Complement and CD4+ T cells drive context-specific corneal sensory neuropathy
Figures
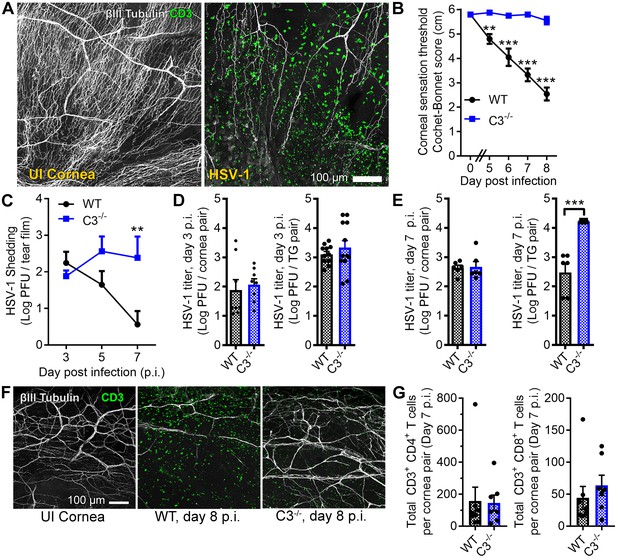
Complement C3 contributes to corneal denervation.
(A) Representative confocal images of cornea flat-mounts showing corneal nerves (βIII Tubulin, white) and T cells (CD3, green) in healthy uninfected (UI) and HSV-1-infected corneas 8 days post infection (p.i.). (B) Corneal mechanosensory function in WT and C3-/- mice following ocular HSV-1 infection (n = 6–8 mice/group; three independent experiments). (C) Viral titers shed in the tear film of WT and C3-/- mice at the indicated times p.i. (n = 5 mice per group; two independent experiments). Viral titers in the corneas and trigeminal ganglia (TG) of WT and C3-/- mice are shown at days 3 and 7 p.i. in (D) and (E), respectively. (F) Representative confocal images of stromal nerve fibers and T cells in healthy and HSV-infected corneas of WT and C3-/- mice as in (A). (G) Flow cytometry-based quantification of CD4+ and CD8+ T cells in HSV-infected corneas at day 7 p.i. (n = 7–8 mice/group; four independent experiments). Statistical differences were determined using two-way ANOVAs with Bonferroni posttests (B, C) or Student’s T tests (D, E, G).
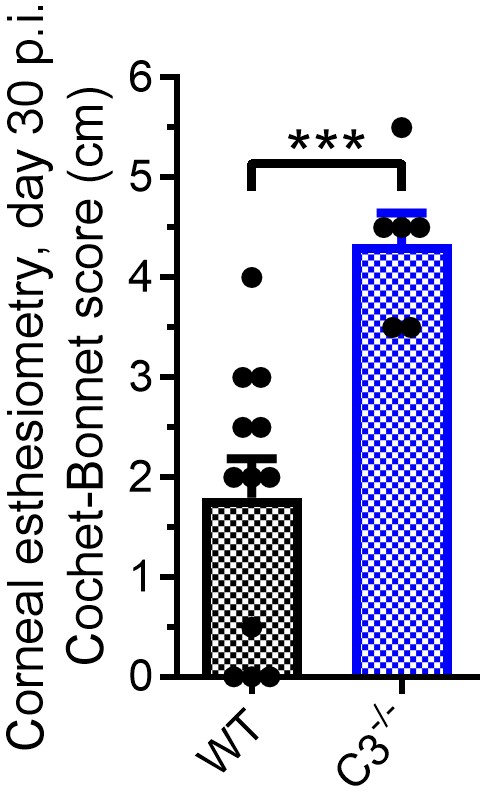
C3-/-mice retain corneal sensation through viral latency.
WT and C3-/- mice were infected with 1 × 104 p.f.u. HSV-1 McKrae per eye and corneal sensation thresholds measured at day 30 post infection when HSV-1 is considered to be latent in animal infection models (n = 6–12 eyes/group; Student’s T-test).
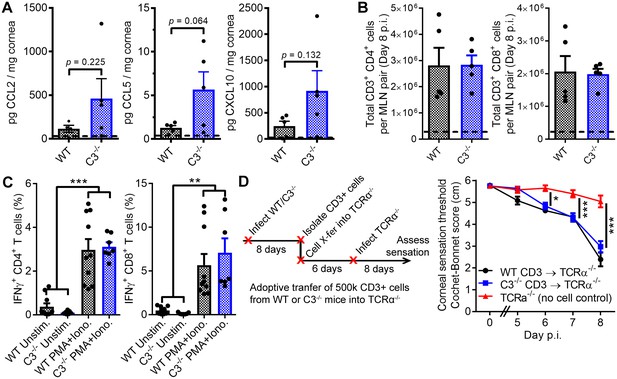
T cells facilitate corneal sensation loss.
(A) Chemokine concentrations in HSV-1-infected corneas from WT and C3-/- mice at day five post infection (p.i.). (B) T cell expansion in the eye-draining mandibular lymph nodes of WT and C3-/- mice. For panels (A) and (B), dashed lines reflect the average value for uninfected WT controls (n = 5 mice per group; two independent experiments; Student’s T). (C) IFNγ expression following stimulation with PMA and ionomycin using T cells harvested from WT or C3-/- mice at day eight post infection (n = 7 unstimulated and 10 activated replicates from three independent experiments; one-way ANOVA, Bonferroni). (D) Adoptive transfer schematic and corneal sensation measurements in TCRα-/- mice following reconstitution with purified splenic T cells from HSV-infected WT and C3-/- mice (n = 5–9 TCRα-/- mice/group; three independent experiments; two-way ANOVA, Bonferroni).
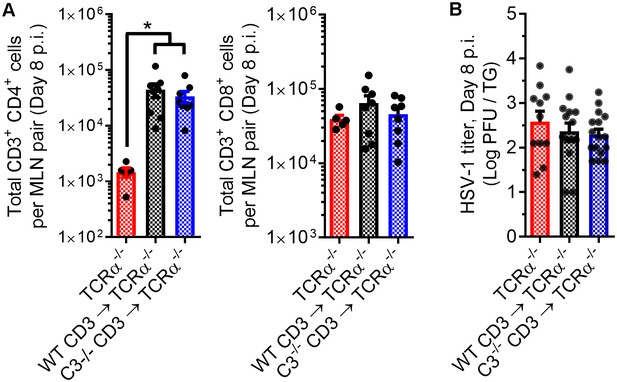
T cell engraftment in recipient TCRα-/- mice.
(A) T cell reconstitution was confirmed by evaluating the total number of T cells in the eye-draining mandibular lymph nodes of HSV-1-infected TCRα-/- mice. (B) Impact of exogenous T cell reconstitution in TCRα-/- mice as determined by HSV-1 titers in the trigeminal ganglia (TG). Data reflect summaries of 5–9 mice/group across three independent experiments (one-way ANOVA, Bonferroni).
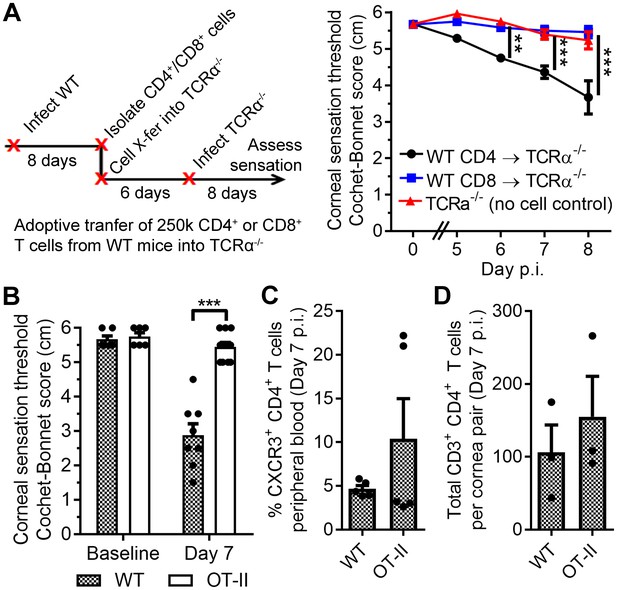
Antigen-specific CD4 T cells drive corneal sensation loss.
(A) Adoptive transfer schematic and corneal sensation measurements in TCRα-/- mice following reconstitution with purified splenic CD4 or CD8 T cells from HSV-infected WT mice (n = 6–7 TCRα-/- mice/group; two independent experiments; two-way ANOVA, Bonferroni). (B) Corneal sensation measurements at baseline and day seven post infection (p.i.) in WT and OT-II mice following ocular HSV-1 infection (n = 4–6 mice/group; three independent experiments). (C) Percentage of CXCR3-expressing CD4 T cells in peripheral blood from WT and OT-II mice at day 7 p.i.; (n = 5 mice/group; two independent experiments). (D) Verification of CD4 T cell infiltration into corneas of WT and OT-II mice at day 7 p.i. (n = 3 mice/group; two independent experiments). Data in panels B-D were analyzed using Student’s T tests.
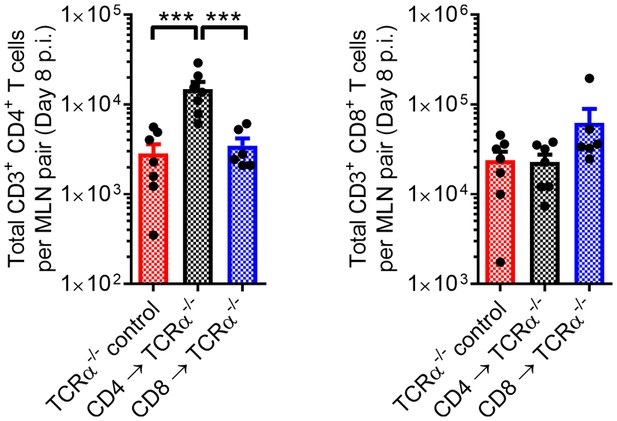
Engraftment of donor T cells into TCRα-/- mice.
T cell reconstitution was confirmed by evaluating the total number of T cells in the eye-draining mandibular lymph nodes (MLN) of HSV-1-infected TCRα-/- mice by flow cytometry 8 days post HSV-1 infection (n = 6–7 TCRα-/- mice/group; two independent experiments; one-way ANOVA, Bonferroni).
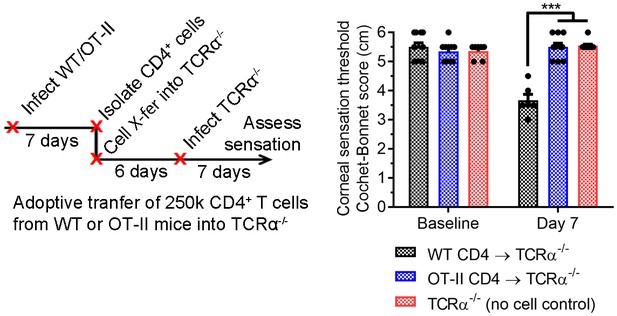
Antigen-specific donor CD4 T cells drive sensation loss during HSV-1 keratitis.
Adoptive transfer schematic and corneal sensation measurements at baseline and day seven post infection in HSV-1-infected TCRα-/- mice following reconstitution with purified splenic CD4 T cells from HSV-infected WT or OT-II mice (n= 5 TCRα-/- mice/group; two independent experiments; one-way ANOVA comparing each timepoint, Bonferroni posttest).
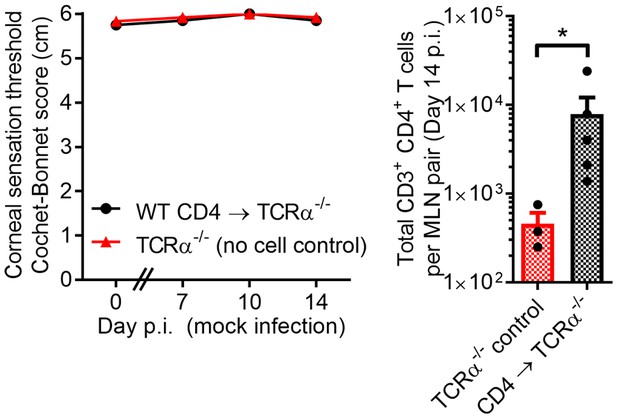
Active corneal HSV-1 infection is required to drive sensation loss in herpetic keratitis.
Corneal sensation in TCRα-/- mice following reconstitution with purified CD4 T cells from HSV-1 infected WT mice as in Figure 3A, with recipient TCRα-/- mice undergoing mock infection (corneal scratch injury). T cell engraftment was confirmed by flow cytometry using MLN from TCRα-/- mice 14 days after mock infection (n = 3–5 TCRα-/- mice/group; two independent experiments; two-way ANOVA, Student’s T test).
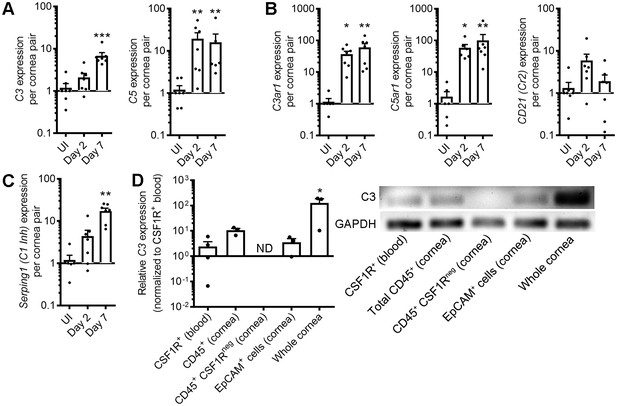
Corneal HSV-1 infection enhances local complement synthesis.
Gene expression of complement effectors (A), receptors (B), and regulators (C) upregulated in the corneas of B6 mice during acute HSV-1 infection (n = 7 WT mice/group; two independent experiments; Kruskal-Wallis, Dunn’s multiple comparisons test). (D) Relative C3 expression among selected cornea-resident and infiltrating cell subsets at day three post-infection (n = 3–4 pooled samples from two mice each for cell subsets or three independent cornea pairs; two independent experiments; one-way ANOVA, Bonferroni; ND, not detected/amplification cycle >35). Final PCR products were resolved on an agarose gel to verify amplification. Data are relative to GAPDH expression and normalized to uninfected control samples for panels A-C or to purified CSF1R-expressing peripheral blood monocytes/macrophages in panel D.
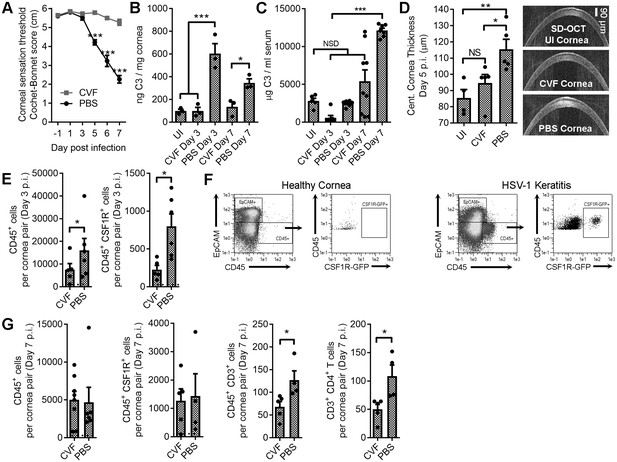
Local C3 depletion prevents HSV-associated corneal sensation loss.
B6 mice were given PBS (vehicle) or 5.0 μg cobra venom factor (CVF) via subconjunctival injection to degrade C3, and ocularly infected with HSV-1 18 hr later. C3 depletion was maintained by daily topical treatment (eyedrop) containing 0.5 μg CVF. (A) Corneal sensation following HSV-1 infection in animals treated with CVF or PBS (n = 5–11 mice/group; three independent experiments; two-way ANOVA, Bonferroni). Impact of CVF treatment on C3 protein concentrations in the cornea (B) and serum (C) (n= 3 cornea pairs, 5–9 serum samples/timepoint; 2–3 independent experiments; one-way ANOVA, Tukey). (D) Corneal edema measurements (central corneal thickness) determined via spectral domain optical coherence tomography (SD-OCT) on uninfected (UI) or HSV-1 infected mice treated with CVF or PBS at day 5 p.i. (n = 4–5 mice/group; two experiments; one-way ANOVA, Newman-Keuls). (E) Impact of CVF treatment on total leukocyte (CD45+) and monocyte/macrophage (CSF1R+) infiltration into the corneas of CVF and PBS-treated MaFIA (CSF1R-GFP) mice at day 3 p.i. (n = 5–6 mice/group; two independent experiments; Student’s T). (F) Representative flow plots showing cell populations in healthy and HSV-1 corneas from panel E. (G) Impact of CVF treatment on leukocyte infiltration into the corneas of HSV-1 infected mice at day 7 p.i. (n = 4–5 mice/group; two independent experiments; Student’s T). Note: Total CD45+ graph in panel G reflects data from two technical replicates. Dashed lines in panels E and G reflect cell number recorded in healthy uninfected mice.
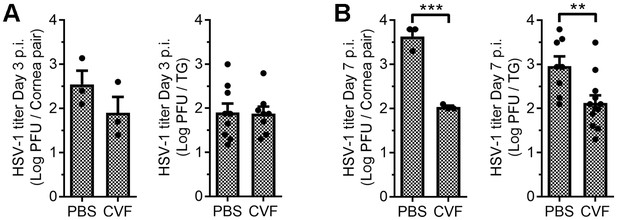
Impact of ocular cobra venom factor treatment on HSV-1 titers.
The ocular surface of B6 mice was treated with PBS or CVF and animals ocularly infected with HSV-1 as in Figure 5. Systemic effects of CVF treatment were evaluated in terms of viral burden in the corneas and trigeminal ganglia (TG) at days 3 (A) and 7 (B) post infection (p.i.). Data in reflect three cornea pairs and 8–9 TG specimens per timepoint; data are composite from two independent experiments and analyzed using Student’s T tests.
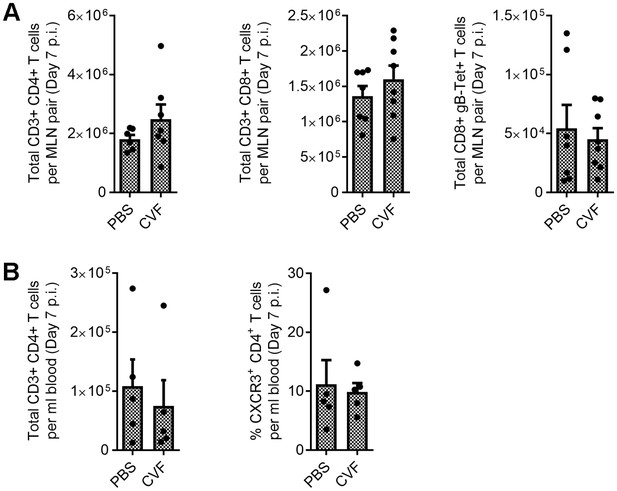
Immunologic impacts of ocular cobra venom factor treatment.
The ocular surface of B6 mice was treated with PBS or CVF and animals ocularly infected with HSV-1 as in Figure 5. Immunologic impact of CVF treatment was assessed by flow cytometry on the eye-draining mandibular lymph nodes (MLN) (A) to assess CD4+, CD8+, and HSV-1 glycoprotein B (gB)-specific CD8+ T cell numbers or blood (B) to evaluate CD4 count and percentage of activated (CXCR3+) CD4 T cells at day 7 p.i. Data in panel A reflects 6–8 mice/group; panel B reflects five samples/group; data are composite from two independent experiments and analyzed using Student’s T tests.
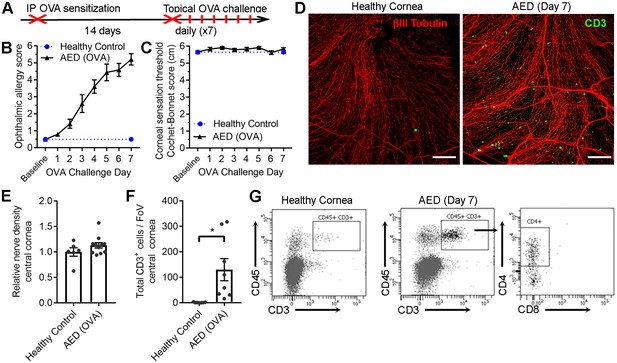
Chronic ocular allergy does not provoke frank corneal sensation loss.
(A) Schematic of allergic eye disease induced by ovalbumin (OVA) immunization followed by topical ocular OVA challenge. Healthy and OVA-challenged B6 mice were evaluated for signs of ophthalmic allergy (B) and corneal sensation thresholds (C). (D) Representative confocal images of cornea flat mounts from healthy and OVA-challenged mice showing nerves (βΙII Tubulin, red) and infiltrating T cells (CD3, green) in the central cornea at 20x magnification (scale bar = 100 μm). (E) Morphometric analysis of nerve densities in the central cornea based on confocal images. Normalization is based on the average volume in the healthy control group. (F) Quantification of total CD3+ cells per field of view. (G) Flow cytometry was used to confirm CD4 T cell infiltration into the corneas of OVA-challenged mice. Data in panels B – F reflect 5 to 6 mice per group; flow plots in panel G reflect pooled digests of corneas from three mice. Data in panels E and F were analyzed using Student’s T tests.
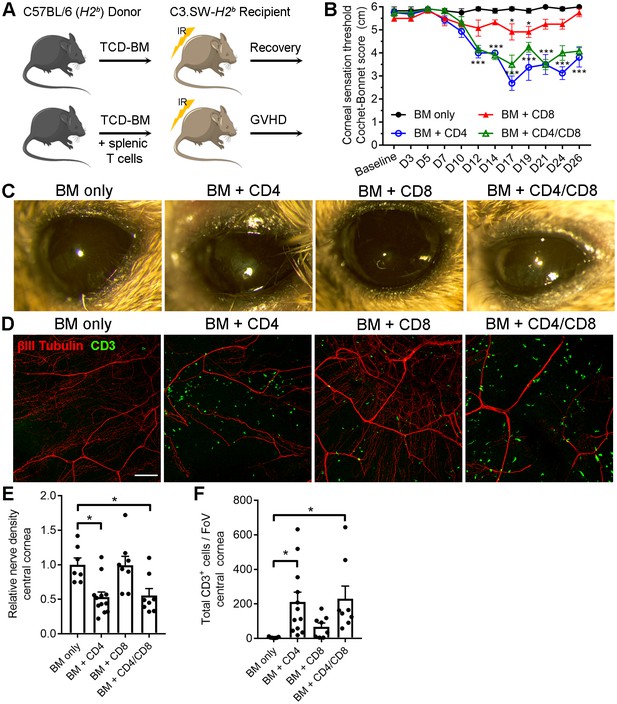
CD4 T cells drive corneal sensation loss in ocular GVHD.
(A) Schematic of GVHD induction using T cell depleted bone marrow (TCD-BM) or TCD-BM with splenic T cells isolated from C57BL/6 donors and transferred into C3.SW-H2 b recipients. (B) Corneal sensation measurements in C3.SW-H2 b mice following reconstitution with BM only or with BM and 1.3x10 6 CD4, CD8, or CD4 and CD8 T cells. (C) External photographs showing CD4-dependent ocular surface morbidities consistent with corneal sensation loss. (D) Representative confocal images of cornea flat mounts from each group of mice at the study endpoint showing nerves (βΙII Tubulin, red) and infiltrating T cells (CD3, green) in the central cornea at 20x magnification (scale bar = 100 μm). (E) Morphometric analysis of nerve densities based on confocal images. Normalization is based on the average volume in the BM only control group. (F) Quantification of total CD3 + cells per field of view. Data reflect independent measurements of corneas from 3-4 mice per group across 2 experiments; sensation data were evaluated by two-way ANOVA with Bonferroni posttests; data in panels E and F were analyzed by one-way ANOVA with Bonferroni posttests. Figure was generated using Servier Medical Art (http://smart.servier.com/) under a Creative Commons 3.0 license.
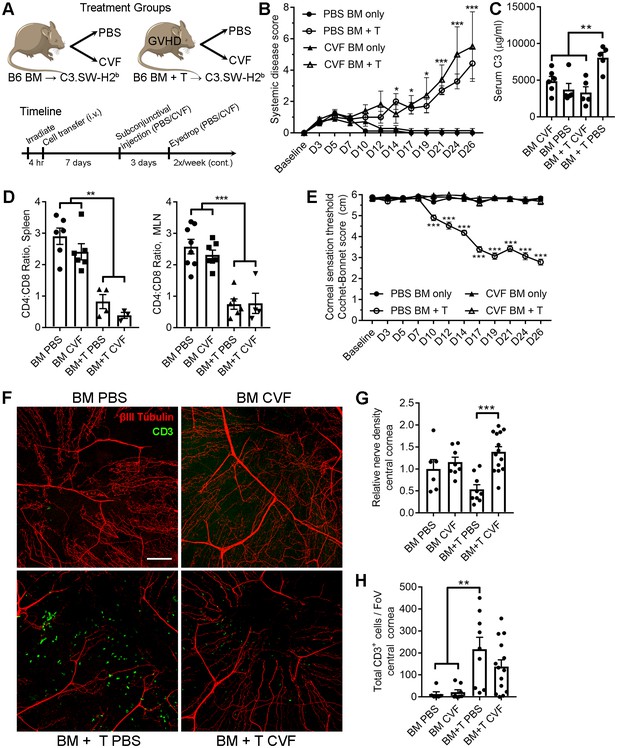
Local C3 depletion prevents corneal sensation loss in ocular GVHD.
(A) Experiment schematic of GVHD induction using T cell depleted bone marrow (TCD-BM) or TCD-BM with splenic T cells isolated from C57BL/6 donors and transferred into C3.SW-H2 b recipients. Each group was subsequently given a subconjunctival injection containing PBS (vehicle) or 5.0 μg cobra venom factor (CVF) to degrade C3. C3 depletion was maintained by topical treatment (eyedrop) containing 2.0 μg CVF twice weekly. (B) Systemic disease scores in each cohort (n=8 mice per group; two-way ANOVA, Bonferroni). (C) Impact of CVF treatment on serum C3 protein concentrations in each group at the experiment endpoint (n=5-6 samples/group; one-way ANOVA, Bonferroni). (D) Evaluation of CD4:CD8 ratios in secondary lymphoid organs by flow cytometry to confirm onset of GVHD at experimental endpoints (n=3-8 mice/group; 2 independent experiments; one-way ANOVA, Bonferroni). (E) Longitudinal corneal sensation measurements in each group of C3.SW-H2 b mice. (F) Representative confocal images of cornea flat mounts from each group of C3.SW-H2 b mice at the study endpoint showing nerves (βΙII Tubulin, red) and infiltrating T cells (CD3, green) in the central cornea at 20x magnification (scale bar = 100 μm). (G) Morphometric analysis of nerve densities based on confocal images. Normalization is based on the average volume in the BM only control group. (H) Quantification of total CD3 + cells per field of view. Data in panels E – H reflect 3-8 mice/group; 2 independent experiments; two-way ANOVA with Bonferroni posttests. Figure was generated using Servier Medical Art (http://smart.servier.com/) under a Creative Commons 3.0 license.
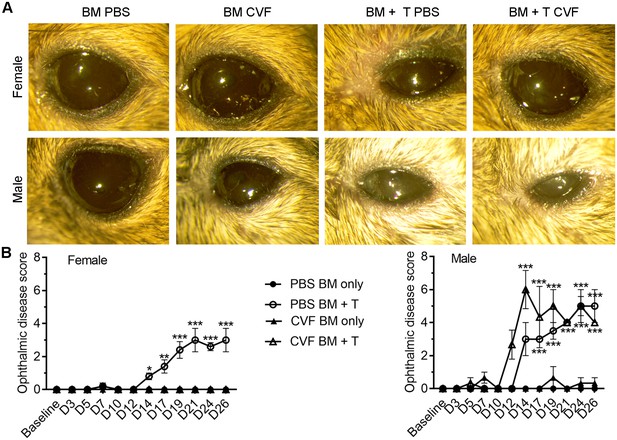
Sex-biased effects of local C3 depletion on ocular GVHD severity.
Models of GVHD were established as described in Figure 8 with cohorts of bone marrow (BM) only controls and GVHD (BM + T cells) mice exposed to ocular treatment with PBS (vehicle) or cobra venom factor (CVF) to degrade complement C3. (A) Representative external photographs showing ocular surface morbidities in each treatment cohort from Figure 8 at the experiment endpoint. Note the sex-biased impact in the CVF-treated GVHD group where treatment limits eyelid swelling in female but not male mice. (B) Ophthalmic disease scores for each experimental group by sex (n = 5 females or three males per group; two-way ANOVA, Bonferroni).
Tables
Complement regulatory factor expression in the cornea during HSV-1 infection
https://doi.org/10.7554/eLife.48378.012| Gene | Uninfected | Day 2 p.i. | Day 7 p.i. |
|---|---|---|---|
| C4bp | ND | ND | ND |
| Crry (CD46 homolog) | 1.193 ± 0.338 | 0.817 ± 0.172 | 1.137 ± 0.331 |
| CD55 (DAF) | 1.087 ± 0.201 | 1.035 ± 0.199 | 0.791 ± 0.251 |
| CD59a (MIRL) | 1.078 ± 0.192 | 0.611 ± 0.123 | 0.728 ± 0.191 |
| Cfi | ND | ND | ND |
| Cr1l | 1.026 ± 0.096 | 0.7442 ± 0.222 | 0.889 ± 0.133 |
-
Data are expressed as mean ± SEM; n = 7 samples/group. Gene expression is standardized to internal GAPDH expression and relative to uninfected control tissue. Abbreviations: C4bp, C4 binding protein; CD, cluster of differentiation; Cfi, complement factor I, Cr1l, complement C3b/C4b receptor-1-like; DAF, decay accelerating factor; GAPDH, glyceraldehyde 3-phosphate dehydrogenase; MIRL, membrane inhibitor of reactive lysis; ND, not detected (amplification cycle >35); p.i., post-infection.
Ophthalmic Disease Scoring Guide for Ocular GVHD.
https://doi.org/10.7554/eLife.48378.018| Criteria | Grade 0 | Grade 1 | Grade 2 | Grade 3 |
|---|---|---|---|---|
| Eyelid Closure | None | Mild squinting | Partial lid closure | Full lid closure |
| Blepharitis | None | Mild lid margin edema | Moderate lid margin edema with crusting | Gross lid edema, loss of lashes/eyelid fur |
| Meibomian Gland Disease | None | ≤4 plugs | >4 plugs OR cystic plugs | >4 plugs AND cystic plugs |
| Conjunctival Chemosis | None | Mild edema | Moderate edema | Gross edema |
| Conjunctival Hyperemia | None | Mild redness | Broad erythema | N/A |
| Mucoid Discharge | None | Mild discharge | Discharge covers the ocular surface | N/A |
| Corneal Opacity | None | Epithelial haze | Focal opacities | Widespread opacity |
| Reagent type (species) or resource | Designation | Source or reference | Identifiers | Additional information |
|---|---|---|---|---|
| Strain, strain background (Mus musculus, C57BL/6J) | Wildtype C57BL/6 (WT) | Jackson Laboratories | Stock # 000664 | |
| Strain, strain background (M. musculus, C57BL/6J) | Complement C3 deficient (C3-/-) | Jackson Laboratories | Stock # 029661 | |
| Strain, strain background (M. musculus, C57BL/6J) | T cell receptor alpha deficient (TCRα-/-) | Jackson Laboratories | Stock # 002116 | |
| Strain, strain background (M. musculus, C57BL/6J) | Transgenic OVA-specific CD4 T cells (OT-II) | Jackson Laboratories | Stock # 004194 | |
| Strain, strain background (M. musculus, C57BL/6J) | Transgenic Csf1r-eGFP (MAFIA) | Jackson Laboratories | Stock # 005070 | |
| Strain, strain background (M. musculus, C57BL/6J) | C3.SW-H2b | Jackson Laboratories | Stock # 000438 | |
| Strain, strain background (Herpes simplex virus type 1, McKrae) | HSV-1 | Macdonald et al., 2012; Watson et al., 2012 | N/A | Originally from Brian Gebhardt |
| Cell line (Cercopithecus aethiops) | Vero | American Type Culture Collection (ATCC) | Cat. # CCL-81 | see Materials and methods |
| Chemical compound, drug | Cobra Venom Factor, Naja naja kaouthia | Millipore-Sigma | Cat. # 233552 | |
| Commercial assay or kit | Anti-mouse CD90.2 IMag particles | BD | Cat. # 551518 | |
| Commercial assay or kit | Anti-mouse CD4 microbeads | Miltenyi Biotec | Cat. # 130-049-201 | |
| Commercial assay or kit | Anti-mouse CD8 microbeads | Miltenyi Biotec | Cat. # 130-116-478 | |
| Commercial assay or kit | Anti-mouse CD90.2 microbeads | Miltenyi Biotec | Cat. # 130-049-101 | |
| Commercial assay or kit | Anti-mouse CD45 microbeads | Miltenyi Biotec | Cat. # 130-052-301 | |
| Commercial assay or kit | Anti-mouse EpCAM microbeads | Miltenyi Biotec | Cat. # 130-105-958 | |
| Antibody | Rabbit polyclonal IgG (anti-mouse beta-III tubulin) | Abcam | Cat. # ab18207 | (1:500) |
| Antibody | AlexaFluor647 donkey polyclonal IgG (anti-rabbit IgG) | Jackson Immunoresearch | Cat. # 711-605-152 | (1:1000) |
| Antibody | Anti-mouse CD3e FITC (Clone: 145–2 C11) | eBioscience (ThermoFisher) | Cat. # 11-0031-82 | (1:500) |
| Other | Roche Liberase TL | Sigma Aldrich | Cat. # 5401020001 | see Materials and methods |
| Commercial assay or kit | C3 ELISA | Abcam | Cat. # ab157711 | |
| Commercial assay or kit | Milliplex MAP Luminex Array | EMD Millipore | Custom Design | |
| Recombinant DNA reagent | iScript cDNA synthesis kit | Biorad | Cat. # 1708891 | |
| Recombinant DNA reagent | Sso Advanced SYBR Green qPCR Supermix | Biorad | Cat. # 1725270 | |
| Other | Trizol | ThermoFisher | Cat. # 15596026 | see Materials and methods |
| Software, algorithm | Prism 6 | GraphPad | N/A | |
| Software, algorithm | Imaris x64 (8.2.1) | Bitplane | N/A |
Additional files
-
Transparent reporting form
- https://doi.org/10.7554/eLife.48378.021





