The mesoscale organization of syntaxin 1A and SNAP25 is determined by SNARE–SNARE interactions
Figures
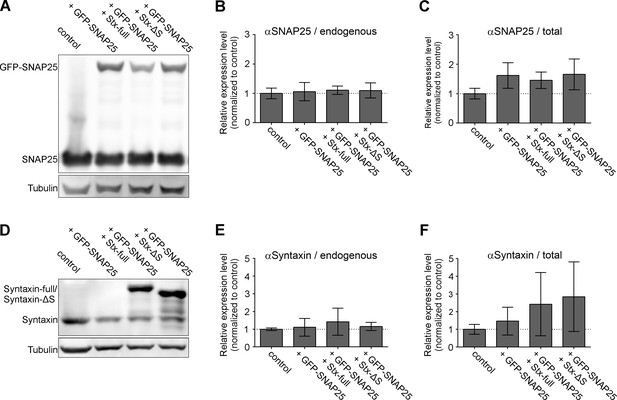
Expression levels of endogenous SNAREs and overexpressed GFP-SNAP25, Stx-full, and Stx-ΔS.
PC12 cells are transfected with GFP-SNAP25 alone or in combination with Stx-full/Stx-ΔS. The sample is split for parallel analysis of expression levels by Western Blot and molecular accessibility to GFP-SNAP25 (Figure 2). (A) Western Blot analyzing the expression levels of endogenous SNAP25 (25 kDa) and GFP-SNAP25 (52 kDa) using an antibody raised against the N-terminal SNARE domain of SNAP25. Band intensities are related to the tubulin light chain bands, followed by normalization to control (untransfected PC12 cells). (B) Quantification of the 25 kDa band of endogenous SNAP25 and (C) total SNAP25 (endogenous SNAP25 and the 52 kDa band of GFP-SNAP25). (D) Western Blot analysis of endogenous syntaxin 1A (33 kDa), overexpressed syntaxin 1A (Stx-full; ~38 kDa with a triple myc-tag fused via a linker region to the C-terminus) and Stx-ΔS (~35 kDa; Stx-full lacking the N-terminal half of the SNARE domain) using an antibody detecting the N-terminal domain of syntaxin 1A. (E) Normalized band intensities of endogenous syntaxin. (F) Signal of endogenous syntaxin (without or with GFP-SNAP25 overexpression) and endogenous syntaxin 1A + Stx-full or Stx-ΔS with GFP-SNAP25 overexpression. Values are given as means ± standard deviation (SD). Statistical analysis showed no significant difference between any of the conditions (n = 3 experiments; two-tailed unpaired t-test, not significant = p > 0.05).
-
Figure 1—source data 1
Raw Western Blot images and annotated full blots for Figure 1A, D.
- https://cdn.elifesciences.org/articles/69236/elife-69236-fig1-data1-v2.zip
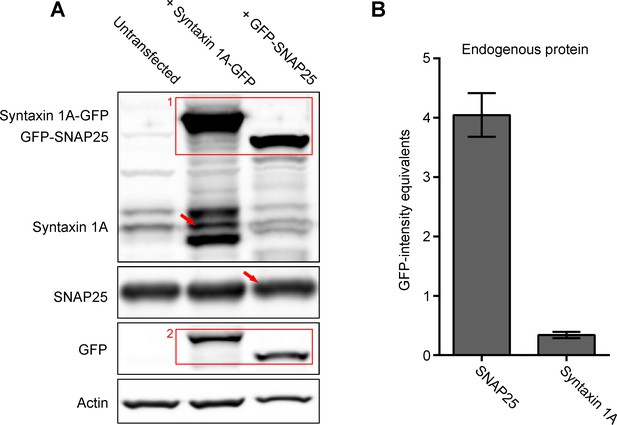
Ratio between endogenous SNAP25 and syntaxin 1A.
Western Blot analysis immunostaining syntaxin 1A-GFP/GFP-SNAP25 bands with an antibody raised against the respective SNARE (red box #1) and stained again after stripping the membrane with an anti-GFP antibody (red box #2). From the band intensities, we obtain a conversion factor allowing us to express the endogenous band intensities in GFP signal equivalents, and by this determine the SNAP25:syntaxin 1A ratio. (A) Representative Western Blot from PC12 cells expressing either no construct (left lane), syntaxin 1A-GFP (middle lane), or GFP-SNAP25 (right lane), using for syntaxin 1A and SNAP25 the same mouse monoclonal antibodies as in Figure 1, for actin a polyclonal rabbit antibody and for GFP a polyclonal rabbit anti-GFP antibody. Different from Stx-full expression (compare to Figure 1), we observe degradation bands of syntaxin 1A-GFP (see above and below the endogenous syntaxin 1A band indicated by a red arrow). For the conversion factor only non-degraded syntaxin 1A-GFP is taken into account. The SNARE-antibody band intensity from the overexpressed construct (red box #1; syntaxin 1A-GFP ~60 kDa; GFP-SNAP25 ~52 kDa) is normalized to actin and then divided by the respective actin normalized GFP-band intensity (red box #2), which yields GFP-conversion factors of 1.85 and 2.49 for syntaxin 1A and SNAP25, respectively. (B) Quantification of GFP-intensity equivalents. The band intensities of the endogenous SNARE proteins (red arrows) are divided by the respective conversion factors, yielding their GFP-intensity equivalents. The ratio of the GFP equivalents equals the ratio of the endogenous SNARE proteins, resulting in a 12-fold excess of SNAP25 over syntaxin 1A. Values are given as means ± standard deviation (SD; n = 3 experiments). Western Blots are shown using a linear grey scale lookup table at arbitrary scaling.
-
Figure 1—figure supplement 1—source data 1
Raw Western Blot images and annotated full blots for Figure 1—figure supplement 1A.
- https://cdn.elifesciences.org/articles/69236/elife-69236-fig1-figsupp1-data1-v2.zip
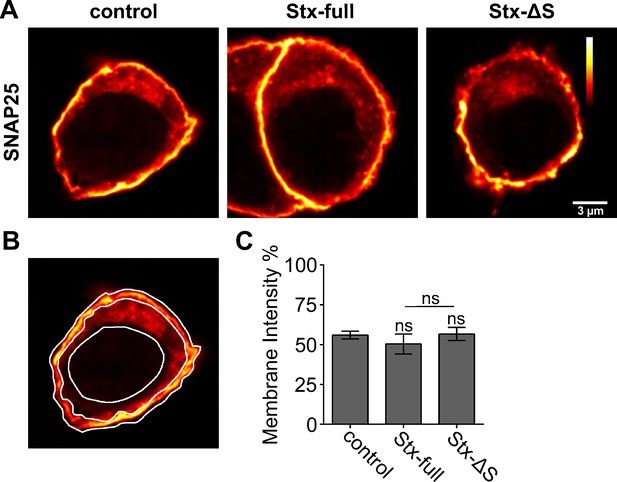
GFP-SNAP25 targeting to the plasma membrane.
PC12 cells transfected with GFP-SNAP25 alone (control) or together with Stx-full/Stx-ΔS were analyzed to quantify the amount of GFP-SNAP25 localizing to the plasma membrane. (A) Confocal micrographs illustrate optical sections of the cell’s equatorial planes, imaging the GFP-tag of GFP-SNAP25. Images are shown at the same scaling applying the ‘red hot’ look up table, displaying increasingly brighter pixel intensities employing a colour code from black to red to yellow to white (see vertical scale on right image). (B) For analysis of the plasma membrane fraction, three regions of interest (ROIs) were manually drawn: ‘outer rim’, ‘inner rim’, and ‘nucleus’. For details regarding the calculation, see Materials and methods. (C) Average GFP-SNAP25 intensity at the plasma membrane in percent. Values are given as means ± standard deviation (SD; n = 3 experiments; 20 cells per condition and experiment; two-tailed unpaired t-test compares (1) control to Stx-full/Stx-ΔS and (2) Stx-full to Stx-ΔS; ns = not significant = p > 0.05).
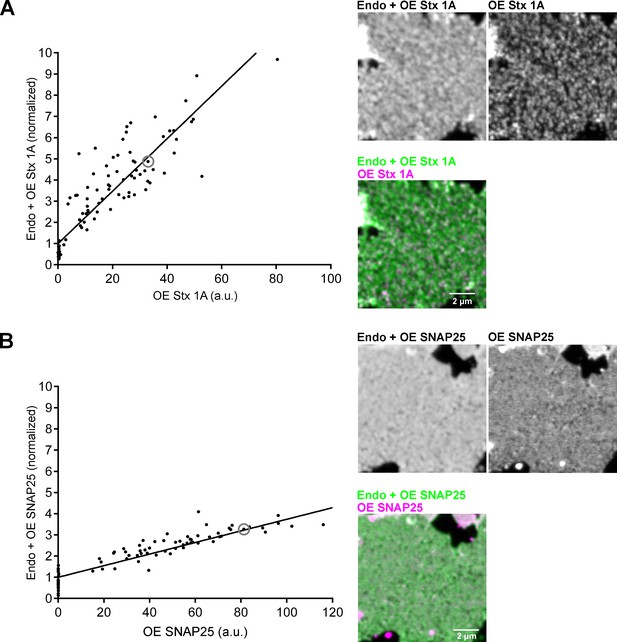
Elevation of SNAP25 and syntaxin 1A over endogenous levels in plasma membrane sheets.
(A) Stx-full or (B) GFP-SNAP25 is expressed in PC12 cells, cell-free membrane sheets are generated, fixed, stained for endogenous/overexpressed (Endo + OE) and overexpressed protein (OE), and imaged by confocal microscopy. (A) Endogenous syntaxin 1A/Stx-full (Endo + OE Stx1A; stained with an antibody raised against the N-terminal domain [HPC-1] in combination with a secondary goat anti-mouse antibody coupled to StarRed) plotted against Stx-full (OE Stx1A; stained with an anti-myc antibody in combination with a secondary donkey anti-rabbit antibody coupled to AlexaFluor594) or (B) endogenous SNAP25/GFP-SNAP25 (Endo + OE SNAP25; stained using an antibody raised against SNAP25’s C-terminus in combination with a secondary goat-anti-rabbit antibody coupled to StarRed) plotted against GFP-SNAP25 (OE SNAP25; GFP intensity). Antibodies raised against syntaxin 1A and SNAP25 visualize endogenous plus overexpressed protein ([Endo + OE]StarRed) whereas anti myc-staining against Stx-full and GFP-fluorescence from GFP-SNAP25 visualize only the overexpressed protein (OEAlexaFluor/GFP). On single membrane sheets, average region of interest (ROI) intensities in the channels ‘Endo + OE’ and ‘OE’ are measured manually and corrected for background. Plotting (Endo + OE)StarRed against OEAlexaFluor/GFP and line fitting yields regression lines (with slopes of 0.12 and 0.03 for syntaxin 1A and SNAP25, respectively) from which y-axis intercepts (OEAlexaFluor/GFP = 0) correspond to the average signal of the endogenous proteins, used for normalization of the ‘Endo + OE’ values. Values from three biological replicates are pooled, each including at least 30 membrane sheets. Images are shown at arbitrary scaling. Circles mark the membrane sheets shown for illustration.
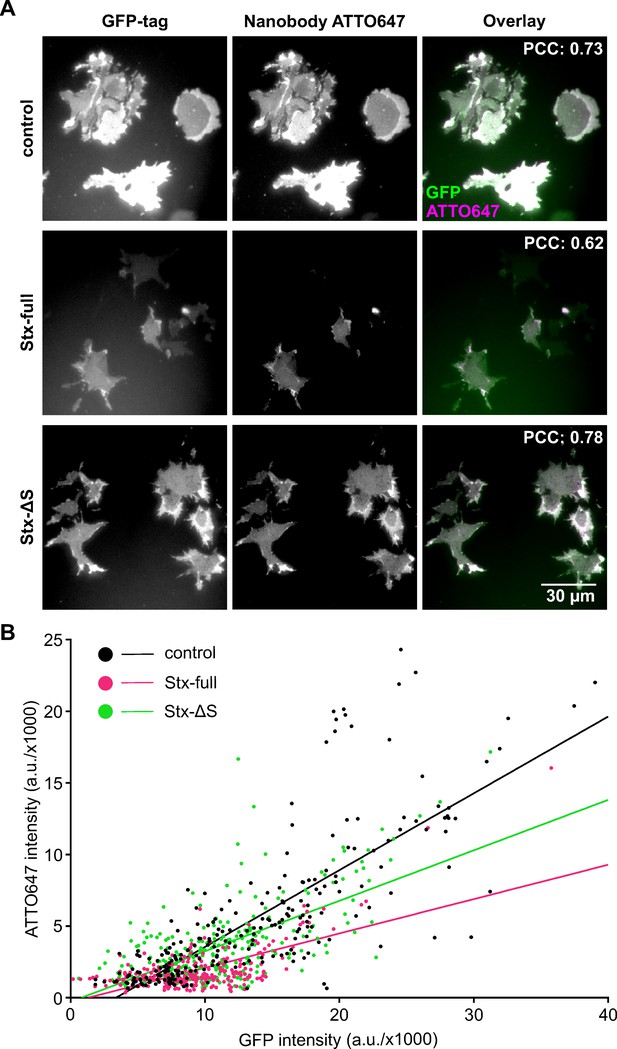
Syntaxin 1A overexpression reduces nanobody accessibility to GFP-SNAP25.
Cells from the same preparations as used for Western Blot analysis (Figure 1) were unroofed, leaving behind glass-adhered plasma membrane sheets that are fixed and stained with an ATTO647-labelled GFP nanobody. (A) Epifluorescence micrographs recording GFP- (left) and ATTO647 fluorescence (middle) arising from GFP-SNAP25 and anti-GFP nanobody (ATTO647 labelled), respectively. Membrane sheets are generated from cells expressing GFP-SNAP25 alone (control) or together with Stx-full/Stx-ΔS. Images from one channel are shown at the same scaling employing a linear lookup table. The average Pearson correlation coefficient (PCC) between the two channels is stated in the top right corner, for statistics see Figure 2—figure supplement 2A. (B) From individual membrane sheets, mean ATTO647 intensity is plotted against mean GFP intensity, followed by fitting of a linear regression line (slope values are 0.54 [control], 0.24 [Stx-full], and 0.35 [Stx-ΔS]). Two-tailed t-test shows a p value < 0.0001 between control and Stx-full/Stx-ΔS and between Stx-full and Stx-ΔS slopes. Per condition, membrane sheets from three experiments are pooled (control, n = 245; Stx-full, n = 266; Stx-ΔS, n = 269 membrane sheets). For the average GFP signal intensity, see Figure 2—figure supplement 2B.

GFP-SNAP25 detection by an anti-GFP nanobody.
PC12 membrane sheets generated from cells expressing GFP-SNAP25 were stained with an anti-GFP nanobody labelled with ATTO647 (same as in Figure 2), followed by confocal microscopy imaging. Left panels show overview and magnified view of the GFP fluorescence, middle panels the anti-GFP nanobody’s ATTO647 fluorescence. For overlay of magnified views see bottom right. Top, right: cartoon illustrating the topology of an anti-GFP nanobody–SNAP25 complex. Due to technical limitations, the Pearson correlation coefficient (PCC) between two channels visualizing a double tagged protein is much lower than 1, in the range of 0.6–0.8. Here, we find an even lower PCC of 0.5 due to the noisy GFP channel, but overall the two patterns look very similar. Images are shown at arbitrary scaling. PCC = 0.50 ± 0.42 (mean ± standard deviation [SD]; n = 3 experiments, 5–20 sheets per experiment).
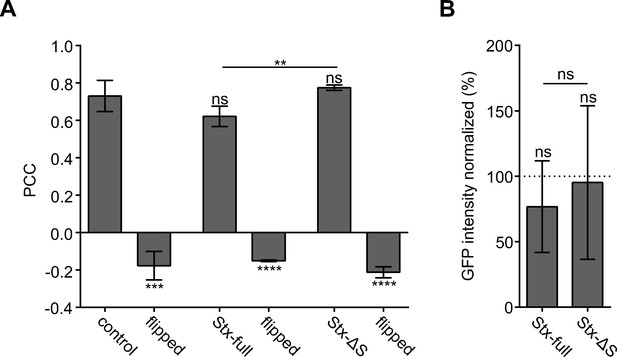
Pearson correlation coefficients (PCCs) and average GFP fluorescence intensity.
(A) PCC statistical significance analysis. In a PCC control (‘flipped’) mimicking non-related images the GFP channel is flipped vertically and horizontally. Values are given as means ± standard deviation (SD; n = 3 experiments, 16–42 membranes per experiment and condition). Two-tailed Student’s t-tests compare (1) control to Stx-full/Stx-ΔS, (2) control/Stx-full/Stx-ΔS to the respective flipped images, and (3) Stx-full to Stx-ΔS. (B) Average GFP intensity from membrane sheets in Figure 2, normalized to control (set to 100%). Values are given as means ± SD (n = 3 experiments, 63–123 membranes per experiment and condition). Two-tailed Student’s t-tests compare (1) control to Stx-full/Stx-ΔS and (2) Stx-full to Stx-ΔS; ns = not significant, p > 0.05, *p < 0.05, **p < 0.01, ***p < 0.001, ****p < 0.0001.
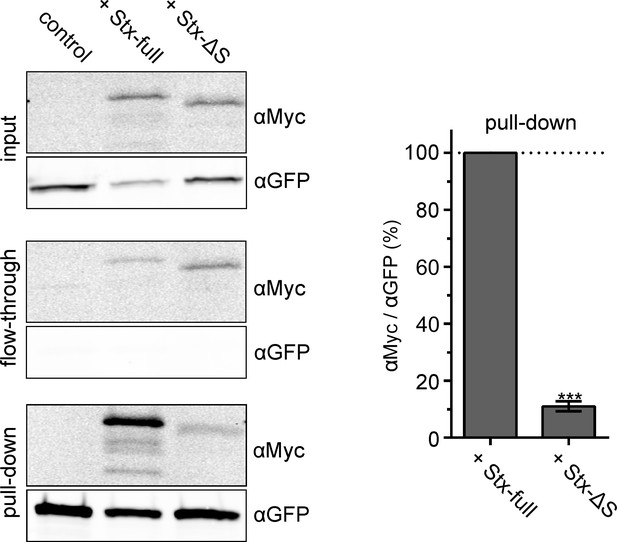
Interaction between SNAP25 and Stx-full/Stx-ΔS.
HepG2 cells that do not endogenously express syntaxin 1A and SNAP25 express GFP-SNAP25 alone (control) or together with Stx-full/Stx-ΔS. For immunoprecipitation of GFP-SNAP25, cell lysate is incubated with anti-GFP agarose beads. Left, Western Blot detecting Stx-full/Stx-ΔS using an anti-Myc-tag antibody (αMyc) and GFP-SNAP25 using an anti-GFP antibody (αGFP) in the input (cell lysate), flow-through and pull-down. Right, band quantification of pull-down. The αMyc-band intensity is related to the αGFP-band intensity and Stx-full is set to 100 %. Values are given as means ± standard deviation (SD; n = 3 experiments). Two-tailed paired t-test compares Stx-full to Stx-ΔS; ***p < 0.001.
-
Figure 3—source data 1
Raw Western Blot images and annotated full blot for Figure 3.
- https://cdn.elifesciences.org/articles/69236/elife-69236-fig3-data1-v2.zip
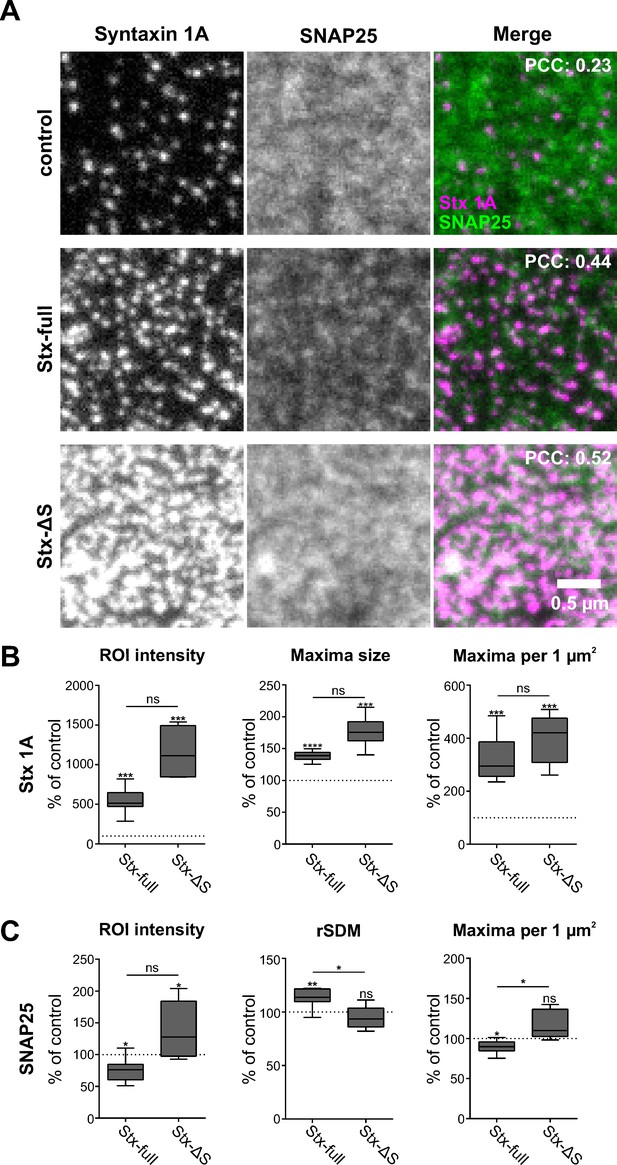
Syntaxin clusters change the distribution of SNAP25.
Membrane sheets are generated from PC12 cells expressing GFP-SNAP25 alone (control) or together with Stx-full/Stx-ΔS. Endogenous syntaxin/syntaxin constructs are recognized by an antibody raised against the molecule’s N-terminus, visualized by an AlexaFluor594-coupled secondary antibody; GFP-SNAP25 is visualized by an ATTO647-labelled GFP nanobody. (A) Representative images from the syntaxin 1A and SNAP25 channels of the control (only endogenous syntaxin), Stx-full (+ additional syntaxin), and Stx-ΔS (+ additional syntaxin with a shortened SNARE domain) condition. For magnified views see Figure 4—figure supplement 4. Top right corner, average Pearson correlation coefficient (PCC) between the two channels; for statistics see Figure 4—figure supplement 5. (B) Change of the syntaxin 1A staining pattern after Stx-full/Stx-ΔS expression. Staining intensity (region of interest [ROI] intensity), maxima size, and maxima density are related to the average control values (set to 100%). (C) SNAP25 staining pattern after Stx-full/Stx-ΔS expression. SNAP25 staining intensity (ROI intensity), clustering degree (relative standard deviation of the mean [rSDM]; 100% = 0.47), and maxima density are related to the control. For the relation between rSDM and Stx-full expression level, see Figure 4—figure supplement 6. Values are shown as box plots (n = 6–9 experiments, including three experimental preparations used as well for Figure 2; at least 15 membrane sheets were imaged per condition and experiment), showing the median, the 25th and 75th percentile (box), and the minimum and maximum value. Two-tailed paired Student’s t-test compare (1) control to Stx-full/Stx-ΔS and (2) Stx-full to Stx-ΔS; ns = not significant, p > 0.05, *p < 0.05, **p < 0.01, ***p < 0.001. After exchanging the spectral properties of the labels using ATTO647 for the syntaxin and ATTO594 for the SNAP25 channel, the same trends are observed. The syntaxin 1A maxima are sharper and more defined than SNAP25 maxima as well (Figure 4—figure supplement 7).
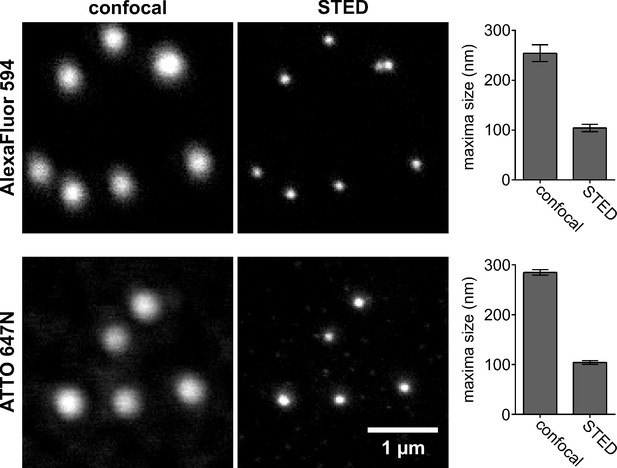
Size of the point spread functions in the AlexaFluor594 and ATTO647N channels.
To determine the point spread functions (PSFs) of the STED microscope system, we used HPV16 pseudoviruses (PsVs) that have a defined size of ~60 nm (Baker et al., 1991). PsVs are stained using a primary antibody (16L1-312F) raised against the L1 capsid protein, followed by staining with an AlexaFluor594- (used for syntaxin 1A STED imaging) or ATTO647N (used for SNAP25 STED imaging)-coupled secondary antibody. PsVs are imaged using the same excitation and depletion laser power settings as in Figures 4 and 5. The PsVs maxima size was determined exactly as the size of syntaxin 1A/SNAP25 maxima. In the STED mode, the PsVs sizes in the images are reduced in the AlexaFluor594 channel from 254 to 104 nm (upper panels) and in the ATTO647N channel from 285 to 104 nm (lower panels). Values are given as means ± standard deviation (SD; n = 20 images, pooled from two technical replicates [PsV values from one image are averaged]; 431–1496 PsVs per replicate). After primary and secondary antibody staining, we assume an increase in the PsV particle diameter by ~20 nm as described before for microtubule staining (Aquino et al., 2011). Thus, calculating the PSFs from the measured sizes, we assume ~80 nm as the PsVs physical size, using the following formula: PSF = . The calculated PSFs in the AlexaFluor594 and ATTO647 channels are 67 and 66 nm, respectively.
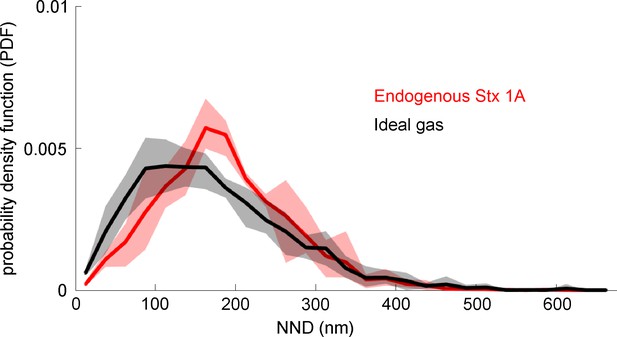
Comparing real syntaxin 1A maxima positions to a uniformly random distribution.
The observed distribution of endogenous syntaxin 1A is compared to a calculated distribution expected for an ideal gas. The analysis of the probability density function of the nearest-neighbour distances (NNDs) on real (red line; endogenous Stx1A) and randomized positions (black line, ideal gas control) is using data from the control condition shown in Figure 4A. The analysis includes 74 image pairs, for each real image a randomized/ideal gas image was created using the same maxima number, yet assuming that maxima positions are independent from each other and uniformly random distributed (for details see methods). For each single image, the NND was calculated and the corresponding probability density function (PDF) was constructed. Values are given as means ± standard deviation (SD; shaded area; n = 4; using those experiments from Figure 4 which include all three conditions).
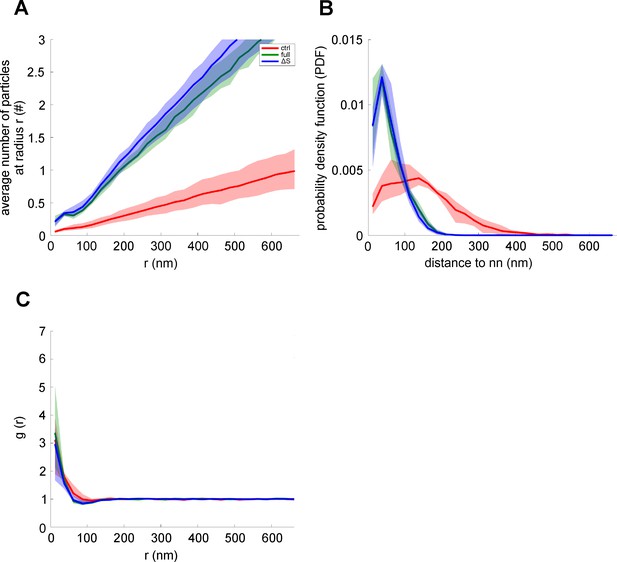
Average number of syntaxin 1A maxima at a specific distance around SNAP25 maxima, probability density function, and radial pair distribution function.
(A) Average number of syntaxin 1 A maxima ± 15 nm around the given radius (r) from a given SNAP25 maxima. As expected, compared to control (red; ctrl), increasing the total number of Stx-full (green; full), and Stx-ΔS (blue; ΔS) maxima yields a steeper increase in particles with radial distance. (B) Probability density function (PDF) over the distance of SNAP25 maxima to the nearest syntaxin 1 A maxima. The PDF illustrates the probability of finding the nearest syntaxin 1 A maxima at a given distance from a SNAP25 maxima. (C) Radial pair distribution function (g(r)) of SNAP25 and syntaxin 1 A maxima. For all plots, SNAP25 is considered the reference maxima. Values are given as means ± standard deviation (SD; shaded area; n = 4; using those experiments from Figure 4 which include all three conditions).
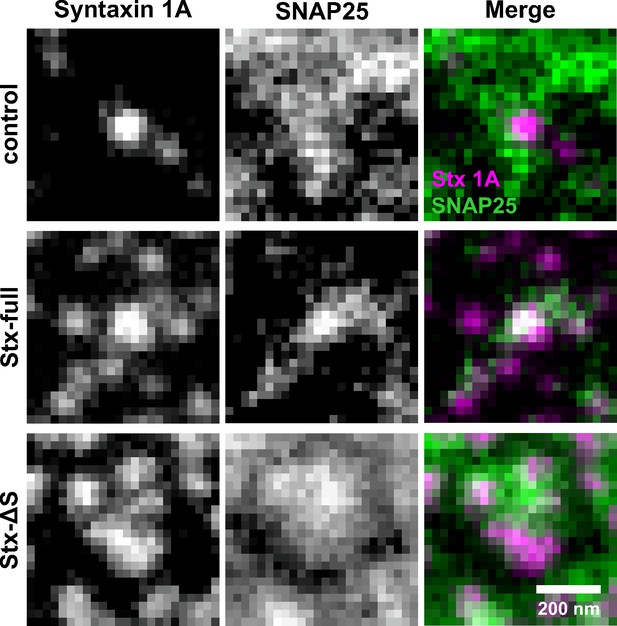
SNAP25 accumulates at syntaxin clusters.
Magnified views from images shown in Figure 4, at different arbitrary scaling. SNAP25 and syntaxin 1A signals are close to each other, but their centres do not perfectly coincide.
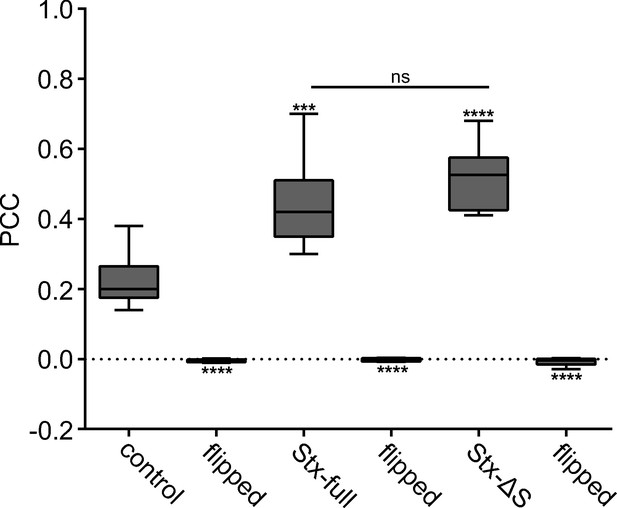
Pearson correlation coefficient (PCC) and corresponding controls.
Details regarding the average PCC values stated in Figure 4, showing that there is a significant difference between the control and Stx-full/Stx-ΔS, but not between Stx-full and Stx-ΔS. A randomization control was added (‘flipped’) where the Stx-channel is flipped vertically and horizontally before calculating the PCC. All values are shown as box plots (n = 6–9 experiments), showing the median, the 25th and 75th percentile (box), and the minimum and maximum value. Two-tailed Student’s t-test compare (1) control to Stx-full/Stx-ΔS, (2) control/Stx-full/Stx-ΔS to the respective flipped condition, and (3) Stx-full to Stx-ΔS; ns = not significant, p > 0.05, ***p < 0.001, ****p < 0.0001.
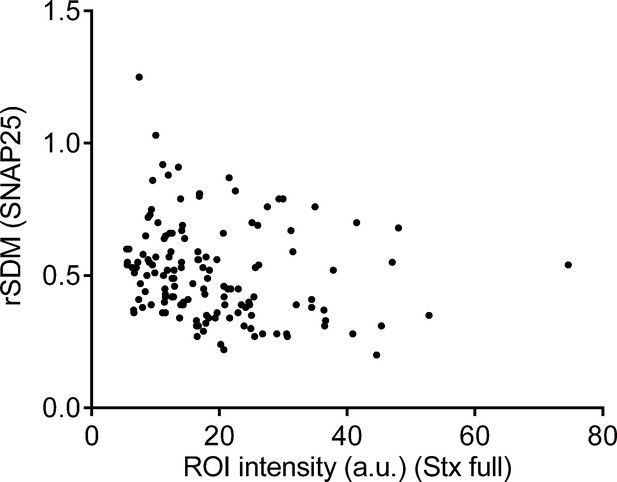
Relative standard deviation of the mean (rSDM) dependence from syntaxin expression level.
Plotting from individual membrane sheets the rSDM of the SNAP25 channel against the region of interest (ROI) intensity of the syntaxin 1A channel. Plotted are values of 139 single membrane sheets from the Stx-full condition in Figure 4.
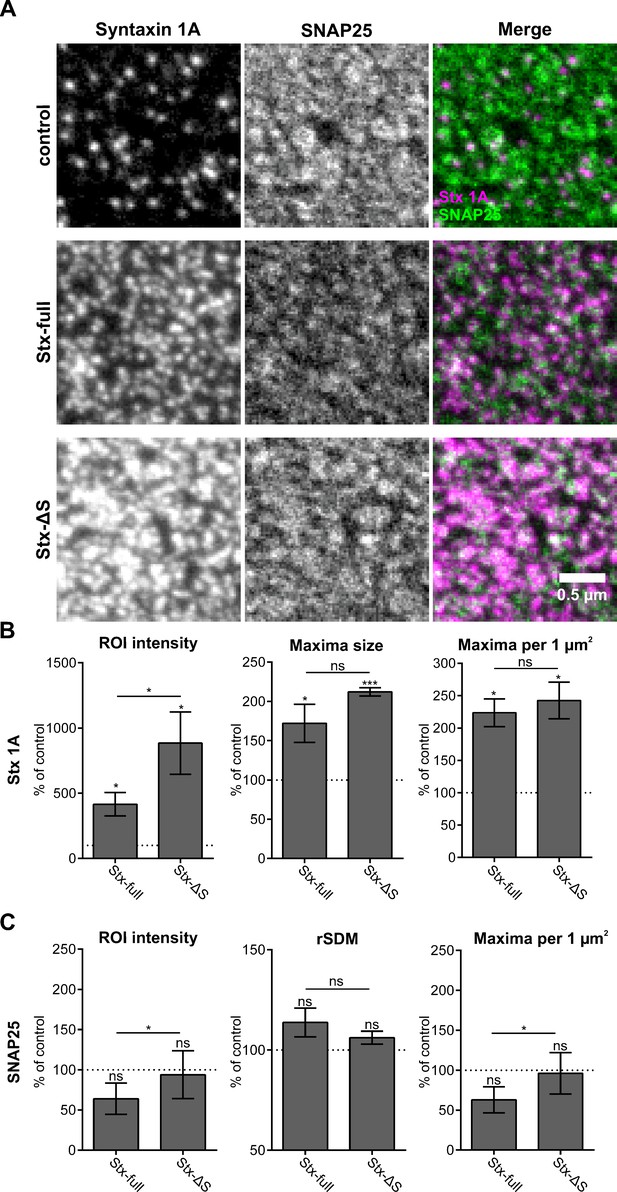
STED analysis with exchanged spectral properties of the SNAP25 and syntaxin 1A channels.
The experiment in Figure 4 was repeated switching the spectral properties of the syntaxin 1 A/SNAP25 stainings to test whether observations were independent of the wavelength. (A) Images displaying recordings from the syntaxin 1 A channel (HPC-1 antibody visualized with a goat anti-mouse ATTO647 secondary antibody), SNAP25 channel (anti-GFP nanobody coupled with ATTO594), and overlays. As in Figure 4, syntaxin 1 A maxima are sharply defined and cover only a small area, whereas SNAP25 maxima appear more diffuse and are more widespread. (B, C) Compared to Figure 4, the same trends in region of interest (ROI) intensity, maxima number, maxima size, and relative standard deviation of the mean (rSDM) are also apparent after switching fluorophores. All values are given as means ± standard deviation (SD), related to the control set to 100 % (n = 3 experiments, 20 membrane sheets were imaged per condition and experiment). Two-tailed Student’s t-test compare (1) control to Stx-full/Stx-ΔS and (2) Stx-full to Stx-ΔS; ns = not significant, p > 0.05, *p < 0.05, **p < 0.01, ***p < 0.001.
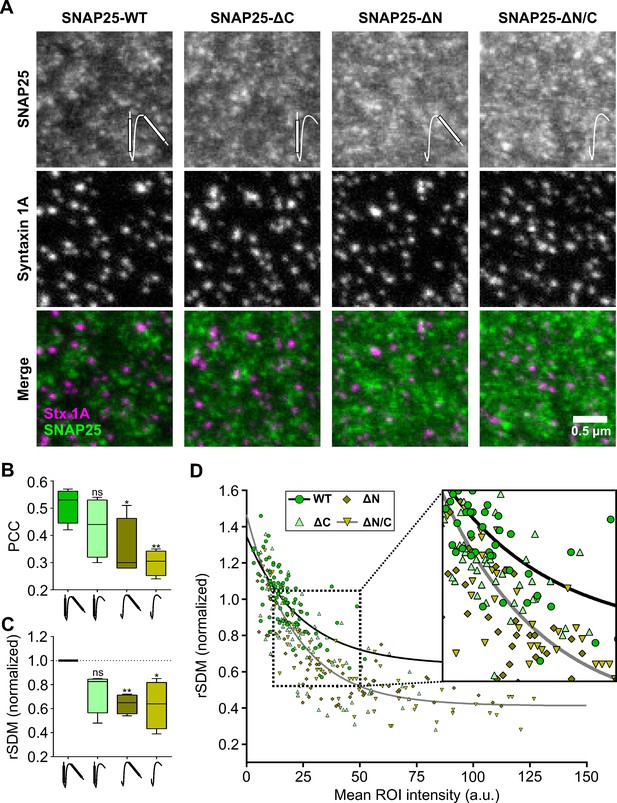
SNAP25 crowds bind to syntaxin 1A clusters via the N-terminal SNARE domain of SNAP25.
Stx-full (syntaxin 1A is stained by the HPC-1 antibody visualized by an AlexaFluor594-coupled secondary antibody) is overexpressed in PC12 cells together with GFP-SNAP25 or GFP-SNAP25 lacking either the C-terminal SNARE domain (SNAP25-ΔC), the N-terminal SNARE domain (SNAP25-ΔN), or both (SNAP25-ΔN/C) (visualized by an anti-GFP nanobody coupled to ATTO647). Membrane sheets are generated and analyzed exactly as in Figure 4. (A) Representative images from the different channels and overlays. Please note that SNAP25-wildtype (SNAP25-WT) staining was on average 40 % dimmer than for example SNAP25-ΔN. Pictograms in the upper image row illustrate the SNARE domains of SNAP25 that are depicted as white bars. (B) Pearson correlation coefficient (PCC) between GFP and syntaxin 1A channel; for control see Figure 5—figure supplement 1. (C) Clustering degrees normalized to SNAP25-WT (100 % = 0.523). Values are shown as box plots (n = 4 experiments; at least 10 membrane sheets were imaged per condition and experiment), showing the median, the 25th and 75th percentile (box), and the minimum and maximum value. Two-tailed Student’s t-test compare SNAP25-WT to SNAP25-ΔC/SNAP25-ΔN/SNAP25ΔN/C; ns = not significant, p > 0.05, *p < 0.05, **p < 0.01. (D) From individual membrane sheets, the degree of clustering (relative standard deviation of the mean, rSDM) is plotted against the mean region of interest (ROI) intensity of the GFP staining. Black and grey lines illustrate fits of a non-linear regression line (y = (a − b)e−d*x + b) to the values of SNAP25-WT (a = 1.35, b = 0.64, d = 0.042) and SNAP25-ΔN/C (a = 1.47, b = 0.41, d = 0.046), respectively.
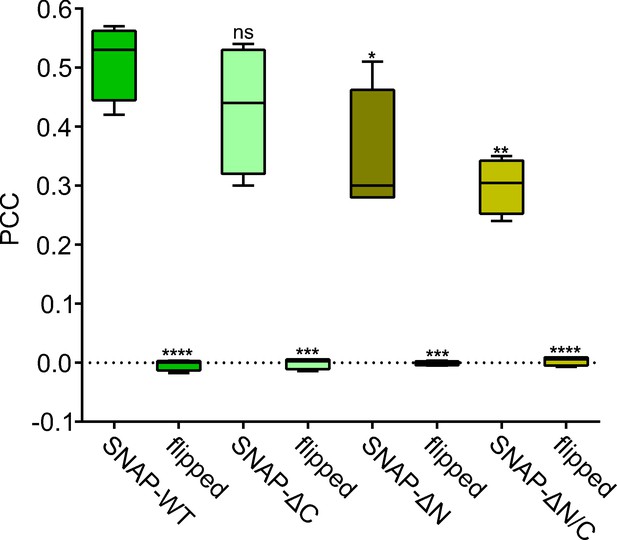
Pearson correlation coefficient (PCC) controls.
PCC between SNAP25/ SNAP25 deletion constructs and syntaxin 1A from Figure 5B and the respective controls (‘flipped’; for explanation see legend of Figure 4—figure supplement 5). Two-tailed Student’s t-test compare (1) SNAP25-WT to SNAP25-ΔC/SNAP25-ΔN/SNAP25-ΔN/C and (2) SNAP25-WT/SNAP25-ΔC/SNAP25-ΔN/SNAP25-ΔN/C to their respective flipped images, ns = not significant, p > 0.05, *p < 0.05, **p < 0.01, ***p < 0.001, ****p < 0.0001.
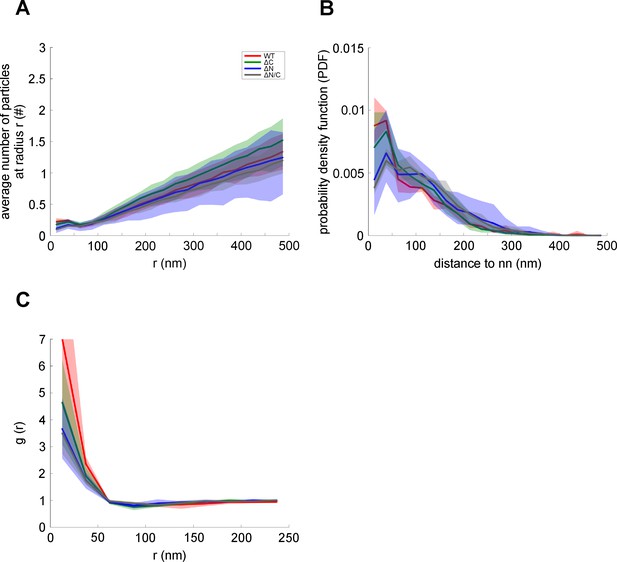
Average number of syntaxin 1A maxima at a specific distance around SNAP25/SNAP25 deletion constructs maxima, probability density function, and radial pair distribution function.
(A) Average number of syntaxin 1A maxima ± 15 nm around the given radius (r) from a given SNAP25 (WT; red), SNAP25-ΔC (ΔC; green), SNAP25-ΔN (ΔN; blue), or SNAP25-ΔN/C (ΔN/C; gray) maxima. (B) Probability density function (PDF) over the distance of SNAP25 maxima to the nearest syntaxin 1A maxima, illustrating the probability of finding the nearest syntaxin 1A maxima at a given distance from a SNAP25 maxima. It is lowest for the deletion variants missing either the N-terminal domain (SNAP25-ΔN) or both domains (SNAP25-ΔN/C). (C) Radial pair distribution function (g(r)) of SNAP25 and syntaxin 1A maxima. For all plots, SNAP25 is considered the reference maxima. Values are given as means ± standard deviation (SD; shaded area; n = 4; data from experiments shown in Figure 5).
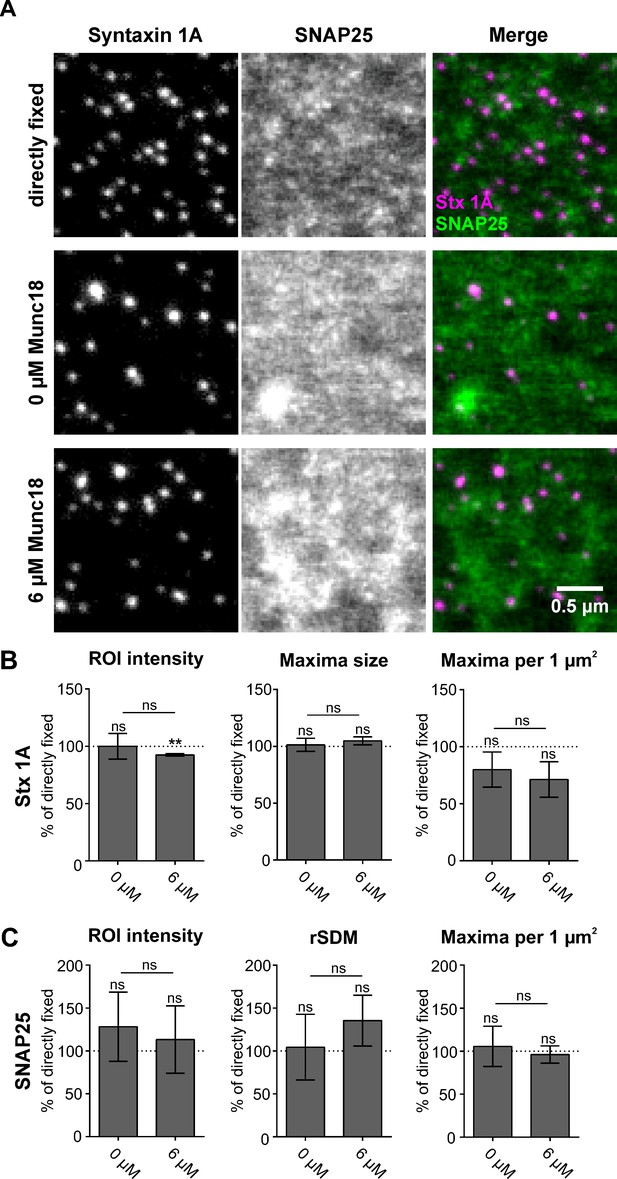
Munc18-1 does not influence SNAP25 mesoscale organization.
Membrane sheets from PC12 cells expressing GFP-SNAP25 were directly fixed or incubated at room temperature (RT) for 15 min in control buffer (0 µM Munc18) or with 6 µM Munc18, followed by immunostaining and STED microscopy analysis (as in Figure 4). (A) Example images illustrating the three conditions. Again, syntaxin 1 A is visualized by the HPC-1 antibody in combination with an AlexaFluor594-coupled secondary antibody, whereas GFP-SNAP25 is visualized by an anti-GFP nanobody coupled to ATTO647. (B) Analysis of syntaxin 1 A average region of interest (ROI) intensity, maxima size, and maxima per µm2, related to the average of directly fixed membrane sheets (set to 100%). (C) Analysis of SNAP25 average ROI intensity, clustering degree (relative standard deviation of the mean, rSDM), and maxima per µm2, related to directly fixed membrane sheets. Values are given as means ± standard deviation (SD; n = 3 experiments, at least 10 sheets were imaged per condition and experiment). Two-tailed Student’s t-test compare (1) control to 0 µM Munc18 /6 µM Munc18 and (2) 0 µM Munc18 to 6 µM Munc18; ns = not significant, p > 0.05, **p < 0.01.
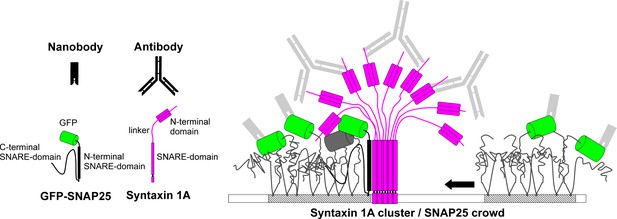
Model of SNAP25 crowd recruitment to a syntaxin 1A cluster.
Syntaxin 1A molecules form tightly packed clusters resembling a bunch of flowers. SNAP25 is enriched in cholesterol-dependent lipid phases (shaded membrane). In the unbound crowd (right), the GFP-tags are able to move freely. Hence, it is less likely that two GFPs are close enough for self-quenching. Moreover, in the unbound crowd all GFPs are accessible to the nanobody. This is different in the bound crowd (left). Here, the N-terminal SNARE domain of SNAP25 interacts with the surface of the syntaxin 1A cluster, positioning the crowd close to the trunk such that some of the GFP-tags are shielded under the cluster’s roof and are no longer accessible to nanobody binding. The GFPs pack more densely and are immobilized, which promotes self-quenching (see grey GFP barrel). One syntaxin 1A cluster can recruit several SNAP25 crowds, explaining how syntaxin controls a surplus of SNAP25. The legend shows the binding probes, GFP-SNAP25 and syntaxin 1A. SNARE domains of SNAP25 are unstructured, unless they participate in an interaction with syntaxin 1A during which they become alpha-helical (in the cartoon alpha-helicity is indicated by drawing the SNARE domain as a rectangle).
Tables
| Reagent type (species) or resource | Designation | Source or reference | Identifiers | Additional information |
|---|---|---|---|---|
| Cell line (rattus norvegicus) | PC12 cells | Other, Weber et al., 2017 | Gift from Rolf Heumann (Bochum, Germany), similar to clone 251; cell line maintained in lab | |
| Cell line (Homo sapiens) | HepG2 cells | Cell line services | Cat# 300198; RRID:CVCL_0027 | Cell line maintained in lab |
| Strain, strain background (Escherichia coli) | BL21 (DEf3) | Stratagene (Agilent Technologies), Parisotto et al., 2014 | ||
| Recombinant DNA reagent | Stx-full | Merklinger et al., 2017 | N/A | Rat syntaxin 1A (NP_446240.2) in pEGFP-N1 vector (Clontech) |
| Recombinant DNA reagent | Stx-ΔS | Merklinger et al., 2017 | N/A | Rat syntaxin 1 A deletion construct (aa 191–2818 missing) in pEGFP-N1 vector (Clontech) |
| Recombinant DNA reagent | GFP-SNAP25; SNAP25-WT | Halemani et al., 2010 | N/A | Rat SNAP25B (NP_112253.1) in pEGFP-C1 vector (Clontech) |
| Recombinant DNA reagent | SNAP25-ΔN | Halemani et al., 2010 | N/A | Rat SNAP25B deletion construct (aa 14–79 missing) in pEGFP-C1 vector (Clontech) |
| Recombinant DNA reagent | SNAP25-ΔC | Halemani et al., 2010 | N/A | Rat SNAP25B deletion construct (aa 143–202 missing) in pEGFP-C1 vector (Clontech) |
| Recombinant DNA reagent | SNAP25-ΔN/C | Halemani et al., 2010 | N/A | Rat SNAP25B deletion construct (aa 14–79 and 143–202 missing) in pEGFP-C1 vector (Clontech) |
| Peptide, recombinant protein | Munc18; Munc18-1 | Parisotto et al., 2014 | N/A | cDNA of rat Munc18-1 subcloned into the pEG(KG) vector (gift of Dr. Richard Scheller) |
| Antibody | anti-HPC-1 (mouse monoclonal) | Sigma-Aldrich | cat# S0664; RRID:AB_477483 | IF: (1:500)WB: (1:1000) |
| Antibody | anti-SNAP25 (clone 71.1, mouse monoclonal) | Synaptic-Systems | cat# 111-011 | WB: (1:1000) |
| Antibody | anti-SNAP25 (rabbit polyclonal) | Synaptic Systems | cat# 111-002 | IF: (1:200) |
| Antibody | anti-α-Tubulin (rabbit polyclonal) | Cell Signaling | cat# 2144 S | WB: (1:5000) |
| Antibody | anti-β-actin (clone 13E5, rabbit polyclonal) | Cell Signaling | cat# 4970 | WB: (1:5000) |
| Antibody | anti-GFP (rabbit polyclonal) | Chromotek | cat# PABG1; RRID:AB_2749857 | WB: (1:2000) |
| Antibody | anti-myc (clone Myc.A7, mouse monoclonal) | Invitrogen | cat# MA1-21316; RRID:AB_558473 | WB: (1:1000) |
| Antibody | 16L1-312F (mouse monoclonal) | Aliquot kindly provided by Luise Florin, University Medical Centre Mainz, Knappe et al., 2007 | IF: (1:500) | |
| Antibody | anti-GFP nanobody (labelled with ATTO647) | Chromotek | cat# gba647n-100 | IF: (1:200) |
| Antibody | anti-GFP nanobody (labelled with ATTO594) | Chromotek | cat# gba594n-100 | IF: (1:200) |
| Commercial assay or kit | anti-GFP agarose beads | Chromotek | cat# gta-20 | (25 µl beads per probe) |
| Chemical compound, drug | isopropyl β-D-1-thiogalactopyranoside | Peqlab brand | cat# 730–1497 | (0.3 mM) |
| Software, algorithm | ImageJ | NIH; Schindelin et al., 2012, | RRID:SCR_003070 | |
| Software, algorithm | MatLab | MathWorks | RRID:SCR_001622 | |
| Software, algorithm | GraphPad Prism 6 | GraphPad software | RRID:SCR_002798 |
Additional files
-
Transparent reporting form
- https://cdn.elifesciences.org/articles/69236/elife-69236-transrepform1-v2.docx
-
Source data 1
Statistical reporting.
- https://cdn.elifesciences.org/articles/69236/elife-69236-supp1-v2.xlsx





