Measuring ligand efficacy at the mu-opioid receptor using a conformational biosensor
Figures
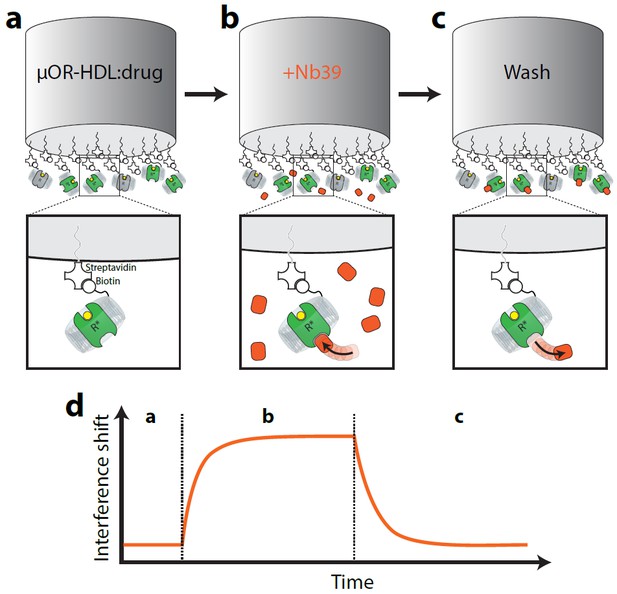
Cartoon schematic of interferometry assay to detect µ-OR:Nb39 interactions.
Briefly, (a) Biotin-conjugated rHDL particles containing µ-OR are loaded on streptavidin-coated tips and incubated with saturating ligand (or vehicle) for 10 min. (b) Probe is exposed to Nb39 for five min in the presence of ligand or vehicle until steady state is reached. (c) In the presence of ligand, probe is moved to well containing no Nb39 to monitor dissociation for 5 min.
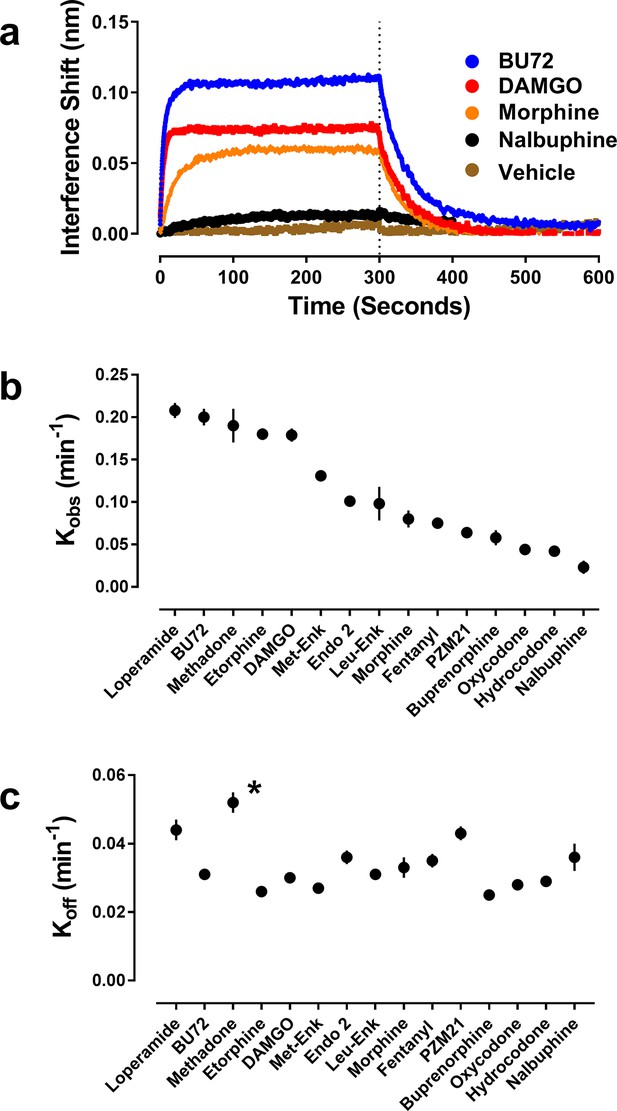
Orthosteric ligand-mediated Nb39 association and dissociation in µ-OR-rHDL.
As described in the Materials and methods, the association and dissociation of Nb39 (1 µM) was measured using an OctetRed® instrument. Shown is a representative experiment comparing four orthosteric agonists at 30 µM (a) Using GraphPad Prism 6.02, one-phase association lines were fit and the calculated kobs (b) and koff (c) for each ligand are plotted (±s.e.m.). Rate constants are means of multiple independent experiments as listed in see Table 1. *Dissociation of Nb39 from the methadone-bound receptor is statistically different from all other ligands (one-way ANOVA with Tukey’s post-hoc test).
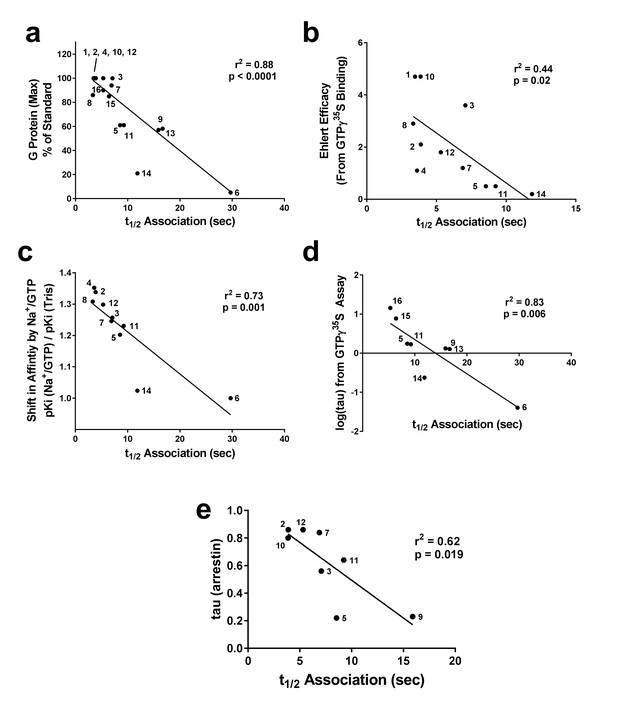
Correlation of association times of Nb39 with various measures of agonist efficacy.
The t1/2 of association of Nb39 in the presence of saturating agonist was measured and is plotted against (a) maximal stimulation of GTPγ35S binding by agonist, (b) the calculated Ehlert efficacy (Ehlert, 1985) values for each agonist to activate G protein, (c) the shift in affinity of the agonist as measured by radioligand competition binding in the absence or presence of Na+/Guanine nucleotide, and (d) the τ value using data from GTPγ35S assays (Livingston and Traynor, 2014) or (e) β-arrestin recruitment assays; McPherson et al., 2010), analyzed with the Black-Leff operational model (Black and Leff, 1983). Data used to compile correlation graphs is listed in the source data table. The ligands are: (1) BU72, (2) DAMGO, (3) Leu-Enk, (4) L-methadone, (5) Morphine, (6) Nalbuphine, (7) Endomorphin 2, (8) Loperamide, (9) Oxycodone, (10) Etorphine, (11) Fentanyl, (12) Met-Enk, (13) Hydrocodone, (14) Buprenorphine, (15) Morphine + BMS-986122, (16) Morphine + BMS-986187. Correlation analysis was performed using GraphPad Prism 6.02.
-
Figure 3—source data 1
List of values used to construct correlation graphs.
Ligand number refers to the list in legend to Figure 3; the t1/2 for association of NB39 is from Table 1; Maximum stimulation of [35S]GTPγS binding by each ligand is taken from Livingston and Traynor (2014); Ehlert’s efficacy value (e, Ehlert, 1985) determined for each ligand using [35S]GTPγS assay data and ligand affinities, taken from Livingston and Traynor (2014); Shift in ligand affinity in the presence of 100 mM NaCl and 10 μM GTPγS, from Livingston and Traynor (2014); Efficacy (τ) values for stimulation of [35S]GTPγS binding (Livingston and Traynor, 2014) calculated according to Black and Leff (1983); Efficacy (τ) values for arrestin recruitment taken from McPherson et al. (2010).
- https://doi.org/10.7554/eLife.32499.006
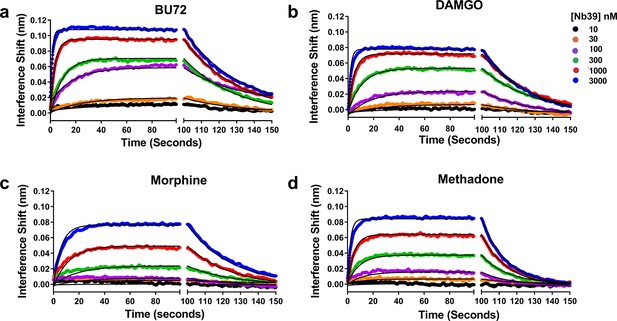
Association and dissociation of a range of Nb39 concentrations induced by various agonists.
The association of six different concentrations of Nb39 was measured in the presence of saturating ligand concentrations. Utilizing global regression analysis, the Kd of Nb39 (nM) for ligand bound receptor was calculated as follows: BU72 (144 ± 4), DAMGO (194 ± 9), Morphine (944 ± 13), Methadone (580 ± 3). The Kon values (min−1, M−1 × 10−5) were BU72 (2.35 ± 0.45), DAMGO (17.8 ± 0.5), Morphine (0.43 ± 0.05), and Methadone (1.21 ± 0.07) and the Koff values (min−1) BU72 (0.034 ± 0.0003), DAMGO (0.034 ± 0.003), morphine (0.04 ± 0.003), methadone (0.07 ± 0.0002).
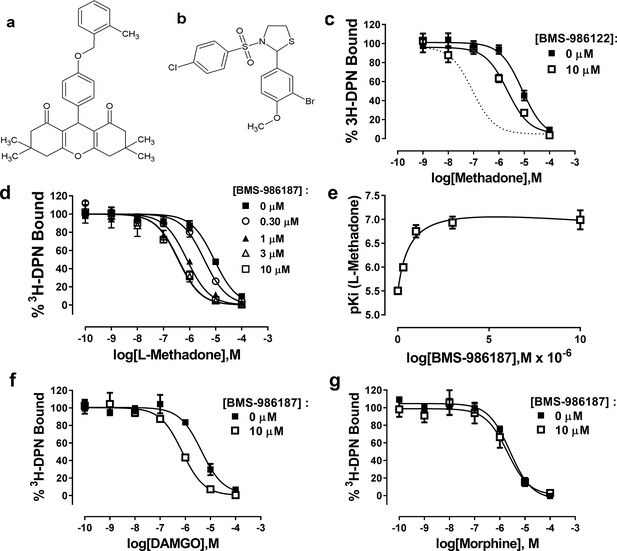
Allosteric modulation of µ-OR-rHDL by small molecule PAMs.
Structures of (a) BMS-986187 and (b) BMS-986122. The ability of BMS-986122 (c) or BMS-986187 (d) to enhance the binding affinity of L-methadone was measured using displacement of the orthosteric antagonist 3H-diprenorphine. The dotted line in (c) shows the effect previously obtained in membranes from C6μ cells (Livingston and Traynor, 2014). The effect of BMS-986187 on L-methadone affinity is plotted in (e). Enhancement of the affinity of DAMGO or morphine in the presence of 10 µM BMS-986122 is shown in (f) and (g), respectively. All plotted points are means ±s.e.m. of three (morphine) or five independent experiments (all other), each in duplicate. Nonlinear regression analysis with GraphPad Prism 6.02 was utilized to determine the affinity of the ligands. Hill slopes were not significantly different from unity.
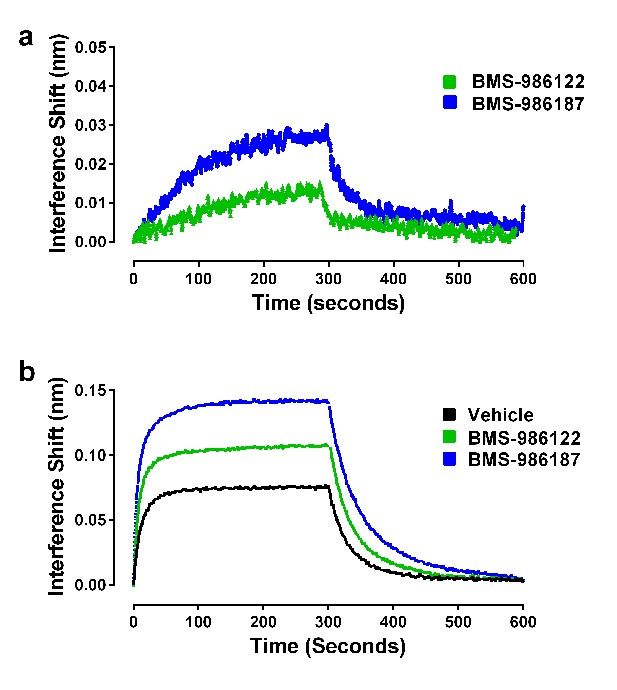
Effects of allosteric ligands on binding kinetics of Nb39.
The association and dissociation of Nb39 (1 µM) was measured as described in the Materials and methods. (a) Shown is a representative experiment of data in Table 1, comparing two allosteric agonists at 30 µM. (b) Representative experiment comparing the kinetics of Nb39 binding in the presence of morphine in the absence (black), or presence of 30 µM BMS-986122 (green), or 30 µM BMS-986187 (blue).
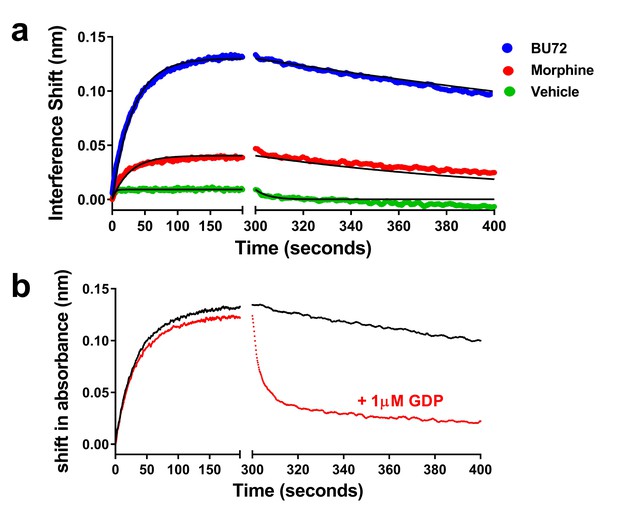
Association and dissociation of heterotrimeric G protein and the effect of GDP on dissociation.
(a) Utilizing a global fit analysis the affinity (Kd) of G protein for μ-receptor was determined as 13 ± 1 nM for the BU72-bound receptor and 2.9 ± 0.8 nM for the morphine-bound receptor, though these are pseudo-affinity constants as the G protein binding was nearly irreversible on the time-scale studied. Only in the presence of nucleotide (b) did the BU72-bound heterotrimer rapidly dissociate.
Tables
Association and dissociation kinetics of Nb39 to µ-OR-rHDL in the presence of various agonists.
kobs and koff were determined for each independent experiment (number of individual experiments indicated in ‘n’ column) and averaged. One-phase association and single-phase exponential decay models were used. Half-time values (t1/2) numbers were calculated from the respective k values (t1/2 = 0.693/k). A one-way ANOVA was performed followed by a Tukey post-hoc test. Methadone was found to be statistically different compared to all other orthosteric ligands other than loperamide and PZM21. Both loperamide and PZM21 were also found to be statistically different from several other ligands, though not as many as methadone.
| Ligand | kobs ± SEM (min−1) | t1/2ass (sec) | koff (min−1) | t1/2dis (sec) | N |
|---|---|---|---|---|---|
| BU72 | 0.20 ± 0.01 | 3.5 | 0.031 ± 0.001 | 22 | 14 |
| DAMGO | 0.179 ± 0.008 | 3.9 | 0.030 ± 0.001 | 23 | 6 |
| Leu-Enk | 0.098 ± 0.02 | 7.1 | 0.031 ± 0.001 | 23 | 9 |
| L-Methadone | 0.19 ± 0.02 | 3.6 | 0.052 ± 0.003 | 13 | 6 |
| Morphine | 0.08 ± 0.01 | 8.5 | 0.033 ± 0.003 | 21 | 7 |
| Nalbuphine | 0.023 ± 0.008 | 30 | 0.036 ± 0.004 | 19 | 11 |
| PZM21 | 0.064 ± 0.004 | 11 | 0.043 ± 0.002 | 16 | 6 |
| Endomorphin 2 | 0.101 ± 0.002 | 6.9 | 0.036 ± 0.002 | 19 | 6 |
| Loperamide | 0.208 ± 0.009 | 3.2 | 0.044 ± 0.003 | 16 | 6 |
| Oxycodone | 0.044 ± 0.001 | 16 | 0.028 ± 0.001 | 25 | 9 |
| Etorphine | 0.180 ± 0.007 | 3.9 | 0.026 ± 0.001 | 27 | 6 |
| Fentanyl | 0.075 ± 0.005 | 9.2 | 0.035 ± 0.002 | 20 | 6 |
| Met-Enk | 0.131 ± 0.004 | 5.3 | 0.027 ± 0.001 | 26 | 6 |
| Hydrocodone | 0.042 ± 0.004 | 17 | 0.029 ± 0.001 | 24 | 6 |
| Buprenorphine | 0.058 ± 0.009 | 12 | 0.025 ± 0.001 | 27 | 6 |
| Naloxone | n/a | ---- | n/a | ---- | 3 |
| Diprenorphine | n/a | ---- | n/a | ---- | 3 |
| BMS-986122 | 0.012 ± 0.001 | 56 | 0.027 ± 0.003 | 25 | 10 |
| BMS-986187 | 0.025 ± 0.006 | 28 | 0.037 ± 0.005 | 19 | 11 |
Alteration in Nb39 kinetics in the presence of µ-PAMs
https://doi.org/10.7554/eLife.32499.010| Morphine (1 µM Nb39) | ||||||
|---|---|---|---|---|---|---|
| kobs (min−1) | t1/2Assoc (sec) | koff (min−1) | t1/2Diss(sec) | N | ||
| Vehicle | 0.08 ± 0.01 | 8.5 | 0.033 ± 0.001 | 21 | 3 | |
| BMS-986122 | 0.11 ± 0.001 | 6.4 | 0.032 ± 0.0002 | 22 | 3 | |
| BMS-986187 | 0.13 ± 0.01 *** | 5.3 | 0.024 ± 0.0004 | 29 | 3 | |
| L-Methadone (100 nM Nb39) | ||||||
| kobs (min−1) | t1/2Assoc (sec) | koff (min−1) | t1/2Diss(sec) | n | ||
| Vehicle | 0.087 ± 0.009 | 8.0 | 0.050 ± 0.004 † | 14 | 7 | |
| BMS-986122 | 0.077 ± 0.007 | 9.0 | 0.047 ± 0.004 ++ | 15 | 7 | |
| BMS-986187 | 0.055 ± 0.004 * | 13 | 0.033 ± 0.002 ** | 21 | 7 | |
| DAMGO (100 nM Nb39) | ||||||
| kobs (min−1) | t1/2Assoc (sec) | koff (min−1) | t1/2Diss(sec) | n | ||
| Vehicle | 0.051 ± 0.003 | 14 | 0.035 ± 0.003 ‡ | 20 | 7 | |
| BMS-986122 | 0.050 ± 0.01 | 13 | 0.029 ± 0.003 | 24 | 7 | |
| BMS-986187 | 0.050 ± 0.004 | 14 | 0.022 ± 0.001* | 31 | 7 | |
-
Values are means from independent experiments (number of individual experiments indicated in ‘n’ column). Analyses were performed by two-way ANOVA with a Tukey post-hoc test.
*Indicates significance compared to vehicle condition for each orthosteric ligand (*p<0.05, **p<0.01, ***p<0.001). ++Indicates p<0.01 as compared to L-methadone/BMS-986187 combination.
-
†Indicates p<0.01 as compared to morphine/vehicle combination.
‡Indicates p<0.01 as compared to DAMGO/vehicle combination.
| Reagent type (species) or resource | Designation | Source or reference | Identifiers | Additional information |
|---|---|---|---|---|
| Peptide, recombinant protein | mu-opioid receptor | Manglik et al. (2012) | OPRM1 | |
| Peptide, recombinant protein | Gbetagamma | Iñiguez-Lluhi et al., 1992 | ||
| Peptide, recombinant protein | Myristoylated Gαi1 | Greentree and Linder, 2004 | ||
| Peptide, recombinant protein | Apolipoprotein-AI | Kuszak et al., 2009 | ||
| Peptide, recombinant protein | [D-Ala2, N-Me-Phe4, Gly5-ol]-Enkephalin acetate salt (DAMGO) | Sigma | E7384 | CAS#100929-53-1 |
| Peptide, recombinant protein | Leu-Enkephalin | Sigma | L9133 | |
| Peptide, recombinant protein | Met-Enkephalin | Sigma | M6638 | CAS#82362-17-2 |
| Peptide, recombinant protein | Endomorphin 2 | Sigma | SCP0133 | |
| Peptide, recombinant protein | Nanobody 39 (Nb39) | Huang et al., 2015 | ||
| Chemical compound, drug | 3H-diprenorphine | Perkin Elmer | NET1121250UC | |
| Chemical compound, drug | Morphine sulfate | National Institute on Drug Abuse, NIH. Drug Supply Catalog | 9300–001 | CAS # 6211-15-0 |
| Chemical compound, drug | (R)-Methadone | National Institute on Drug Abuse, NIH. Drug Supply Catalog | 9250–005 | CAS# 125-58-6 |
| Chemical compound, drug | Buprenorphine | National Institute on Drug Abuse, NIH. Drug Supply Catalog | 9064–110 | CAS# 53152-21-9 |
| Chemical compound, drug | BU72 | Huang et al. (2015) | ||
| Chemical compound, drug | Diprenorphine | Other | CAS# 14357-78-9: Opioid Research Center, U Michigan | |
| Chemical compound, drug | Etorphine | Other | CAS# 14521-96-1: Opioid Research Center, U Michigan | |
| Chemical compound, drug | Fentanyl | National Institute on Drug Abuse, NIH. Drug Supply Catalog | 9801–001 | CAS# 1443-54-5 |
| Chemical compound, drug | Hydrocodone | Other | CAS# 125-29-1:Opioid Research Center, U Michigan | |
| Chemical compound, drug | Loperamide | Other | CAS # 34552-83-5: Opioid Research Center, U Michigan | |
| Chemical compound, drug | Nalbuphine | Other | CAS# 23277-43-2: Opioid Research Center, U Michigan | |
| Chemical compound, drug | Naloxone | Sigma | PHR1802 | CAS# 51481-60-8 |
| Chemical compound, drug | Oxycodone | Other | CAS# 76-42-6: Opioid Research Center, U Michigan | |
| Chemical compound, drug | PZM21 | Manglik et al. (2016) | ||
| Chemical compound, drug | BMS-986187 | Bristol Myers Squib; | Burford et al. (2015) | CAS# 684238-37-7 |
| Chemical compound, drug | BMS-986122 | Bristol Myers Squib; | Burford et al. (2013) | CAS# 313669-88-4 |
| Software, algorithm | GraphPad Prism 6.0 | GraphPad, La Jolla, CA | https://www.graphpad.com/scientific-software/prism/ | |
| Software, algorithm | Octet Data Analysis 7.0 software | Pall Forte Bio | https://shop.fortebio.com/site-license-octet-data-analysis-software-version-7.x.html |
Additional files
-
Transparent reporting form
- https://doi.org/10.7554/eLife.32499.012





