGCTGCGGTCAGATC-3′ |
| Sequence-based reagent | Conjugated DNA_R 18 base pair | This paper, Son et al., 2020 | Oligonucleotide | 5′-Cholesterol-TEG-TCGACGCCAGTCTAG-3′ |
| Sequence-based reagent | Conjugated DNA_F 48 base pair | This paper, Son et al., 2020 | Oligonucleotide | 5′-Alexa647-N-GATCTGACCGCAGCTATCT |
| Sequence-based reagent | Conjugated DNA_R 48 base pair | This paper, Son et al., 2020 | Oligonucleotide | 5′-Cholesterol-TEG-CTAGACTGGCGTCGATA |
| Sequence-based reagent | Conjugated DNA_F 63 base pair | This paper, Son et al., 2020 | Oligonucleotide | GACGTACTGCTATTGCTA GCGATTCCAT- |
| Sequence-based reagent | Conjugated DNA_R 63 base pair | This paper, Son et al., 2020 | Oligonucleotide | 5′-Cholesterol-TEG-ATGGAATCGCTAGC AATAGCAGTACGTCTACA TGAAGTCGTTGATTCGAC GCCAGTCTAGTAG-3′ |
| Peptide, recombinant protein | Fibrinogen from human plasma, Alexa Fluor 647 | Thermo Fisher | Cat# F35200 | (20 μg/ml) |
| Peptide, recombinant protein | Wheat germ agglutinin, Alexa Fluor 488 | Thermo Fisher | Cat# W11261 | (1:250) |
| Peptide, recombinant protein | Wheat germ agglutinin, Alexa Fluor 647 | Thermo Fisher | Cat# W32466 | (1:250) |
| Peptide, recombinant protein | IdeS | Hansa Biopharma | ||
| Software, algorithm | FluoroDist | This paper, GitHub | https://github.com/nordenfeltLab/FluoroDist.jl (Kumra Ahnlide, 2022; copy archived at swh:1:rev:053e516413277cf488ef9db1a24ce576e24d18f9) | Simulated fluorescence images with accurate surface distances |
| Software, algorithm | RegisterQD | GitHub, Greer, 2019 | https://github.com/Holylab/RegisterQD.jl | Image registration with the QuadDIRECT optimization algorithm |
| Software, algorithm | Site Localization | This paper, GitHub | https://github.com/nordenfeltLab/SiteLocalization | Binding site localization on non-homogeneous cell surfaces using topological image averaging |