Analysis of the mechanosensor channel functionality of TACAN
Figures
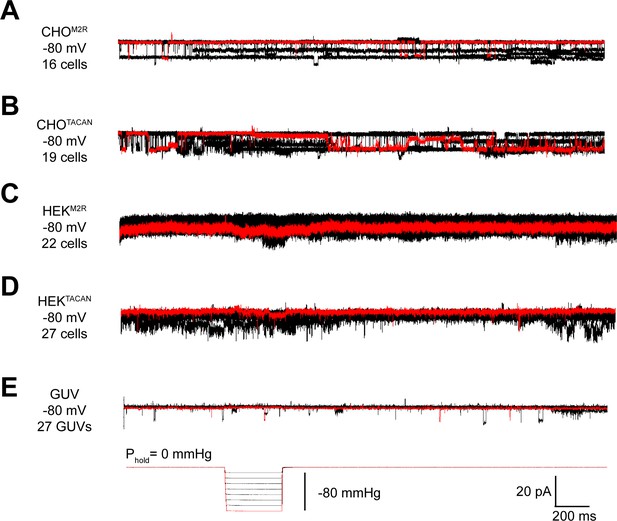
TACAN does not produce mechanically evoked currents.
(A, B) Representative excised inside-out patch recordings of M2 muscarinic receptor (M2R, A) and TACAN (B) transfected into CHO-K1 cells. (C, D) Representative excised inside-out patch recordings of M2R (C) and TACAN (D) transfected into piezo-1 knockout HEK-293T cells. (E) Representative excised inside-out patch recording of TACAN reconstituted in giant unilamellar vesicles (GUVs). All recordings were performed with identical pipette and bath solution containing 10 mM HEPES pH 7.4, 140 mM KCl, and 1 mM MgCl2 (~300 Osm/L). Traces were obtained holding at –80 mV with a pressure pulse protocol shown at the bottom: 0 to –80 mmHg with 10 mmHg step. Traces colored in red represent the observed currents with –80 mmHg pressure pulse.
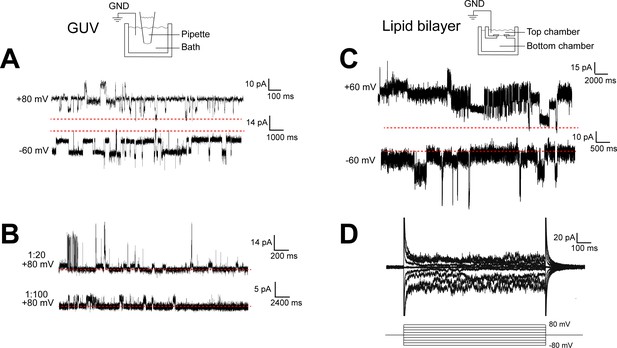
TACAN produces heterogenous currents in reconstituted systems.
(A, B) Representative recordings of TACAN from excised giant unilamellar vesicle (GUV) patches. Symmetrical buffers (10 mM HEPES pH 7.4, 140 mM KCl, 1 mM MgCl2) were used in pipette and bath. The dashed red lines indicate the baseline currents. (A) Traces from GUVs at 1:20 protein-to-lipid ratio (w/w) holding at +80 mV and –60 mV. (B) Traces from GUVs at 1:20 and 1:100 protein-to-lipid ratio (w/w) holding at +80 mV. (C, D) Representative traces of TACAN reconstituted in a lipid bilayer. Symmetrical buffers (10 mM HEPES pH 7.4, 150 mM KCl) were used in top and bottom chambers. The dashed red lines indicate the baseline currents. (C) Traces while holding at +60 mV and –60 mV. (D) Traces recorded during a voltage family from –80 to +80 mV in 20 mV increment.
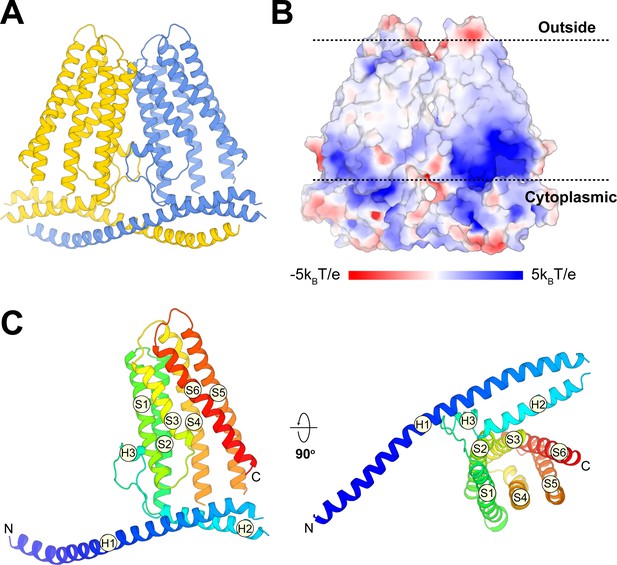
Overall structure of TACAN.
(A) Cartoon representation of the TACAN dimer with each protomer colored uniquely. (B) Surface charge distribution and the possible orientation of TACAN, blue and red representing the positive and negative charges, respectively. The membrane is demarcated by dashed lines. (C) Tertiary structure of TACAN protomer viewed from the side and the cytoplasmic side. The protein is colored rainbow from N-terminus (blue) to C-terminus (red). The six transmembrane helices (S1–S6), two horizontal helices (H1 and H2), as well as a short helix (H3) in between are labeled.
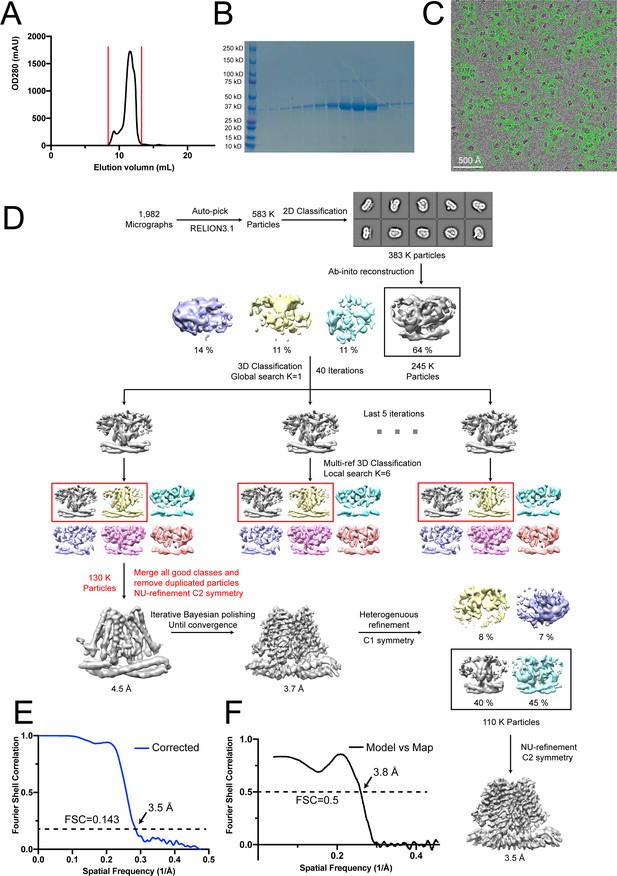
Cryo-EM analysis of wild-type TACAN.
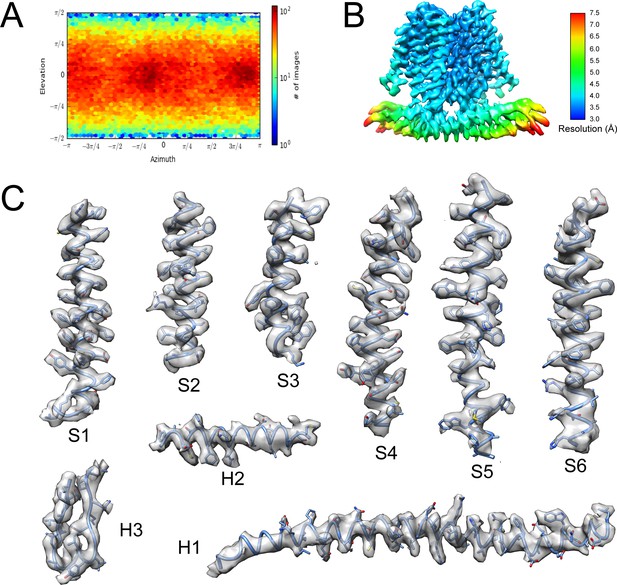
(A) Size-exclusion chromatography of TACAN on a Superdex 200 Increase 10/300 GL column. (B) SDS- PAGE of fractions from size-exclusion chromatography between the red vertical lines shown in (A). (C) Representative cryo-EM image of the TACANWT, selected particles are circled in green, and the scale bar is 50 nm. (D) Cryo-EM data processing workflow for TACANWT. (E) Gold-standard Fourier shell correlation (FSC) curve after correction for masking effects. The resolution was estimated based on the FSC = 0.143 criterion. (F) FSC curve of the refined model versus EM map. The resolution was estimated based on the FSC = 0.5 criterion.
Representative density in the cryo-EM map of wild-type TACAN.
(A) The angular distribution of final reconstruction. (B) Local resolution map of TACANWT. (C) Cryo-EM densities for selected regions of TACANWT (contour level 4.4 in COOT).
Structural analysis of wild-type TACAN.
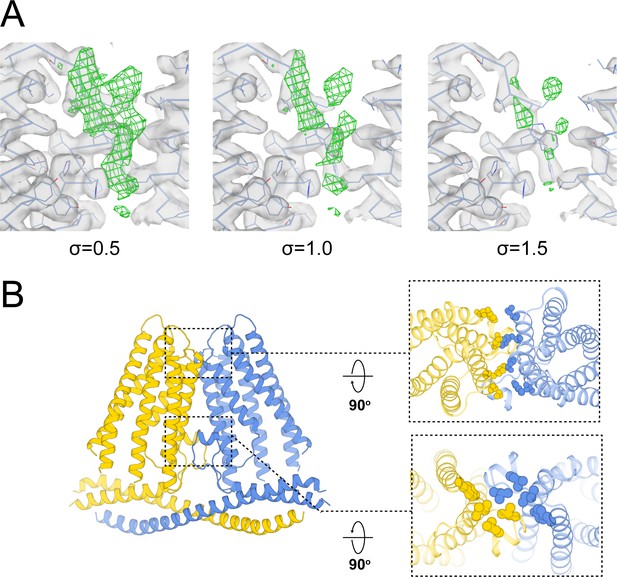
(A) The non-protein density (green mesh) observed in TACANWT is shown at different contour levels (UCSF Chimera). The surrounding protein density is shown as gray surface. (B) Interactions of TACAN protomers at the dimer interface. The protomers are colored uniquely, and the interfacial residues are shown in spheres.
TACAN shares structural homology to the fatty acid elongase ELOVL7.
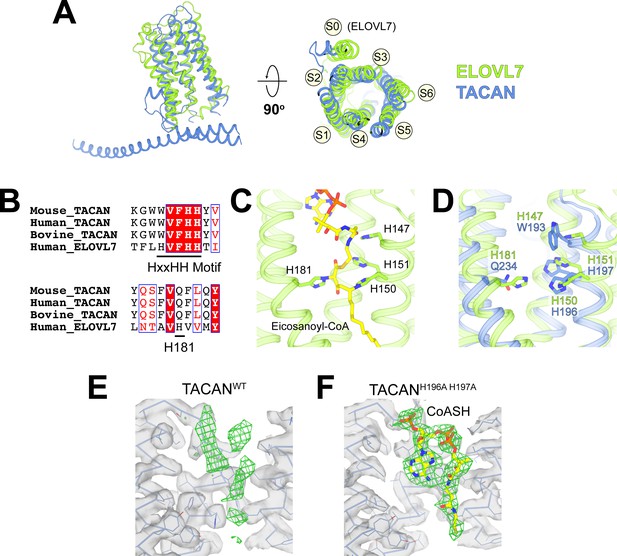
(A) Superposition between TACAN (blue) and ELOVL7 (green) protomers. Transmembrane helices are labeled to correspond with the topology of TACAN. The extra transmembrane helix in ELOVL7 is labeled as S0. (B) Sequence alignment of TACAN from different species and human ELOVL7 with conserved residues highlighted. The catalytically important HxxHH motif (His147, His150, and His151) and His181 in ELOVL7 are underlined. (C) Structure details of the interactions between the HxxHH motif, His181 (sidechains shown as sticks), and eicosanoyl-CoA (shown as sticks) in ELOVL7 (PDB: 6Y7F). His150 and His181 are covalently linked to eicosanoyl-CoA. (D) Zoom-in view of the ELOVL7 (green) catalytic center with TACAN (blue) superimposed. (E, F) The non-protein density (green mesh) in the narrow tunnel of wild-type (E) and His196Ala, His197Ala mutant of TACAN (F). Protein density is represented as transparent surface (gray) with protein shown as lines and ribbons. The two maps are shown at the same contour level. CoASH in mutant TACAN is shown as sticks and colored according to atom type.
Cryo-EM analysis of the His196Ala His197Ala mutant TACAN.
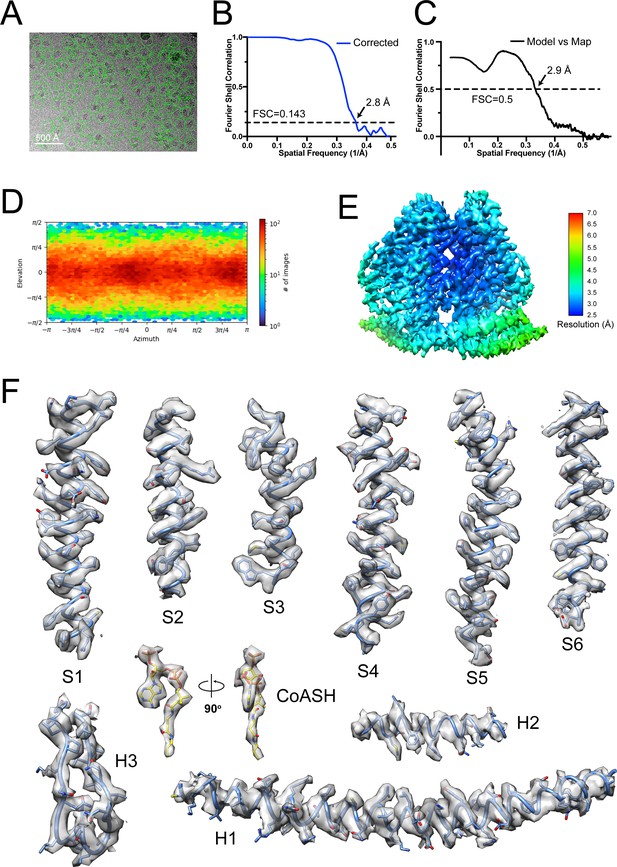
(A) Representative cryo-EM image of the TACANH196A H197A, selected particles are circled in green, and the scale bar is 50 nm. (B) Gold-standard Fourier shell correlation (FSC) curve after correction for masking effects. The resolution was estimated based on the FSC = 0.143 criterion. (C) FSC curve of the refined model versus EM map. The resolution was estimated based on the FSC = 0.5 criterion. (D) The angular distribution of final reconstruction. (E) Local resolution map of TACANH196A H197A. (F) Cryo-EM densities for selected regions of TACANH196A H197A (contour level 5.2 in COOT). The CoASH molecule is colored according to atom type.
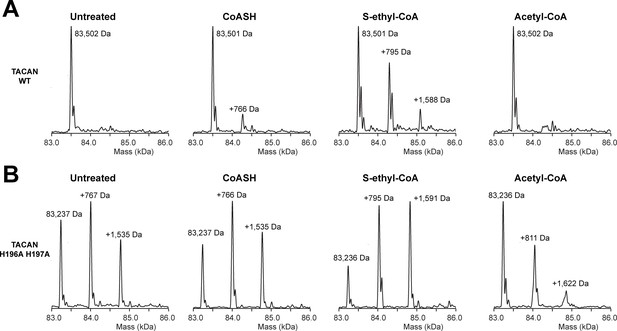
Native mass spectrometry indicates the presence of coenzyme A in the mutant TACAN sample.
(A, B) Mass species detected in purified wild-type (A) and His196Ala, His197Ala mutant (B) TACAN protein without treatment (“untreated”), or incubated with CoASH (MW = 767.5 Da), S-ethyl-CoA (MW = 795.6 Da), or acetyl-CoA (MW = 809.6 Da).
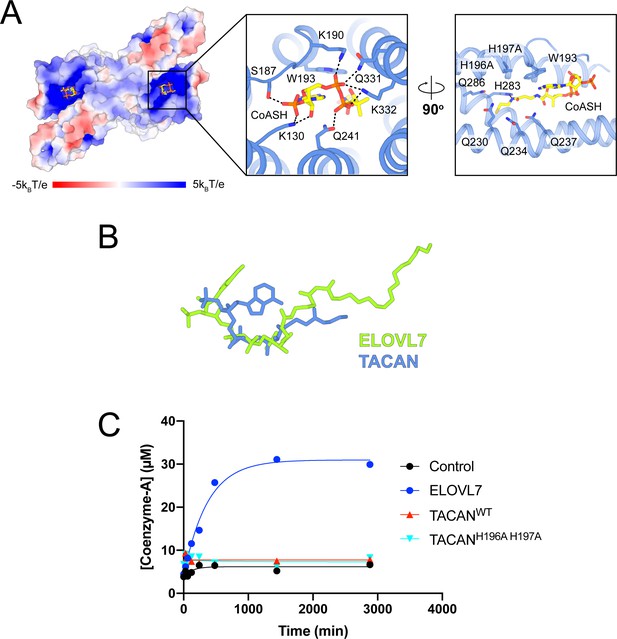
TACAN is co-purified with coenzyme A molecules.
(A) Binding details of CoASH in TACANH196A H197A. Surface charge is represented from blue (positive charges) to red (negative charges). Neighboring residues are shown as sticks, and the hydrogen bonding is indicated by a black dash. A CoASH molecule is shown as sticks and colored according to atom type. (B) Conformation comparison of eicosanol-CoA in ELOVL7 (green) and CoASH in TACAN (blue) based on the alignment in Figure 4A. (C) CoASH releasing activity of ELOVL7 and TACAN. Proteoliposomes of TACAN and ELOVL7 reconstituted in soy L-α-phosphatidylcholine (soy-PC) at 1:50 protein-to-lipid ratio (w/w) with 10 μg protein and 500 μg soy-PC were used. Empty proteoliposomes made of soy-PC were used as control. ELOVL7 showed significant activity, while neither the wild-type nor His196Ala His197Ala mutant of TACAN showed any activity.
Tables
Cryo-EM data collection and refinement statistics, related to Figures 3 and 4.
| TACANWT | TACANH196A H197A | |
|---|---|---|
| EMDB ID | EMD-24107 | EMD-24108 |
| PDB ID | 7N0K | 7N0L |
| Data collection | ||
| Microscope | Titan Krios | |
| Detector | K2 summit | K3 summit |
| Voltage (kV) | 300 | 300 |
| Pixel size (Å) | 1.03 | 0.515 |
| Total electron exposure(e-/Å2) | 75.4 | 56.6 |
| Defocus range (μm) | 0.7–2.1 | 0.8–2.2 |
| Micrographs collected | 2,071 | 10,541 |
| Reconstruction | ||
| Final particle images | 110,090 | 155,946 |
| Pixel size (Å) | 1.03 | 1.03 |
| Box size (pixels) | 256 | 256 |
| Resolution (Å)(FSC = 0.143) | 3.5 | 2.8 |
| Map sharpening B-factor (Å2) | –20 | –3.4 |
| Model composition | ||
| Non-hydrogen atoms | 5,156 | 5,272 |
| Protein residues | 626 | 626 |
| Ligands | 0 | 2 |
| Metals | 0 | 0 |
| Refinement | ||
| Model-to-map CC (mask) | 0.77 | 0.80 |
| Model-to-map CC (volume) | 0.73 | 0.81 |
| R.m.s deviations | ||
| Bond length (Å) | 0.003 | 0.003 |
| Bond angles (°) | 0.54 | 0.52 |
| Validation | ||
| MolProbity score | 2.09 | 2.22 |
| Clash score | 7.86 | 9.10 |
| Ramachandran plot | ||
| Outliers (%) | 0 | 0 |
| Allowed (%) | 0.98 | 1.95 |
| Favored (%) | 99.02 | 98.05 |
| Rotamer outliers (%) | 7.46 | 9.23 |
| C-beta deviations (%) | 0 | 0 |
-
FSC: Fourier shell correlation.
| Reagent type (species) or resource | Designation | Source or reference | Identifiers | Additional information |
|---|---|---|---|---|
| Gene (Mus musculus TMEM120A) | M. musculus TACAN | Synthetic | Synthesized at GeneWiz. | |
| Gene (Homo sapiens TMEM120A) | H. sapiens TACAN | Synthetic | Synthesized at GeneWiz. | |
| Gene (Homo sapiens ELOVL7) | H. sapiens ELOVL7 | Synthetic | Synthesized at GeneWiz. | |
| Strain, strain background (Escherichia coli) | DH10Bac | Thermo Fisher Scientific | 10361012 | |
| Recombinant DNA reagent | TACAN-eGFP BacMam | This study | ||
| Recombinant DNA reagent | ELOVL7-eGFP BacMam | This study | ||
| Recombinant DNA reagent | Halo-M2R-eGFP BacMam | This study | ||
| Cell line (Spodoptera frugiperda) | Sf9 | ATCC | CRL-1711 | Cells purchased from ATCC, and we have confirmed there is no mycoplasma contamination |
| Cell line (Chinese hamster) | CHO-K1 | ATCC | CRL-9618 | Cells purchased from ATCC, and we have confirmed there is no mycoplasma contamination |
| Cell line (Homo sapiens) | HEK293S GnTI- | ATCC | CRL-3022 | Cells purchased from ATCC, and we have confirmed there is no mycoplasma contamination |
| Cell line (Homo sapiens) | Piezo1 knockout HEK293T | https://digitalcommons.rockefeller.edu/cgi/viewcontent.cgi?article=1422&context=student_theses_and_dissertations | We have confirmed there is no mycoplasma contamination | |
| Chemical compound, drug | SF-900 II SFM medium | Gibco | 11330-032 | |
| Chemical compound, drug | L-Glutamine (100×) | Gibco | 25030-081 | |
| Chemical compound, drug | Pen Strep | Gibco | 15140-122 | |
| Chemical compound, drug | Grace’s insect medium | Gibco | 11605-094 | |
| Chemical compound, drug | Freestyle 293 medium | Gibco | 12338-018 | |
| Chemical compound, drug | DMEM/F-12 medium | Gibco | 11605-094 | |
| Chemical compound, drug | DMEM | Gibco | 11965-118 | |
| Chemical compound, drug | Fetal bovine serum | Gibco | 16000-044 | |
| Chemical compound, drug | Cellfectin II reagent | Invitrogen | 10362100 | |
| Chemical compound, drug | FuGENE HD transfection reagent | Promega | E2312 | |
| Chemical compound, drug | Cholesteryl hemisuccinate (CHS) | Anatrace | CH210 | |
| Chemical compound, drug | n-Decyl-β-D-maltopyranoside (DM) | Anatrace | D322S | |
| Chemical compound, drug | Lauryl maltose neopentyl glycol (LMNG) | Anatrace | NG310 | |
| Chemical compound, drug | Digitonin | Millipore Sigma | 300410 | |
| Chemical compound, drug | Coenzyme A trilithium salt (CoASH) | Sigma-Aldrich | C3019 | |
| Chemical compound, drug | Acetyl coenzyme A sodium salt (acetyl-CoA) | Sigma-Aldrich | A2056 | |
| Chemical compound, drug | S-Ethyl-coenzyme A sodium salt (S-ethyl-CoA) | Jena-Biosciences | NU-1168 | |
| Chemical compound, drug | Malonyl coenzyme A lithium salt (malonyl-CoA) | Sigma-Aldrich | M4263 | |
| Chemical compound, drug | Stearoyl coenzyme A lithium salt (stearoyl-CoA) | Sigma-Aldrich | S0802 | |
| Chemical compound, drug | (1H, 1H, 2H, 2H-Perfluorooctyl)phosphocholine (FFC8) | Anatrace | F300F | |
| Commercial assay or kit | CNBr-activated Sepharose beads | GE Healthcare | 17-0430-01 | |
| Commercial assay or kit | Superdex 200 Increase 10/300 GL | GE Healthcare Life Sciences | 28990944 | |
| Commercial assay or kit | R1.2/1.3 400 mesh Au holey carbon grids | Quantifoil | 1210627 | |
| Commercial assay or kit | Coenzyme A (CoA) Assay Kit | Sigma-Aldrich | MAK034 | |
| Software, algorithm | RELION 3.0 | https://doi.org/10.7554/eLife.42166.001 | http://www2.mrc-lmb.cam.ac.uk/relion | |
| Software, algorithm | RELION 3.1 | https://doi.org/10.1101/798066 | http://www2.mrc-lmb.cam.ac.uk/relion | |
| Software, algorithm | MotionCor2 | https://doi.org/10.1038/nmeth.4193 | http://msg.ucsf.edu/em/software/motioncor2.html | |
| Software, algorithm | Gctf 1.0.6 | https://doi.org/10.1016/j.jsb.2015.11.003 | https://www.mrc-lmb.cam.ac.uk/kzhang/Gctf/ | |
| Software, algorithm | CtfFind4.1.8 | https://doi.org/10.1016/j.jsb.2015.08.008 | http://grigoriefflab.janelia.org/ctffind4 | |
| Software, algorithm | CryoSPARC 2.9.0 | https://doi.org/10.7554/eLife.46057.001 | https://cryosparc.com/ | |
| Software, algorithm | COOT | https://doi.org/10.1107/S0907444910007493 | http://www2.mrc-lmb.cam.ac.uk/personal/pemsley/coot | |
| Software, algorithm | PHENIX | https://doi.org/10.1107/S0907444909052925 | https://www.phenix-online.org | |
| Software, algorithm | Adobe Photoshop version 16.0.0 (for figure preparation) | Adobe Systems, Inc. | ||
| Software, algorithm | GraphPad Prism version 8.0 | GraphPad Software | ||
| Software, algorithm | MacPyMOL: PyMOL v2.0 Enhanced for Mac OS X | Schrodinger LLC | https://pymol.org/edu/?q=educational/ | |
| Software, algorithm | Chimera | https://doi.org/10.1002/jcc.20084 | https://www.cgl.ucsf.edu/chimera/download.html | |
| Software, algorithm | Serial EM | https://doi.org/10.1016/j.jsb.2005.07.007 | http://bio3d.colorado.edu/SerialEM | |
| Software, algorithm | pClamp | Axon Instruments, Inc | ||
| Software, algorithm | Thermo Xcalibur Qual Browser (v. 4.2.47) | Thermo Fisher Scientific | ||
| Software, algorithm | UniDec v. 4.2.0 | Marty et al., 2015; Reid et al., 2018 | https://github.com/michaelmarty/UniDec/releases |





