Polo-like kinase Cdc5 regulates Spc72 recruitment to spindle pole body in the methylotrophic yeast Ogataea polymorpha
Figures
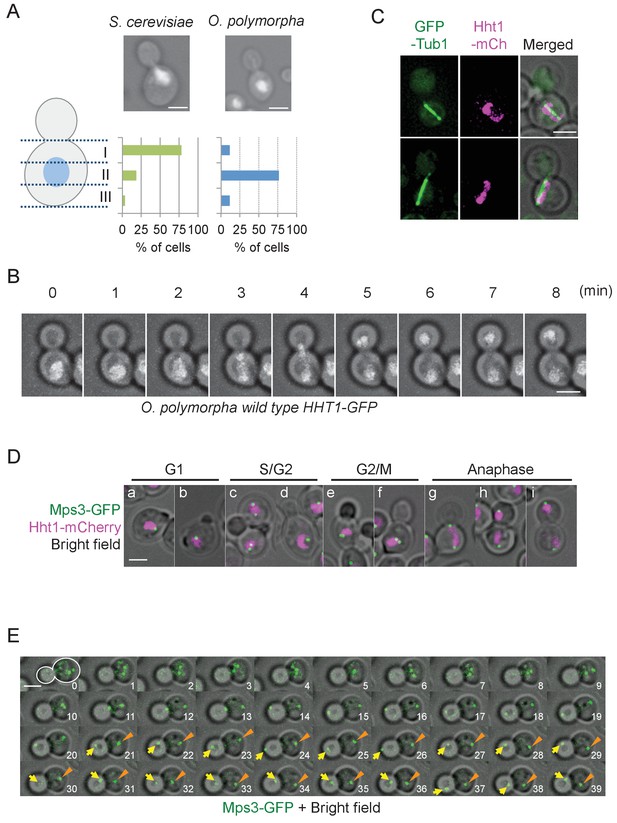
Nuclear positioning in O. polymorpha.
(A) Nucleus is positioned in the cell centre in pre-anaphase cells of O. polymorpha. S. cerevisiae strain YPH499 and O. polymorpha type strain CBS4732 were grown in YPDS at 30°C. DNA was stained with DAPI. The positions of nuclei were as outlined in the cartoon shown on the left side of the subfigure. Scale bar, 2 µm. N = 60 (YPH499), 55 (CBS4732). Result of a similar experiment using HHT1-GFP cells is shown in Figure 1—figure supplement 2. (B) Time-lapse microscopy of histone H3 gene (HHT1)-GFP cells (HPH31). Anaphase onset judged by Hht1-GFP was observed at the 3 min timepoint. Shown are a merged figure of bright field images and deconvolved and projected GFP images. Scale bar, 2 µm. (C) Early anaphase cells (HPH164) with a single DNA mass along the spindle grown in YPDS at 30°C. Microtubules and DNA are visualized by GFP-Tub1 and Hht1-mCherry fluorescence, respectively. Scale bar, 2 µm. (D) Early anaphase cells (HPH1678) grown in YPDS at 30°C. SPB and DNA are visualized by Mps3-GFP and Hht1-mCherry fluorescence, respectively. Scale bar, 2 µm. (E) Time lapse microscopy of cells expressing SPB-GFP maker. MPS3-GFP cells (HPH1681) were grown in SD complete medium at 30°C. Consecutive sections were taken every 60 s. Shown are representative images of cells with an inappropriate angled spindle against the polarity axis. Yellow arrows and orange arrowheads point SPB. Shown are deconvolved and projected GFP images merged with bright field image. Scale bar, 2 µm. Another example is shown in Figure 1—figure supplement 6.
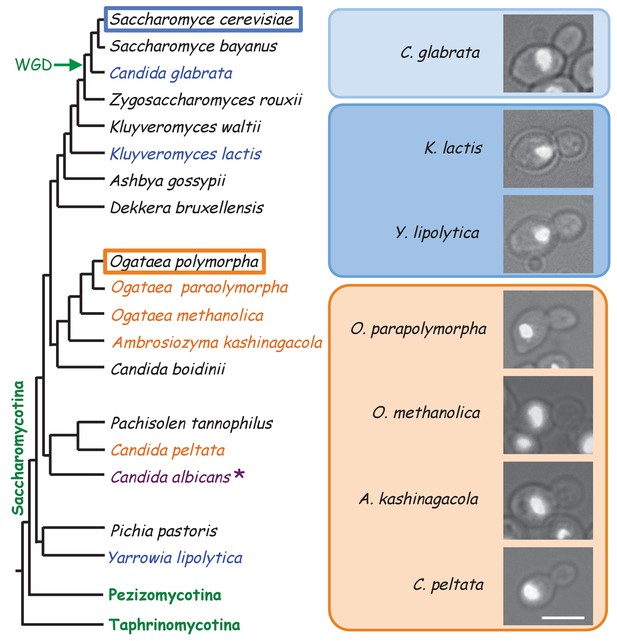
Nuclear position in different yeasts and their phylogenetic relationship.
The Phylogenetic tree is based on Kurtzman et al., 2011 and is not drawn to scale. Different yeast strains growing in YPDS at 30°C were fixed with ethanol and DNA was visualized with DAPI. Yeast strains used were BY21165, BY21167, BY23876, BY5243, JCM9829, JCM 10237, and JCM15019. * the regulation of nuclear position is reported not as strict as that in S. cerevisiae (Martin et al., 2004; Finley et al., 2008).
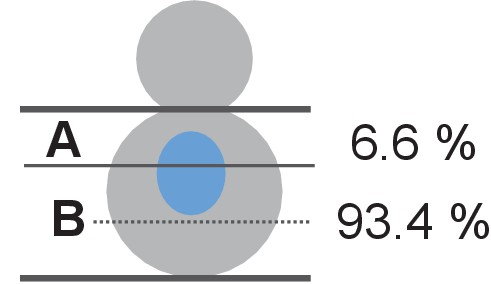
Nuclear positioning in O. polymorpha.
Nucleus is positioned in the cell centre in pre-anaphase cells of O. polymorpha. HHT1-GFP cells (HPH31) were grown in YPDS at 30°C. Position of nuclei was categorized into two: (A) the bud proximal one third of the mother cell body; (B) the bud distal two third of the mother cell as outlined in the cartoon. Percentage of cells that nucleus positioned in each categories are given next to the cartoon. N = 61.
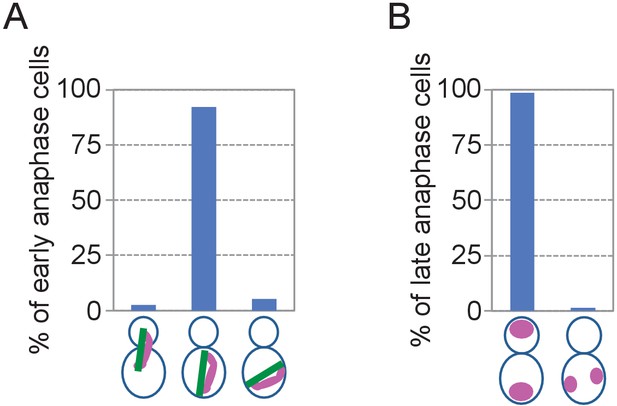
Nuclear segregation in O.polymorpha.
(A) Quantification of Figure 1C. Early anaphase cells were categorized into three based on the position and orientation of the spindle. Left, cells with spindle inserted into the bud. Centre, cells with oriented spindle in the mother cell body. Right, cells with misoriented spindle in the mother cell body. n = 38. (B) Nuclear segregation is successful in O. polymorpha cells in Figure 1C. Late anaphase cells of wild-type (HPH164) judged by two fully segregated DNAs and an elongated spindle were subject to the analysis. Position of two segregated DNAs was categorized into two: left, one DNA mass was segregated into the bud; right, two DNA masses remained in the mother cell body. n = 72.
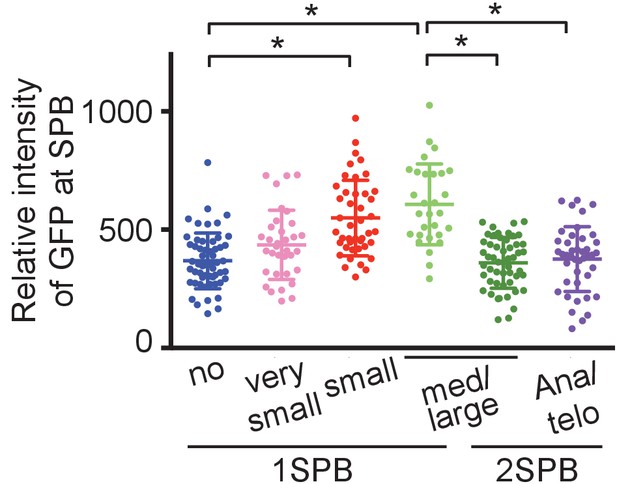
SPB duplication initiates at the timing of bud emergence in O. polymorpha.
Quantification of the Mps3-GFP SPB signal in cells carrying MPS3-GFP cells (HPH1681). Cells were grown in YPDS medium at 30°C. Cells were categorized based on the bud size and the number of SPB signal. Signal intensities were background-subtracted. Statistical significance was determined by the student t-test and is indicated by an asterisk. Error bars indicate SD. N = 255.

Time lapse microscopy of cells expressing SPB-GFP maker.
MPS3-GFP cells (HPH1681) were grown in SD complete medium at 30°C. Consecutive sections were taken every 60 s. Orange arrowhead point separated SPB signals. Shown are deconvolved and projected GFP images merged with bright field image. Scale bar, 2 µm.

Behaviour of SPB during spindle alignment and mitosis.
Another example of time lapse microscopy shown in Figure 1E. MPS3-GFP cells (HPH1681) were grown in SD complete medium at 30°C. Consecutive sections were taken every 60 s. Yellow arrows and orange arrowheads point SPB. Shown are deconvolved and projected GFP images merged with bright field image. Scale bar, 2 µm.
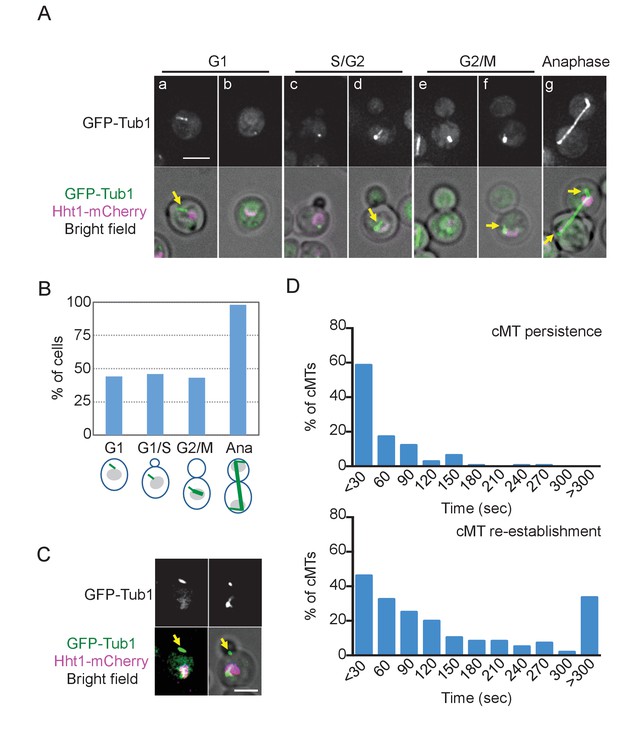
O. polymorpha cells contain fewer cytoplasmic microtubules (cMTs).
(A) Wild-type cells (HPH164) were grown in YPDS medium at 30°C. Microtubules and DNA are visualized by GFP-Tub1 and Hht1-mCherry fluorescence, respectively. cMTs are marked by yellow arrows. a, b: unbudded G1 cell; c, d: preanaphase cell with monopolar nuclear MTs; e, f: preanaphase cell with bipolar spindle; g: anaphase cell. Scale bar, 2 µm. (B) Quantification of (A). G1, S/G2, G2/M, and anaphase represent unbudded cells, small budded cells with single unduplicated SPB, medium budded cells with 2 SPB, and large budded cells with an SPB in both the mother and the bud, and large budded cells with elongated spindle, respectively. n > 50 cells for each category. Ana, anaphase. (C) Cells containing the cMT detached from the SPB in (A). Scale bar, 2 µm. (D) Analysis of the duration of cMT persistence and cMT re-establishment time at SPBs in preanaphase cells with a bipolar spindle by time-lapse microscopy. GFP-TUB1 cells (HPH194) were grown in SD complete medium at 30°C. Consecutive sections were taken every 30 s. Duration of continuous cMT presence was scored as cMT persistance, while the time between the loss of cMT and the acquisition of a new cMT was scored as cMT re-establishment. Total recording time was 38460 s.
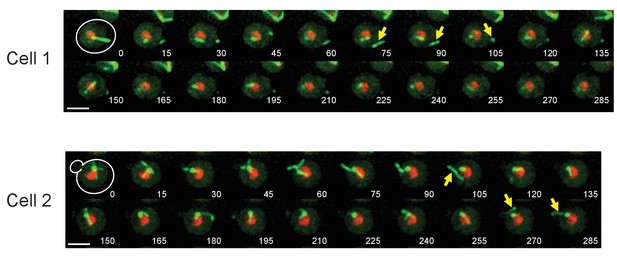
Time-lapse microscopy of GFP-TUB1 HHT1-mCherry cells.
Representative images of time-lapse microscopy in GFP-TUB1 HHT1-mCHerry cells (HPH164). Cells were grown in SD complete medium at 30°C. Consecutive sections were taken every 30 s. Yellow arrows point cMTs detached from SPB. Scale bar, 2 µm.
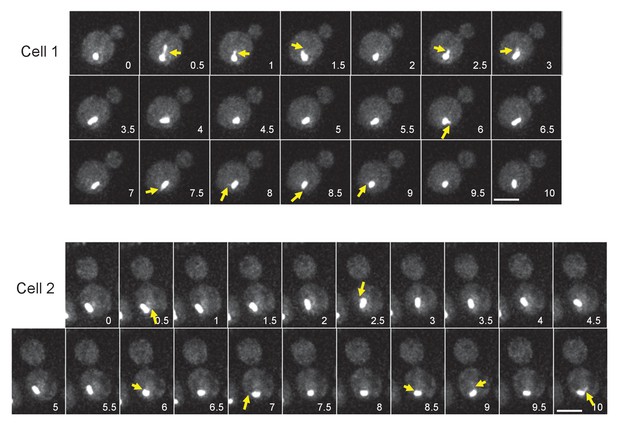
Time-lapse microscopy of GFP-TUB1 cells.
Representative images of time-lapse microscopy in (D). GFP-TUB1 cells (HPH194) were grown in SD complete medium at 30°C. Consecutive sections were taken every 30 s. Yellow arrows point cMTs. Scale bar, 2 µm.
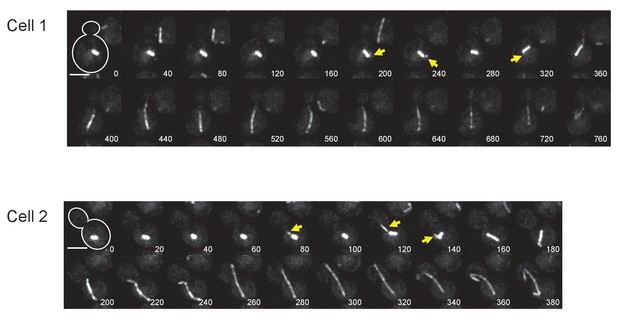
Time-lapse microscopy of GFP-TUB1 cells.
Representative images of time-lapse microscopy of GFP-TUB1 cells (HPH194). Cells were grown in SD complete medium at 30°C. Consecutive sections were taken every 40 s for Cell1 and 20 s for Cell2. Yellow arrows point cMTs in pre-anaphase stages. Scale bar, 2 µm.
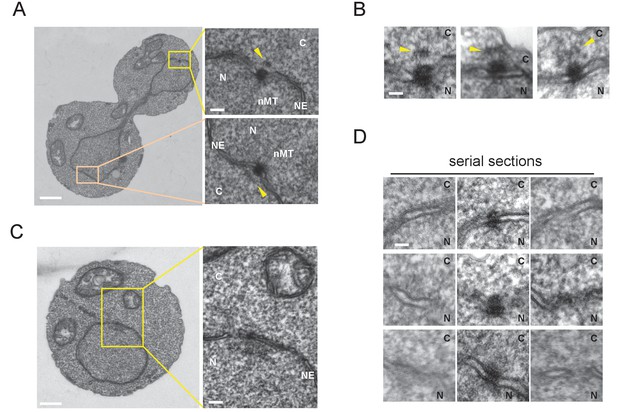
Cytoplasmic structure of SPB is regulated during the cell cycle.
Electron microscopy (EM) of thin serial sections of cells in G1 and anaphase. Wild-type cells (BY4329) were grown to log phase at 30°C in YPDS and then prepared for EM. Indicated are the cytoplasm (C), nucleus (N), nuclear envelope (NE), and nuclear microtubules (nMT). (A, B) Representative SPBs in anaphase cells (n = 10). The SPB in the mother does not appear in the section shown as the main image. The pale orange rectangle in the mother merely indicates the position of the lower inset which is the image of the section containing the SPB in the mother. (C, D) representative SPBs in unbudded G1 cells (n = 10). Consecutive three sections are shown in (D). Scale bars of the main images in (A) and (C) represent 1 µm. Scale bars of the insets in (A) and (C), i.e., (B) and (D), represent 100 nm.
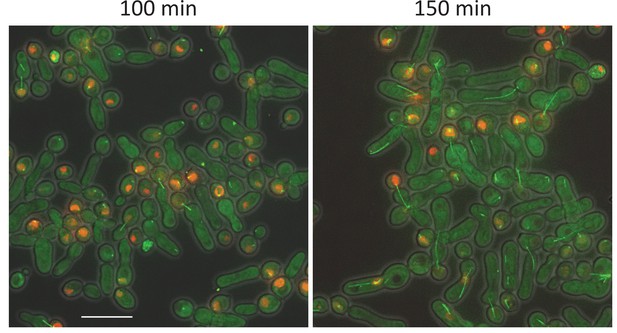
Phenotype of O. polymorpha cdc28-as cells.
Logarithmically growing cdc28-as GFP-TUB1 (HPH1210) cells in YPDS medium were treated wtih 5 µM IAA for the indicated time. Inhibition of Cdc28 kinase caused elongated bud morphologies as observed in S. cerevisiae. However, cells were not arrested completely and appearance of short spindle and segregated nuclei at 150 timepoint indicate cell cycle progression, albeit slowly.
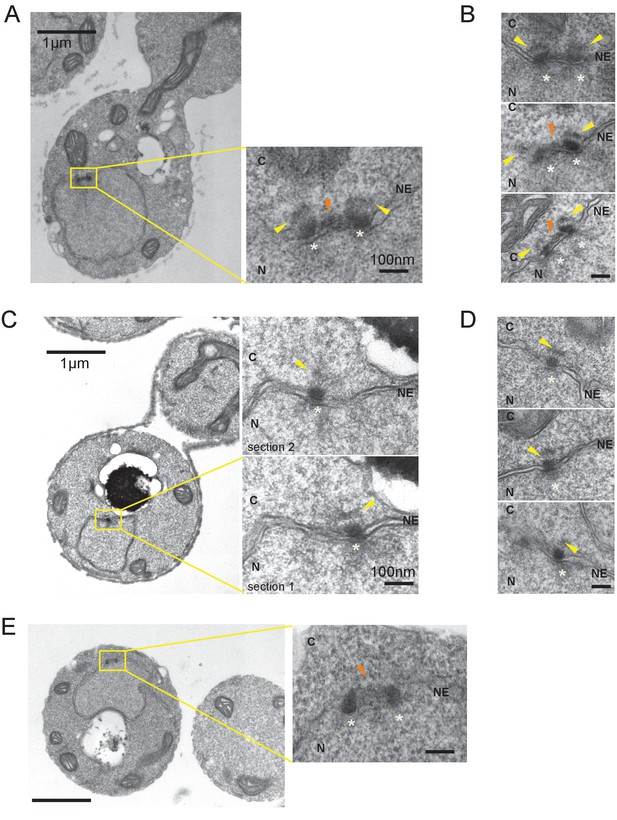
Side-by-side SPBs contains outer plaque.
Electron microscopy (EM) of thin serial sections of cells with side-by-side SPBs and nocodazole arrested cells.
Indicated are the cytoplasm (C), nucleus (N), and nuclear envelope (NE). Yellow arrowheads indicate outer plaques. SPB is marked with white star. Scale bars of the main images in (A) and (C) represent 1 µm. Scale bars of the insets in (A) and (C), i.e., (B) and (D), represent 100 nm. (A, B) Representative images of cells with side-by-side SPBs. Cdc28-as cells (HPH1150) grown in YPDS at 30°C were treated with 5 µM 1NM-PP1 for 110 min before prepared for EM. N = 6. (C, D) Representative images of SPBs in nocodazole arrested cells. Log phase cells of wild-type (BY4330) grown in YPDS at 30°C were treated with nocodazole for 2 hr until cells were arrested before prepared for EM. N = 6. (E) Image of one of side-by-side SPBs in (A, B) from top angle.
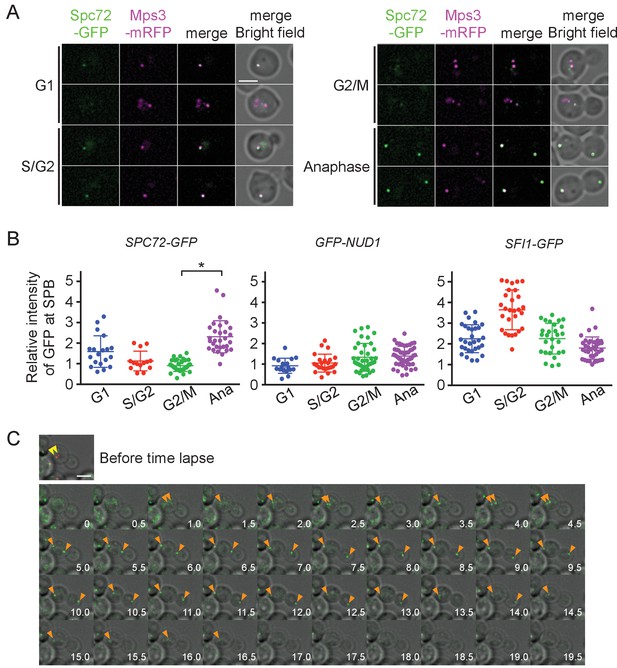
Accumulation of Spc72 at SPBs is cell cycle dependent.
(A) Cell cycle dependent localization of Spc72-GFP. SPC72-GFP MPS3-mRFP cells (HPH1394) were grown in SD complete medium at 30°C. Cell cycle stages are as shown in Figure 2B. Mps3-mRFP is a marker for SPB. Scale bar, 2 µm. (B) Quantification of the Spc72/Nud1/Sfi1 SPB signal of cells at different cell cycle stages. Yeast strains HPH972, HPH1396, and HPH1400 were used. Signal intensities were background-subtracted. Statistical significance of the difference between 2 SPBs and anaphase was determined by the student t-test and is indicated by an asterisk. Error bars indicate SD. n = 95, 127, and 124 for Spc72, Nud1, and Sfi1, respectively. Note that intensity of some of Sc72-GFP signals in G1 was high, because it decreased only gradually at SPB during the end of mitosis and the following G1 as shown in (C). (C) Time-lapse microscopy of SPC72-GFP MPS3-mRFP cells (HPH1394). Images were taken every 30 s. RFP signal was captured only before staring the time-lapse series. Anaphase onset judged by sudden spindle elongation was observed at the 5 min timepoint. Spindle orientation was corrected between 4.5 min and 5 min timepoints. Yellow arrowheads indicate the position of Mps3-mRFP before the image capture. Orange arrowheads indicate Spc72-GFP signals at SPBs. Shown are deconvolved and projected images. Scale bar, 2 µm. Another example is presented in Figure 4—figure supplement 1.
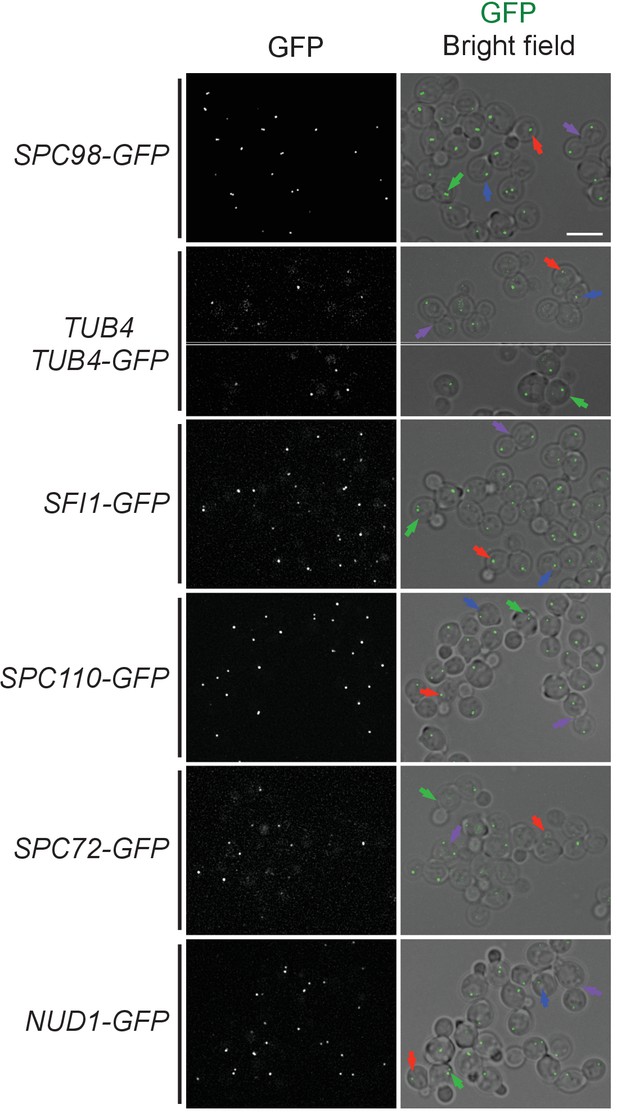
SPB localization of O. polymorpha orthologues of SPB components and γ-TuSC components.
Cells were logarithmically growing in YPDS expressing GFP-fusion proteins of Spc98, Tub4, Sfi1, Spc110, Spc72, and Nud1 orthologues (HPH475, HPH41, HPH1400, HPH1405, HPH1394, and HPH1396). Cells at different cell cycle stages are marked with arrows. G1, blue; 1SPB, red; 2SPBs, green; Anaphase, purple. Shown are deconvolved and projected images. Scale bar, 5 µm. GFP intensity is not scaled among the strains. Note that while all GFP-fusion proteins were sole source of these proteins, TUB4-GFP cells carried both untagged TUB4 gene at endogenous locus and PTUB4-TUB4-GFP gene at exogenous locus.
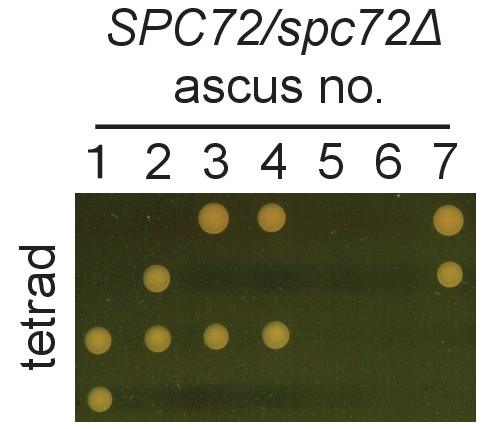
SPC72 of O. polymorpha is essential for growth.
Ascospores derived from heterozygous SPC72/spc72Δ diploid were subjected to tetrad dissection anlaysis. Dissected spores were incubated on YPDS plate at 30 for 3 days.

Time-lapse microscopy of SPC72-GFP cells.
Time-lapse microscopy of SPC72-GFP MPS3-mRFP cells (HPH1394) as in Figure 4C. Images were taken every 30 s. RFP signal was capture only before staring the time-lapse series. Anaphase onset judged by sudden spindle elongation was observed at the 6.5 min timepoint. Spindle orientation was corrected between 5.5 min and 6.5 min timepoints. Yellow arrowheads indicate the position of Mps3-mRFP before the image capture. Orange arrowheads indicate the first appearance of Spc72-GFP signals at SPBs. Shown are deconvolved and projected images. Scale bar, 2 µm.

Time-lapse microscopy of SPC72-GFP cells.
(A, B) Time-lapse microscopy of SPC72-GFP MPS3-mRFP cells (HPH1394) as in Figure 4C. Images were taken every 1 min. Images were captured only for GFP and bright field. Spc72-GFP signal at SPBs are indicated by yellow arrowheads. Spc72-GFP signals appeared in the neighbouring cell at later timepoints are indicated by orange arrowheads. in (B), weak GFP signal in the mother cell body persisted until the end of the time-lapse series. Shown are deconvolved, projected images of Spc72-GFP overlayed with bright field images. Scale bar, 2 µm.
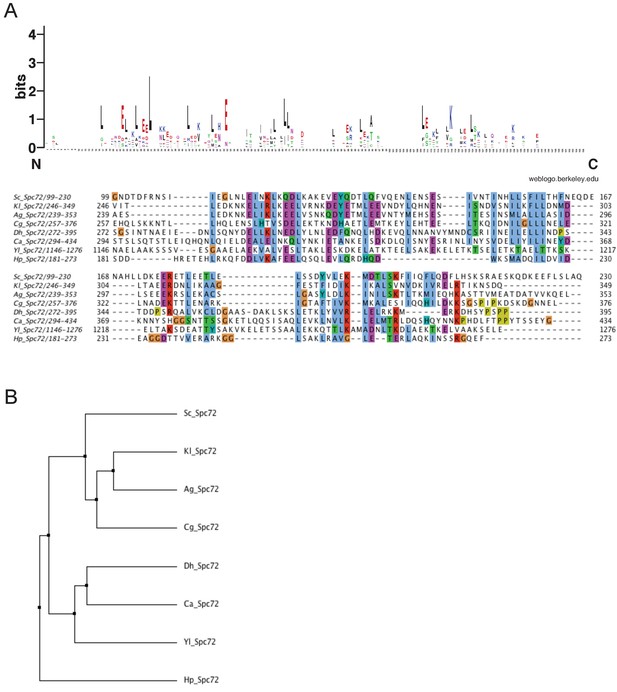
Stu2 binding domain of Spc72 is conserved among budding yeasts.
A Multiple sequence alignment of the Stu2 binding domain of Spc72 among budding yeasts. Amino acid sequences of the Stu2 binding domain of Spc72 in S. cerevisiae, K. lactis, Asibia gossippi, C. glabrata, D. hanseni, C. albicans, Y. lipolytica, and O. polymorpha (H. polymorpha) were aligned using the MAFFT algorism built into Jalview software. Residues are marked according to the ClustalX colour scheme. The occurrence of each amino acid in each position of the CM1 motif is presented with Weblogo 2.0. B Average distance tree of the Stu2 binding domain in (A) using PAM250 in Jalview software.
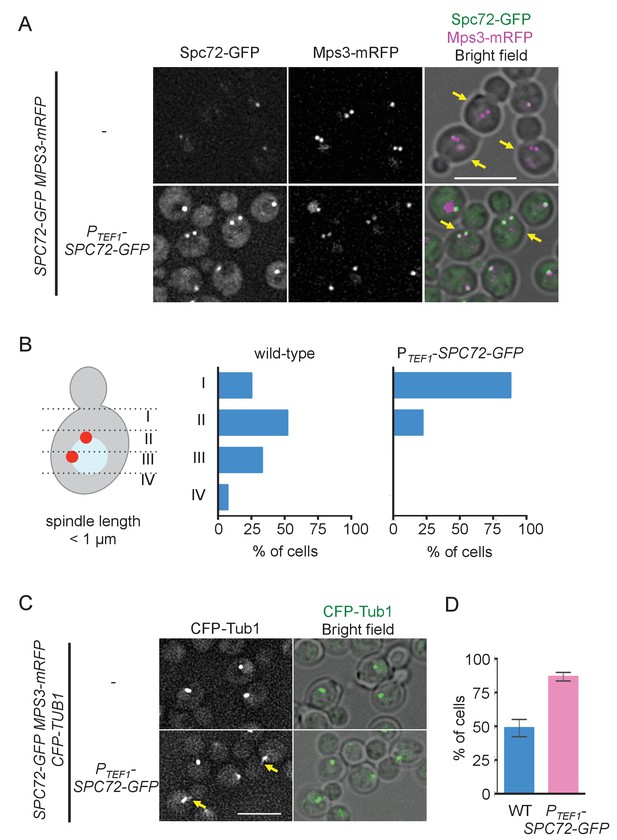
Overexpression of Spc72 converts the O. polymorpha type of nuclear position to the S. cerevisiae type.
(A) Overexpressed Spc72-GFP were recruited to SPB at all stages of the cell cycle. SPC72-GFP was expressed from a strong promoter of the TEF1 gene (HPH1393). Enrichment of Spc72 to SPB was evident in G1 cells and cells with short spindles (SPB distance <1 µm, yellow arrows) compared with images of wild-type cells (HPH1394). Mps3-mRFP marks SPB. Scale bar, 5 µm. (B) SPB is positioned close to the bud neck in G2/M cells carrying PTEF1-SPC72-GFP. The position of the SPB closer to the bud was as outlined in the cartoon shown on the left side of the subfigure. Strains used were HPH1393 (n = 110) and HPH1394 (n = 117). (C) Overexpression of Spc72 stimulated cMT acquisition. MTs were visualized with CFP-TUB1 in wild type and cells overexpressing SPC72-GFP (HPH1653 and HPH1652, respectively) were grown in YPDS medium at 30°C. Images were captured only for CFP and brightfield. Scale bar, 5 µm. (D) Quantification of (C). Presence/absence of cMTs was scored in cells with short spindle. Shown is the average of three independent experiments. Error bars indicate SD. n > 100. Average of three independent experiments.
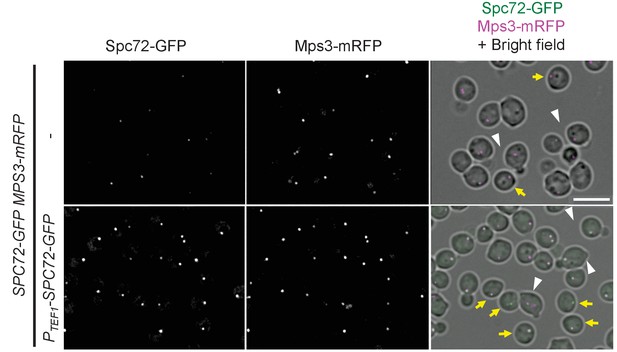
Spc72-GFP binds to SPBs in G1 and G1/S cells when overexpressed.
Additional images of Figure 5F. Unbudded G1 cells and G1/S cells are indicated by yellow arrows and white arrowheads respectively. Scale bar, 5 µm.
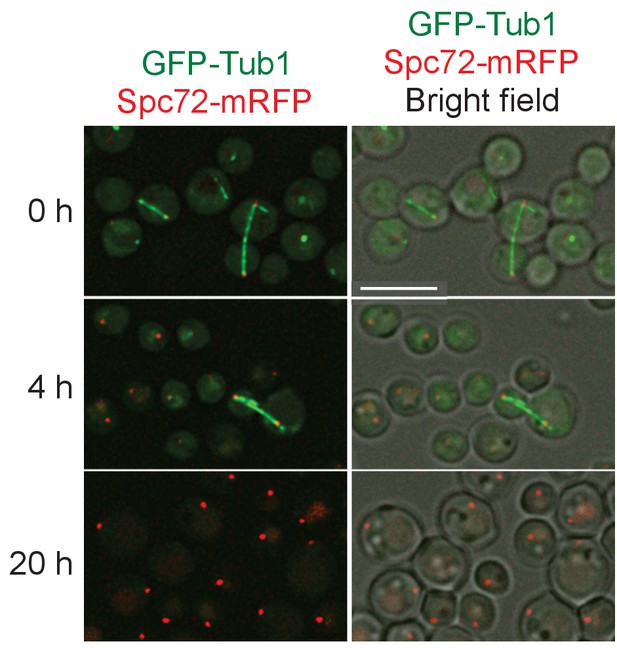
Spc72-mRFP accumulate at SPB in starvation conditions.
Wild-type cells carrying SPC72-mRFP GFP-TUB1 (HPH449) grown in YPDS medium were transferred to NaKG medium and incubated for the indicated time. Scale bar, 5 µm.
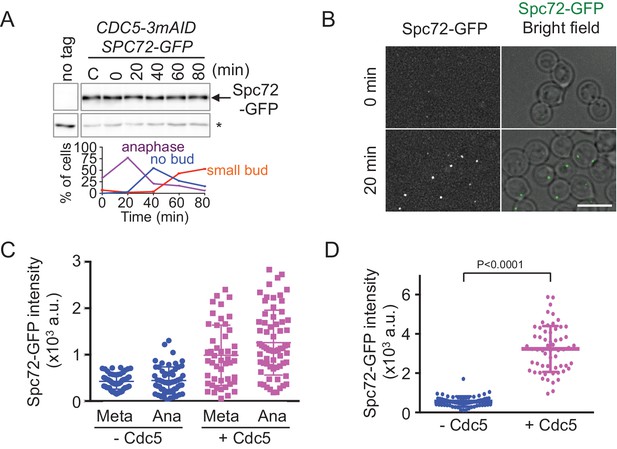
The recruitment of Spc72 to SPBs is dependent on the polo-like Cdc5 kinase.
(A) Abundance of Spc72 does not fluctuate during the cell cycle. Logarithmically growing SPC72-GFP CDC5-3mAID PCDC28-OsTIR cells (HPH1380) were synchronized with IAA followed by release. Samples were analysed by immunoblotting for Spc72-GFP. Comparable intensities of unspecific signal in immunoblotting (asterisk) indicate equal loading of samples. Budding index and mitotic index (DAPI) were determined over time. n > 100 cells per time point. (B) SPC72-GFP CDC5-3mAID PCDC28-OsTIR cells (HPH1380) were synchronized and released as in (A). Images were captured without fixation. Shown are deconvolved and projected images. Time after release is indicated on the left. Scale bar, 5 µm. (C) Quantification of Spc72-GFP intensity at SPBs in (B). Signal intensities were background-subtracted. Statistical significance of the difference between 2 SPBs and anaphase was determined by the student t-test. – Cdc5: before the release;+Cdc5: 20 min after the release. Error bars indicate SD. n > 50 cells per time point. (D) Accumulation of Spc72 at SPBs in metaphase depends on Cdc5 function. SPC72-GFP CDC5-3mAID PCDC28-OsTIR cells (HPH1380) were arrested with nocodazole in the presence (− Cdc5) or absence (+Cdc5) of IAA and Spc72-GFP signal at SPBs was quantified. Signal intensities were background-subtracted. Error bars indicate SD. n > 50 cells.
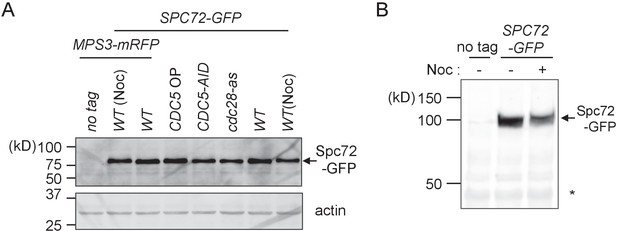
Spc72 protein level does not change at different cell cycle stages.
(A) Logarithmically growing wild type (HPH399, HPH1394), CDC5-3mAID (HPH1380), PTEF1-CDC5∆53 (HPH1542), cdc28-as (HPH1212) cells with SPC72-GFP as well as no-tag control cells (HPH448) were grown in YPDS medium. IAA and 1NM-PP1 was added to cdc28-as and CDC5-3mAID cells respectively and incubated for 90 min prior to cell harvest (Figure 3—figure supplement 1). Wild type strains were incubated in YPDS medium containing nocodazole for 2.5 hr. Total cell extracts were analysed in a polyacrylamide gel followed by immunoblotting using anti-GFP antibody or anti-actin antibody. (B) Logarithmically growing SPC72-GFP cells (HPH399) were incubated in YPDS medium containing nocodazole for 2 hr. Total cell extracts were analysed in a polyacrylamide gel followed by immunoblotting using anti-GFP antibody. Comparable intensities of unspecific signal in immunoblotting (asterisk) indicate equal loading of samples.
Localization of Spc72 to SPBs depends on Cdc5 activity.
(A) Bright field images of CDC5-3mAID cells incubated with or without IAA in (D). Most of cells were arrested with large bud, and no re-budding was observed in both cultures. Scale bar, 10 µm. (B) Images of cells in Figure 6D. Deconvolved and projected images are shown. Scale bar, 5 µm.
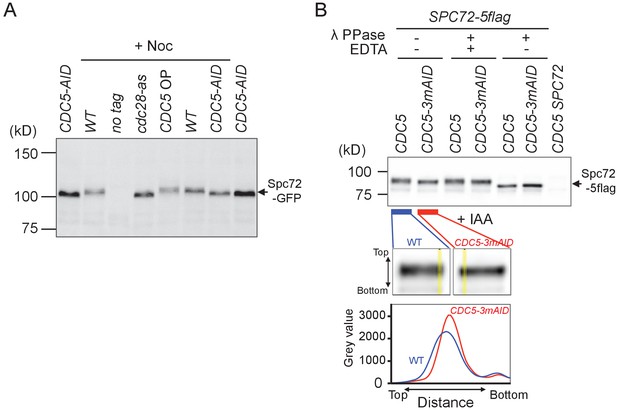
Spc72 phosphorylation is partly dependent on Cdc5.
(A) Logarithmically growing wild type (HPH1394), CDC5-3mAID (HPH1380), PTEF1-CDC5∆53 (HPH1542), cdc28-as (HPH1212) cells with SPC72-GFP as well as wild type control cells were arrested in YPDS containing nocodazole for 2.5 hr. After 1 hr incubation in nocodazole-containing medium, IAA or 1NM-PP1 was added and incubated for 90 min prior to cell harvest (Figure 3—figure supplement 1). Total cell extracts were analysed in a polyacrylamide gel followed by immunoblotting using anti-GFP antibody. Total cell extract prepared from CDC5-3mAID (HPH1380) cells treated with IAA was loaded on both ends of the gel as control. (B) Logarithmically growing SPC72-5flag CDC5-3mAID PCDC28-OsTIR (HPH1430) cells, SPC72-5flag PCDC28-OsTIR (HPH1436) cells, and wild-type cells (HPH466) were arrested in YPDS containing nocodazole for 2.5 hr. After growing 1.5 hr in nocodazole-containing medium, IAA was added and incubated for 60 min prior to cell harvest. Spc72-5flag was immunoprecipitated followed by treatment of 100 units of λ phosphatase in the presence or absence of 50 mM EDTA and then analysed in polyacrylamide gels followed by immunoblotting using anti-flag antibody. The flag tag was used for the efficiency of immunoprecipitation and lower unspecific signals in immunoblotting. Upper panel: immunoblotting to detect Spc72-5flag protein. Lower panel: linescan of yellow lines in the enlarged images of the upper panel. Broader intensity distribution on the line of wild type indicates the presence of slower migrating species in Spc72-5flag protein band compared with that of CDC5-AID.
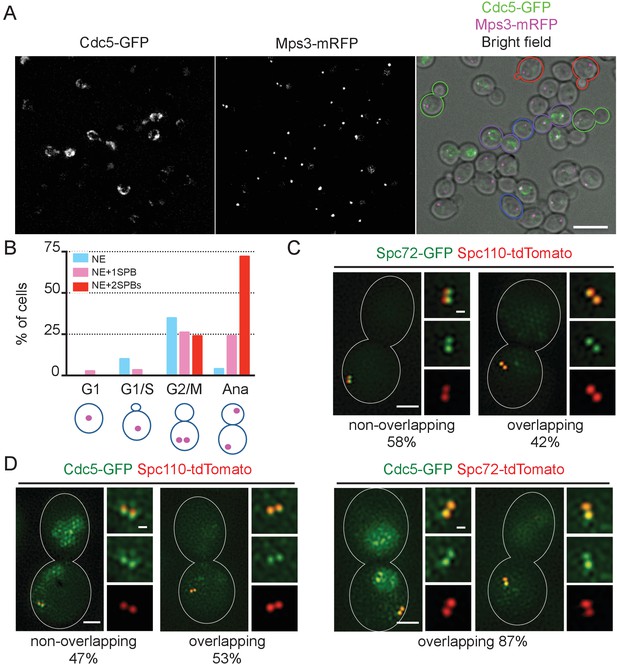
Localization of polo-like Cdc5 kinase in the nucleus, the nuclear envelope, and at the SPB is cell cycle dependent.
(A) CDC5-GFP MPS3-mRFP cells (HPH1562) were grown in YPDS at 30°C. Blue, red, green, and purple cell contours mark G1, S/G2, G2/M, and anaphase cells, respectively. Shown is a projected image after deconvolution. Scale bar, 5 µm. (B) Quantification of Cdc5-GFP localization in (A). The position of SPB (magenta) was as outlined in the cartoon shown at the bottom of the subfigure. n > 30 cells for each cell cycle stage. Ana, anaphase. (C) SIM images of Spc72-GFP and Spc110-tdTomato in nocodazole-arrested cells. SPC72-GFP SPC110-tdTomato cells (HPH1581) grown in SC medium at 30°C were arrested in metaphase with nocodazole. Scale bars, 1 µm and 0.2 µm in the large and small images, respectively. n = 78. (D) SIM images of Cdc5-GFP together with either Spc110-tdTomato or Spc72-tdTomato in nocodazole-arrested cells. CDC5-GFP SPC110-tdTomato and CDC5-GFP SPC72-tdTomato cells grown in SC medium at 30°C were arrested in metaphase with nocodazole. Scale bars, 1 µm and 0.2 µm in the large and small images, respectively. Strains used were HPH1583 (n = 75) and HPH1575 (n = 55). Diffused nuclear signal of Cdc5-GFP were also observed in all cells (Figure 7—figure supplement 2).
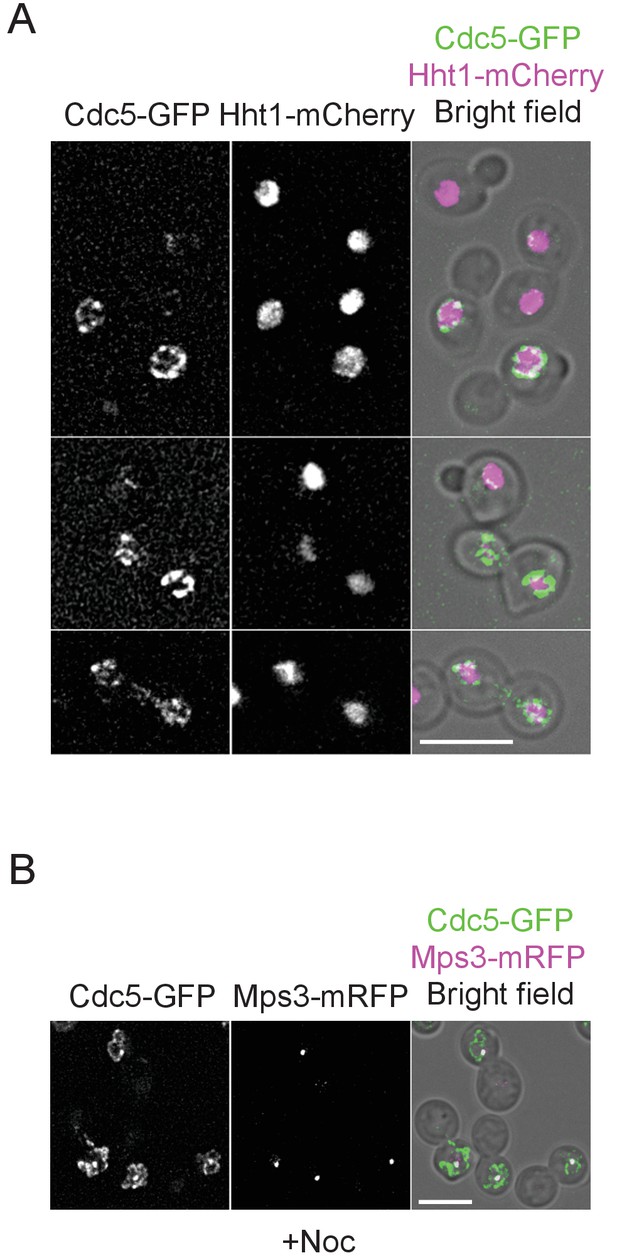
Localization of Cdc5-GFP.
(A) CDC5-GFP HHT1-mCherry cells (HPH1564) were grown in YPDS at 30°C. Shown are projected images after deconvolution. Scale bar, 5 µm. (B) Cdc5 localize at SPB in nocodazole-arrested cells. CDC5-GFP MPS3-mRFP cells (HPH1562) were grown in YPDS at 30°C and incubated in the presence of nocodazole for 150 min. Shown is a projected image after deconvolution. Scale bar, 5 µm.
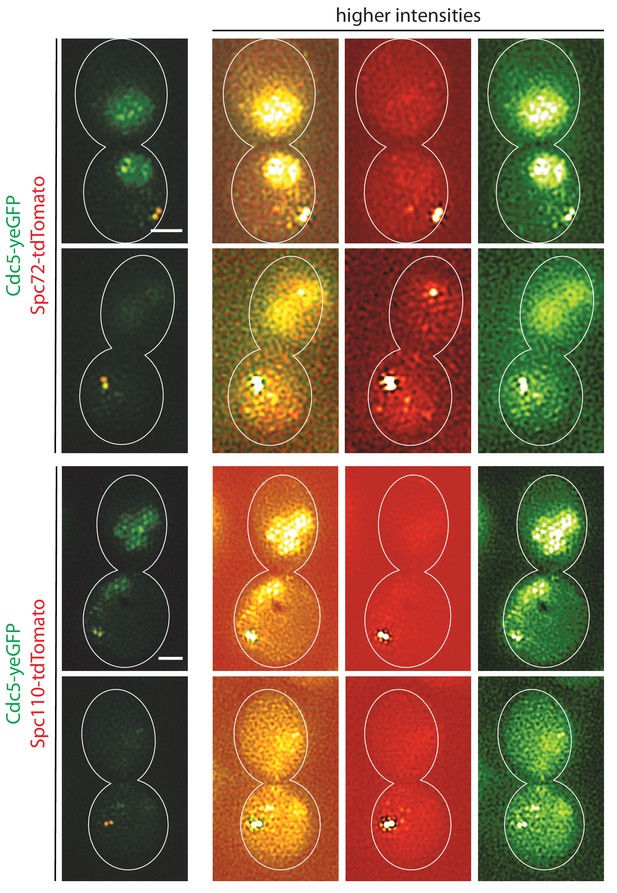
Localization of Cdc5-GFP.
SIM images of high intensities in Figure 7D. Nuclear GFP signals are easily visible. Scale bars, 1 µm.
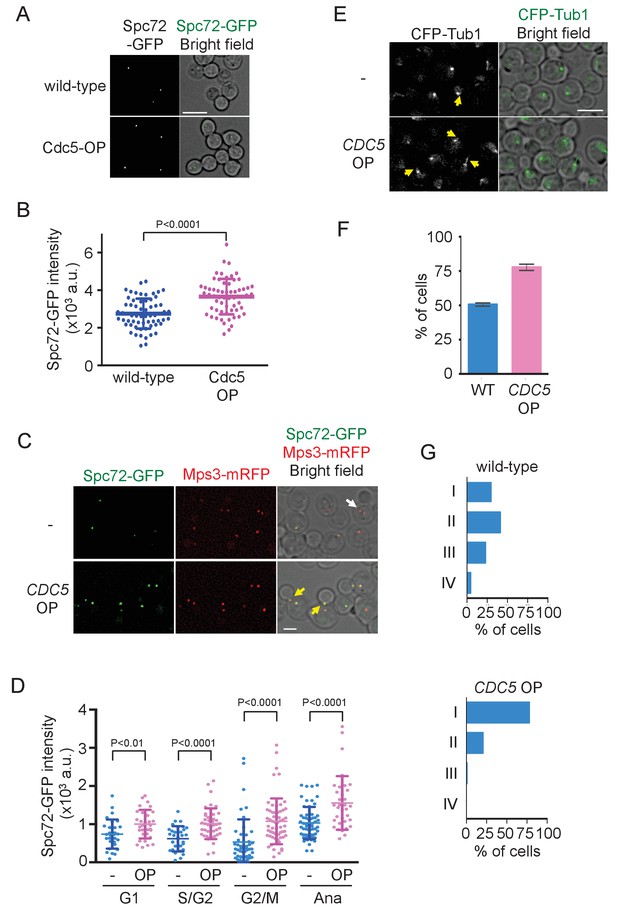
Overexpression of Cdc5 kinase promotes early association of Spc72 to SPBs.
(A) Overexpression of CDC5 enhances SPB binding of Spc72. SPC72-GFP MPS3-mRFP (HPH1394) and SPC72-GFP MPS3-mRFP PTEF1-CDC5∆53 cells (HPH1542) were arrested in metaphase with nocodazole. Deconvolved and projected images are shown. Scale bar, 5 µm. (B) Spc72-GFP signal at SPBs in (A) was quantified. Signal intensities were background-subtracted. Error bars indicate SD. n > 50 cells. (C) Spc72-GFP was recruited to SPB at earlier stages of the cell cycle when CDC5 gene is overexpressed. SPC72-GFP MPS3-mRFP (HPH1394, -) and SPC72-GFP MPS3-mRFP PTEF1-CDC5∆53 cells (HPH1542, CDC5 OP) were grown in YPDS medium. Enrichment of Spc72 to SPB and alignment of the spindle along the mother-bud axis were evident in preanaphase cells (yellow arrows) overexpressing CDC5, compared with wild-type cells (white arrow). Shown are deconvolved and projected images. Mps3-mRFP marks SPB. Scale bar, 2 µm. (D) Quantification of Spc72-GFP intensity at SPBs in (C). –: wild-type; OP: CDC5 overexpression. Signal intensities were background-subtracted. Statistical significances of the difference between wild-type (-) and CDC5 overexpressing cells (CDC5 OP) were determined by the student t-test. Error bars indicate SD. (E) Overexpression of Spc72 stimulated cMT acquisition. MTs were visualized with CFP-TUB1 in wild type and CDC5 overexpressing cells (HPH1680 and HPH1673, respectively) were grown in YPDS medium at 30°C. Images were captured only for CFP and brightfield. Scale bar, 5 µm. (F) Quantification of (E). Presence/absence of cMTs was scored in cells with short spindle. Shown is the average of three independent experiments. Error bars indicate SD. n > 100. (G) SPB is positioned close to the bud neck in G2/M cells overexpressing CDC5. The position of the SPB closer to the bud was as outlined in the cartoon shown in Figure 5B. Strains used were HPH1394 (n = 125) and HPH1542 (n = 101).
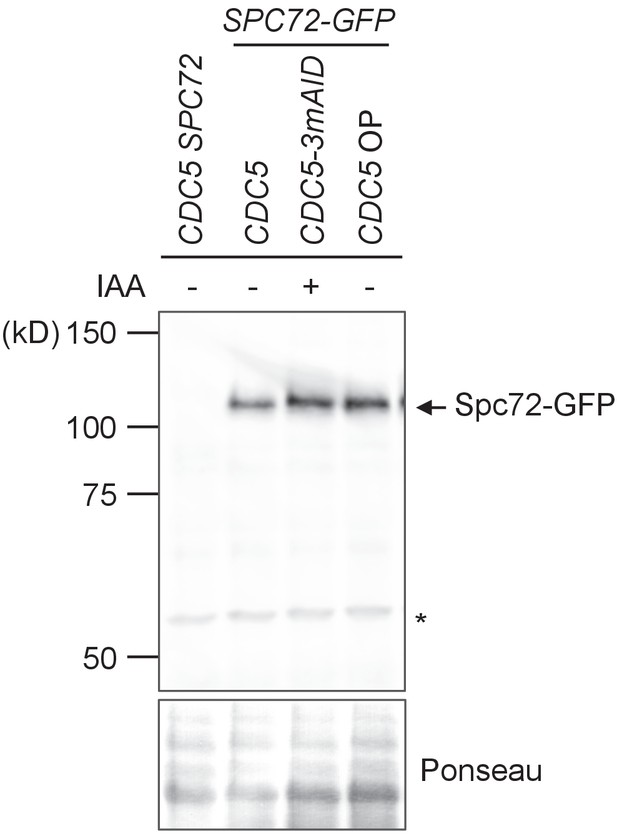
Spc72-GFP protein level was not affected by CDC5 overexpression.
Logarithmically growing wild type (HPH1394), CDC5-3mAID (HPH1380), PTEF1-CDC5∆53 (HPH1542) with SPC72-GFP as well as wild type control cells (HPH448) were grown in YPDS medium. IAA was added and incubated for 90 min prior to cell harvest of HPH1380. Total cell extracts were analysed in a polyacrylamide gel followed by immunoblotting using anti-GFP antibody.
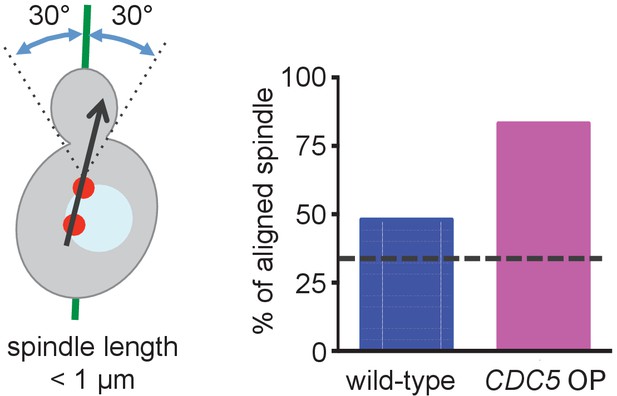
Preanaphase spindle was aligned along the mother-bud axis in cells overexpressing CDC5.
Quantification of the spindle orientation in Figure 8F. Spindle orientation was measured based on the line expanding two SPBs as outlined in the cartoon shown on the left side of the figure. The expected percentage from random orientation (33%) is indicated by the dashed line in the figure.
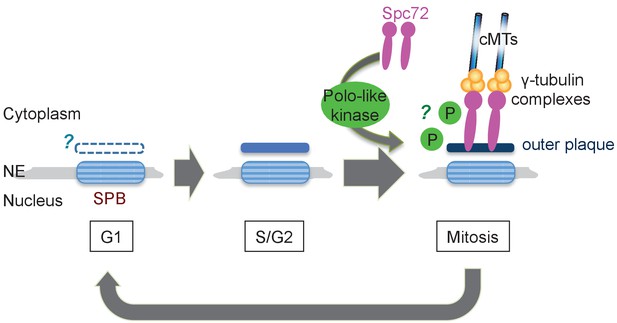
Model of the SPB cycle in O. polymorpha.
See Discussion for details.
Tables
Yeast strains and plasmids
https://doi.org/10.7554/eLife.24340.037| Name | Genotype/species/construction | Source or reference |
|---|---|---|
| O. polymorpha strains | ||
| BY4329 | leu1-1 | NBRP |
| BY21401 | CBS4732 Type strain | NBRP |
| HPH31 | HHT1::pHM713 ura3-1 | this study |
| HPH41 | ura3-1 pHM719 | this study |
| HPH164 | HHT1::pHM726 TUB1:: pHM737 leu1-1 | this study |
| HPH194 | TUB1::pHM737 leu1-1 | this study |
| HPH206 | Δdyn1::natNT2 leu1-1 | this study |
| HPH207 | Δdyn1::natNT2 leu1-1 | this study |
| HPH221 | wild type | this study |
| HPH222 | Δbub2::hphNT1 | this study |
| HPH223 | Δkar9::natNT2 | this study |
| HPH224 | Δkar9::natNT2 Δbub2::hphNT1 | this study |
| HPH225 | leu1-1 | this study |
| HPH399 | SPC72-GFP-hphNT1 | this study |
| HPH449 | SPC72-mRFP-hphNT1 TUB1:: pHM737 leu1-1 | this study |
| HPH466 | wild type | this study |
| HPH475 | hphNT1-PSPC98-GFP-SPC98 | this study |
| HPH972 | SPC72-GFP-hphNT1 | this study |
| HPH1150 | Δcdc28::natNT2::pHM878 ura3-1 | this study |
| HPH1210 | Δcdc28::natNT2::pHM878 TUB1:: pHM737 HHT1::pHM713 ura3-1 | this study |
| HPH1380 | SPC72-GFP-hphNT1 CDC5-3mAID-natNT2 ura3-1::pHM922 | this study |
| HPH1393 | SPC72-GFP-hphNT1 MPS3-mRFP-kanMX6 ura3-1::pHM859 | this study |
| HPH1394 | SPC72-GFP-hphNT1 MPS3-mRFP-kanMX6 | this study |
| HPH1396 | hphNT1-PNUD1-GFP-NUD1 | this study |
| HPH1400 | SFI1-GFP-hphNT1 | this study |
| HPH1405 | SPC110-GFP-hphNT1 | this study |
| HPH1430 | SPC72-5flag-hphNT1 CDC5-3mAID-natNT2 ura3-1::pHM922 | this study |
| HPH1542 | SPC72-GFP-hphNT1 MPS3-mRFP-kanMX6 TEF1::pHM950::pHM956 | this study |
| HPH1562 | CDC5-GFP-hphNT1 MPS3-mRFP-kanMX6 | this study |
| HPH1564 | CDC5-GFP-hphNT1 MPS3-mRFP-kanMX6 HHT1::pHM726 | this study |
| HPH1575 | CDC5-GFP-hphNT1 SPC72-tdTomato-hphMX | this study |
| HPH1581 | SPC72-tdTomato-hphMX SPC110-GFP-hphNT1 | this study |
| HPH1583 | CDC5-GFP-hphNT1 SPC110-tdTomato-natNT2 | this study |
| HPH1678 | MPS3-GFP-hphNT1 HHT1::pHM713 | this study |
| other yeast strains | ||
| BY21467 | S. cerevisiae YPH499 | NBRP |
| BY21165 | Kluyveromyces lactis NH27 | NBRP |
| BY21167 | Yarrowia lipolytica T22 | NBRP |
| BY23876 | Candida glabrata YAT3377 | NBRP |
| BY5243 | Ogataea parapolymorpha DL-1 | NBRP |
| JCM9829 | Candida peltata | JCM |
| JCM 10237 | Ogataea methanolica | JCM |
| JCM15019 | Ambrosiozyma kashinagacola | JCM |
| plasmids | ||
| pHM713 | pREMI-Z carrying HHT1-GFP and HHF1(histoneH4) | this study |
| pHM719 | pKS144 carrying TUB4-GFP | this study |
| pHM726 | pREMI-Z carrying HHT1-mCherry and HHF1(histoneH4) | this study |
| pHM737 | pRS305 carrying PTUB1-GFP-TUB1 | this study |
| pHM859 | pBSII carring HpURA3 and PTEF1-HpSPC72-GFP | this study |
| pHM878 | pBSII carring HpURA3 and cdc28-as | this study |
| pHM922 | pBSII carring HpURA3 and PCDC28-OsTIR | this study |
| pHM950 | pFA6a-natNT2 carrying PTEF1-HpCDC5Δ53 | this study |
| pHM956 | pFA6a-natNT2 carrying PTEF1-HpCDC5Δ53 and zeo | this study |
Additional files
-
Transparent reporting form
- https://doi.org/10.7554/eLife.24340.038





