Phosphorylation of tyrosine 90 in SH3 domain is a new regulatory switch controlling Src kinase
Figures
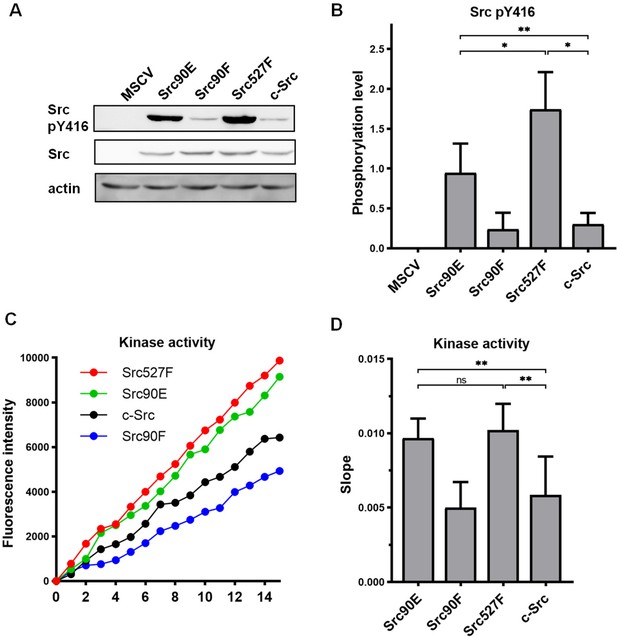
Substitutions mimicking the Y90 phosphorylation state affect Src kinase activity.
(A, B) Lysates from SYF fibroblasts stably expressing the Src variants were analysed by immunodetection on western blots using antibodies against Src pY416, total Src, and actin as a loading control. After densitometric analysis of the blots, ratio between Y416-phosphorylated Src and total Src was calculated. MSCV represents SYF cells bearing empty pMSCV-EGFP vector. (C, D) Kinase assay (Omnia Y Peptide 2 Kit) was performed from lysates of SYF cells expressing the Src variants. Kinase activity was measured as an increase of fluorescence depicted in C (showing representative results from one experiment). The data are shown as means with standard deviation out of minimum four independent experiments. Statistical significance was calculated by one-way ANOVA.
-
Figure 1—source data 1
Unedited immunoblots and source datasets for graphs in Figure 1.
- https://cdn.elifesciences.org/articles/82428/elife-82428-fig1-data1-v2.zip
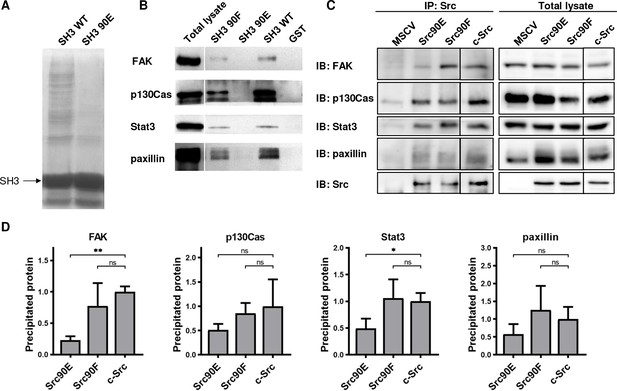
Phosphomimicking mutation 90E reduces the SH3 domain ligand binding capacity.
The variants of Src SH3 domains (90F, 90E, wild type [WT]) were expressed as recombinant GST-SH3 fusion constructs in bacteria, isolated and used for pull-down from HeLa lysates. (A) Precipitated samples were run on SDS-PAGE electrophoresis and stained with Coomassie Brilliant Blue. Loading of GST-SH3 constructs was equal and is visible as a large band with molecular weight of around 33 kDa. (B) Western blotting of the precipitates was followed by immunodetection by antibodies against FAK, p130Cas, Stat3, or paxillin. (C, D) Immunoprecipitation of the full-length Src variants from SYF lineages followed by immunodetection of selected SH3-binding proteins in precipitates and total cell lysates. MSCV indicates SYF cell expressing empty pMSCV-EGFP construct, which serve as a control. Quantification was performed from at least three independent experiments and represents a normalised ratio between immunoprecipitated protein and total protein. Statistical significance was calculated by one-way ANOVA.
-
Figure 2—source data 1
Unedited immunoblots and source datasets for graph in Figure 2.
- https://cdn.elifesciences.org/articles/82428/elife-82428-fig2-data1-v2.zip
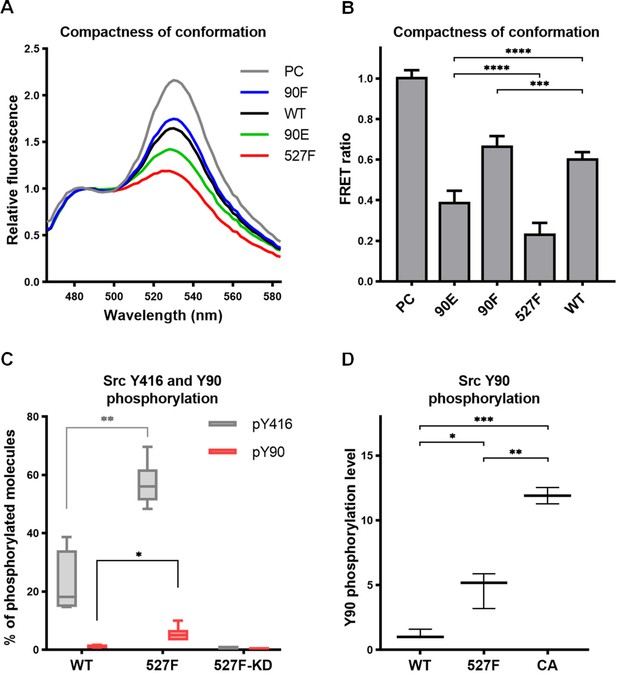
Phosphorylation of Y90 represents an autoactivatory mechanism that opens the Src structure.
(A, B) Analysis of effects mediated by the Y90 substitutions on the Src kinase structure using SrcFRET sensor. SrcFRET sensor variants with Y90 and/or Y527 mutations were expressed in U2OS cells. Fluorescence emission spectra were recorded in native cell lysates. (A) Representative emission spectra normalised to emission maximum of CFP. PC indicates positive control for FRET (construct where CFP and mCit are connected by a short linker). (B) Bar graph shows ratio of normalised mCit (525 nm) and CFP (486 nm) emission. The data are shown as means with standard deviation out of minimum four independent experiments. Statistical significance was calculated by one-way ANOVA. (C, D) Quantitative mass spectrometry (MS) analysis of Src phosphorylation. SrcFRET constructs were expressed in U2OS cells, immunoprecipitated and analysed on MS. (C) Absolute values of Y416 and Y90 phosphorylation were determined using internal peptide standards and depicted on the plot as percentage of phosphorylated tyrosines from the total pool of given tyrosine. (D) Normalised ratio between phopho-peptide and non-modified base peptide was plotted. The data are shown as means with standard deviation from three independent experiments. Statistical significance was calculated by one-way ANOVA.
-
Figure 3—source data 1
Source datasets for graphs in Figure 3.
- https://cdn.elifesciences.org/articles/82428/elife-82428-fig3-data1-v2.zip
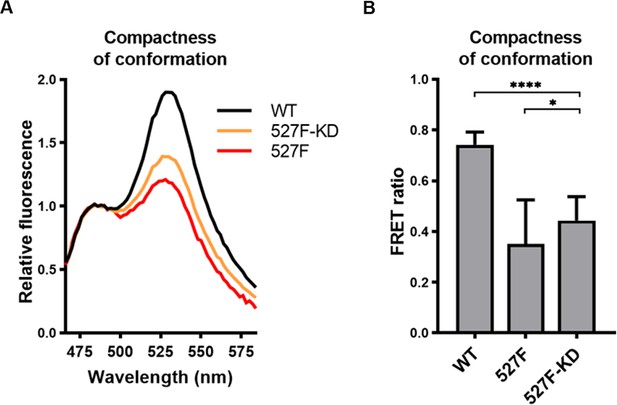
Activated Y527F Src carrying a kinase-dead mutation retains an open conformation.
Indicated SrcFRET sensor variants were expressed in U2OS cells and fluorescence emission spectra were recorded in native cell lysates. (A) Representative emission spectra normalised to emission maximum of CFP. (B) Bar graph shows ratio of mCit (525 nm) and CFP (486 nm) emission. The data are shown as means with standard deviation out of five independent experiments. Statistical significance was calculated by one-way ANOVA.
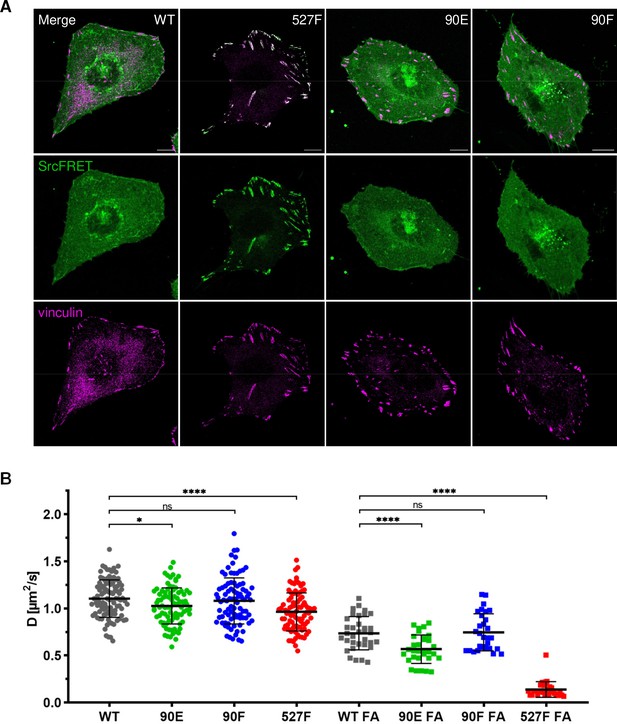
Y90 phosphorylation state does not affect Src localisation but alters its mobility within cytoplasmic membrane.
(A) U2OS cells transiently expressing the FRET constructs were fixed, stained for vinculin (focal adhesions marker, magenta) and imaged by confocal microscopy. SrcFRET fluorescence (mCit) is depicted in green. Scale bar: 10 µm. (B) Diffusion coefficients of membrane associated SrcFRET variants outside and inside focal adhesions. U2OS cells were cotransfected with SrcFRET constructs and mCherry-vinculin. Mobility of the SrcFRET variants within the membrane was measured by imaging fluorescence correlation spectroscopy (ImFCS) combined with TIRF microscopy. Membrane regions within focal adhesions were identified by the presence of mCherry-vinculin. Data from three independent experiments are shown as a scatter plot with the mean represented by the middle horizontal line. Statistical significance was calculated by one-way ANOVA.
-
Figure 4—source data 1
Source dataset for graph in Figure 4B.
- https://cdn.elifesciences.org/articles/82428/elife-82428-fig4-data1-v2.zip
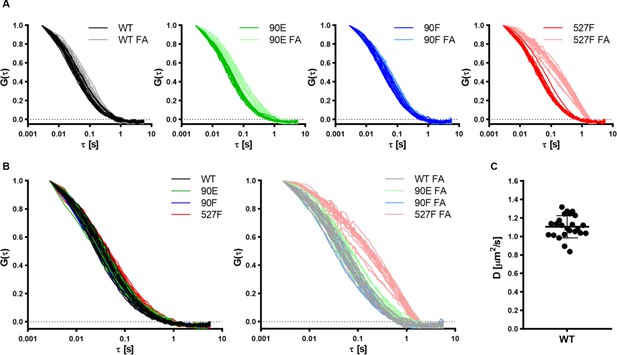
Y90 phosphorylation state does not affect Src localisation but alters its mobility within cytoplasmic membrane.
U2OS cells were cotransfected with the SrcFRET constructs and mCherry-vinculin. Mobility of the SrcFRET variants within the membrane was measured by imaging fluorescence correlation spectroscopy (ImFCS) using TIRF microscopy. Membrane regions within focal adhesions (FAs) were identified by the presence of mCherry-vinculin. (A, B) Autocorrelation curves comparing mobility of the Src variants outside of FAs and within FA sites. Shift of ‘FA’ correlation curves to the right indicates longer diffusion times. (C) Diffusion coefficient of SrcFRET wild type (WT) measured by line-scan fluorescent correlation spectroscopy used for calibration of ImFCS data.
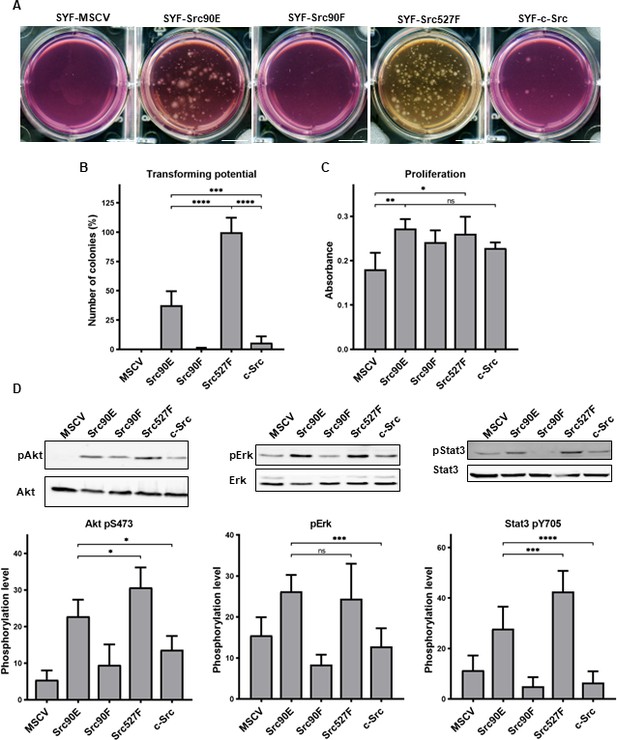
Src90E has transforming potential.
(A, B) Soft agar assay was performed with SYF fibroblasts stably expressing the Src variants. Cells were cultivated in 0.4% agar. After 14 days the number of colonies was determined. Scale bars: 10 mm. The graph shows relative amounts of colonies compared to SYF-Src527F. (C) Proliferation rate determined by alamarBlue assay. Equal number of cells was seeded into 98-well plate in tetraplicates for every lineage. After 3 days, change of absorbance at 570 nm (referenced to 600 nm) of added alamarBlue solution was measured. (D) Activity within PI3K/Akt, Ras/MAPK, and Stat3 pathways evaluated as the phosphorylation rate of Akt, Erk, and Stat3. Lysates from SYF fibroblasts stably expressing the Src variants were standardised to overall protein. Immunodetection on western blots was performed with antibodies against Akt, Akt pS473, Erk 1/2, pErk 1/2, Stat3, and Stat3 pY705. Rates between phosphorylated form and total protein were calculated from at least three independent experiments and plotted as means with standard deviation. Statistical significance was determined by one-way ANOVA.
-
Figure 5—source data 1
Unedited immunoblots and source datasets for graphs in Figure 5.
- https://cdn.elifesciences.org/articles/82428/elife-82428-fig5-data1-v2.zip
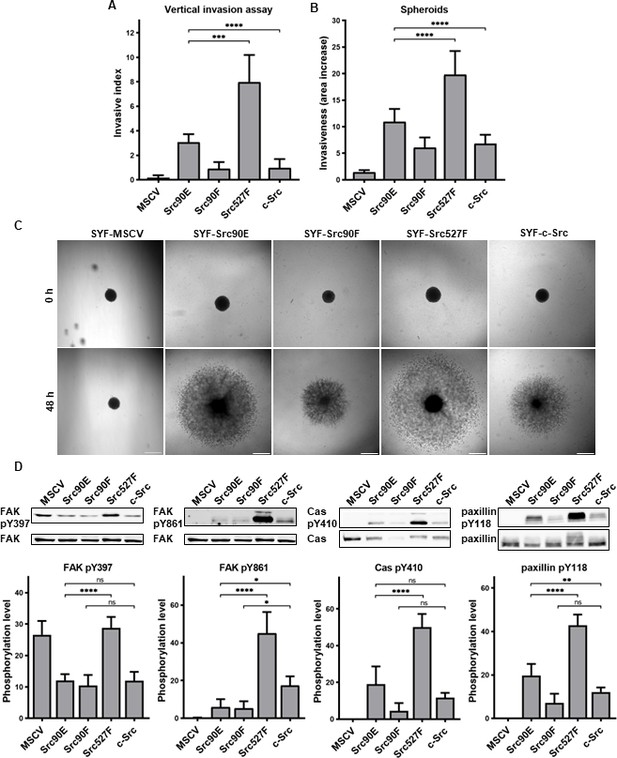
Phosphomimicking mutation 90E increases cell invasiveness.
(A) Vertical invasion assay performed with SYF fibroblasts stably expressing the Src variants. Cells were seeded on top of 1.5% collagen. After 3 days, z-stack pictures were taken, and number of cells was counted in each level. Cellular invasiveness was determined as invasive index representing weighted arithmetic mean of invasion depth. (B, C) Spheroid invasion assay. SYFs grown as spheroids were embedded in 1.5% collagen. Images were taken immediately after seeding and 48 hr later. Scale bars: 500 μm. Ratio between spheroid area at 48 hr and 0 hr was determined. (D) Phosphorylation levels of FAK, p130Cas, and paxillin were detected in lysates from SYF cells stably expressing the Src variants. Ratio between phosphorylated form and total protein was determined based on immunodetection of total FAK, p130Cas, paxillin, and their phosphorylated forms (FAK pY397, FAK pY861, p130Cas pY410, paxillin pY118). All the data in bar graphs are shown as means with standard deviation out of minimum three independent experiments. Statistical significance was calculated by one-way ANOVA.
-
Figure 6—source data 1
Unedited immunoblots and source datasets for graphs in Figure 6.
- https://cdn.elifesciences.org/articles/82428/elife-82428-fig6-data1-v2.zip
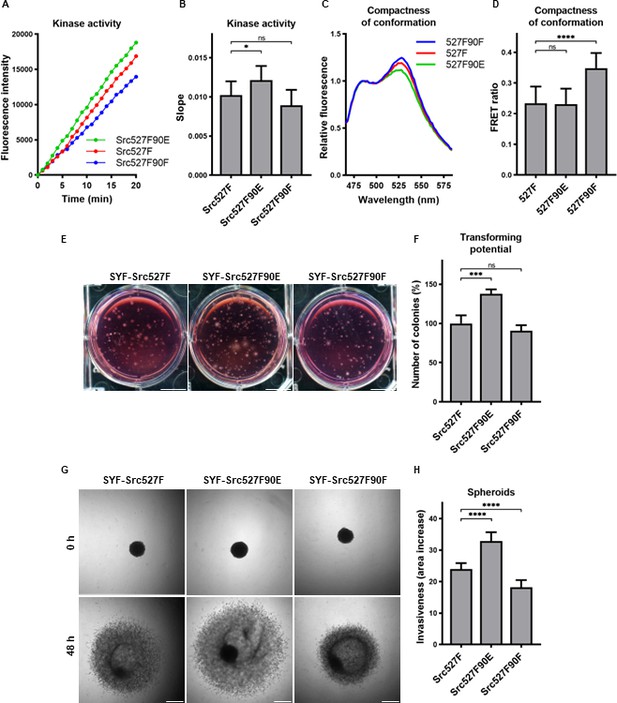
Effect of 527F-activated Src can be modulated by Y90 phosphorylation.
(A, B) Catalytic activity of Src527F and the double-mutated variants was analysed using kinase assay (Omnia Y Peptide 2 Kit). Reactions were performed from lysates of SYF cells expressing indicated Src variants. Kinase activity was measured as an increase of fluorescence depicted in Figure 6B (showing representative results from one experiment). (C, D) Effect of Y90 substitutions on compactness of the Src527F structure. SrcFRET527F sensor variants with Y90 mutations were expressed in U2OS cells. Fluorescence emission spectra were recorded in native cell lysates. (C) Representative emission spectra normalised to emission maximum of CFP. PC indicates positive control for FRET. (D) Bar graph shows ratio of normalised mCit (525 nm) and CFP (486 nm) emission. (E, F) Transforming potential was assessed by soft agar assay with SYF fibroblasts stably expressing indicated Src variants. Cells were cultivated in 0.4% agar. After 14 days, the number of colonies was counted. Scale bars: 10 mm. The graph shows relative number of colonies compared to SYF-Src527F. (G, H) Invasiveness of the SYF cell lines was evaluated with spheroid invasion assay. SYFs grown as spheroids were embedded in 1.5% collagen. Images were taken immediately after seeding and 48 hr later. Scale bars: 500 μm. Ratio between spheroid area at 48 hr and 0 hr was determined. The data in bar graphs are shown as means with standard deviation out of minimum three independent experiments. Statistical significance was calculated by one-way ANOVA.
-
Figure 7—source data 1
Source datasets for graphs in Figure 7.
- https://cdn.elifesciences.org/articles/82428/elife-82428-fig7-data1-v2.zip
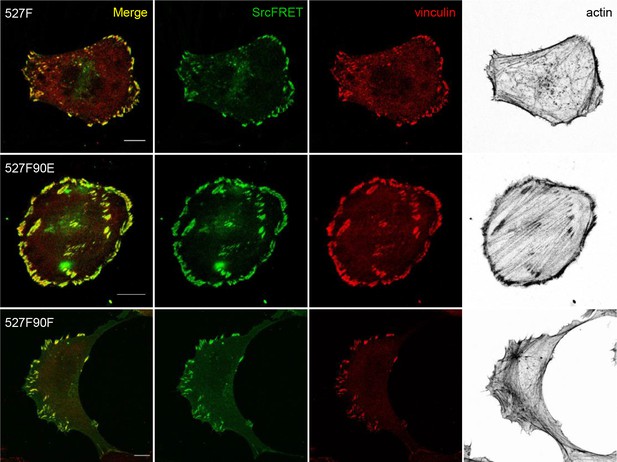
Localisation of double-mutated SrcFRET constructs.
U2OS cells transiently expressing the SrcFRET constructs were fixed, stained for vinculin (focal adhesions marker, magenta) and F-actin (grey), and imaged by confocal microscopy. SrcFRET fluorescence (mCit) is depicted in green. Scale bar: 10 µm.





