The RAM signaling pathway links morphology, thermotolerance, and CO2 tolerance in the global fungal pathogen Cryptococcus neoformans
Figures
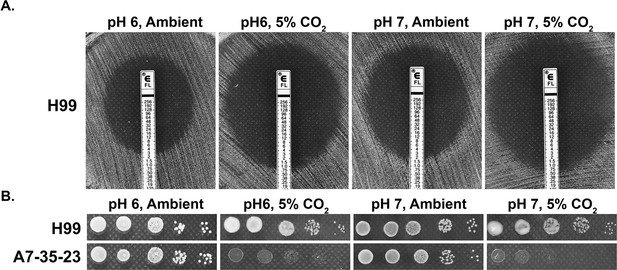
CO2 sensitivity is not simply due to lowered medium pH.
(A) H99 cells were plated onto RPMI solid medium buffered to either pH 6 or pH 7. Fluconazole containing E-test strips were placed onto the lawn of H99 cells, and the plates were incubated at 37°C in ambient air or in 5% CO2. The larger the halo surrounding the E-strip, the more sensitive the cells are to fluconazole. The intercept value of the halo with the E-strip is the minimal inhibitory concentration. (B) Cells of the previously identified CO2-tolerant H99 and CO2-sensitive A7-35-23 were serial diluted, spotted onto RPMI media buffered to pH 6 or pH 7, and incubated at 37°C in ambient air or in 5% CO2.
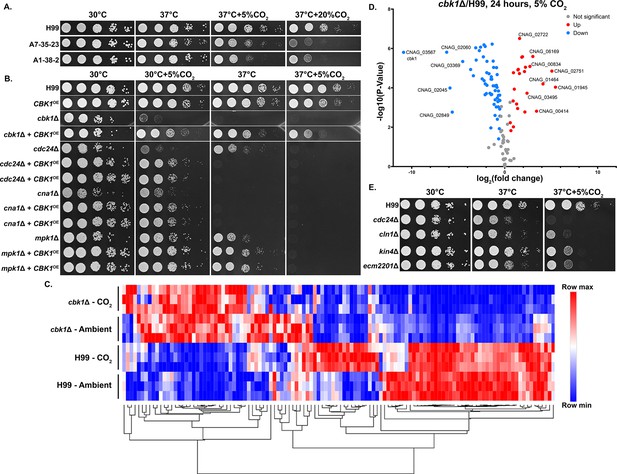
The Regulator of Ace2 and Morphogenesis (RAM) pathway effector kinase Cbk1 is critical for CO2 tolerance.
(A). The clinical reference strain H99 and environmental strains A7-35-23 and A1-38-2 were grown overnight in yeast peptone dextrose (YPD), serially diluted, and spotted onto solid YPD media plates. Photographs were taken 2 days after incubation in the indicated condition. (B) This serial dilution spotting assay was similarly performed for H99 and the mutants indicated. Two independent overexpression transformants for each mutant background were included as biological replicates. (C) Heatmap showing normalized total RNA counts of NanoString targets in H99 and cbk1Δ cultured at either ambient or 5% CO2, red indicates higher and blue indicates lower transcript abundance. (D) Volcano plot showing significantly differentially expressed transcripts (p-value of <0.05) in the cbk1Δ compared to H99 in the 5% CO2 condition. (E) Serial dilution spotting assay of H99 and four of the mutants found in the deletion set screening to be CO2 sensitive which also correspond to significantly downregulated genes shown in the volcano plot.

The cAMP(cyclic AMP) pathway is not essential for CO2 tolerance.
The reference strain H99 and the indicated gene deletion mutants were grown overnight in yeast peptone dextrose (YPD), serially diluted, and spotted onto solid YPD media plates. Photographs were taken 2 days after incubation in the indicated condition.

Confirming overexpression of CBK1.
The relative transcript levels of the CBK1 gene in the cbk1Δ, CBK1OE strain compared to in the wild-type (WT) strain was assayed by RT-PCR. The transcript level of CBK1 in the WT strain background was set to 1 for normalization. The housekeeping gene TEF1 was used as an endogenous control. Three biological and three technical replicates were used for each sample. Student’s t-test was used to determine significance level. ‘****’ refers to a p</i>-value<0.0001.
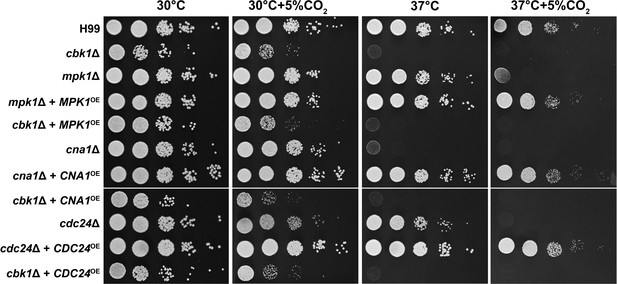
Overexpression of CDC24, MPK1, or CNA1 does not restore growth at host CO2 or temperature levels.
The strains above were grown overnight in yeast peptone dextrose (YPD), serially diluted, and spotted onto solid YPD media plates. Photographs were taken 2 days after incubation in the indicated condition.
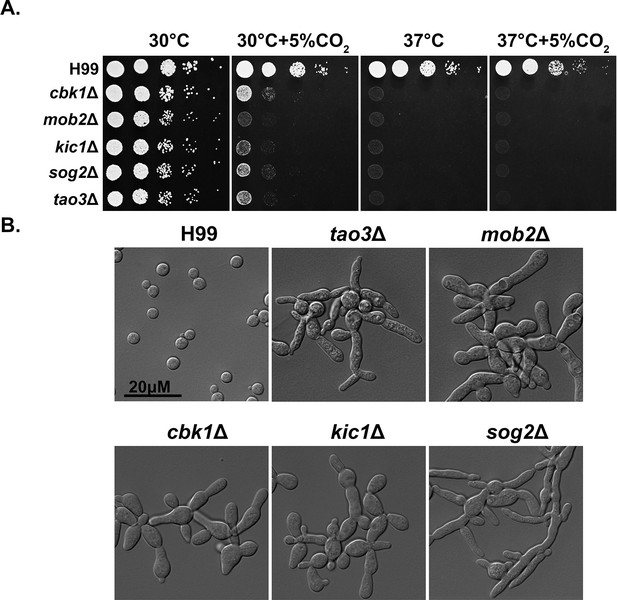
The Regulator of Ace2 and Morphogenesis (RAM) pathway is critical for normal morphology, thermotolerance, and CO2 tolerance.
(A) Cryptococcus neoformans WT H99 and RAM pathway mutants were serially diluted, spotted onto yeast peptone dextrose (YPD) medium, and incubated for 2 days at the indicated condition. (B) The cellular morphology of C. neoformans WT H99 and RAM pathway mutants cultured in YPD medium.
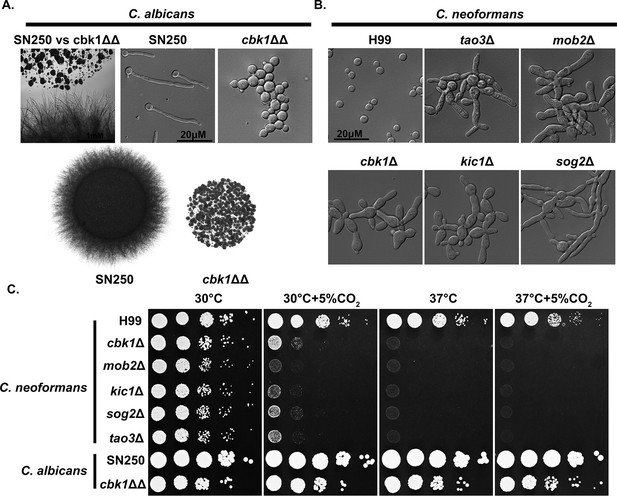
Conserved and divergent roles of the Regulator of Ace2 and Morphogenesis (RAM) pathway in ascomycete Candida albicans and basidiomycete Cryptococcus neoformans.
(A) The colony and cellular morphology of C. albicans WT strain SN250 and the homozygous cbk1ΔΔ mutant grown on RPMI. (B) The cellular morphology of C. neoformans WT H99 and RAM pathway mutants cultured in yeast peptone dextrose (YPD) medium. (C) C. neoformans and C. albicans WT and RAM pathway mutants were serially diluted, spotted onto YPD medium, and incubated for 2 days at the indicated condition. The C. neoformans data in this figure were taken from Figure 3.
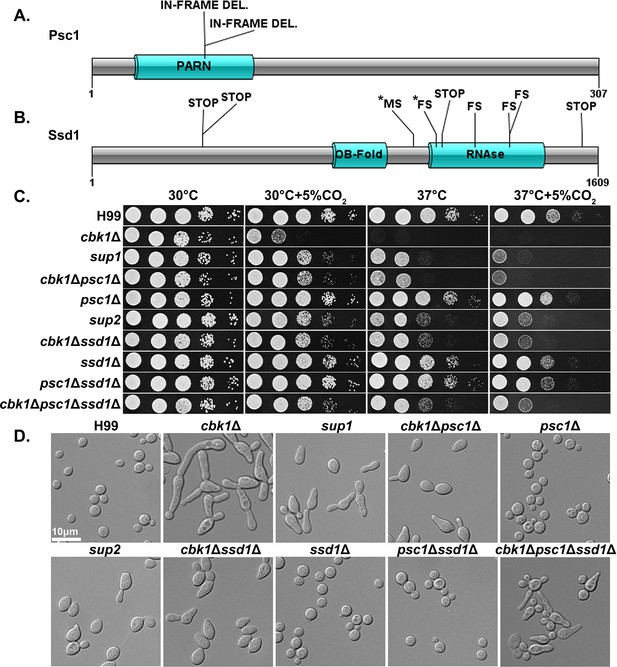
Natural suppressors of the Regulator of Ace2 and Morphogenesis (RAM) pathway cbk1Δ mutant restore multiple defects.
(A) Protein diagram of Psc1 showing the effects and positions of suppressor mutations in the two sup1 type natural suppressors. (B) Protein diagram of Ssd1 and the effects and positions of suppressor mutations in Ssd1 in the nine sup2 type natural suppressors. STOP indicates a non-sense mutation, MS a missense mutation, and FS a frameshift mutation. Asterisks (*) indicate mutations in the same suppressor strain. (C) Serial dilutions of H99 and the mutant strains were spotted onto yeast peptone dextrose (YPD) agar media and incubated for 2 days in the indicated condition to observe growth. (D) The cellular morphology of H99 and the mutant strains in liquid YPD cultures were examined under microscope.
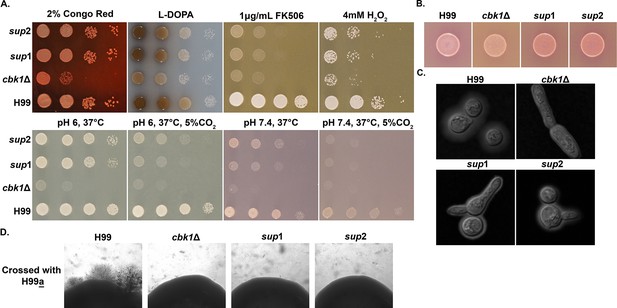
Phenotypic characterization of cbk1Δ suppressor mutants.
(A and B) The strains above were grown overnight in yeast peptone dextrose (YPD), serially diluted, and spotted onto the indicated solid media. YPD +2% congo red was used to assay cell wall stress tolerance. YPD +1 μg/mL FK506 was used to assay growth under calcineurin inhibition. YPD +4 mM H2O2 was used to assay oxidative stress tolerance. L-DOPA media was used to assay melaninization. RPMI media buffered with MOPS(3-(N-Morpholino)propanesulfonate) was used to test tolerance to 37°C or 37°C+5% CO2 at pH 6 and pH 7.4. Photographs were taken 2 days of incubation. Christensen Urea Agar was used to assay urease activity, indicated by change in media coloration from yellow to pink. (C) The cells incubated for 2 days on RPMI pH 7.4, 37°C+5% CO2, were stained with India Ink and observed under the microscope to check capsule size. (D) Cells were grown overnight in liquid culture and then diluted to the concentration of OD600=0.01. Equal volumes of the indicated MATa and MATα cells were mixed, and 3 μL was spotted onto V8 pH 5.0 solid medium. After incubation at 22°C for 2 weeks in the dark, mating colonies were examined with a SZX16 stereoscope (Olympus).
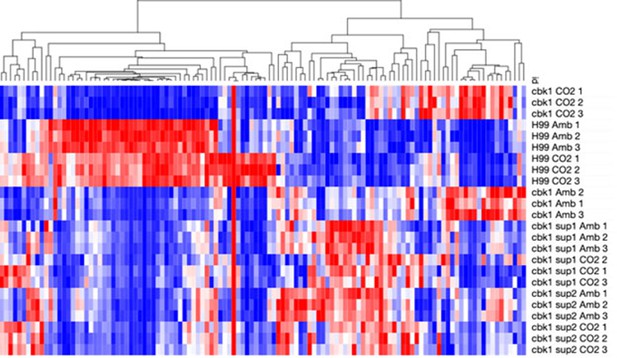
Suppressor mutants do not restore transcript levels of NanoString targets in cbk1Δ.
Heatmap showing normalized total RNA counts of NanoString targets in H99, cbk1Δ, sup1, and sup2 cultured at either ambient or 5% CO2. Red indicates higher and blue indicates lower transcript abundance.
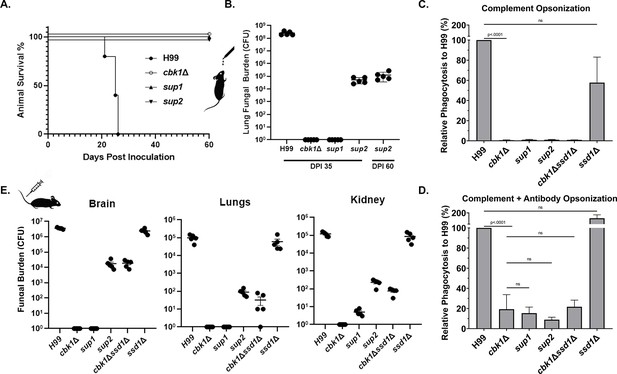
Suppressor mutants are partially restored for phagocytosis and can disseminate in the intravenous infection model of cryptococcosis.
(A) Mice were infected with 1×104 fungal cells intranasally, and their survival was monitored for 60 days post-infection. (B) At day 35 post-infection (DPI 35) and at the time of termination (DPI 60), 5 out of 10 mice per group for the cbk1Δ mutant, sup1 and sup2 groups were harvested for brains, kidneys, and lungs. For H99 infected mice, they were euthanized at their clinical end point (all before DPI 26). Tissue homogenate was serially diluted and plated onto YNB(yeast nitrogen base) medium to count the colony-forming units (CFUs) to measure the fungal burden per organ. (C) Murine macrophage J774A.1 cells were co-incubated with 2×106 cryptococcal cells opsonized with serum from naïve mouse for 2 hr. Non-adherent or phagocytosed cells were washed, and cryptococcal cells were released and then serially diluted before plating onto YNB medium for measurement of CFUs. (D) The same as above, except opsonization, was performed with serum of mice vaccinated against cryptococcosis. (E) Mice were challenged with 1×105 cryptococcal cells intravenously. At day 5 post-infection, five mice per group were sacrificed. Brains, kidneys, and lungs of euthanized mice were dissected and homogenized. Serial dilutions were plated to count CFUs for quantification of fungal burden per organ.
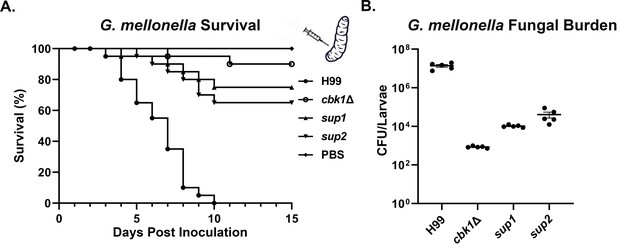
Suppressor mutants partially restore virulence in the G. mellonella model.
(A) G. mellonella larvae were infected with 5×104 fungal cells of the indicated strain, and their survival was monitored for 15 days post-inoculation. The cbk1Δ group survival curve was not significantly different from sup1 (p-value=0.2) and was significantly different from the sup2 curve (p-value=0.05). (B) At day 5 post-inoculation (DPI 5), 5 out of 25 larvae per group for the H99, the cbk1Δ mutant, sup1, and sup2 groups were homogenized, serially diluted, and plated to count the colony-forming units (CFUs) and measure the fungal burden of each larva. The fungal burden of the sup1 and sup2 groups was significantly higher than the cbk1Δ group (p-value<0.001).
Tables
| Reagent type (species) or resource | Designation | Source or reference | Identifiers | Additional information |
|---|---|---|---|---|
| Genetic reagent (Cryptococcus neoformans KN99α, matα) | WT strain: H99 | Nielsen et al., 2003 | ||
| Genetic reagent (C. neoformans KN99α, matα) | WT strain: H99a | Nielsen et al., 2003 | ||
| Genetic reagent (C. neoformans KN99α, matα) | cbk1Δ | Walton et al., 2006 | FJW9 | H99alpha, CBK1::NATr |
| Genetic reagent (C. neoformans KN99α, matα) | mob2Δ | Walton et al., 2006 | FJW10 | H99alpha, MOB2::NATr |
| Genetic reagent (C. neoformans KN99α, matα) | kic1Δ | Walton et al., 2006 | FJW8 | H99alpha, KIC1::NATr |
| Genetic reagent (C. neoformans KN99α, matα) | tao3Δ | Walton et al., 2006 | AI136 | MATalpha, TAO3:: NATr |
| Genetic reagent (C. neoformans KN99α, matα) | sog2Δ | Walton et al., 2006 | AI131 | MATalpha, SOG2:: NATr |
| Genetic Reagent (Candida albicans, SN250) | SN250 | Wakade et al., 2020 | SN250 | HIS-, LEU-, ARG- |
| Genetic Reagent (C. albicans, SN250) | cbk1ΔΔ | Wakade et al., 2020 | ΔΔcbk1 | SN250, ΔΔcbk1: HIS-, LEU-, ARG- |
| Genetic reagent (C. neoformans KN99α, matα) | cna1Δ | FGSC deletion set Plate 46 Well E12 | cna1Δ | H99alpha CNA1::NATr |
| Genetic reagent (C. neoformans KN99α, matα) | cdc24Δ | FGSC deletion set Plate 33 Well C4 | cdc24Δ | H99alpha, CDC24::NATr |
| Genetic reagent (C. neoformans KN99α, matα) | mpk1Δ | FGSC deletion set Plate 11 Well A5 | mpk1Δ | H99alpha, MPK1::NATr |
| Genetic reagent (C. neoformans KN99α, matα) | CBK1OE | This study. | BC1449 | H99alpha, PCTR4-CBK1-mCherry- NEOr |
| Genetic reagent (C. neoformans KN99α, matα) | cdc24Δ, CBK1OE | This study. | BC1281 | H99alpha, CDC24::NATr, PCTR4-CBK1-mCherry- NEOr |
| Genetic reagent (C. neoformans KN99α, matα) | cdc24Δ, CBK1OE | This study. | BC1282 | H99alpha, CDC24::NATr, PCTR4-CBK1-mCherry- NEOr |
| Genetic reagent (C. neoformans KN99α, matα) | cna1Δ, CBK1OE | This study. | BC1283 | H99alpha, CNA1::NATr, PCTR4-CBK1-mCherry- NEOr |
| Genetic reagent (C. neoformans KN99α, matα) | cna1Δ, CBK1OE | This study. | BC1284 | H99alpha, CNA1::NATr, PCTR4-CBK1-mCherry- NEOr |
| Genetic reagent (C. neoformans KN99α, matα) | mpk1Δ, CBK1OE | This study. | BC1285 | H99alpha, MPK1::NATr, PCTR4-CBK1-mCherry- NEOr |
| Genetic reagent (C. neoformans KN99α, matα) | mpk1Δ, CBK1OE | This study. | BC1286 | H99alpha, MPK1::NATr, PCTR4-CBK1-mCherry- NEOr |
| Genetic reagent (C. neoformans KN99α, matα) | cdc24Δ, CDC24OE | This study. | BC650 | H99alpha, CDC24::NATr, PCTR4-mNeonGreen-CDC24-NEOr |
| Genetic reagent (C. neoformans KN99α, matα) | cbk1Δ, CBK1OE | This study. | BC669 | H99alpha, CBK1::NATr, PCTR4-CBK1-mCherry- NEOr |
| Genetic reagent (C. neoformans KN99α, matα) | mpk1Δ, MPK1OE | This study. | BC1356 | H99alpha, MPK1::NATr, PGPD1-mNeonGreen-MPK1-NEOr |
| Genetic reagent (C. neoformans KN99α, matα) | cna1Δ, CNA1OE | Kozubowski et al., 2011 | LK214 | H99a, CNA1::NEOr, PH3-GFP-CNA1-NATr |
| Genetic reagent (C. neoformans KN99α, matα) | cbk1Δ, CDC24OE | This study. | BC1357 | H99alpha, CBK1::NATr, PCTR4-mNeonGreen-CDC24-NEOr |
| Genetic reagent (C. neoformans KN99α, matα) | cbk1Δ, MPK1OE | This study. | BC1358 | H99alpha, CBK1::NATr, PGPD1-mNeonGreen-MPK1-NEOr |
| Genetic reagent (C. neoformans KN99α, matα) | cbk1Δ, CNA1OE | This study. | BC1359 | H99alpha, CBK1::NATr, PH3-GFP-CNA1-NATr |
| Genetic reagent (C. neoformans KN99α, matα) | sup1 | This study. | BC1068 | H99alpha, CBK1::NATr,SUP1 |
| Genetic reagent (C. neoformans KN99α, matα) | sup2 | This study. | BC1076 | H99alpha, CBK1::NATr,SUP2 |
| Genetic reagent (C. neoformans KN99α, matα) | cbk1Δssd1Δ | This study. | BC1239 | H99alpha, CBK1::NATr, SSD1::NEOr |
| Genetic reagent (C. neoformans KN99α, matα) | ssd1Δ | This study. | BC1241 | H99alpha, SSD1::NEOr |
| Genetic reagent (C. neoformans KN99α, matα) | cbk1Δpsc1Δ | This study. | BC1369 | H99alpha, CBK1::NATr, PSC1::HYGr |
| Genetic reagent (C. neoformans KN99α, matα) | psc1Δ | This study. | BC1393 | H99alpha, PSC1::HYGr |
| Genetic reagent (C. neoformans KN99α, matα) | cac1Δ | Bahn et al., 2006 | YSB42 | H99alpha, CAC1::NATr STM#159 |
| Genetic reagent (C. neoformans KN99α, matα) | aca1Δ | FGSC deletion set Plate 32 Well H6 | aca1Δ | H99alpha, ACA1::NATr |
| Genetic reagent (C. neoformans KN99α, matα) | pkr1Δ | FGSC deletion set Plate 33 Well H7 | pkr1Δ | H99alpha, PKR1::NATr |
| Genetic reagent (C. neoformans KN99α, matα) | pde1Δ | FGSC deletion set Plate 10 Well A12 | pde1Δ | H99alpha, PDR1::NATr |
| Genetic reagent (C. neoformans KN99α, matα) | pde2Δ | FGSC deletion set Plate 11 Well H7 | pde2Δ | H99alpha, PDR2::NATr |
| Genetic reagent (C. neoformans KN99α, matα) | gpa1Δ | Bahn et al., 2005 | YSB83 | H99alpha, GPA1::NATr STM#5 |
| Genetic reagent (C. neoformans KN99α, matα) | cln1Δ | This study. | BZ36 | H99alpha, CLN1::NATr |
| Genetic reagent (C. neoformans KN99α, matα) | ecm2201Δ | FGSC deletion set Plate 2 Well A10 | ecm2201Δ | H99alpha, ECM2201::NATr |
| Genetic reagent (C. neoformans KN99α, matα) | kin4Δ | Bahn Kinase Deletion Set Plate 4 Well G8 (Lee et al., 2016) | kin4Δ | H99alpha, KIN4::NATr |
| Recombinant DNA reagent | pPZP-NATcc | Dickinson et al., 2013 | pPZP-NATcc | |
| Recombinant DNA reagent | pDD162 | Walton et al., 2006 | pDD162 | |
| Recombinant DNA reagent | pFZ1-CDC24 | This study. | PCTR4-2-mNeonGreen-CDC24(H99)-NEOr | |
| Recombinant DNA reagent | pXC-CBK1-mCh | This study. | PCTR4-2-CDC24(H99)-mCherry-NEOr | |
| Recombinant DNA reagent | LKB61 | Kozubowski et al., 2011 | PGPD1-mCherry-CNA1-HYGr | |
| Recombinant DNA reagent | pUC19-MPK1-mNG | This study. | PGPD1-MPK1-mNeonGreen-NEOr | |
| Cell line (Mus. Musculus, macrophage cell line J774A.1) | J774A.1 | American Type Culture Collection | ATCC TIB-67 | |
| Chemical Compound and drug | Hygromycin | Research Products International | Cat. NO.: H75000 | |
| Chemical Compound and drug | G418 | Research Products International | Cat. NO.: G64000 | |
| Chemical Compound and drug | Nourseothricin | Jena Bioscience | Cat. NO.: AB-102–25 G | |
| Software and algorithm | Graphpad Prism 9 | Graphpad | ||
| Software and algorithm | nSolver software version 4.0 | NanoString Technologies | ||
| Software and algorithm | Trim Galore v0.6.5 | Krueger, 2021 | ||
| Software and algorithm | BWA aligner v0.7.17 | Li, 2013 | ||
| Software and algorithm | SAMtools v1.10 | Li et al., 2009a | ||
| Software and algorithm | Picard Tools v2.16.0 | Broad_Institute, 2022 | ||
| Software and algorithm | bcftools v1.13 | Danecek et al., 2021 | ||
| Software and algorithm | Illustrator of biological sequences (IBS) | Liu et al., 2015 |
Additional files
-
Supplementary file 1
Hits from forward genetic screening.
- https://cdn.elifesciences.org/articles/82563/elife-82563-supp1-v2.docx
-
MDAR checklist
- https://cdn.elifesciences.org/articles/82563/elife-82563-mdarchecklist1-v2.docx
-
Source data 1
NanoString probe targets.
- https://cdn.elifesciences.org/articles/82563/elife-82563-data1-v2.xlsx





