Bulk flow of cerebrospinal fluid observed in periarterial spaces is not an artifact of injection
Figures
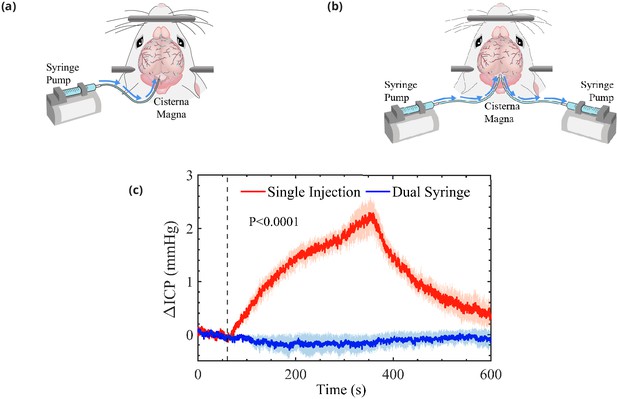
Schematic representation of the cisterna magna injection.
(a) The single-injection protocol for injection of 10 μL at 2 μL/min and (b) the dual-syringe protocol for simultaneous injection and withdrawal of 20 μL at 2 μL/min. The effect of single-injection and dual-syringe tracer infusion upon intracranial pressure (ICP) is shown in (c). The ICP was monitored continuously during injection of cerebrospinal fluid (CSF) tracers into the CM of mice. Injection begins at 60 s, indicated by the vertical dashed line. Single-injection infusion of 10 μL at a rate of 2 μL/min resulted in a mild change of ∼2.5 mmHg in ICP, whereas little or no change in ICP was observed during the simultaneous injection and withdrawal in the dual-syringe protocol. Repeated measures two-way analysis of variance (ANOVA) was performed; interaction p-value < 0.0001; n = 5 mice for single-injection and n = 6 mice for dual-syringe. The shaded regions above and below the plot lines indicate the standard error of the mean (SEM).
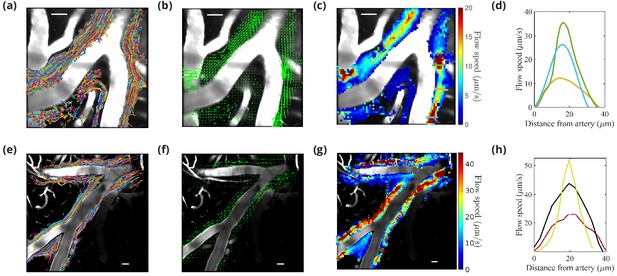
Particle tracking velocimetry in the (PVSs) surrounding cortical branches of the MCA using the single-injection method (panels in first row [Mestre et al., 2018b]) and the new dual-syringe method (second row).
The superimposed particle tracks shown in panels (a) and (e) have similar, continuous spatial distributions and show similar sizes of the perivascular spaces. The time-averaged velocity fields shown in panels (b) and (f) both show net flow of fluid in the same direction as the blood flow. The flow-speed distributions plotted in panels (c) and (g) show comparable speeds, with the fastest flow at the center of the imaged periarterial space and the slowest flow near the boundaries. Panels (d) and (h) show average flow-speed profiles across the corresponding colored lines spanning the PVS in panels (c) and (g), smoothed by interpolation. The parabolic-like nature of these velocity profiles is what is expected for viscous flow in an open channel. Scale bars indicate 50 μm. Figure panels (a, b, c, and d) reproduced from Figure 1, Mestre et al., 2018a, Nature Communications, published under the Creative Commons Attribution 4.0 International Public License (CC BY 4.0; https://creativecommons.org/licenses/by/4.0/).
© 2018, Mestre et al. Figure panels (a), (b), (c), and (d) reproduced from Figure 1, Mestre et al., 2018a, Nature Communications, published under the Creative Commons Attribution 4.0 International Public License.
Particles infused using dual-syringe method are transported downstream in pial perivascular spaces (PVSs).
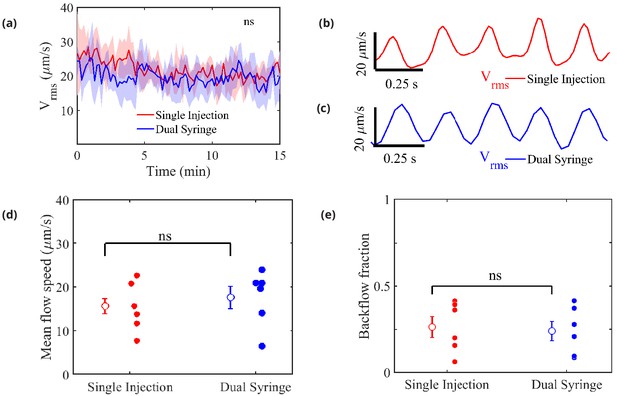
Measured flow characteristics.
Panel (a) shows over the course of the velocity measurements for both infusion methods. Repeated measures two-way ANOVA was performed; ns, not significant; n = 5 mice for single-injection and n = 6 mice for dual-syringe. The solid lines represent the mean value of and the shaded area represents the standard error of the mean within each time bin. The pulsatility of typical measured flows is depicted in panels (b) and (c). Panel (d) shows mean downstream flow speeds and panel (e) shows backflow fractions for the individual experiments, with overall mean values shown as open circles (and bars showing the standard error of the mean). The nearly identical values for the two protocols demonstrate that the flow is independent of the injection method employed. Unpaired Student’s t-test was performed; n = 5 or 6 mice per group; ns, not significant; mean ± SEM.
-
Figure 3—source data 1
Source data for panels (a, d), and (e).
- https://cdn.elifesciences.org/articles/65958/elife-65958-fig3-data1-v2.docx
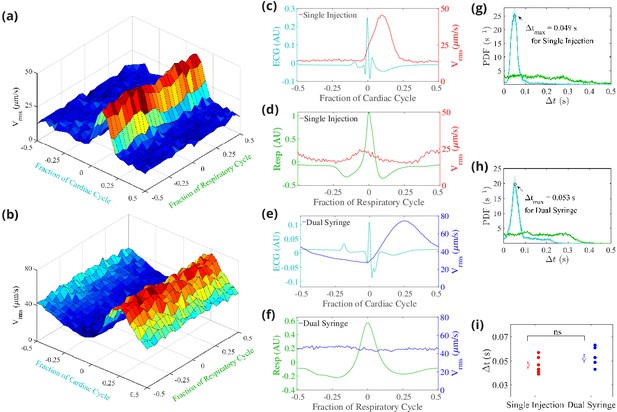
Cerebrospinal fluid (CSF) velocity variations over the cardiac and respiratory cycles.
Panels (a) and (b) show the measured conditionally averaged over the cardiac and respiratory cycles, based on the synchronized measurements of ECG, respiration, and velocity, for the single-injection (a) and the dual-syringe (b) protocols. Panel (c) for single injection and panel (e) for dual syringe both show that the peaks in the ECG are immediately followed by peaks in , indicating a strong correlation between heart rate and fluid motion in both injection protocols. No consistent trends are seen when is averaged over the respiratory cycle, as shown in panels (d) and (f). Panels (g) and (h) show the mean and the standard error of the mean of probability density functions of the delay time between the peak in the cardiac (cyan) or respiration (green) cycle and the subsequent peak in , for single-injection (n = 5) and dual-syringe (n = 6) methods respectively. Panel (i) shows the average between peaks in the cardiac cycle and for both protocols; in both, the peak in typically occurs ∼0.05 s after the peak in the cardiac cycle. Unpaired Student’s t-test was performed; n = 5 or 6 mice per group; ns, not significant; mean ± SEM. Figure panel (a) reproduced from Figure 3 Mestre et al., 2018b, Nature Communications, published under the Creative Commons Attribution 4.0 International Public License (CC BY 4.0; https://creativecommons.org/licenses/by/4.0/).
© 2018, Mestre et al. Figure panel (a) reproduced from Figure 3 Mestre et al., 2018b, Nature Communications, published under the Creative Commons Attribution 4.0 International Public License.
-
Figure 4—source data 1
Data for panel (i).
- https://cdn.elifesciences.org/articles/65958/elife-65958-fig4-data1-v2.docx
Tables
| Reagent type (species) or resource | Designation | Source or reference | Identifiers | Additional information |
|---|---|---|---|---|
| Strain strain background (Mus musculus, C57BL/6NCrl) | Male wild type (WT) | Charles River, 027 | RRID: IMSR_CRL:27 | |
| Software, algorithm | MATLAB | Mathworks | RRID: SCR001622 | |
| Software, algorithm | GraphPad Prism 8 | GraphPad Software | RRID: SCR002798 | |
| Other | FluoSpheres | Invitrogen | Cat: #13081 | Particles are 1.0 μm in diameter with excitation/emission at 580/605 nm |
| Other | Artificial CSF | Sigma-Aldrich | Components in aCSF (concentrations in mM): 126.0 NaCl 3 KCl, 2 MgSO4, 10.0 dextrose, 26.0 NaHCO3, 1.25 NaH2PO4, 2 CaCl2 |





