A coordinated transcriptional switching network mediates antigenic variation of human malaria parasites
Figures
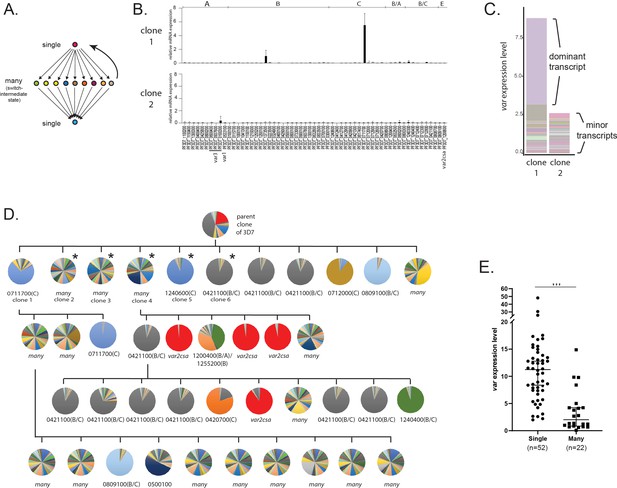
Detection of ‘single’ and ‘many’ var expression profiles in cultured parasite populations.
(A) Schematic representation of the single-many-single (SMS) model for var gene transcriptional switching. Each circle represents expression of an individual var gene and arrows represent switches in expression. Switching events are hypothesized to transition from activation of a single gene (top) to a broad range of genes (middle) to a different single gene (bottom) or back to the original gene (reverse arrow). (B) var gene expression profiles for two clonal populations that display the single phenotype (clone 1, top) or the many phenotype (clone 2, bottom). var transcription levels were determined using a standardized quantitative real-time RT-PCR (qRT-PCR) protocol with the expression of each individual var gene displayed as relative copy number in the histogram. Error bars represent standard deviation of three biological replicates. (C) Total var expression for the two subclones shown in B, with transcripts from each individual var gene shown in a different color. For clone 1, transcripts from the dominant gene are marked, while both clones display similar levels of minor transcripts. (D) Clone tree of wildtype 3D7 parasites. Pie charts display the var expression profile for each subcloned population with each slice of the pie representing the expression level of a single var gene. Vertical and horizontal lines delineate sequential rounds of subcloning by limiting dilution. For parasite populations that display a dominantly expressed var gene, the annotation number is shown below the pie chart with the var type shown in parenthesis. The five subclones marked with an asterisk are further analysed in Figure 2. (E) Total var expression levels as determined by qRT-PCR for 74 subclones (see Figure 1—figure supplement 1). The median ± 95% confidence interval is shown, and an unpaired t-test indicates a ***p < 0.0001.
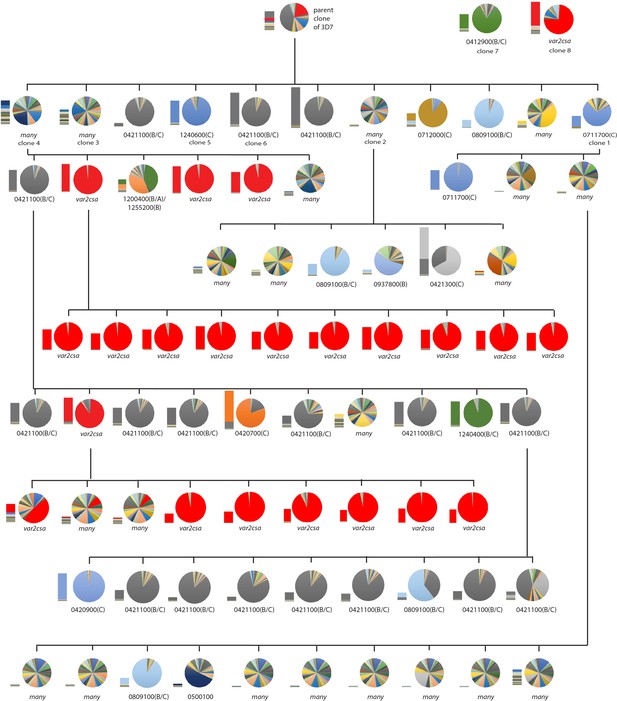
Clone tree of wildtype 3D7 parasites.
Pie charts display the var expression profile for each subcloned population with each slice of the pie representing the expression level of a single var gene. Vertical and horizontal lines delineate sequential rounds of subcloning by limiting dilution. For parasite populations that display a dominantly expressed var gene, the annotation number is shown below the pie chart with the var type shown in parenthesis. A stacked bar graph is included next to each pie chart which displays the total var expression level, with transcripts from each individual var gene shown in a different color.
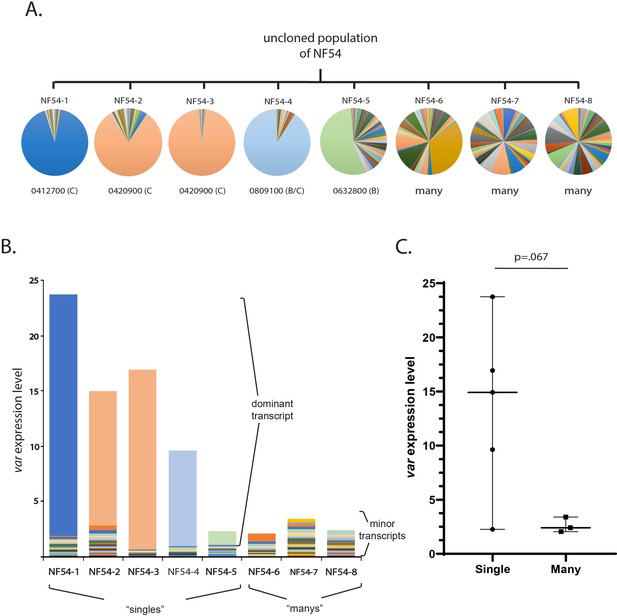
Examination of var gene expression profiles of recent clones of NF54.
(A) Cloning by limiting dilution was performed using an uncloned, heterogenous population of the NF54 isolate. Eight individual clonal populations were generated and allowed to expand for 5 weeks, at which point they were synchronized and RNA isolated from ring stages. var gene expression profiles were generated and displayed as pie charts with the dominantly expressed var gene indicated below the chart for clones 1–4. (B) Total var expression levels for all eight clones shown in A, with transcripts for each var gene shown in a different color. (C) Total var expression levels as determined by quantitative real-time RT-PCR (qRT-PCR) for all eight NF54 subclones. The median ± 95% confidence interval is shown, and an unpaired t-test indicates a p value of 0.0067.
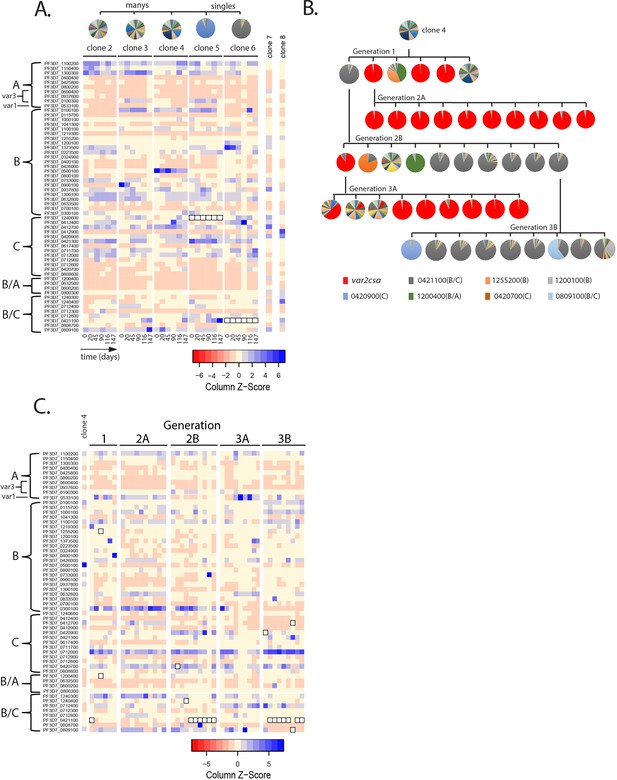
Detection of var gene minor transcript expression profiles in clonal populations overtime.
(A) Heatmap of var minor transcripts for clones 2–6. The clones were all derived from the same parent population as shown in Figure 1D. Pie charts show the initial var gene expression profiles above the heatmap. The annotation numbers for each var gene are shown to the left of the heatmap and organized according to var type (left, var2csa is considered separately in Figure 3). Six time points are included for each clone. Two genetically identical parasite populations (clones 7 and 8) also originally derived from 3D7 but grown separately for several years are shown for comparison. For clones 5 and 6, the dominant transcript (PF3D7_1240600 and PF3D7_0421100, respectively, marked by black boxes in the heatmap) was removed from the analysis to enable visualization of the minor transcripts. (B) Clone tree of wildtype 3D7. The tree is organized into three ‘subclone generations’ derived from initial clone 4 (top row). Pie charts display the var expression profile for each subclone. (C) Heatmap of var minor transcripts for the individual clones from each generation shown in B with the pattern of the parent population (clone 4) shown at the left for comparison. The annotation numbers for each var gene are shown to the left of the heatmap, and the gene order was organized according to var type. The order from left to right of each column in the heatmap corresponds to the order from left to right of the pie charts for each subclone generation shown in B. For parasites expressing the ‘single’ phenotype, the dominant transcript (marked by black boxes in the heatmap) was removed to enable visualization of the minor transcripts.
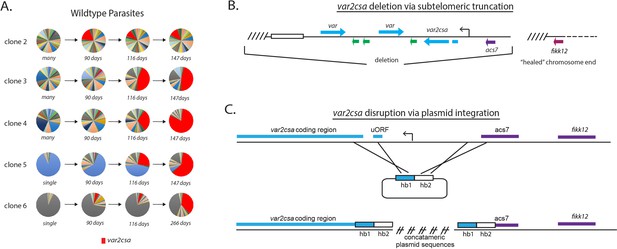
Convergence to var2csa expression in long-term cultures and targeted deletion of the var2csa locus.
(A) Changes in var expression over time in five clonal parasite populations, three ‘manys’ (clones 2–4) and two ‘singles’ (clones 5 and 6), over several months of continuous culture. Expression of var2csa is shown in red. (B) Schematic diagram showing the truncation of the end of chromosome 12 by telomere healing. Telomere repeats are shown as slanted lines and the telomere-associated repeat elements (TAREs) are shown as an open box. The interior of the chromosome is displayed as a dashed line. A ~60 kb deletion, including three var genes (blue), three rif genes (green), and the acs7 gene is shown. (C) Schematic diagram showing the var2csa locus and the plasmid containing homology blocks for targeted integration (hb1 and hb2). The crossed lines linking the plasmid to the chromosome signify sites of double cross over recombination leading to deletion of approximately 2.5 kb upstream of the va2csa gene, including the promoter.
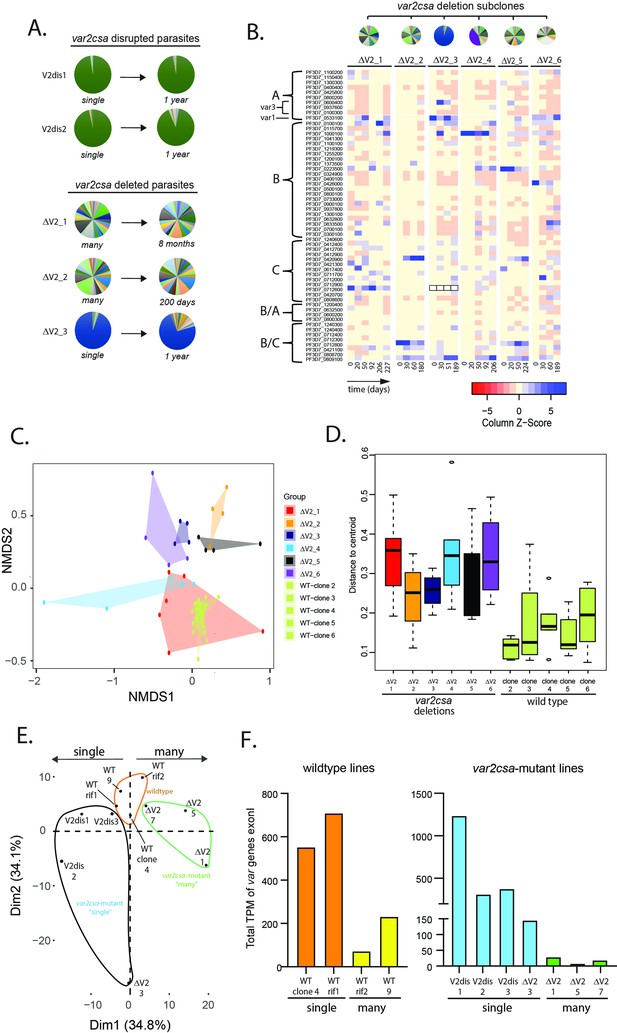
Alterations in var gene expression after deletion or disruption of var2csa.
(A) Changes in var expression profile over time in five clonal parasite populations containing deletions or disruption of the var2csa locus, including two in which the locus was disrupted by plasmid integration (V2dis1 and V2dis2, top) and three in which the locus was deleted by chromosomal truncation (ΔV2_1, ΔV2_2, and ΔV2_3). V2dis1, V2dis2, and ΔV2_3 display a ‘single’ phenotype while ΔV2_1 and ΔV2_2 express many var genes. (B) Heatmap displaying the pattern of var minor transcripts over several time points for five var2csa-deleted lines. The clones were all derived from the same parent population, and pie charts showing the initial var gene expression profiles are shown above the heatmap. The annotation numbers for each var gene are shown to the left of the heatmap, organized according to var type. For ΔV2_3, the dominant transcript (Pf3D7_0712600, marked by black boxes in the heatmap) was removed to enable visualization of the minor transcripts. (C) Non-metric multidimensional scaling (NMDS) plot based on Bray–Curtis dissimilarities displaying the expression profiles of the var minor transcripts for the six var2csa-deleted lines alongside five wildtype lines. (D) The variability within each cluster shown in C is inferred by its area on the plot (as defined by the mean distance from the group centroid for each time point) and displayed in a boxplot. (E) Transcriptomic analysis of wildtype and var2csa-mutated parasite lines. Principal component analysis (PCA) is shown based on the normalized expression level of 5721 genes log10(fragments per kilobase of transcript per million mapped fragments (FPKM) + 1). RNAseq derived whole transcriptomes were obtained from 11 clonal parasite populations: two wildtype lines (WT 3.1 and WT 7) and two WT lines with a control plasmid integration into a rif gene (WT_rif1 and WT_rif2) are displayed in orange; four var2csa-mutated lines displaying a ‘single’ phenotype (V2dis1, V2dis2, V2dis3, and ΔV2_3) are displayed in blue; and three var2csa-mutated lines displaying a “many” phenotype (ΔV2_1, ΔV2_5, and ΔV2_7) are displayed in green. All analyses were performed using data obtained from synchronized populations ~16 hr after red cell invasion. (F) Comparisons of normalized var transcript counts from var2csa-mutated and wildtype lines displaying either the single (left) or many (right) phenotypes.
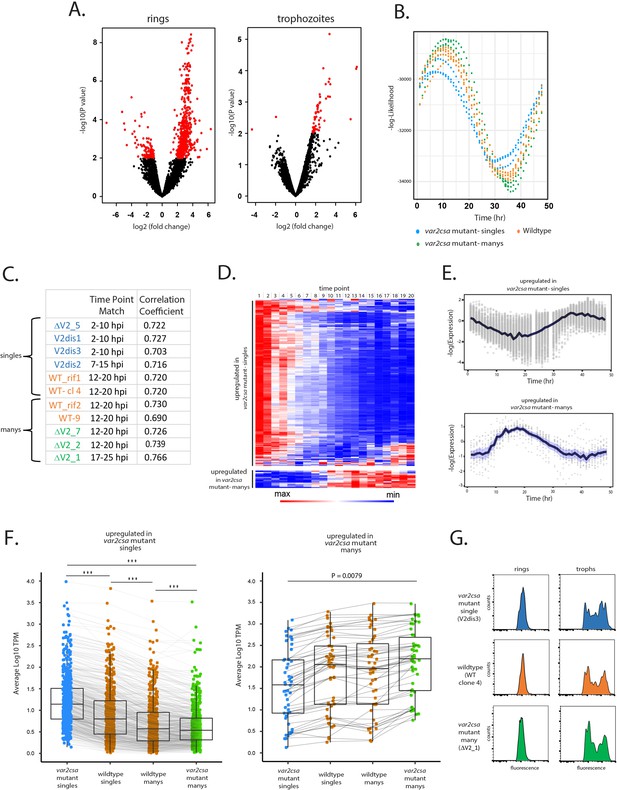
Parasites displaying ‘single’ vs. ‘many’ var expression profiles differ in replication cycle progression.
(A) Volcano plots showing differentially expressed genes derived from whole transcriptome comparisons of var2csa-mutated ‘single’ and ‘many’ parasite lines. Differentially expressed genes are shown in red. For ring-stage parasites (left), 562 transcripts displayed higher expression levels in the single lines, while 49 were higher in the many lines. For trophozoite-stage parasites, only 51 genes were differentially expressed. (B) Estimation of cell cycle position using the method of Lemieux et al., 2009. Using the transcriptome profiles in Supplementary file 8 (Bártfai et al., 2010), the likelihood (vertical axis) of each sample having been derived from a particular time point of the cycle (horizontal axis) is displayed. Eleven parasite lines are shown, with wildtype parasites in orange, var2csa-mutated, ‘single’ parasites in blue and var2csa-mutated, ‘many’ parasites in green type. (C) Table showing estimates of replication cycle progression for all 11 parasite lines. Estimates of the approximate time point within the 48-hr asexual cycle for each population were obtained by comparison with a modeled parasite population over a simulated infection time course using established datasets (Poran et al., 2017). The best time point match is shown for each population along with the correlation coefficient. Wildtype parasites are designated with orange type, var2csa-mutated, ‘single’ parasites with blue type and var2csa-mutated, ‘many’ parasites with green type. (D) Heatmap displaying changes in expression over the asexual replicative cycle for 611 differentially expressed genes according to the 48-hr asexual cycle of the P. falciparum HB3 transcriptome datasets defined by Bozdech et al., 2003; Supplementary file 9. Genes upregulated in var2csa-mutated ‘single’ lines are shown on top and those from var2csa-mutated ‘many’ lines are displayed on the bottom. (E) Average expression levels across the 48-hr asexual cycle of HB3 for 611 differentially expressed genes. Genes upregulated in var2csa-mutated, ‘single’ lines are shown on top and those upregulated in var2csa-mutated, ‘many’ lines are shown on the bottom. (F) Left panel, transcript expression levels for 559 differentially expressed genes that display higher expression in ‘single’ parasites. Average, normalized transcript levels were obtained from var2csa-mutated, ‘single’ parasites (blue), wildtype ‘single’ and ‘many’ parasites (orange, left and right, respectively), and var2csa-mutated, ‘many’ parasites (green). Box and whisker plots display the mean and standard deviation for each dataset. Right panel, analysis of transcript expression levels for 49 differentially expressed genes that display higher expression in ‘many’ parasites. Pairwise comparisons using t-tests with pooled standard deviation. p value adjustment method is from Bonferroni (significance codes: 0 ‘***’, 0.001 ‘**’, 0.01 ‘*’). (G) Monitoring of DNA content of infected cells by flow cytometry. Populations of var2csa-mutant ‘singles’ (top), wildtype (middle), and var2csa-mutant ‘manys’ (bottom) were tightly synchronized then assayed for DNA content by flow cytometry using Hoechst 33342 staining. Parasites were assayed at 16 and 36 hr after invasion.
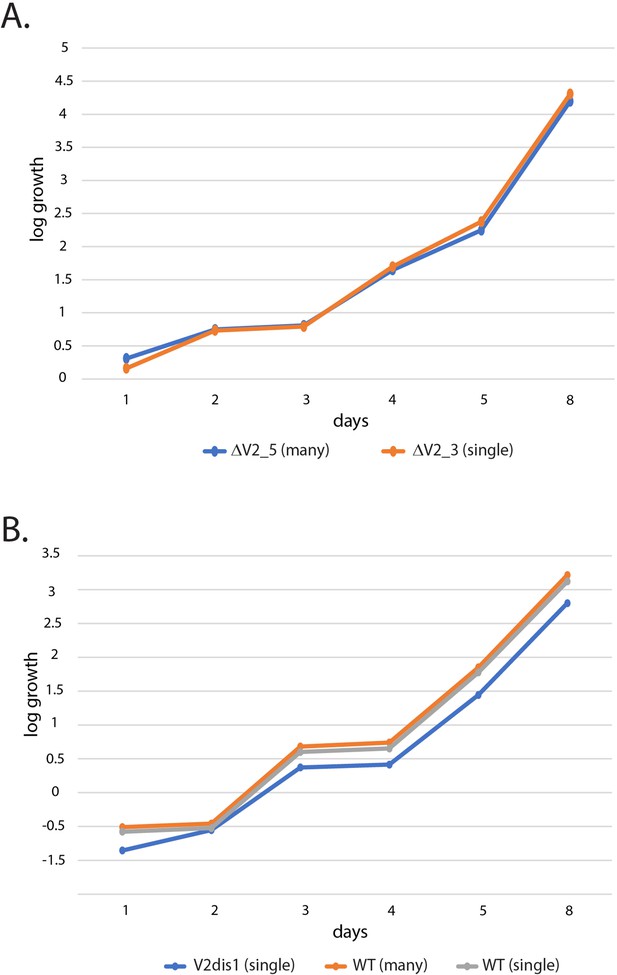
Growth assays of parasites displaying the ‘single’ and ‘many’ var expression phenotypes.
Parasitemias were determined daily by flow cytometry and cultures were diluted by 10-fold whenever the parasitemia reached 0.5%. The daily parasitemia was multiplied by the exponential dilution factor and plotted as log growth over time. (A) Comparison of two clonal lines in which var2csa has been deleted. ΔV2_5 (blue) displays the ‘many’ phenotype while ΔV2_3 (orange) displays the ‘single’ expression profile. (B) Comparison of a var2csa-mutant line (V2dis1) displaying the ‘single’ phenotype with two wildtype lines, one displaying the many phenotype (orange) and one expressing a single dominant var gene (gray).
Tables
| Reagent type (species) or resource | Designation | Source or reference | Identifiers | Additional information |
|---|---|---|---|---|
| Gene (Plasmodium falciparum) | var2csa | EuPathDB | PF3D7_1200600 | |
| Strain, strain background (P. falciparum) | NF54 | Delemarre and van der Kaay, 1979 (PMID:390409) | NF54 | |
| Strain, strain background (P. falciparum) | 3D7 | Walliker et al., 1987 (PMID:3299700) | 3D7 | |
| Transfected construct (P. falciparum) | pL6_eGFP | Ghorbal et al. (PMID:24880488) | CRISPR-targeting plasmid | |
| Transfected construct (P. falciparum) | pUF1_Cas9 | Ghorbal et al. (PMID:24880488) | CRISPR-targeting plasmid | |
| Software, algorithm | aligner HISAT2 | Kim et al., 2019 (PMID:31375807), Afgan et al., 2018 (PMID:29790989) | v.2.2.0 | |
| Software, algorithm | featureCounts | Liao et al., 2014 (PMID:24227677) | Package Rsubread v.2.0.1 | |
| Software, algorithm | DESeq2 | Love et al., 2014 (PMID:25516281) | v.3.14 | |
| Software, algorithm | EdgeR | Chen et al., 2016 (PMID:27508061), McCarthy et al., 2012 (PMID:22287627), Robinson et al., 2010 (PMID:19910308) | v.3.14 | |
| Software, algorithm | R | Rstudio (1.4.1717) | v.4.1.0 | |
| Software, algorithm | vegan | Community Ecology Package | v.2.5-7 | |
| Software, algorithm | PERMANOVA | Community Ecology Package | v.2.5-7 | |
| Software, algorithm | heatmap.2 | gplots | v.2.3.0 |
Additional files
-
Supplementary file 1
List of differentially expressed genes when comparing var2csa-mutant ‘single’ lines to var2csa-mutant ‘many’ lines at ring stages.
Genes upregulated in ‘singles’ are denoted in blue while those upregulated in ‘manys’ are denoted in green. log2 FC = logFold Change, logCPM = log(normalized mean Count per Million), LR = likehood ratio, FDR = adjust p value.
- https://cdn.elifesciences.org/articles/83840/elife-83840-supp1-v2.xlsx
-
Supplementary file 2
List of differentially expressed genes when comparing var2csa-mutant ‘single’ lines to var2csa-mutant ‘many’ lines in trophozoite stage.
Genes upregulated in ‘singles’ are denoted in blue while those upregulated in ‘manys’ are denoted in green. logCPM = log10(normalized mean Count per Million), LR = likelihood ratio, FDR = adjust p value.
- https://cdn.elifesciences.org/articles/83840/elife-83840-supp2-v2.xlsx
-
Supplementary file 3
Annotation numbers and gene ontology terms for each of 562 genes upregulated in var2csa-mutant ‘single’ lines when compared to var2csa-mutant ‘many’ lines.
Also shown are expression values described for each gene across the 48-hr asexual replication cycle as established in the published dataset from Bartfai et al. These values were used to establish the approximate position within the standard asexual cycle for each sample.
- https://cdn.elifesciences.org/articles/83840/elife-83840-supp3-v2.xlsx
-
Supplementary file 4
Annotation numbers and gene ontology terms for each of 49 genes upregulated in var2csa-mutant ‘many’ lines when compared to var2csa-mutant ‘single’ lines.
Also shown are expression values described for each gene across the 48-hr asexual replication cycle as established in the published dataset from Bartfai et al. These values were used to establish the approximate position within the standard asexual cycle for each sample.
- https://cdn.elifesciences.org/articles/83840/elife-83840-supp4-v2.xlsx
-
Supplementary file 5
Detailed likelihood of each sample time point using the method described by Poran et al.
All time points refer to the 48-hr asexual replication cycle.
- https://cdn.elifesciences.org/articles/83840/elife-83840-supp5-v2.xlsx
-
Supplementary file 6
Primers used during the study.
The top four primers were used for PCR amplification of specific homology blocks during the construction of the pL6-var2csa-promoter deletion plasmid. (A) Primers for plasmid pL6-var2csa-promoter-deletion and pLcas9 construction. (B) Additional quantitative real-time RT-PCR (qRT-PCR) primers for var transcription detection.
- https://cdn.elifesciences.org/articles/83840/elife-83840-supp6-v2.xlsx
-
Supplementary file 7
Summary of the total number of reads and mapped reads for the transcriptome analysis of various ‘single’ and ‘many’ expression lines, including var2csa-mutant lines (ΔV2_1, V2dis3, ΔV2_7, V2dis1, V2dis2, ΔV2_5, and ΔV2_3) and wildtype lines (Subclone 3.1, WT_7, WT_rif2, and WT_rif1).
Analysis of ring stages is shown on the left and trophozoite stages are on the right.
- https://cdn.elifesciences.org/articles/83840/elife-83840-supp7-v2.xlsx
-
Supplementary file 8
RNAseq dataset from Bártfai et al., 2010 used as the reference for estimating the replication cycle time point of each sample population using the method of Poran et al., 2017.
Transcription levels (FPKM) are shown for each gene through eight time points of the Plasmodium falciparum (3D7) asexual cycle.
- https://cdn.elifesciences.org/articles/83840/elife-83840-supp8-v2.xlsx
-
Supplementary file 9
Microarray dataset of Plasmodium falciparum (HB3) gene expression from Bozdech et al., 2003 used as the reference to estimate the replication cycle time point of RNAseq samples using the method of Lemieux et al., 2009.
The form was kindly provided by Dr. Silvia Portugal.
- https://cdn.elifesciences.org/articles/83840/elife-83840-supp9-v2.xlsx
-
MDAR checklist
- https://cdn.elifesciences.org/articles/83840/elife-83840-mdarchecklist1-v2.pdf





