Keratinocyte PIEZO1 modulates cutaneous mechanosensation
Figures
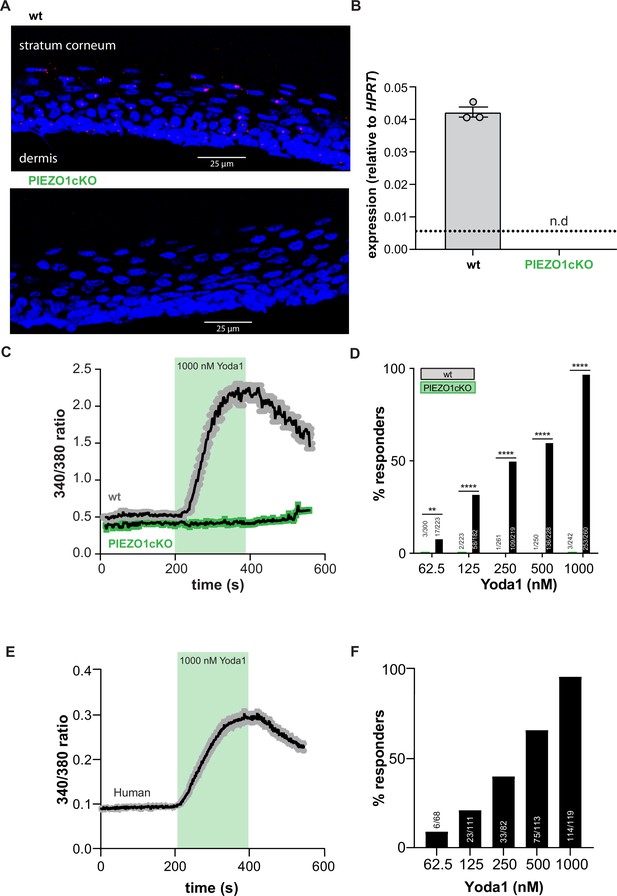
PIEZO1 is functionally expressed in mouse and human keratinocytes.
(A) RNAscope of hindpaw glabrous skin isolated from wildtype and PIEZO1cKO mice targeting PIEZO1 mRNA (blue: DAPI, red: PIEZO1). (B) PIEZO1 gene expression was measured in keratinocytes isolated from wildtype (wt; n=3) and PIEZO1cKO (n=3) mice using quantitative real-time PCR. Expression levels were normalized to HPRT. Piezo1 expression was undetected in PIEZO1cKO samples. (C) Average calcium flux in wildtype (wt) and PIEZO1cKO keratinocytes in response to 1000 nM Yoda1; trace outline is SEM. (D) Percentage of wildtype and PIEZO1cKO keratinocytes that respond to extracellular Yoda1; cells from n=3 mice per genotype; bars are group averages; Chi square. (E) Calcium flux in human keratinocytes in response to 1000 nM Yoda1; trace outline is SEM. (F) Percentage of human keratinocytes that respond to extracellular Yoda1; cells from the skin of n=3 human donors; bars are group averages. All data are mean ± SEM unless otherwise stated. Post-hoc comparisons for all panels: **p<0.01, ****p<0.0001.
-
Figure 1—source data 1
Data for panals Figure 1B-F.
- https://cdn.elifesciences.org/articles/65987/elife-65987-fig1-data1-v2.xlsx
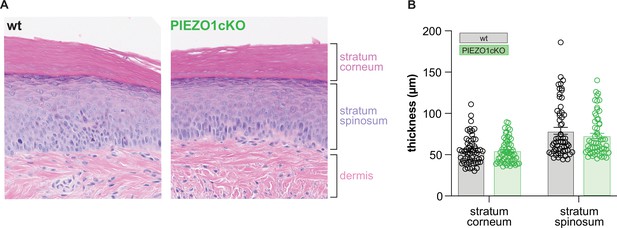
The epidermis of PIEZO1cKO animals has normal morphological features.
(A) H&E stained skin from the hindpaw of wildtype (wt) and PIEZO1cKO mice. (B) Quantification of individual epidermal layer thickness; 2-way ANOVA, n.s. All data are mean ± SEM.
-
Figure 1—figure supplement 1—source data 1
Individual values for epidermal thickness.
- https://cdn.elifesciences.org/articles/65987/elife-65987-fig1-figsupp1-data1-v2.xlsx
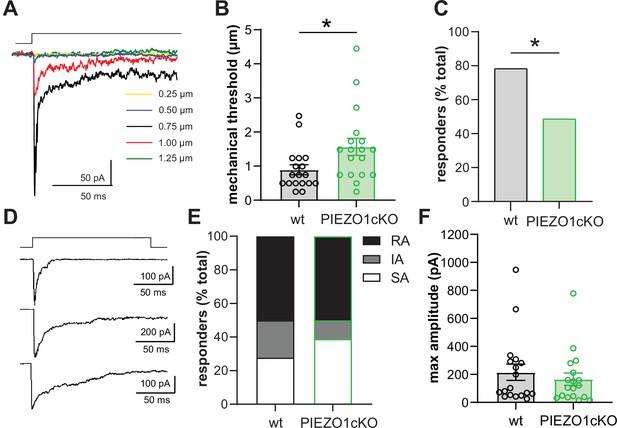
PIEZO1 deletion decreases keratinocyte mechanical sensitivity.
(A) Examples of whole-cell recording of mechanically activated (MA) current (Vh = –40 mV) evoked in keratinocytes by a stepwise increase in membrane indentation depth. (B) The mechanical threshold to evoke MA current by gradual increasing of indentation depth is significantly increased in keratinocytes of PIEZO1cKO mice compared to wild-type controls; Mann-Whitney U-test. (C) PIEZO1 deletion significantly decreased number of keratinocytes that responded to membrane indentation with MA current (wildtype: n=23 cells; PIEZO1cKO: n=37 cells); Chi square test. (D) Representative traces of rapidly adapting (RA), intermediately adapting (IA), and slowly adapting (SA) MA currents induced in keratinocytes in response to membrane indentation. (E) Proportion of keratinocytes that responded to membrane indentation with RA, IA, and SA currents is not affected by PIEZO1 deletion (n=18 recordings of MA current per each group); Chi square and Fisher’s exact post hoc test, n.s. (F) Maximal amplitude of MA currents in wild-type and PIEZO1cKO keratinocytes; Mann-Whitney U-test, n.s. For whole-cell patch clamp experiments, cells were harvested from n=5 mice per group. All data are mean ± SEM. *p<0.05.
-
Figure 2—source data 1
Data for mechanical threshold, percent responders, current profile and max amplitude of wt and PIEZO1cKO keratinocytes.
- https://cdn.elifesciences.org/articles/65987/elife-65987-fig2-data1-v2.xlsx
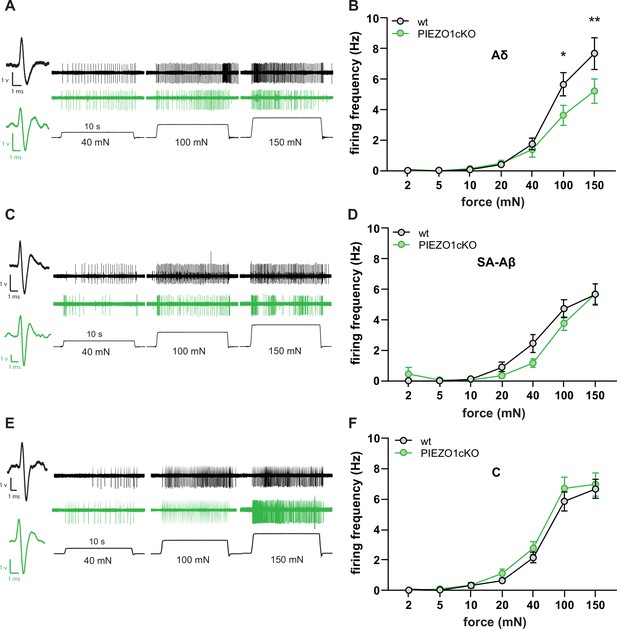
Normal mechanically-induced primary afferent firing requires epidermal PIEZO1 expression.
Ex vivo tibial nerve recordings of Piezo1cKO and wildtype (wt) mice. (A) Aδ fiber example traces. (B) Mean mechanically induced firing rates of Aδ fibers (n=33 wt and 30 PIEZO1cKO fibers). (C) SA-Aβ fiber example traces. (D) Mean mechanically induced firing rates of SA-β fibers (n=33 wt and 28 PIEZO1cKO fibers). (E) C fiber example traces. (F) Mean mechanically induced firing rates of C fibers (n=30 wt and 34 PIEZO1cKO fibers). For all recordings, the mechanical stimulus was applied to the skin for 10 seconds. All data are mean ± SEM; 2-way ANOVA and Sidak post-hoc comparisons for firing frequency panels: *p<0.05, **p<0.01; fibers from n=17–19 mice.
-
Figure 3—source data 1
Data for mechanically induced firing frequency of sensory afferents.
- https://cdn.elifesciences.org/articles/65987/elife-65987-fig3-data1-v2.xlsx
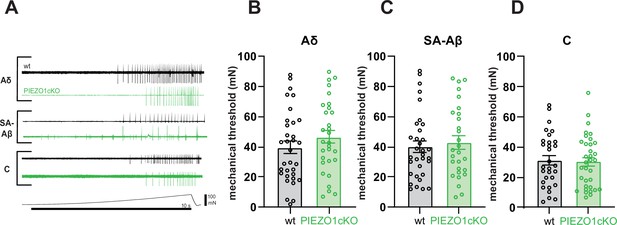
PIEZO1 deletion does not alter sensory fiber mechanical thresholds.
(A) Single unit firing in response to mechanical force ramp (0–100 mN; 10 s). Mechanical thresholds of (B) Aδ fibers, (C) SA-Aβ fibers, and (D) C fibers to ramp stimuli. All data are mean ± SEM; Student’s (two tailed) t test, n.s. Fibers from n=17–19 mice.
-
Figure 3—figure supplement 1—source data 1
Data for mechanical threshold.
- https://cdn.elifesciences.org/articles/65987/elife-65987-fig3-figsupp1-data1-v2.xlsx
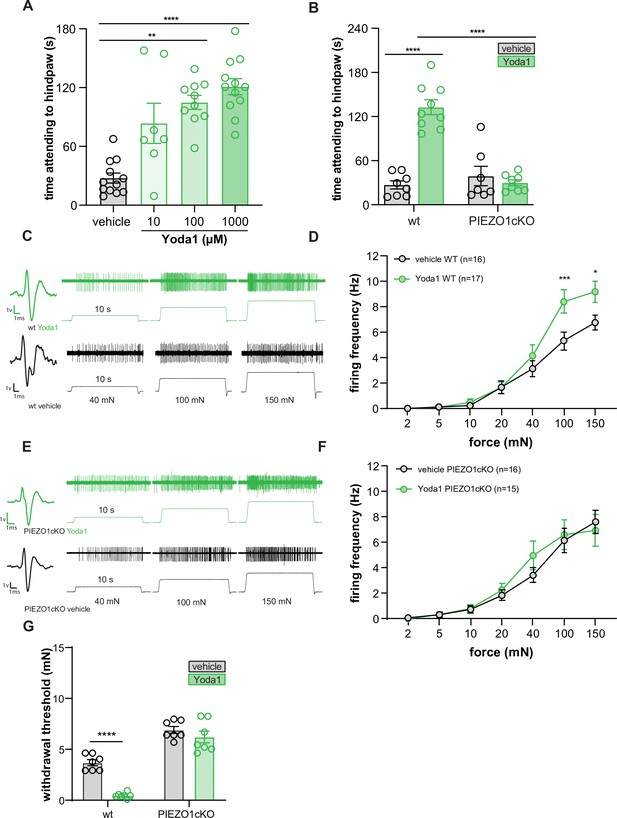
Yoda1 induces paw attending behaviors and C fiber mechanical hypersensitivity.
(A) Yoda1-induced attending behaviors in wildtype mice; Kruskal Wallis test. (B) 1 mM Yoda1 induced attending behaviors in wildtype and PIEZO1cKO mice; 2-way ANOVA. (C) Example traces from wildtype preparations exposed to 1 mM Yoda1 or vehicle during mechanical testing. (D) Mean mechanical firing frequency of C fibers from wildtype animals exposed to 1 mM Yoda1 or vehicle. (E) Example traces of PIEZO1cKO preparations exposed to 1 mM Yoda1 or vehicle during mechanical testing. (F) Mean mechanical firing frequency of C fibers from PIEZO1cKO animals exposed to 1 mM Yoda1 or vehicle. Fibers from n=12–14 mice. For all teased fiber recordings, the mechanical stimulus was applied to the skin for 10 s; 2-way ANOVA. (G) Von Frey mechanical thresholds of wildtype and PIEZO1cKO mice tested 30 min after an injection of 1 mM Yoda1 or vehicle; 2-way ANOVA . All data are mean ± SEM unless otherwise stated. Post-hoc comparisons for all panels: **p<0.01, ***p<0.001, ****p<0.0001.
-
Figure 4—source data 1
Data for time attending to hindpaws, Yoda1 induced firing frequency, and Yoda1 induced mechanical hypersensitivity.
- https://cdn.elifesciences.org/articles/65987/elife-65987-fig4-data1-v2.xlsx
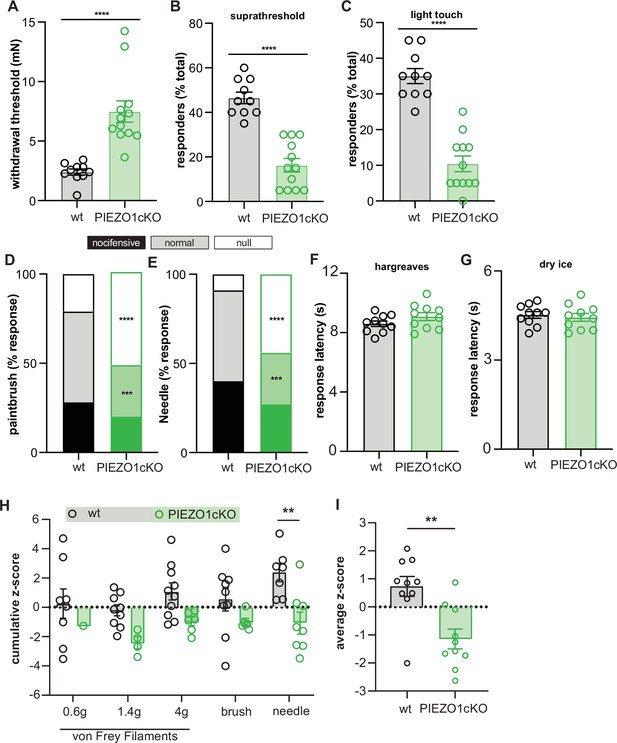
Epidermal Piezo1 is required for normal innocuous and noxious mechanosensation.
(A) Von Frey mechanical thresholds of wildtype and PIEZO1cKO mice; Mann-Whitney U-test. (B) Wildtype and PIEZO1cKO responses to repeated suprathreshold (3.61 mN) von Frey filament stimulation; Mann-Whitney U-test. (C) Wildtype and PIEZO1cKO responses to repeated static light touch (0.6 mN von Frey filament) stimulation; Mann-Whitney U-test. (D) Response characterization to paintbrush swiping across hindpaw; n=10–12; bars are group averages; Chi Square test. (E) Response characterization to noxious needle hindpaw stimulation; n=10–12; bars are group averages; Chi Square test. (F) Withdrawal latency to radiant heat hindpaw stimulation; Student’s (two-tailed) t test, n.s. (G) Withdrawal latency to dry ice hindpaw stimulation; Student’s (two-tailed) t test, n.s. (H) High-speed imaging mechanical sensitivity scores in response to von Frey (0.6 g, 1.4 g, 4 g), brush, and needle stimulation in wildtype and PIEZO1cKO mice; two-way ANOVA . Cumulative z-scores were calculated from paw height, paw velocity, and pain score at each stimulus. (I) Average high-speed imaging mechanical sensitivity score across all stimuli for each animal. All data are mean ± SEM unless otherwise stated. Post-hoc comparisons for all panels: **p<0.01, ***p<0.001, ****p<0.0001.
-
Figure 5—source data 1
Data for behavioral mechanical sensitivity.
- https://cdn.elifesciences.org/articles/65987/elife-65987-fig5-data1-v2.xlsx
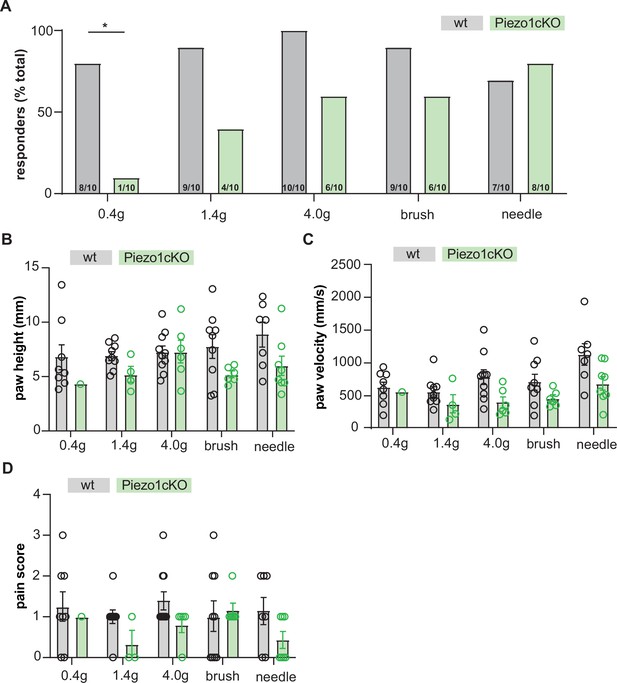
High-speed imaging of PIEZO1cKO and wildtype mice.
(A) Percent of mice responding to a single application of von Frey (0.4 g, 1.4 g, 4.0 g), brush, or needle stimuli with a paw withdrawal; Chi square. (B) Height of paw withdrawal in response to mechanical stimulation. (C) Velocity of paw withdrawal in response to mechanical stimulation. (D) Pain score in response to mechanical stimulation. Pain score was calculated based on whether the animal exhibited paw fluttering, guarding, or jumping behaviors in response to stimulation. All data are mean ± SEM. *p<0.05.
-
Figure 5—figure supplement 1—source data 1
Data for high speed imaging of mechanical sensitivity.
- https://cdn.elifesciences.org/articles/65987/elife-65987-fig5-figsupp1-data1-v2.xlsx
Tables
| Type | WT | Piezo1cKO | P value | |
|---|---|---|---|---|
| Threshold | RA | 0.92±0.19 (n=9) | 1.36±0.31 (n=9) | 0.24 |
| IA | 1.13±0.48 (n=4) | 2.13±0.63 (n=2) | ||
| SA | 0.85±0.22 (n=5) | 1.86±0.48 (n=7) | 0.13 | |
| MAX amplitude | RA | 126.4±38.76 (n=9) | 214±78.62 (n=9) | 0.73 |
| IA | 282.8±136.3 (n=4) | 79.5±40.50 (n=2) | ||
| SA | 320.8±164.8 (n=5) | 128.1±48.03 (n=7) | 0.22 |





