Peer review process
Revised: This Reviewed Preprint has been revised by the authors in response to the previous round of peer review; the eLife assessment and the public reviews have been updated where necessary by the editors and peer reviewers.
Read more about eLife’s peer review process.Editors
- Reviewing EditorErika BachNYU Grossman School of Medicine, New York, United States of America
- Senior EditorRichard WhiteUniversity of Oxford, Oxford, United Kingdom
Reviewer #1 (Public review):
Summary:
This preprint from Shaowei Zhao and colleagues presents results that suggest tumorous germline stem cells (GSCs) in the Drosophila ovary mimic the ovarian stem cell niche and inhibit the differentiation of neighboring non-mutant GSC-like cells. The authors use FRT-mediated clonal analysis driven by a germline-specific gene (nos-Gal4, UASp-flp) to induce GSC-like cells mutant for bam or bam's co-factor bgcn. Bam-mutant or bgcn-mutant germ cells produce tumors in the stem cell compartment (the germarium) of the ovary (Fig. 1). These tumors contain non-mutant cells - termed SGC for single-germ cells. 75% of SGCs do not exhibit signs of differentiation (as assessed by bamP-GFP) (Fig. 2). The authors demonstrate that block in differentiation in SGC is a result of suppression of bam expression (Fig. 2). They present data suggesting that in 73% of SGCs BMP signaling is low (assessed by dad-lacZ) (Fig. 3) and proliferation is less in SGCs vs GSCs. They present genetic evidence that mutations in BMP pathway receptors and transcription factors suppress some of the non-autonomous effects exhibited by SGCs within bam-mutant tumors (Fig. 4). They show data that bam-mutant cells secrete Dpp, but this data is not compelling (see below) (Fig. 5). They provide genetic data that loss of BMP ligands (dpp and gbb) suppresses the appearance of SGCs in bam-mutant tumors (Fig. 6). Taken together, their data support a model in which bam-mutant GSC-like cells produce BMPs that act on non-mutant cells (i.e., SGCs) to prevent their differentiation, similar to what in seen in the ovarian stem cell niche. This preprint from Shaowei Zhao and colleagues presents results that suggest tumorous germline stem cells (GSCs) in the Drosophila ovary mimic the ovarian stem cell niche and inhibit the differentiation of neighboring non-mutant GSC-like cells. The authors use FRT-mediated clonal analysis driven by a germline-specific gene (nos-Gal4, UASp-flp) to induce GSC-like cells mutant for bam or bam's co-factor bgcn. Bam-mutant or bgcn-mutant germ cells produce tumors in the stem cell compartment (the germarium) of the ovary (Fig. 1). These tumors contain non-mutant cells - termed SGC for single-germ cells. 75% of SGCs do not exhibit signs of differentiation (as assessed by bamP-GFP) (Fig. 2). The authors demonstrate that block in differentiation in SGC is a result of suppression of bam expression (Fig. 2). They present data suggesting that in 73% of SGCs BMP signaling is low (assessed by dad-lacZ) (Fig. 3) and proliferation is less in SGCs vs GSCs. They present genetic evidence that mutations in BMP pathway receptors and transcription factors suppress some of the non-autonomous effects exhibited by SGCs within bam-mutant tumors (Fig. 4). They show data that bam-mutant cells secrete Dpp, but this data is not compelling (see below) (Fig. 5). They provide genetic data that loss of BMP ligands (dpp and gbb) suppresses the appearance of SGCs in bam-mutant tumors (Fig. 6). Taken together, their data support a model in which bam-mutant GSC-like cells produce BMPs that act on non-mutant cells (i.e., SGCs) to prevent their differentiation, similar to what in seen in the ovarian stem cell niche.
Strengths:
(1) Use of an excellent and established model for tumorous cells in a stem cell microenvironment
(2) Powerful genetics allow them to test various factors in the tumorous vs non-tumorous cells
(3) Appropriate use of quantification and statistics
Weaknesses:
(1) What is the frequency of SGCs in nos>flp; bam-mutant tumors? For example, are they seen in every germarium, or in some germaria, etc or in a few germaria.
This concern was addressed in the rebuttal. The line number is 106, not line 103.
(2) Does the breakdown in clonality vary when they induce hs-flp clones in adults as opposed to in larvae/pupae?
This concern was addressed in the rebuttal. However, these statements are no on lines 331-335 but instead starting on line 339. Please be accurate about the line numbers cited in the rebuttal. They need to match the line numbers in the revised manuscript.
(3) Approximately 20-25% of SGCs are bam+, dad-LacZ+. Firstly, how do the authors explain this? Secondly, of the 70-75% of SGCs that have no/low BMP signaling, the authors should perform additional characterization using markers that are expressed in GSCs (i.e., Sex lethal and nanos).
The authors did not perform additional staining for GSC-enriched protein like Sex lethal and nanos.
(4) All experiments except Fig. 1I (where a single germarium with no quantification) were performed with nos-Gal4, UASp-flp. Have the authors performed any of the phenotypic characterizations (i.e., figures other than figure 1) with hs-flp?
In the rebuttal, the authors stated that they used nos>flp for all figures except for Fig. 1I. It would be more convincing for them to prove in Fig. 1 than there is not phenoytpic difference between the two methods and then switch to the nos>FLP method for the rest of the paper.
(5) Does the number of SGCs change with the age of the female? The experiments were all performed in 14-day old adult females. What happens when they look at young female (like 2-day old). I assume that the nos>flp is working in larval and pupal stages and so the phenotype should be present in young females. Why did the authors choose this later age? For example, is the phenotype more robust in older females? or do you see more SGCs at later time points?
The authors did not supply any data to prove that the clones were larger in 14-day-old flies than in younger flies. Additionally, the age of "younger" flies was not specified. Therefore, the authors did not satisfactorily answer my concern.
(6) Can the authors distinguish one copy of GFP versus 2 copies of GFP in germ cells of the ovary? This is not possible in the Drosophila testis. I ask because this could impact on the clonal analyses diagrammed in Fig. 4A and 4G and in 6A and B. Additionally, in most of the figures, the GFP is saturated so it is not possible to discern one vs two copies of GFP.
In the rebuttal, the authors stated that they cannot differential one vs two copies of GFP. They used other clone labeling methods in Fig. 4 and 6. I think that the authors should make a statement in the manuscript that they cannot distinguish one vs two copies of GFP for the record.
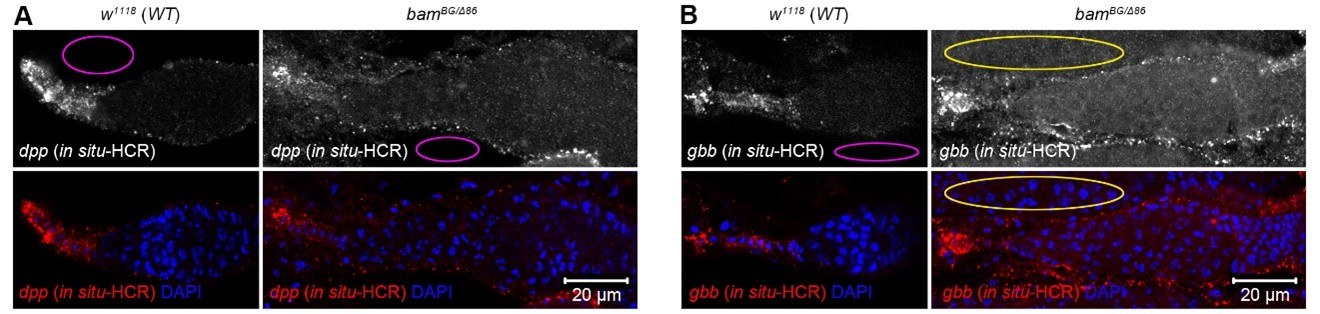
(7) More evidence is needed to support the claim of elevated Dpp levels in bam or bgcn mutant tumors. The current results with dpp-lacZ enhancer trap in Fig 5A,B are not convincing. First, why is the dpp-lacZ so much brighter in the mosaic analysis (A) than in the no-clone analysis (B); it is expected that the level of dpp-lacZ in cap cells should be invariant between ovaries and yet LacZ is very faint in Fig. 5B. I think that if the settings in A matched those in B, the apparent expression of dpp-lacZ in the tumor would be much lower and likely not statistically significantly. Second, they should use RNA in situ hybridization with a sensitive technique like hybridization chain reactions (HCR) - an approach that has worked well in numerous Drosophila tissues including the ovary.
The HCR FISH in Fig.5 of the revised manuscript needs an explanation for how the mRNA puncta were quantified. Currently, there is no information in the methods. What is meant but relative dpp levels. I think that the authors should report in and unbiased manner "number" of dpp or gbb puncta in TFs. For the germaria, I think that they should report the number of puncta of dpp or gbb divide by the total area in square pixels counted. Additionally, the background fluorescence is noticeably much higher in bamBG/delta86 germaria, which would (falsely) increase the relative intensity of dpp and gbb in bam mutants. Although, I commend the authors for performing HCR FISH, these data are still not convincing to me.
(8) In Fig 6, the authors report results obtained with the bamBG allele. Do they obtain similar data with another bam allele (i.e., bamdelta86)?
The authors did not try any experiments with the bamdelta86 allele, despite this allele being molecularly defined, where the bamBG allele is not defined.
Comments on second revision:
The authors have adequately addressed several points. However, there is still no information in the material and methods for how they measured and quantified the HCR-FISH probe signal. They have the same size region that they use for each genotype, but they do not control for the number of nuclei in each square. I would also be helpful if they provided a different image for the gbb probe stained in the mutant background. It is the only panel that does not have other germaria in very close proximity. I am still not fully convinced of the HCR data, esp for gbb.
Reviewer #2 (Public review):
In the current version, Zhang et al. have made substantial improvements to the manuscript. It is now easier to read, and the data are more solid compared with the previous version, supporting their conclusion that tumor GSCs secrete stemness factors (BMPs and Dpp) to suppress the differentiation of neighboring wild-type GSCs. This study should benefit a broad readership across developmental biology, germ cell biology, stem cell biology, and cancer biology.
Comments on revision:
If the exact number of germaria was not recorded (as described), an approximate number can be provided in the Materials and Methods; for example, stating that more than 10 germaria were analyzed per biological replicate.
Reviewer #3 (Public review):
Zhang et al. investigated how germline tumors influence the development of neighboring wild-type (WT) germline stem cells (GSC) in the Drosophila ovary. They report that germline tumors generated by differentiation-arrested mutations (bam and bgcn) inhibit the differentiation of neighboring WT GSCs by arresting them in an undifferentiated state, resulting from reduced expression of the differentiation-promoting factor Bam. They find that these tumor cells produce low levels of the niche-associated signaling molecules Dpp and Gbb, which suppress bam expression and consequently inhibit the differentiation of neighboring WT GSCs non-cell-autonomously. Based on these findings, the authors propose that germline tumors mimic the niche to suppress the differentiation of the neighboring wild-type germline stem cells.
Strengths:
The study uses a well-established in vivo model to addresses an important biological question concerning the interaction between germline tumor cells and wild-type (WT) germline stem cells in the Drosophila ovary. If the findings are substantiated, this study could provide valuable insights that are applicable to other stem cell systems.
Weaknesses:
The authors have addressed some of my concerns in the revised submission. However, the data presented do not allow the authors to distinguish whether the failed differentiation of WT stem cells/germline cells results from "arrested differentiation due to the loss of the differentiation niche" or from "direct inhibition by tumor-derived expression of niche-associated molecules Dpp and Gbb". The critical supporting data, HCR in situ results, are not sufficiently convincing.