IRAK1-dependent Regnase-1-14-3-3 complex formation controls Regnase-1-mediated mRNA decay
Figures
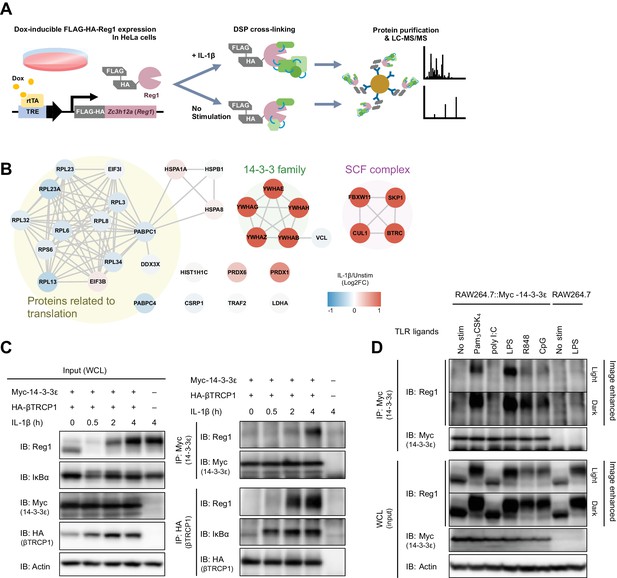
IL-1β or TLR1/2/4/7/8/9-ligand stimulation induces Regnase-1-14-3-3 interaction.
(A) Schematic illustration of the DSP-crosslinking workflow. (B) Protein-protein interaction of the Regnase-1 (Reg1)-associating proteins. Each node represents Regnase-1 associating protein. The proteins whose association with Regnase-1 is weakened or enhanced in IL-1β-stimulated cells are colored in blue or red, respectively. (C) Immunoblot analysis of immunoprecipitates (IP: Myc or IP: HA) and WCL (whole cell lysates) from HeLa cells transiently expressing Myc-14-3-3ε and HA-βTRCP1 stimulated with IL-1β (10 ng/ml) for indicated time. (D) Immunoblot analysis of immunoprecipitates (IP: Myc) and WCL from RAW264.7 or RAW264.7 stably expressing Myc-14-3-3ε stimulated with Pam3CSK4 (10 ng/ml), poly I:C (100 μg/ml), LPS (100 ng/ml), R848 (100 nM), or CpG DNA (1 μM) for 4 hr.
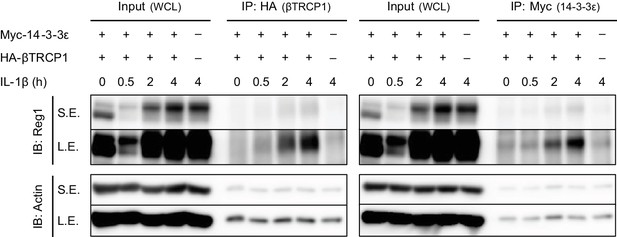
Regnase-1 binds to 14-3-3 and βTRCP in response to IL-1β stimulation.
Immunoblot analysis of immunoprecipitates (IP: Myc or IP: HA) and WCL from HeLa cells transiently expressing Myc-14-3-3ε and HA-βTRCP1 stimulated with IL-1β (10 ng/ml) for indicated time. The immunoprecipitates and corresponding WCL samples, both of which were used in Figure 1C, were applied to the same gel and the membranes were blotted with anti Regnase-1 antibody as well as anti-Actin antibody.
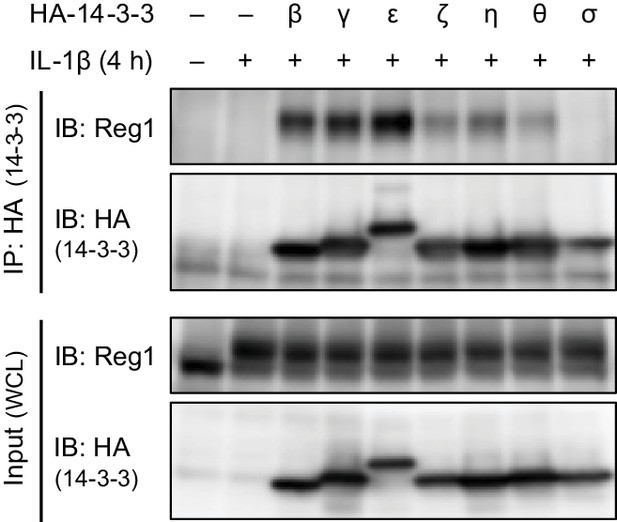
Regnase-1 binds to 14-3-3β/γ/ε/ζ/η/θ but not 14-3-3σ.
Immunoblot analysis of immunoprecipitates (IP: HA) and WCL from HeLa cells transiently expressing HA-14-3-3β, γ, ε, ζ, η, θ, or σ stimulated with IL-1β (10 ng/ml) for 4 hr.
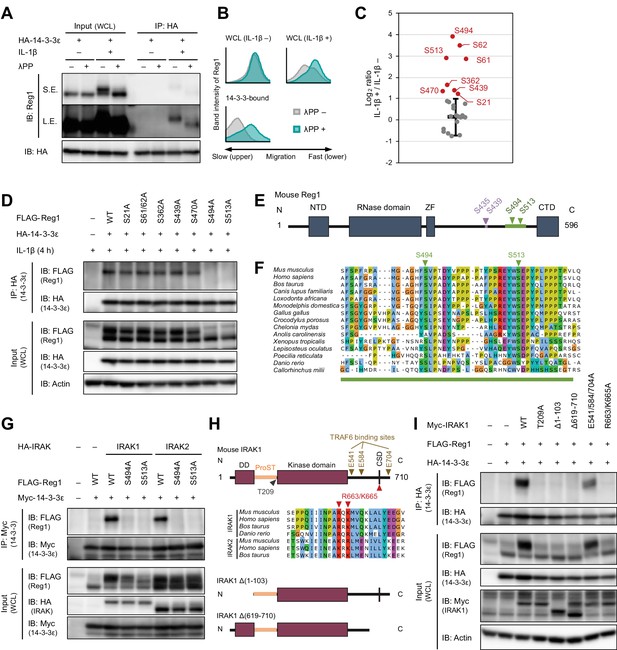
IL-1β-induced phosphorylation of Regnase-1 at S494 and S513 is necessary for Regnase-1-14-3-3 binding.
(A) Immunoblot analysis of λPP-treated immunoprecipitates (IP: HA) and WCL from HeLa cells transiently expressing HA-14-3-3ε stimulated with IL-1β (10 ng/ml) for 4 hr. S.E.: short exposure, L.E.: long exposure. (B) The intensity of Regnase-1-bands in (A). (C) Quantitation of phosphosites on Regnase-1 in HeLa cells stimulated with or without IL-1β (10 ng/ml) for 4 hr. Each dot shows phosphosite quantitative ratio between IL-1β + and IL-1β -. Phosphosites with log2 ratio > one were colored with red. Black horizontal line shows Regnase-1 protein quantitative ratio derived from the average of non-phosphopeptide quantitative ratios, and its error bars show the standard deviation. (D) Immunoblot analysis of immunoprecipitates (IP: HA) and WCL from HeLa cells transiently expressing HA-14-3-3ε and FLAG-Regnase-1-WT or indicated mutants stimulated with IL-1β (10 ng/ml) for 4 hr. (E) Schematic illustration of Regnase-1 protein. The amino acid sequence including S494 and S513 shown in (F) is highlighted in green. NTD: N-terminal domain, ZF: Zinc finger domain, CTD: C-terminal domain. (F) The amino acid sequences including S494 and S513 of Regnase-1 from mouse and other indicated vertebrates. (G) Immunoblot analysis of immunoprecipitates (IP: Myc) and WCL from HeLa cells transiently expressing Myc-14-3-3ε, HA-IRAK1/2, and FLAG-Regnase-1-WT or indicated mutants. (H) Schematic illustration of IRAK1 protein. The amino acid sequence in CSD of IRAK1 and IRAK2 from mouse and other indicated vertebrates are also shown. DD: Death domain, CSD: C-terminal structural domain. (I) Immunoblot analysis of immunoprecipitates (IP: HA) and WCL from HeLa cells transiently expressing FLAG-Regnase-1-WT, HA-14-3-3ε, and Myc-IRAK1-WT or indicated mutants.
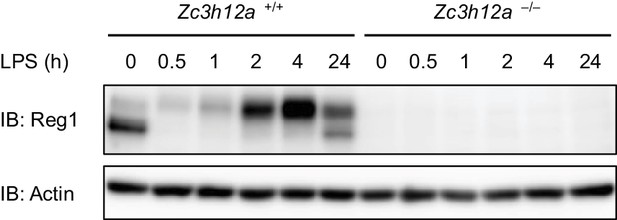
Regnase-1 bands migrate slower in LPS-stimulated samples.
Immunoblot analysis of Zc3h12aWT/WT and Zc3h12a–/– thioglycollate-elicited PECs stimulated with LPS (100 ng/ml) for indicated time.
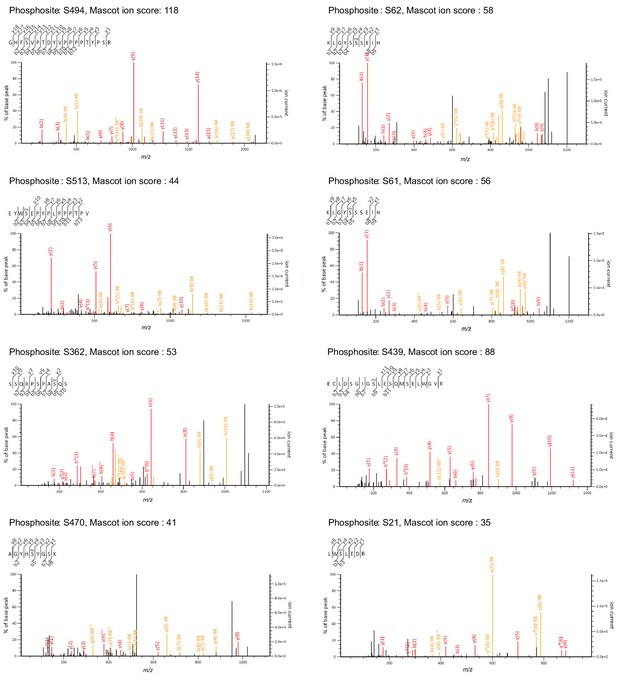
Candidate spectra of Regnase-1 phosphopeptides with confident site localization.
Only quantitatively altered phosphopeptides are shown. Fragment ions containing the N-(b-type ions) or C-(y-type ions) terminus are labeled with red (without neutral loss) or orange (with neutral-loss).
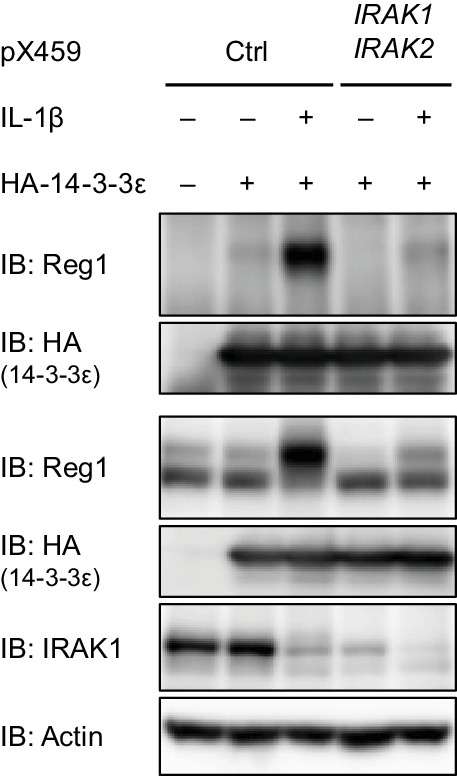
Regnase-1-14-3-3 interaction is impaired in IRAK1/2-depleted cells.
HeLa cells were transfected with pX459 to knockout IRAK1 and IRAK2. After the selection with puromycin, the cells were transfected with 14-3-3ε and stimulated with IL-1β (10 ng/ml) for 4 hr. Immunoblot analysis of immunoprecipitates (IP: HA) and WCL is shown.
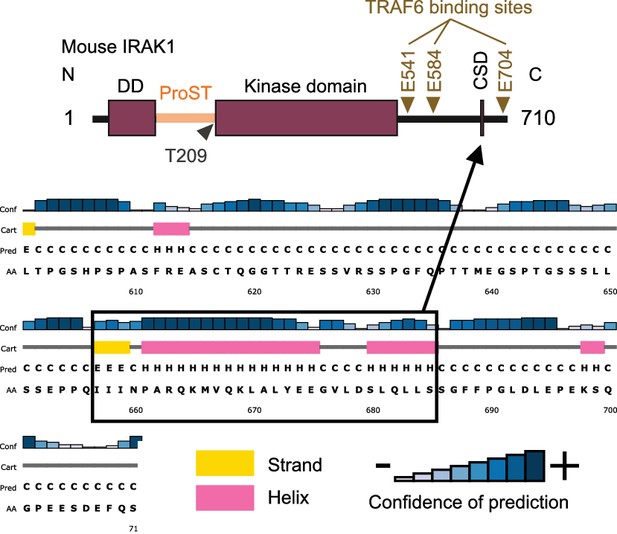
Schematic illustration of IRAK1.
The result of secondary structure prediction is shown below. DD: Death domain, CSD: C-terminal structural domain.
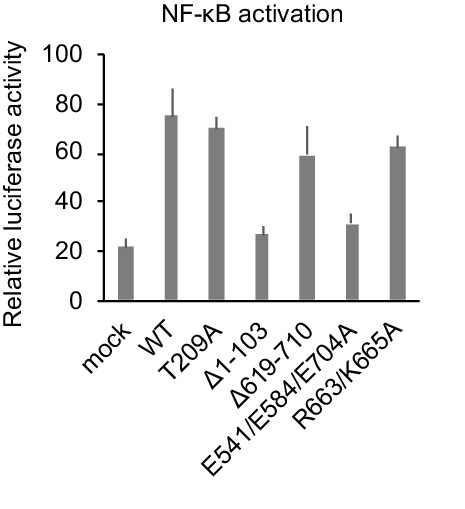
R663/K665A mutation does not abrogate IRAK1-mediated NF-κB activation.
Luciferase activity of HeLa cells transiently transfected with NF-κB luciferase reporter plasmid together with expression plasmids of IRAK1-WT or indicated mutants.
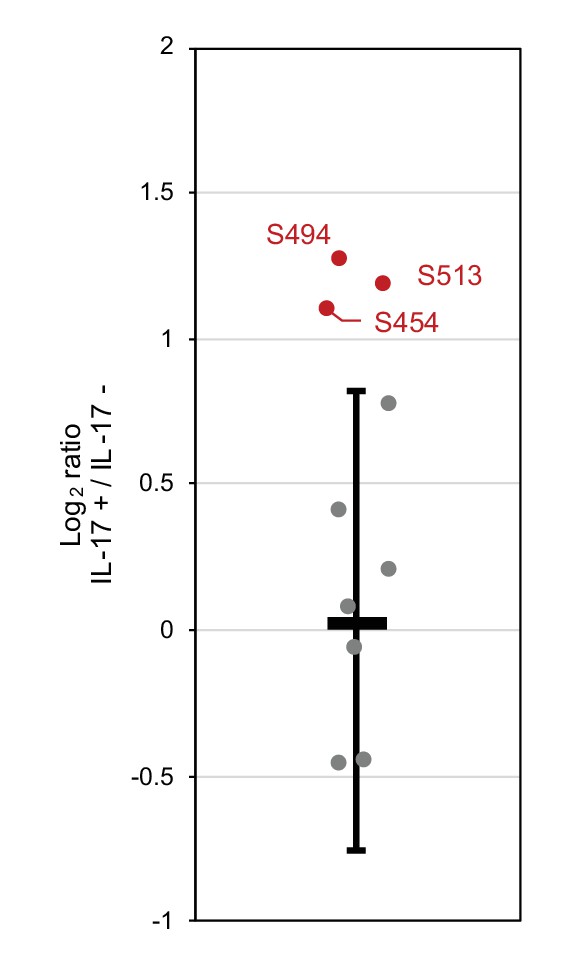
IL-17A stimulation induces phosphorylation at S494 and S513 of Regnase-1.
Quantitation of phosphosites on Regnase-1 in HeLa cells stimulated with or without IL-17A (50 ng/ml) for 4 hr. Each dot shows phosphosite quantitative ratio between IL-17A+ and IL-17A-. Phosphosites with log2 ratio > 1 were colored with red. Black horizontal line shows Regnase-1 protein quantitative ratio derived from the average of non-phosphopeptide quantitative ratios, and its error bars show the standard deviation.
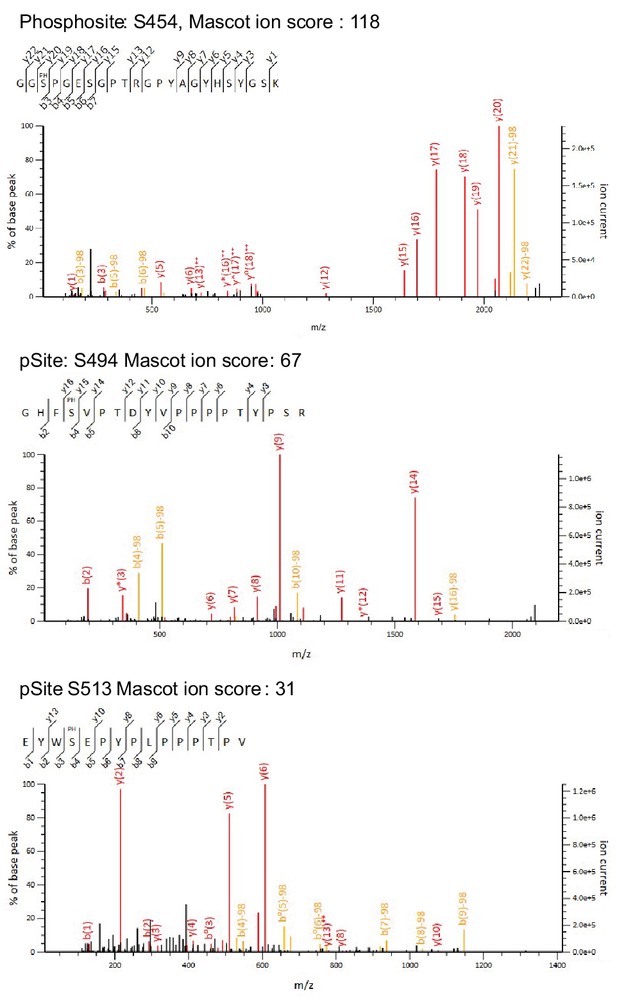
Candidate spectra of Regnase-1 phosphopeptides with confident site localization.
Only quantitatively altered phosphopeptides are shown. Fragment ions containing the N-(b-type ions) or C-(y-type ions) terminus are labeled with red (without neutral loss) or orange (with neutral-loss).
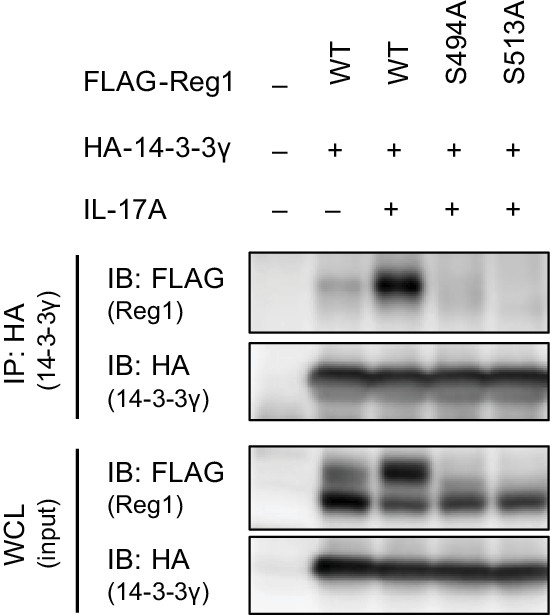
IL-17A stimulation induces Regnase-1-14-3-3 association.
Immunoblot analysis of immunoprecipitates (IP: HA) and WCL from HeLa cells transiently expressing HA-14-3-3γ and FLAG-Regnase-1-WT or indicated mutants stimulated with IL-17A (50 ng/ml) for 4 hr.
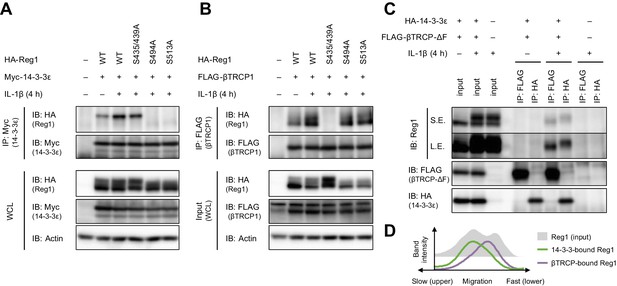
βTRCP binds to 14-3-3-free Regnase-1.
(A) Immunoblot analysis of immunoprecipitates (IP: Myc) and WCL from HeLa cells transiently expressing Myc-14-3-3ε and HA-Regnase-1-WT or indicated mutants stimulated with IL-1β (10 ng/ml) for 4 hr. (B) Immunoblot analysis of immunoprecipitates (IP: FLAG) and WCL from HeLa cells transiently expressing FLAG-βTRCP1 and HA-Regnase-1-WT or indicated mutants stimulated with IL-1β (10 ng/ml) for 4 hr. (C) Immunoblot analysis of immunoprecipitates (IP: FLAG or HA) and WCL from HeLa cells transiently expressing FLAG-βTRCP-ΔF and HA-14-3-3ε stimulated with IL-1β (10 ng/ml) for 4 hr. S.E.: short exposure, L.E.: long exposure. (D) The intensity of Regnase-1-bands in (C).
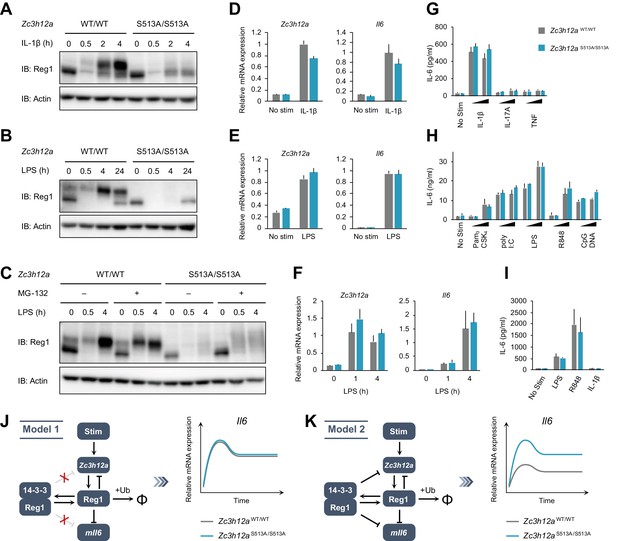
The S513A mutation destabilizes Regnase-1 protein but does not affect target mRNA abundance.
(A–C) Immunoblot analysis of Zc3h12aWT/WT and Zc3h12aS513A/S513A MEFs stimulated with IL-1β (10 ng/ml) (A), BMDMs stimulated with LPS (100 ng/ml) (B), and thioglycollate-elicited PECs stimulated with LPS (100 ng/ml) (C) for indicated time. PECs were pretreated with MG-132 (5 μM) 2 hr before the stimulation. (D)-(F) mRNA expression of Zc3h12a and Il6 in Zc3h12aWT/WT and Zc3h12aS513A/S513A MEFs stimulated with IL-1β (10 ng/ml) for 4 hr (D), BMDMs stimulated with LPS (100 ng/ml) for 4 hr (E), and thioglycollate-elicited PECs stimulated with LPS (100 ng/ml) for indicated time (F). (G)-(I) IL-6 secretion in Zc3h12aWT/WT and Zc3h12aS513A/S513A MEFs stimulated with IL-1β (10 ng/ml), IL-17A (50 ng/ml), or TNF (10 ng/ml) for 24 hr (G), BMDMs stimulated with Pam3CSK4 (1 or 10 ng/ml), poly I:C (10 or 100 μg/ml), LPS (10 or 100 ng/ml), R848 (10 or 100 nM), or CpG DNA (0.1 or 1 μM) for 24 hr (H), and thioglycollate-elicited PECs stimulated with LPS (100 ng/ml), R848 (100 nM), or IL-1β (10 ng/ml) for 24 hr (I). (J) Schematic representation of Model 1 in which 14-3-3-bound Regnase-1 does not have the function of degrading its target mRNAs. This model could explain the experimental observations. (K) Schematic representation of Model 2 in which 14-3-3-bound Regnase-1 maintains some ability to degrade its target mRNAs. This model is not consistent with the experimental observations. In (D)-(I), bars represent mean values of biological replicates (n = 3), and error bars represent standard deviation. Data is representative of two independent experiments, each with three biological replicates.
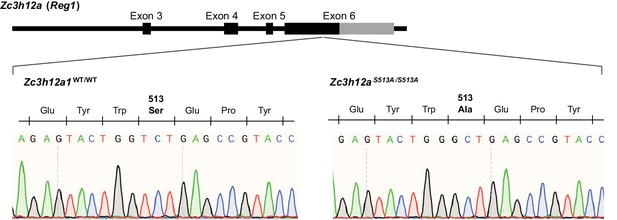
Schematic illustration of Zc3h12a gene in mice.
The result of Sanger sequencing around S513 of Regnase-1 are shown below.
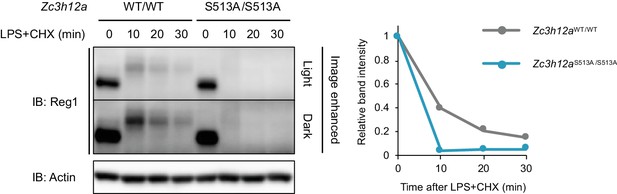
The protein stability of Regnase-1-WT and S513A.
Immunoblot analysis of Zc3h12aWT/WT and Zc3h12aS513A/S513A thioglycollate-elicited PECs treated with LPS (100 ng/ml) and CHX (100 μg/ml) for indicated time. CHX and LPS were added at the same time.
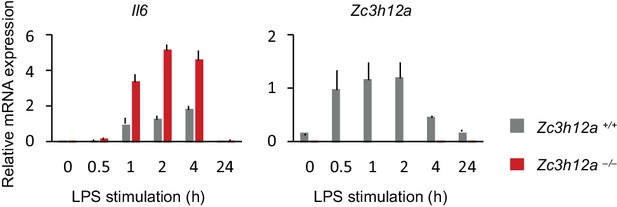
Il6 expression in Zc3h12a–/– PECs mRNA expression of Il6 and Zc3h12a in Zc3h12aWT/WT and Zc3h12a–/– thioglycollate-elicited PECs stimulated with LPS (100 ng/ml) for indicated time.
Bars represent mean values of biological replicates (n = 3), and error bars represent standard deviation.
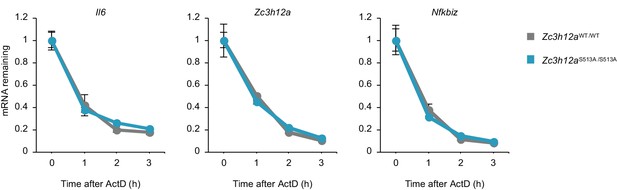
The stability of Regnase-1 target mRNAs.
The remaining mRNA levels of indicated genes in Zc3h12aWT/WT and Zc3h12aS513A/S513A MEFs. Actinomycin D (ActD, 10 μg/ml) was added after 4 hr stimulation with IL-1β (10 ng/ml). Dots represent mean values of biological replicates (n = 3), and error bars represent standard deviation.
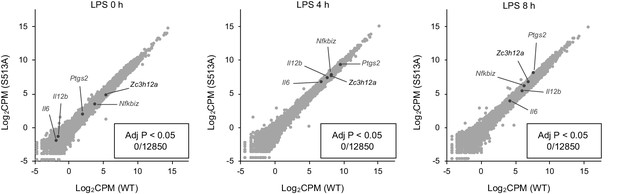
S513A mutation of Regnase-1 does not affect gene expression.
Transcriptome analysis of Zc3h12aWT/WT and Zc3h12aS513A/S513A thioglycollate-elicited PECs stimulated with LPS (100 ng/ml) for indicated time. Several known Regnase-1 target transcripts are annotated. None of transcripts shows significant (adjusted p value < 0.05) difference between Zc3h12aWT/WT and Zc3h12aS513A/S513A.
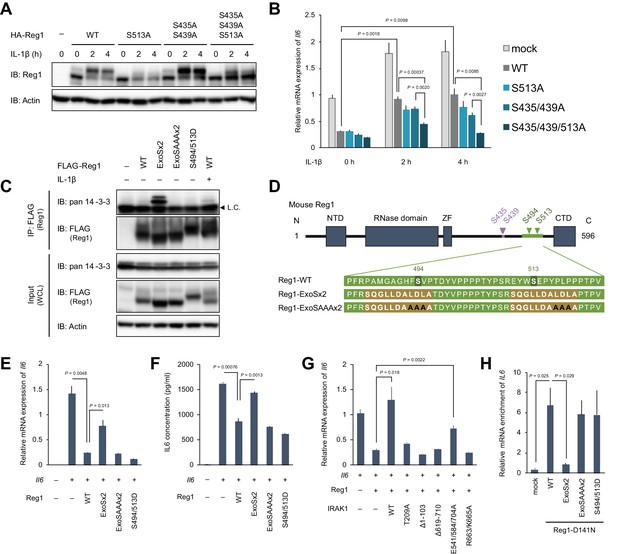
14-3-3 bound to phospho-S494 and S513 inactivates Regnase-1 by inhibiting Regnase-1-mRNA binding.
(A) Immunoblot analysis of ZC3H12A-KO HeLa cells transiently expressing Regnase-1-WT or indicated mutants. Cells were stimulated with IL-1β (10 ng/ml) for indicated time. (B) mRNA expression of Il6 in HeLa cells transiently expressing Regnase-1-WT or indicated mutants together with Il6. Cells were stimulated with IL-1β (10 ng/ml) for indicated time. (C) Immunoblot analysis of immunoprecipitates (IP: FLAG) and WCL from HeLa cells transiently expressing FLAG-Regnase-1-WT or indicated mutants. For the IL-1β stimulation, cells were stimulated with IL-1β (10 ng/ml) for 4 hr. L.C.: light chain. (D) Schematic illustration of Regnase-1 and the amino acid sequences of Regnase-1-WT, -ExoSx2, and ExoSAAAx2. NTD: N-terminal domain, ZF: Zinc finger domain, CTD: C-terminal domain. (E) mRNA expression of Il6 in HeLa cells transiently expressing Regnase-1-WT or indicated mutants together with Il6. (F) Secreted IL6 concentration in (E). (G) mRNA expression of Il6 in HeLa cells transiently expressing Regnase-1-WT and IRAK1-WT or indicated mutants together with Il6. (H) The amount of IL6 mRNAs immunoprecipitated with FLAG-Regnase-1-D141N or other indicated mutants in HeLa cells. In (B), (E)-(H), bars represent mean values of biological replicates (n = 3), and error bars represent standard deviation. p-Values were calculated using unpaired, two-sided t-test. Data is representative of two independent experiments, each with three biological replicates.
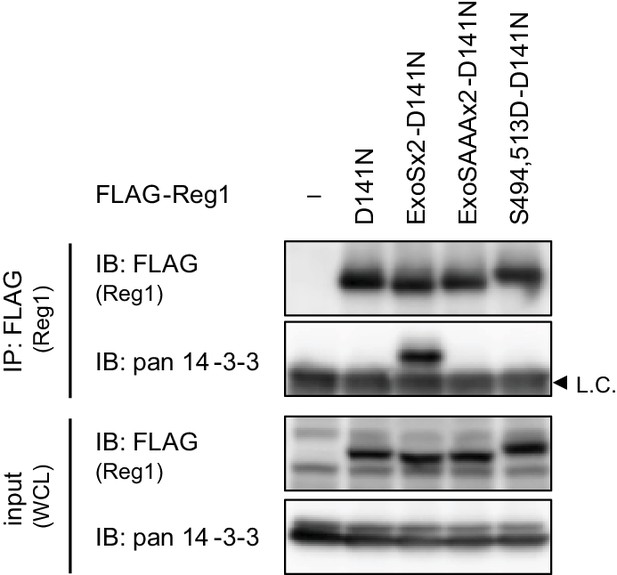
Regnase-1-ExoSx2-D141N binds to 14-3-3.
Immunoblot analysis of immunoprecipitates (IP: FLAG) and WCL from HeLa cells transiently expressing FLAG-Regnase-1-D141N or indicated mutants. L.C.: light chain.
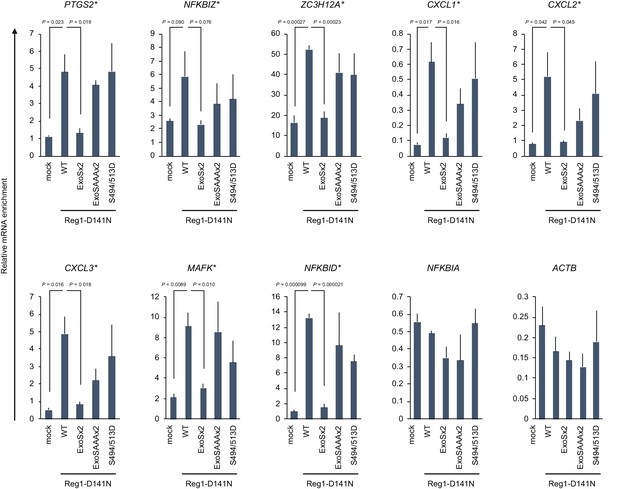
Regnase-1-ExoSx2-D141N failed to bind to target mRNAs.
The amount of mRNAs immunoprecipitated with FLAG-Regnase-1-D141N or other indicated mutants in HeLa cells. The binding targets of Regnase-1 are denoted with *. Bars represent mean values of biological replicates (n=3), and error bars represent standard deviation. p-Values were calculated using unpaired, two-sided t-test.
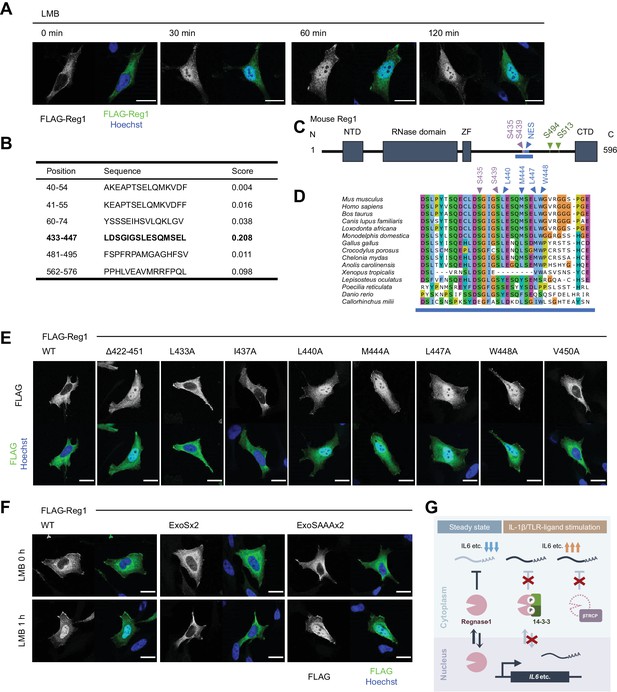
14-3-3 inhibit nuclear-cytoplasmic shuttling of Regnase-1.
(A) Immunofluorescence analysis of HeLa cells transiently expressing FLAG-Regnase-1-WT treated with Leptomycin B (LMB) (10 ng/ml) for indicated time. (B) The result of NES prediction of Regnase-1 by LocNES. Higher score indicates higher probability. (C) Schematic illustration of Regnase-1. The amino acid sequence shown in (D) is highlighted in blue. NTD: N-terminal domain, ZF: Zinc finger domain, CTD: C-terminal domain. (D) The amino acid sequences including S435/S439 and NES of Regnase-1 from mouse and other indicated vertebrates. (E) Immunofluorescence analysis of HeLa cells transiently expressing FLAG-Regnase-1-WT or indicated mutants. (F) Immunofluorescence analysis of HeLa cells transiently expressing FLAG-Regnase-1-WT or indicated mutants treated with LMB (10 ng/ml) for 1 hr. (G) Model of 14-3-3- and βTRCP-mediated regulation of Regnase-1. In the steady state, Regnase-1 shuttles between the nucleus and the cytoplasm and degrades target mRNAs such as Il6. Under IL-1β or TLR-ligands stimulation, two different regulatory mechanisms suppress the activity of Regnase-1 not to disturb proper expression of inflammatory genes; βTRCP induces protein degradation of Regnase-1 and 14-3-3 inhibits nuclear-cytoplasmic shuttling and mRNA recognition of Regnase-1. In (A), (E), and (F), white scale bars indicate 20 μm.
Tables
| Reagent type (species) or resource | Designation | Source or reference | Identifiers | Additional information |
|---|---|---|---|---|
| Gene (Mus musculus) | Zc3h12a | NA | Gene ID: 230738 | |
| Gene (Homo sapiens) | ZC3H12A | NA | Gene ID: 80149 | |
| Strain, strain background (Mus musculus) | Zc3h12aWT/WT | CLEA Japan | C57BL/6 | C57BL/6JJcl |
| Strain, strain background (Mus musculus) | Zc3h12a-/- | https://doi.org/10.1038/nature07924 | ||
| Strain, strain background (Mus musculus) | Zc3h12aS513A/S513A | this paper | generated using CRISPR-Cas9 system | |
| Sequence-based reagent | DNA oligo (for pX330) | this paper | CACCGCGGCTCAGACCAGTACTCTC | for Zc3h12aS513A/S513A generation |
| Sequence-based reagent | DNA oligo (for pX330) | this paper | AAACGAGAGTACTGGTCTGAGCCGC | for Zc3h12aS513A/S513A generation |
| Sequence-based reagent | Donor single strand oligo | this paper | GAAGGACAGGAGTGGGTGGGGGTAATGGGTACGGCTCAGCCCAGTACTCTCTGGATGGGTAGGTGGGTGGCGGGGGCACA | for Zc3h12aS513A/S513A generation |
| Sequence-based reagent | DNA oligo (for pX459-IRAK1KO) | https://doi.org/10.1093/bioinformatics/btu743 | CACCGGTCTGGTCGCGCACGATCA | |
| Sequence-based reagent | DNA oligo (for pX459-IRAK1KO) | https://doi.org/10.1093/bioinformatics/btu743 | AAACTGATCGTGCGCGACCAGACC | |
| Sequence-based reagent | DNA oligo (for pX459-IRAK2KO) | https://doi.org/10.1038/nbt.3437 | CACCGAAAACCGCAAAATCAGCCAG | |
| Sequence-based reagent | DNA oligo (for pX459-IRAK2KO) | https://doi.org/10.1038/nbt.3437 | AAACCTGGCTGATTTTGCGGTTTTC | |
| Antibody | Anti-mouse-Regnase-1 (rabbit polyclonal) | MBL life science | custom antibody production (1:1000) | |
| Antibody | Anti-human-Regnase-1 (rabbit polyclonal) | Atlas Antibodies | Cat # HPA032053 | (1:500) |
| Antibody | Anti-14-3-3 (pan) (mouse monoclonal) | Santa Cruz Biotechnology | Cat # sc-1657 | (1:1000) |
| Antibody | Anti-IκB-α (rabbit polyclonal) | Santa Cruz Biotechnology | Cat # sc-371 | (1:1000) |
| Antibody | Anti-IRAK1 (mouse monoclonal) | Santa Cruz Biotechnology | Cat # sc-5288 | (1:500) |
| Antibody | Anti-FLAG (mouse monoclonal) | Sigma | Cat # F3165 | (WB:1:2000, IF:1:5000) |
| Antibody | Anti-FLAG (rabbit polyclonal) | Sigma | Cat # F7425 | (1:2000) |
| Antibody | Anti-HA (mouse monoclonal) | Sigma | Cat # H3663 | (1:2000) |
| Antibody | Anti-HA (rabbit polyclonal) | Sigma | Cat # H6908 | (1:2000) |
| Antibody | Anit-Myc (mouse monoclonal) | Sigma | Cat # M4439 | (1:2000) |
| Antibody | Anti-Myc (rabbit polyclonal) | Sigma | Cat # C3956 | (1:2000) |
| Antibody | Anti-β-Actin-HRP (mouse monoclonal) | Santa Cruz Biotechnology | Cat # sc-47778-HRP | (1:2000) |
| Antibody | Anti-Mouse IgG-HRP F(ab')2 (sheep polyclonal) | cytiva | Cat # NA9310-1ML | (1:5000) |
| Antibody | Anti-Rabbit IgG-HRP F(ab')2 (donkey polyclonal) | cytiva | Cat # NA9340-1ML | (1:5000) |
| Antibody | F(ab')2-anti-Mouse IgG (H+L)-AF488 (Goat polyclonal) | Invitrogen | Cat # A11017 | (1:2000) |
| Recombinant DNA reagent | pX330-U6-Chimeric_BB-CBh-hSpCas9 | Addgene | RRID:Addgene_42230 | |
| Recombinant DNA reagent | pSpCas9(BB)-2A-Puro (PX459) V2.0 | Addgene | RRID:Addgene_62988 | |
| Recombinant DNA reagent | pMD2.G | Addgene | RRID:Addgene_12259 | |
| Recombinant DNA reagent | pMDLg/pRRE | Addgene | RRID:Addgene_12251 | |
| Recombinant DNA reagent | pRSV-Rev | Addgene | RRID:Addgene_12253 | |
| Recombinant DNA reagent | pInducer20 | Addgene | RRID:Addgene_44012 | |
| Recombinant DNA reagent | pInducer20-puro | this paper | NeoR of pInducer20 (Addgene_44012) was replaced with PuroR | |
| Recombinant DNA reagent | pFLAG-CMV2 | Sigma | Cat # E7033 | |
| Recombinant DNA reagent | pEGFP-C1 | Clontech | ||
| Peptide, recombinant protein | FLAG Peptide | Sigma | Cat # F3290 | |
| Peptide, recombinant protein | HA peptide | MBL Life science | Cat # 3320 | HA tagged Protein PURIFICATION KIT |
| Peptide, recombinant protein | recombinant human IL-1β | R and D Systems | Cat # 201-LB-005 | |
| Peptide, recombinant protein | recombinant mouse IL-1β | BioLegend | Cat # 575102 | |
| Peptide, recombinant protein | recombinant human IL-17A | BioLegend | Cat # 570502 | |
| Peptide, recombinant protein | recombinant human TNF | BioLegend | Cat # 570104 | |
| Commercial assay or kit | Dynabeads Protein G | Invitrogen | Cat # 10004D | |
| Commercial assay or kit | Lambda Protein Phosphatase | NEB | Cat # P0753S | |
| Commercial assay or kit | Signal Enhancer HIKARI | nacalai tesque | Cat # 02270-81 | |
| Commercial assay or kit | Immobilon Forte Western HRP Substrate | Millipore | Cat # WBLUF0500 | |
| Commercial assay or kit | TRIzol Reagent | Invitrogen | Cat # 15596018 | |
| Commercial assay or kit | RNA Clean and Concentrator-5 | Zymo Research | Cat # R1014 | |
| Commercial assay or kit | PowerUp SYBR Green Master Mix | Applied Biosystems | Cat # A25742 | |
| Commercial assay or kit | IL-6 Mouse Uncoated ELISA Kit | Invitrogen | Cat # 88-7064-88 | |
| Chemical compound, drug | DSP (dithiobis(succinimidyl propionate)) | TCI | Cat # D2473 | |
| Chemical compound, drug | Pam3CSK4 | InvivoGen | Cat # tlrl-pms | |
| Chemical compound, drug | poly I:C | cytiva | Cat # 27473201 | |
| Chemical compound, drug | LPS | InvivoGen | Cat # tlrl-smlps | |
| Chemical compound, drug | R848 | InvivoGen | Cat # tlrl-r848-5 | |
| Chemical compound, drug | CpG DNA | InvivoGen | Cat # tlrl-1668-1 | ODN 1668 |
| Chemical compound, drug | MG-132 | Sigma | Cat # 474790 | |
| Chemical compound, drug | Actinomycin D | Sigma | Cat # A9415 | |
| Chemical compound, drug | Leptomycin B | Sigma | Cat # L2913 |
Additional files
-
Source data 1
Raw data of the results of immunoblotting are zipped in Source Data Files.
- https://cdn.elifesciences.org/articles/71966/elife-71966-data1-v3.zip
-
Supplementary file 1
Primer sequences used in qPCR.
- https://cdn.elifesciences.org/articles/71966/elife-71966-supp1-v3.xlsx
-
Transparent reporting form
- https://cdn.elifesciences.org/articles/71966/elife-71966-transrepform-v3.docx





