Bladder-cancer-associated mutations in RXRA activate peroxisome proliferator-activated receptors to drive urothelial proliferation
Figures
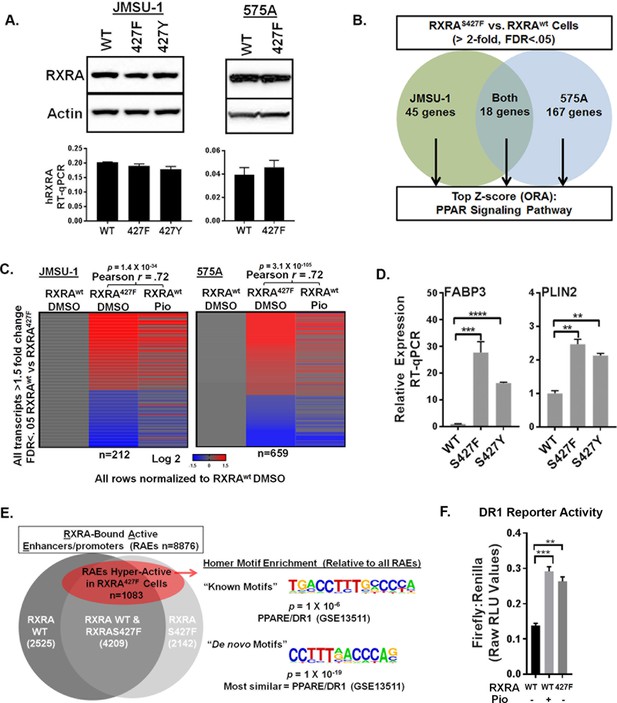
RXRA hot-spot mutations induce the PPAR signaling pathway by activating enhancer/promoters with a canonical PPAR response element.
(A) JMSU-1 and 575A cells were transduced with pBABE retrovirus to express indicated RXRA alleles and expression confirmed by western blot (top) or RT-qPCR in triplicate ± SD (data expressed as a fraction of actin signal). (B) Protein coding transcripts up-regulated greater than or equal to twofold (FDR < 0.05) in cells expressing RXRAS427F compared to cells expressing RXRAwt were identified and then subjected to over representation analysis (ORA, GO-Elite) to discover enriched pathways relative to all other protein coding transcripts identified by RNA-seq. Experiment was done in two bladder cancer cell lines, JMSU-1 or 575A, using three RNA samples, each purified from an independent cell well, for each condition. (See also source data 1). (C) Transcriptome changes induced by RXRAS427F relative to RXRAwt were compared to expression changes of the same transcripts induced by 16 hr of pioglitazone (1 μM) treatment in the RXRAwt expressing cells. D) Relative expression of two PPAR targets with expression of indicated RXRA alleles. RT-qPCR performed in triplicate ±SD. Comparison by Student’s t-test. (E) RAEs were defined by the presence of overlapping ChIP-seq signal for RXRA and H3K27ac. RAEs identified by binding of RXRAwt and/or RXRAS427F are represented in grey. Hyperactive RAEs represented in red had elevated H3K27ac mean peak height in the mutant expressing cells compared to the wild-type cells (FDR < 0.05). All ChIP-seq peak callings were based on data from three independent immuno-precipitations, each utilizing input material from an independent cell plate. HOMER motif analysis was used to identify motifs enriched in hyperactive RAEs relative to the background of non-hyperactive RAEs. Source data 2 specifies number of peaks in each sector of the venn diagram. (F) Activity of a DR1 response element reporter (3X PPRE) transfected into JMSU-1 cells stably expressing either RXRAwt or RXRAS427F. RXRAwt cells were also treated with pioglitazone (1 μM) for 16 hr. For all reporter assays, Firefly luciferase expressing reporter was co-transfected with a constitutive Renilla luciferase expression vector to normalize for transfection efficiency. Data represents mean ± SEM of Firefly to Renilla luciferase signal from three independent experiments done on different days, each performed using triplicate cell wells. Statistical comparisons are by paired t-test.
-
Figure 1—source data 1
GO Elite Over Representation Analysis complete results.
Pathways over-represented in genes up-regulated by RXRAS427Fcompared to RXRAwtin either JMSU-1 or 575A. Analysis was also done using genes up-regulated in both cell lines.
- https://doi.org/10.7554/eLife.30862.004
-
Figure 1—source data 2
ChIP-seq peak numbers used to generate venn diagram in Figure 1E.
- https://doi.org/10.7554/eLife.30862.005
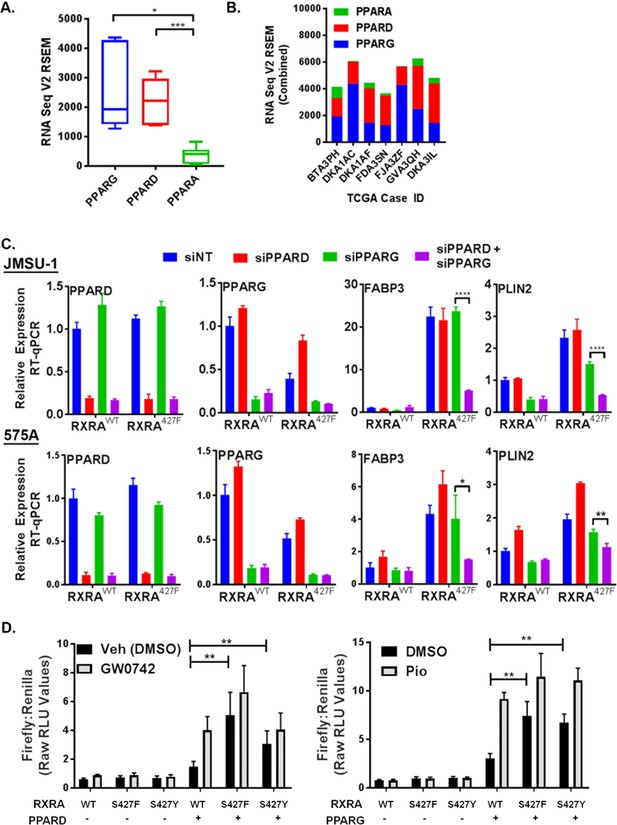
PPARG or PPARD expression is necessary and sufficient for mutant RXRA activity.
A) PPAR RNA expression in RXRA hot-spot mutant clinical samples from the TCGA dataset. Whisker plot shows 25th, median, and 75th percentile. (B) Data from panel A plotted per patient with hot-spot mutation. (C) Effects of siRNA-mediated knock-down of PPARD and PPARG in JMSU-1 and 575A cell lines on two target genes (PLIN2 and FABP3) up-regulated by mutant RXRA. Data by RT-qPCR in triplicate ±SD and indicated comparisons by Student’s t-test. (D) DR1 luciferase reporter activity in UM-UC-3 cells transfected with RXRA ±PPARD or PPARG. Cells were treated with 1 µM of the PPARG agonist pioglitazone or the PPARD agonist GW0742 for 16 hr. Data represents mean ±SEM of Firefly to Renilla luciferase signal from three independent experiments done on different days, each performed using triplicate cell wells. Statistical comparisons are by paired t-test.
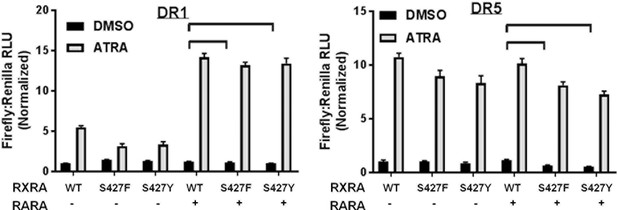
RARA expression is not sufficient for mutant RXRA hyperactivity.
(A) DR1 and DR5 luciferase reporter activity in UM-UC-3 cells transfected with RXRA ±RARA as indicated and treated with 100 nM all-trans-retinoic acid (ATRA) for 16 hr.
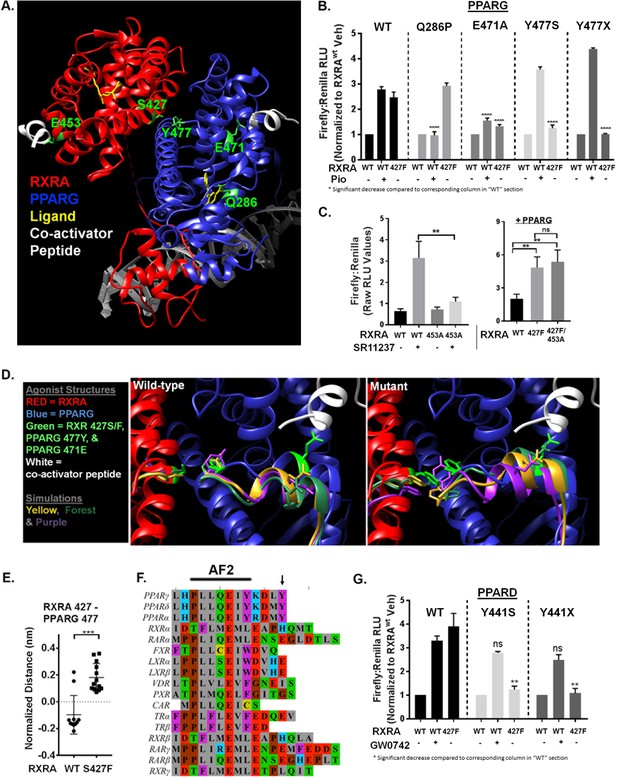
Mutant RXRA induces allosteric activation of PPARs through their terminal tyrosine.
A) RXRA S427 and other amino acids mutated for structure-function studies highlighted in green on a published full-length crystal structure of a RXRA/PPARG heterodimer. (B) DR1 reporter assay in UM-UC-3 co-transfecting indicated RXRA and PPARG alleles. Cells were treated with vehicle (DMSO) or pioglitazone 1 µM for 16 hr. Data represents mean normalized signal ±SEM of three independent experiments done on different days, each performed in triplicate, with data from each experiment normalized to the RXRAwt vehicle condition for each section. Statistical comparisons are by unpaired t-test. (C) Left, reporter assay performed with indicated RXRA alleles only and drug treatment with the RXRA agonist SR11237 (100 nM) for 16 hr. Right, reporter assay with wild-type PPARG co-transfected. Data represent mean ±SEM of Firefly to Renilla luciferase signal from three independent experiments done on different days, each performed using triplicate cell wells. Statistical comparisons are by paired t-test. (D) Published agonist structure of RXRA/PPARG heterodimer (PDB: 1FM6) in red and blue with key residues highlighted in bright green. Top three occupied microstate clusters from simulation experiments are superimposed. (E) Distance from starting agonist structure between alpha carbons of RXR 427 and PPARG 477 in the top 5% most-occupied microstates for wild-type and mutant RXRA. Mean ±SD, comparison is by Student’s t test. (F) Alignment of the AF2 region and C-terminus of all RXRA dimerization partners. Terminal tyrosine unique to PPARs is indicated. (G) Reporter assay similar to B, but using PPARD and the PPARD agonist GW0742.
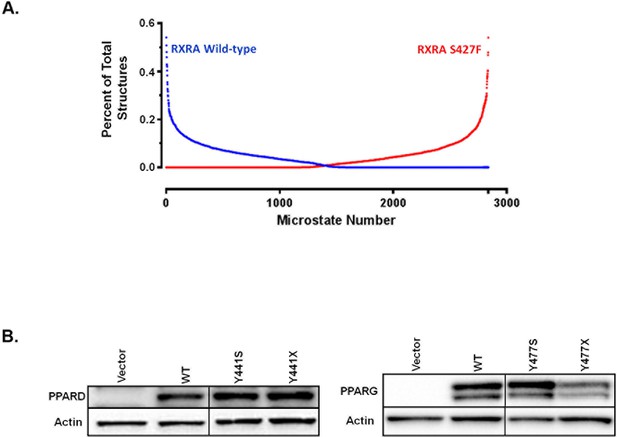
(A) Distribution of micro-state clusters occupied in RXRAwt and RXRAS427F simulations. (B) Western blot confirming expression of PPARD and PPARG with terminal tyrosine mutation or deletion in transfected UM-UC-3 cells. Irrelevant intervening lanes were removed where indicated with vertical bar without further image manipulation.
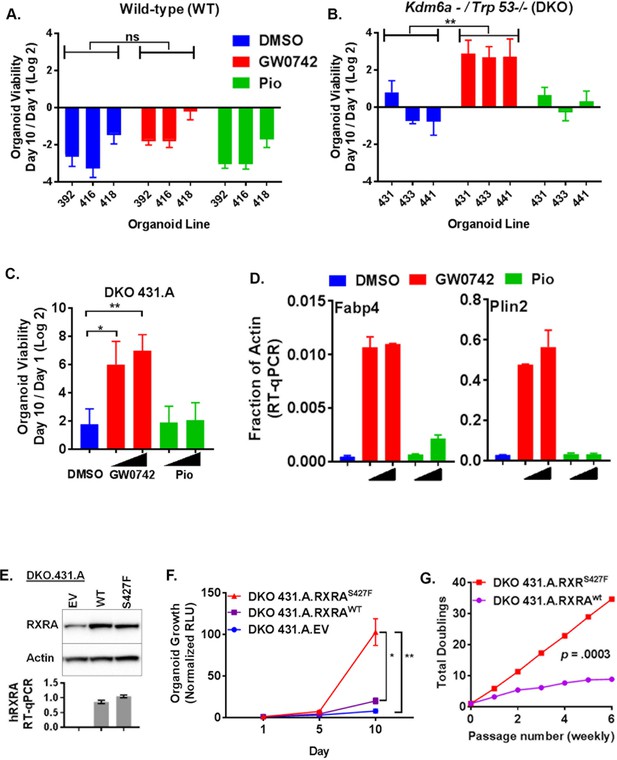
PPARD agonist and mutant RXRA confer growth-factor-independent growth to urothelium in the context of tumor suppressor loss.
(A) Organoids were derived from three independent wild-type mouse bladders and infected with Adeno-Cre. Growth was determined for each line in organoid media without EGF using CellTiter-Glo after treatment with vehicle, GW0742 (100 nM), or pioglitazone (100 nM). Data represent mean ±SEM from three independent experiments, each performed in triplicate cell wells. Statistical comparison is by unpaired t-test. (B) Similar to A but organoids were derived from Trp53flox/flox; Kdm6aflox mice. (C) CellTiter-Glo growth assay in media without EGF of a sub-clone from DKO431 (DKO 431.A) treated with GW0742 (10, 100 nM) or pioglitazone (100, 1000 nM). Mean values ± SEM from three independent experiments, each performed using triplicate organoid wells. Comparison is by paired t-test. (D) DKO 431.A organoids were plated in standard organoid media and then treated with, vehicle, GW0742 (10, 100 nM)) or pioglitazone (100, 1000 nM) for an additional 48 hr. Induction of PPAR target genes was determined by RT-qPCR in triplicate ± SD. (See also Figure 4—figure supplement 1C.) (E) DKO 431.A organoids were infected with a retroviral vector that was empty, expresses RXRAwt, or expresses RXRAS427F and expression of total RXRA (western blot) and human RXRA (RT-qPCR, triplicate +/SD) was determined. (F) Mean CellTiter-Glo signal ±SEM from three identical experiments, each performed using triplicate organoid wells. Comparison of D10 data is by paired t-test. (G) Identical number of indicated organoid cells were plated in media without EGF and then harvested with trypsin weekly and counted in duplicate using a BioRad TC20. Identical numbers of cells were then re-plated and this was repeated for 6 weeks. Total cell number doublings were calculated and plotted. Comparison of doublings is by paired t-test.
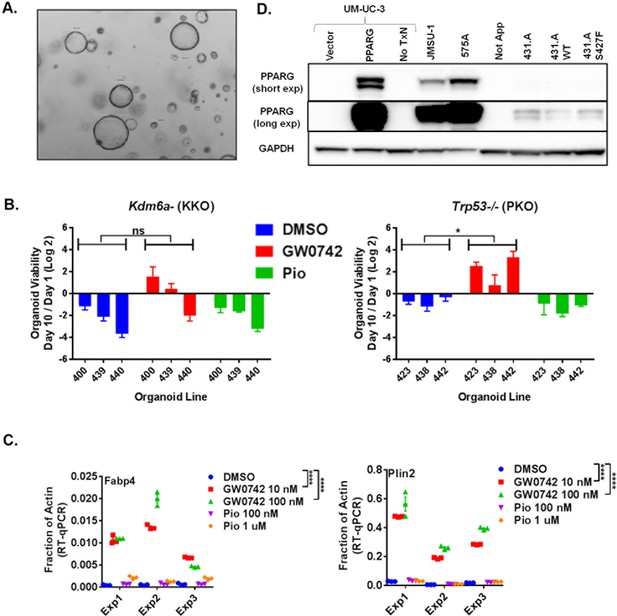
(A) Wild-type urothelial organoids grown for 3 weeks in standard organoid media and imaged with low-power (4X) phase contrast bright-field microscopy. (B) CellTiter-glo growth assay in media without EGF performed identically to those in Figure 4A and Figure 4B, but with organoids generated from mice with singly floxed Trp53 or Kdm6a. Statistical comparisons are by unpaired t test. (C) Aggregated RT-qPCR from three distinct experiments as described in Figure 4D with each data point from each experiment plotted with mean indicated. Statistical comparisons are by two-way ANOVA (Repeated Measures, GraphPad Prism). (D) PPARG western blot of indicated cell lines. PPARG transfected UM-UC-3 included as a positive control. Equivalent protein mass from whole cell lysates were loaded into each well.
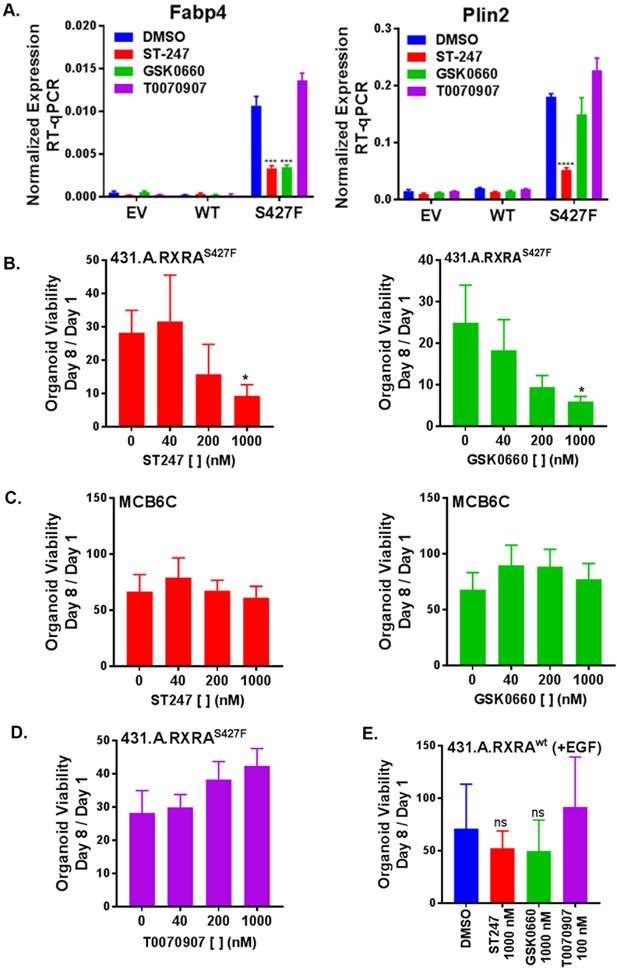
RXRA S427F generates PPARD-dependent urothelial growth.
(A) Retrovirally transduced organoids from Figure 4 were plated for 7 days in standard media and then treated with the indicated PPARD antagonists (1000 nM ST-247, GSK0660) or PPARG antagonist (100 nM T0070907) for 2 days. Expression of PPAR targets was determined by RT-qPCR in triplicate ±SD and comparison is by Student’s t-test to the RXRAS427F DMSO condition. (See also Figure 5—figure supplement 1)) (B-E) CellTiter-Glo growth assay of indicated organoid lines treated with indicated drugs. Plotted is mean signal ±SEM from three independent experiments, each performed using triplicate organoid wells. MCB6C is an organoid line we derived from a carcinogen-induced bladder tumor and lacks RXRA mutation. Organoids were cultured in media without EGF except for panel E where inclusion of EGF is indicated. Comparison is to DMSO condition using paired t-test.
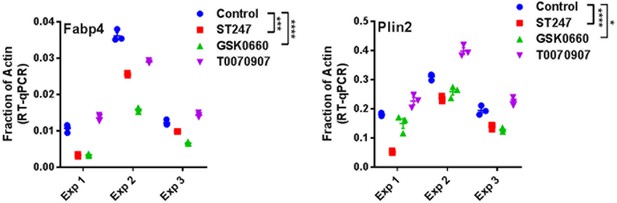
Aggregated RT-qPCR from three distinct experiments as described in Figure 5A with each data point from each experiment plotted with mean indicated.
Statistical comparisons are by two-way ANOVA (repeated measures, GraphPad Prism).
Tables
| Reagent type (species) or resource | Designation | Source or reference | Identifiers | Additional information |
|---|---|---|---|---|
| gene (Homo sapiens) | RXRA | NA | NCBI Gene ID:6256; NM_002957 | |
| gene (H. sapiens) | PPARG | NA | NCBI Gene ID:5468; NM_138711 | |
| gene (H. sapiens) | PPARD | NA | NCBI Gene ID:5467; NM_006238 | |
| strain, strain background (Mus musculus) | Kdm6aF | other | Generated by Dr. Lukas Wartman (Washington University School of Medicine) with ES cells obtained from EUCOMM with the Kdm6atm1a (EUCOMM)Wtsi allele (manuscript in preparation) | |
| strain, strain background (M. musculus) | Trp53Flox; B6.129P2- Trp53tm1brn/J | The Jackson Laboratory | The Jackson Laboratory:008462; RRID:IMSR_JAX:008462 | |
| genetic reagent | Ad5CMVCre-eGFP adenovirus | University of Iowa Viral Vector Core | VVC-U of Iowa:1174 | |
| cell line (M. musculus) | MCB6C | this paper | Clonal organoid line generated from tumor bearing bladder of male C57BL/6 mouse treated with BBN | |
| cell line (M. musculus) | DKO 431.A | this paper | clonal organoid line generated from the urothelium of a male Trp53Flox/Flox;Kdm6aFlox mouse | |
| cell line (M. musculus) | WT | this paper | Organoid lines generated from the urothelium of wild-type male mice resulting from cross between Trp53Flox/+ and Kdm6aFlox/+ mice | |
| cell line (M. musculus) | "Trp53-/-; Kdm6a-"; DKO | this paper | Organoid lines were generated from the urothelium of Trp53Flox/Flox; Kdm6aFlox male mice and then infected with Ad5CMVCre-eGFP adenovirus in vitro | |
| cell line (M. musculus) | "Kdm6a-"; KKO | this paper | Organoid lines were generated from the urothelium of Kdm6aFlox male mice and then infected with Ad5CMVCre-eGFP adenovirus in vitro | |
| cell line (M. musculus) | "Trp53-/-"; PKO | this paper | Organoid lines were generated from the urothelium of Trp53Flox/Flox male mice and then infected with Ad5CMVCre-eGFP adenovirus in vitro | |
| cell line (M. musculus) | DKO 431.A.EV | this paper | DKO 431.A organoid line infected with retrovirus carrying pBABE puro empty vector | |
| cell line (M. musculus) | DKO 431. A.RXRAwt | this paper | DKO 431.A organoid line infected with retrovirus carrying pBABE puro RXRA | |
| cell line (M. musculus) | DKO 431.A.RXRAS427F | this paper | DKO 431.A organoid line infected with retrovirus carrying pBABE puro RXRA S427F | |
| cell line (H. sapiens) | JMSU-1 | other | RRID:CVCL_2081 | obtained from Dr. David Solit (MSKCC) |
| cell line (H. sapiens) | 575A | other | RRID:CVCL_7941 | obtained from Dr. David Solit (MSKCC) |
| cell line (H. sapiens) | UM-UC-3 | other | RRID:CVCL_1783 | obtained from Dr. David Solit (MSKCC) |
| cell line (H. sapiens) | Lenti-X 293T | Clontech | Clontech:632180 | |
| cell line (H. sapiens) | JMSU-1 RXRA WT | this paper | JMSU-1 cell line infected with retrovirus carrying pBABE puro RXRA | |
| cell line (H. sapiens) | JMSU-1 RXRA S427F | this paper | JMSU-1 cell line infected with retrovirus carrying pBABE puro RXRA S427F | |
| cell line (H. sapiens) | JMSU-1 RXRA S427Y | this paper | JMSU-1 cell line infected with retrovirus carrying pBABE puro RXRA S427Y | |
| cell line (H. sapiens) | 575A RXRA WT | this paper | 575A cell line infected with retrovirus carrying pBABE puro RXRA | |
| cell line (H. sapiens) | 575A RXRA S427F | this paper | 575A cell line infected with retrovirus carrying pBABE puro RXRA S427F | |
| antibody | anti-PPARG (81B8) (rabbit monoclonal) | Cell Signaling Technology | Cell Signaling Technology:2443; RRID:AB_823598 | (1:1000) |
| antibody | anti-PPARD (rabbit monoclonal) | Abcam | Abcam:ab178866 | (1:5000) |
| antibody | anti-RXRA (D6H10) (rabbit monoclonal) | Cell Signaling Technology | Cell Signaling Technology:3085 | (1:1200) |
| antibody | anti-beta-Actin (mouse monoclonal) | Sigma-Aldrich | Sigma-Aldrich:A5441; RRID:AB_476744 | (1:50000) |
| antibody | anti-GAPDH (D16H11) (rabbit monoclonal) | Cell Signaling Technology | Cell Signaling Technology:5174; RRID:AB_10622025 | (1:1000) |
| antibody | anti-rabbit IgG, HRP (goat) | Cell Signaling Technology | Cell Signaling Technology:7074; RRID:AB_2099233 | (1:7500) |
| antibody | anti-mouse IgG, HRP (horse) | Cell Signaling Technology | Cell Signaling Technology:7076; RRID:AB_330924 | (1:7500) |
| antibody | anti-RXRA (K8508) (mouse monoclonal) | R&D Systems | R&D Systems: PP-K8508-00; RRID:AB_2182738 | (5 µg) |
| antibody | anti-H3K27Ac (rabbit polyclonal) | Abcam | Abcam:ab4729; RRID:AB_2118291 | (0.4 µg) |
| recombinant DNA reagent | PPRE X3-TK-luc; DR1 reporter (plasmid) | Addgene; PMID 9539737 | Addgene:1015 | plasmid was deposited by Bruce Spiegelman |
| recombinant DNA reagent | pGL3-RARE-luciferase; DR5 reporter (plasmid) | Addgene; PMID 16818722 | Addgene:13458 | plasmid was deposited by T. Michael Underhill |
| recombinant DNA reagent | pRL-SV40 (plasmid) | Promega | Promega:E2231 | |
| recombinant DNA reagent | pCL-ampho (plasmid) | other | obtained from Dr. Charles Sawyers (MSKCC) | |
| recombinant DNA reagent | VSVG (plasmid) | other | obtained from Dr. Charles Sawyers (MSKCC) | |
| recombinant DNA reagent | pCMV6-XL4 RARA (plasmid) | OriGene | OriGene:SC119566 | |
| recombinant DNA reagent | pCMV6-XL4 PPARG (plasmid) | OriGene | OriGene:SC108192 | |
| recombinant DNA reagent | pCMV6-XL4 PPARG Q286P (plasmid) | this paper | Q286 was mutated via site-directed mutagenesis of pCMV6-XL4 PPARG | |
| recombinant DNA reagent | pCMV6-XL4 PPARG E471A (plasmid) | this paper | E471 was mutated via site-directed mutagenesis of pCMV6-XL4 PPARG | |
| recombinant DNA reagent | pCMV6-XL4 PPARG Y477S (plasmid) | this paper | Y477 was mutated via site-directed mutagenesis of pCMV6-XL4 PPARG | |
| recombinant DNA reagent | pCMV6-XL4 PPARG Y477X (plasmid) | this paper | Y477 was deleted via site-directed mutagenesis of pCMV6-XL4 PPARG | |
| recombinant DNA reagent | pCMV6-XL4 empty vector (plasmid) | this paper | generated by digesting pCMV6-XL4 PPARG with NotI to remove PPARG and by ligating the plasmid ends with T4 DNA ligase | |
| recombinant DNA reagent | pBABE puro RXRA (plasmid) | Addgene | Addgene:11441 | deposited by Ronald Kahn |
| recombinant DNA reagent | pBABE puro RXRA S427F (plasmid) | this paper | S427 was mutated via site-directed mutagenesis of pBABE puro empty vector | |
| recombinant DNA reagent | pBABE puro RXRA S427Y (plasmid) | this paper | S427 was mutated via site-directed mutagenesis of pBABE puro empty vector | |
| recombinant DNA reagent | pBABE puro RXRA E453A (plasmid) | this paper | E453 was mutated via site-directed mutagenesis of pBABE puro empty vector | |
| recombinant DNA reagent | pBABE puro RXRA S427F/E453A (plasmid) | this paper | E453 was mutated via site-directed mutagenesis of pBABE puro RXRA S427F | |
| recombinant DNA reagent | pBABE puro empty vector (plasmid) | this paper | generated by digesting pBABE puro RXRA with EcoRI to remove RXRA and by ligating the plasmid ends with T4 DNA ligase | |
| recombinant DNA reagent | pCMV6-XL4 PPARD (plasmid) | this paper | Human PPARD was cloned from JMSU-1 epithelial bladder cancer cells and inserted into the pCMV6-XL4 | |
| recombinant DNA reagent | pCMV6-XL4 PPARD Y441S (plasmid) | this paper | Y441 was mutated via site-directed mutagenesis of pCMV6-XL4 PPARD | |
| recombinant DNA reagent | pCMV6-XL4 PPARD Y441X (plasmid) | this paper | Y441 was deleted via site-directed mutagenesis of pCMV6-XL4 PPARD | |
| sequence-based reagent | ON-TARGETplus Non-targeting Pool (siRNA) | Dharmacon | Dharmacon: D-001810-10-20 | |
| sequence-based reagent | ON-TARGETplus Human PPARG siRNA | Dharmacon | Dharmacon: L-003436-00-0005 | |
| sequence-based reagent | ON-TARGETplus Human PPARD siRNA | Dharmacon | Dharmacon: L-003435-00-0005 | |
| commercial assay or kit | Dual-Glo Luciferase Assay System | Promega | Promega:E2940 | |
| commercial assay or kit | CellTiter-Glo | Promega | Promega:G7571 | |
| commercial assay or kit | Ovation Ultraflow System V2 | NuGen | NuGen:0344-32 | |
| chemical compound, drug | Pioglitazone | Sigma-Aldrich | Sigma-Aldrich:E6910 | |
| chemical compound, drug | GW 0742 | Tocris | Tocris:2229 | |
| chemical compound, drug | SR 11237 | Tocris | Tocris:3411 | |
| chemical compound, drug | all-trans-Retinoic Acid (ATRA) | Sigma-Aldrich | Sigma-Aldrich:R2625 | |
| chemical compound, drug | GSK 0660 | Tocris | Tocris:3433 | |
| chemical compound, drug | ST247 | Sigma-Aldrich | Sigma-Aldrich:SML0424 | |
| chemical compound, drug | T0070907 | Cayman Chemical | Cayman Chemical:10026 | |
| software, algorithm | GROMACS 5.1.3 | DOI: 10.1016/j.softx. 2015.06.001 | RRID:SCR_014565 | |
| software, algorithm | MSMBuilder 2.8 | PMID: 22125474 | ||
| software, algorithm | Chimera | PMID: 15264254; http://www.rbvi.ucsf.edu/chimera | RRID:SCR_004097 | |
| sequence-based reagent | mKDM6A Forward (primer) | this paper | 5' CGAGAAAGGAAATGTG AGAGCAAGG 3' | |
| sequence-based reagent | mKDM6A Reverse 4 (primer) | this paper | 5' CTGGCAGGATATGATA GCAATGTG 3' | |
| sequence-based reagent | oIMR8543 (primer) | The Jackson Laboratory; https://www2.jax.org/protocolsdb/f?p=116:2:0::NO:2:P2_MASTER_PROTOCOL_ID,P2_JRS_CODE:3226,008462 | 5' GGTTAAACCCAGCT TGACCA 3' | |
| sequence-based reagent | oIMR8544 (primer) | The Jackson Laboratory; https://www2.jax.org/protocolsdb/f?p=116:2:0::NO:2:P2_MASTER_PROTOCOL_ID,P2_JRS_CODE:3226,008462 | 5' GGAGGCAGAGACA GTTGGAG 3' | |
| sequence-based reagent | PPARD.qPCR.Fwd.1 (primer) | this paper | 5' ATGCACCAACGA GGCTGATG 3' | |
| sequence-based reagent | PPARD.qPCR.Rev.1 (primer) | this paper | 5' CTGCTCCATGGCT GATCTCC 3' | |
| sequence-based reagent | PPARG fwd1 (primer) | this paper | 5' ATGCCTTGCAGT GGGGATGTC 3' | |
| sequence-based reagent | PPARG rev1 (primer) | this paper | 5' GAGGTCAGCGGA CTCTGGATTC 3' | |
| sequence-based reagent | hPLIN2 fwd1 (primer) | this paper | 5' AGTGCTCTGCCC ATCATCCAG 3' | |
| sequence-based reagent | hPLIN2 rev1 (primer) | this paper | 5' TCACAGCGCCTT TGGCAT TG 3' | |
| sequence-based reagent | FABP4 fwd1 (primer) | this paper | 5' ACTGCAGCTTCCT TCTCACCTTG 3' | |
| sequence-based reagent | FABP4 rev1 (primer) | this paper | 5' TGCCAGCCACTT TCCTGGTG 3' | |
| sequence-based reagent | mPlin2 Fwd1 (primer) | this paper | 5' GTGCCCTGCCC ATCATCC 3' | |
| sequence-based reagent | mPlin2 Rev1 (primer) | this paper | 5' TTACGGCACCTCT GGCACTG 3' | |
| sequence-based reagent | mFabp4 Fwd1 (primer) | this paper | 5' TGCAGCCTTTCTCA CCTGGAAG 3' | |
| sequence-based reagent | mFabp4 Rev1 (primer) | this paper | 5' GCCTGCCACTTTCC TTGTGG 3' | |
| sequence-based reagent | RXRA fwd1 (primer) | this paper | 5' ACAAGACGGAGC TGGGCTG 3' | |
| sequence-based reagent | RXRA rev2 (primer) | this paper | 5' GGCTGCTCTGGGT ACTTGTGC 3' | |
| sequence-based reagent | RXRA E453A SDM For (primer) | this paper | 5' acaccttccttatggccat gctggaggcgccg 3' | |
| sequence-based reagent | RXRA E453A SDM Rev (primer) | this paper | 5' cggcgcctccagcatggcc ataaggaaggtgt 3' | |
| sequence-based reagent | RXRa S427F-F (primer) | this paper | 5' CCG GCT CTG CGC TTT ATC GGG CTC AAA T 3' | |
| sequence-based reagent | RXRa S427F-R (primer) | this paper | 5' CAT TTG AGC CCG ATA AAG CGC AGA GCC G 3' | |
| sequence-based reagent | RXRa S427Y-F (primer) | this paper | 5' CCG GCT CTG CGC TAT ATC GGG CTC AAA T 3' | |
| sequence-based reagent | RXRa S427Y-R (primer) | this paper | 5' CAT TTG AGC CCG ATA TAG CGC AGA GCC G 3' | |
| sequence-based reagent | F hPPARGQ286P (primer) | this paper | 5' ccacggagcgaaacgg gcagccctgaaag 3' | |
| sequence-based reagent | R hPPARGQ286P (primer) | this paper | 5' ctttcagggctgcccgttt cgctccgtgg 3' | |
| sequence-based reagent | F hPPARGE471A (primer) | this paper | 5' agtccttgtagatcgcctg caggagcggg 3' | |
| sequence-based reagent | R hPPARGE471A (primer) | this paper | 5' cccgctcctgcaggcgat ctacaaggact 3' | |
| sequence-based reagent | PPARG Y477S For SDM (primer) | this paper | 5' GAGATCTACAAGGACTTGAG CTAGCAGAGAGTCCTGAGC 3' | |
| sequence-based reagent | PPARG Y477S Rev SDM (primer) | this paper | 5' GCTCAGGACTCTCTGCTAGCT CAAGTCCTTGTAGATCTC 3' | |
| sequence-based reagent | PPARG Y477X For SDM (primer) | this paper | 5' GATCTACAAGGACTTGTAG TAGCAGAGAGTCCTGA 3' | |
| sequence-based reagent | PPARG Y477X Rev SDM (primer) | this paper | 5' TCAGGACTCTCTGCTACTAC AAGTCCTTGTAGATC 3' | |
| sequence-based reagent | PPARD Y441S For (primer) | this paper | 5' AGATCTACAAGGACATGAG CTAACGGCGGCACCCAG 3' | |
| sequence-based reagent | PPARD Y441S Rev (primer) | this paper | 5' CTGGGTGCCGCCGTTA GCTCATGTCCTTGTAGATCT 3' | |
| sequence-based reagent | PPARD Y441X For (primer) | this paper | 5' GATCTACAAGGACATGTGA TAACGGCGGCACCCAGG 3' | |
| sequence-based reagent | PPARD Y441X Rev (primer) | this paper | 5' CCTGGGTGCCGCCGTTATC ACATGTCCTTGTAGATC 3' |
Additional files
-
Transparent reporting form
- https://doi.org/10.7554/eLife.30862.014





