Evolution of a plant gene cluster in Solanaceae and emergence of metabolic diversity
Abstract
Plants produce phylogenetically and spatially restricted, as well as structurally diverse specialized metabolites via multistep metabolic pathways. Hallmarks of specialized metabolic evolution include enzymatic promiscuity and recruitment of primary metabolic enzymes and examples of genomic clustering of pathway genes. Solanaceae glandular trichomes produce defensive acylsugars, with sidechains that vary in length across the family. We describe a tomato gene cluster on chromosome 7 involved in medium chain acylsugar accumulation due to trichome specific acyl-CoA synthetase and enoyl-CoA hydratase genes. This cluster co-localizes with a tomato steroidal alkaloid gene cluster and is syntenic to a chromosome 12 region containing another acylsugar pathway gene. We reconstructed the evolutionary events leading to this gene cluster and found that its phylogenetic distribution correlates with medium chain acylsugar accumulation across the Solanaceae. This work reveals insights into the dynamics behind gene cluster evolution and cell-type specific metabolite diversity.
eLife digest
Plants produce a vast variety of different molecules known as secondary or specialized metabolites to attract pollinating insects, such as bees, or protect themselves against herbivores and pests. The secondary metabolites are made from simple building blocks that are readily available in plants, including amino acids, fatty acids and sugars.
Different species of plant, and even different parts of the same plant, produce their own sets of secondary metabolites. For example, the hairs on the surface of tomatoes and other members of the nightshade family of plants make metabolites known as acylsugars. These chemicals deter herbivores and pests from damaging the plants.
To make acylsugars, the plants attach long chains known as fatty acyl groups to molecules of sugar, such as sucrose. Some members of the nightshade family produce acylsugars with longer chains than others. In particular, acylsugars with long chains are only found in tomatoes and other closely-related species. It remained unclear how the nightshade family evolved to produce acylsugars with chains of different lengths.
To address this question, Fan et al. used genetic and biochemical approaches to study tomato plants and other members of the nightshade family. The experiments identified two genes known as AACS and AECH in tomatoes that produce acylsugars with long chains. These two genes originated from the genes of older enzymes that metabolize fatty acids – the building blocks of fats – in plant cells. Unlike the older genes, AACS and AECH were only active at the tips of the hairs on the plant’s surface. Fan et al. then investigated the evolutionary relationship between 11 members of the nightshade family and two other plant species. This revealed that AACS and AECH emerged in the nightshade family around the same time that longer chains of acylsugars started appearing.
These findings provide insights into how plants evolved to be able to produce a variety of secondary metabolites that may protect them from a broader range of pests. The gene cluster identified in this work could be used to engineer other species of crop plants to start producing acylsugars as natural pesticides.
Introduction
Despite the enormous structural diversity of plant specialized metabolites, they are derived from a relatively small number of primary metabolites, such as sugars, amino acids, nucleotides, and fatty acids (Maeda, 2019). These lineage-, tissue- or cell- type specific specialized metabolites mediate environmental interactions, such as herbivore and pathogen deterrence or pollinator and symbiont attraction (Mithöfer and Boland, 2012; Pichersky and Lewinsohn, 2011). Specialized metabolism evolution is primarily driven by gene duplication (Moghe and Last, 2015; Panchy et al., 2016), and relaxed selection of the resulting gene pairs allows modification of cell- and tissue-specific gene expression and changes in enzymatic activity. This results in expanded substrate recognition and/or diversified product formation (Khersonsky and Tawfik, 2010; Leong and Last, 2017). The neofunctionalized enzymes can prime the origin and diversification of specialized metabolic pathways (Schenck and Last, 2020; Weng et al., 2012; Weng, 2014).
There are many examples of mechanisms that lead to novel enzymatic activities in specialized cell- or tissue-types, however, the principles that govern assembly of multi-enzyme specialized metabolic pathways are less well established. One appealing hypothesis involves the stepwise recruitment of pathway enzymes (Noda-Garcia et al., 2018). In rare cases, non-homologous specialized metabolic enzyme genes occur in proximity to each other in a genomic region, forming a biosynthetic gene cluster (Nützmann et al., 2016; Nützmann and Osbourn, 2014; Rokas et al., 2018). In recent years, an increasing number of specialized metabolic gene clusters (SMGCs) were experimentally identified or bioinformatically predicted in plants (Boutanaev et al., 2015; Castillo et al., 2013; Schläpfer et al., 2017). However, although most experimentally characterized plant SMGCs are co-expressed, the majority of the bioinformatically predicted ones do not show coexpression under global network analysis (Wisecaver et al., 2017).
While examples of SMGCs are still relatively rare in plants, experimentally validated cases were reported for a surprisingly diverse group of pathways. These include terpenes (Chae et al., 2014; Prisic et al., 2004; Qi et al., 2004; Wilderman et al., 2004), cyclic hydroxamic acids (Frey et al., 1997), biosynthetically unrelated alkaloids (Itkin et al., 2013; Winzer et al., 2012), polyketides (Schneider et al., 2016), cyanogenic glucosides (Takos et al., 2011), and modified fatty acids (Jeon et al., 2020). However, whereas each cluster encodes multiple non-homologous enzymes of a biosynthetic pathway, evolution of their assembly is not well understood.
Acylsugars are a group of insecticidal (Leckie et al., 2016) and anti-inflammatory (Herrera-Salgado et al., 2005) chemicals mainly observed in glandular trichomes of Solanaceae species (Fan et al., 2019; Schuurink and Tissier, 2020). These specialized metabolites are sugar aliphatic esters with three levels of structural diversity across the Solanaceae family: acyl chain length, acylation position, and sugar core (Fan et al., 2019). The primary metabolites sucrose and aliphatic acyl-CoAs are the biosynthetic precursors of acylsucroses in plants as evolutionarily divergent as the cultivated tomato Solanum lycopersicum (Fan et al., 2016; Figure 1), Petunia axillaris (Nadakuduti et al., 2017) and Salpiglossis sinuata (Moghe et al., 2017). The core tomato acylsucrose biosynthetic pathway involves four BAHD [BEAT, AHCT, HCBT, DAT (D'Auria, 2006) family acylsucrose acyltransferases (Sl-ASAT1 through Sl-ASAT4), which are specifically expressed in the type I/IV trichome tip cells (Fan et al., 2016; Schilmiller et al., 2015; Schilmiller et al., 2012). These enzymes catalyze consecutive reactions utilizing sucrose and acyl-CoA substrates to produce the full set of cultivated tomato acylsucroses in vitro (Fan et al., 2016).
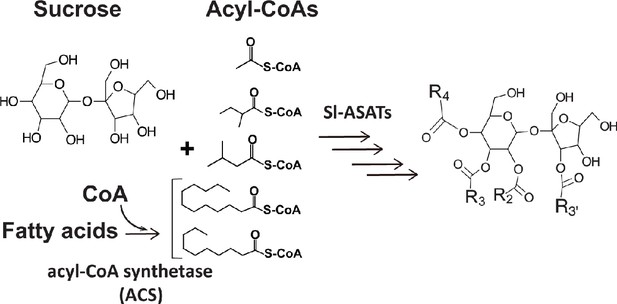
Primary metabolites are biosynthetic precursors of tomato trichome acylsugars.
In cultivated tomatoes, the trichome acylsucroses are synthesized by four Sl-ASATs using the primary metabolites – sucrose and different types of acyl-CoAs – as substrates. In this study we provide evidence that medium chain fatty acids are converted to acyl-CoAs by an acyl-CoA synthetase for medium chain acylsugar biosynthesis.
Co-option of primary metabolic enzymes contributed to the evolution of acylsugar biosynthesis and led to interspecific structural diversification across the Solanum tomato clade. One example is an invertase-like enzyme originating from carbohydrate metabolism that generates acylglucoses in the wild tomato S. pennellii through cleavage of the acylsucrose glycosidic bond (Leong et al., 2019). In another case, allelic variation of a truncated isopropylmalate synthase-like enzyme (IPMS3) – from branched chain amino acid metabolism – leads to acylsugar iC4/iC5 (2-methylpropanoic/3-methylbutanoic acid) acyl chain diversity in S. pennellii and S. lycopersicum (Ning et al., 2015). Acylsugar structural diversity is even more striking across the family. Previous studies revealed variation in acyl chain length (Ghosh et al., 2014; Liu et al., 2017; Moghe et al., 2017): Nicotiana, Petunia and Salpiglossis species were reported to accumulate acylsugars containing only short acyl chains (carbon number, C ≤ 8). In contrast, some species in Solanum and other closely related genera produce acylsugars with medium acyl chains (C ≥ 10). These results are consistent with the hypothesis that the capability to produce medium chain acylsugars varies across the Solanaceae family.
In this study, we identify a metabolic gene cluster on tomato chromosome 7 containing two non-homologous genes – acylsugar acyl-CoA synthetase (AACS) and acylsugar enoyl-CoA hydratase (AECH) – affecting medium chain acylsugar biosynthesis. Genetic and biochemical results show that the trichome enriched AACS and AECH are involved in generating medium chain acyl-CoAs, which are donor substrates for acylsugar biosynthesis. Genomic analysis revealed a syntenic region on chromosome 12, where the acylsucrose biosynthetic Sl-ASAT1 is located (Fan et al., 2016). Phylogenetic analysis of the syntenic regions in Solanaceae and beyond led to evolutionary reconstruction of the origin of the acylsugar gene cluster. We infer that sequential gene insertion facilitated emergence of this gene cluster in tomato. These results provide insights into specialized metabolic evolution through emergence of cell-type specific gene expression, the formation of metabolic gene clusters and illuminates additional examples of primary metabolic enzymes being co-opted into specialized metabolism.
Results
Identification of a metabolic gene cluster that affects tomato trichome medium chain acylsugar biosynthesis
S. pennellii natural accessions (Mandal et al., 2020), as well as the S. lycopersicum M82 ×S. pennellii LA0716 chromosomal substitution introgression lines (ILs) (Eshed and Zamir, 1995), offer convenient resources to investigate interspecific genetic variation that affects acylsugar metabolic diversity (Mandal et al., 2020; Schilmiller et al., 2010). In a rescreen of ILs for S. pennellii genetic regions that alter trichome acylsugar profiles (Schilmiller et al., 2010), IL7-4 was found to accumulate increased C10 medium chain containing acylsugars compared with M82 (Figure 2, A and B). The genetic locus that contributes to the acylsugar phenotype was narrowed down to a 685 kb region through screening selected backcross inbred lines (BILs) (Ofner et al., 2016) that have recombination breakpoints on chromosome 7 (Figure 2C). Because tomato acylsucrose biosynthesis occurs in trichomes, candidate genes in this region were filtered based on their trichome-specific expression patterns. This analysis identified a locus containing multiple tandemly duplicated genes of three families – an acyl-CoA synthetase (ACS), enoyl-CoA hydratase (ECH), and BAHD acyltransferase. Our analysis (Moore et al., 2020) revealed co-expression of four Sl-ASATs (Fan et al., 2019) and three genes at the locus – Solyc07g043630, Solyc07g043660, and Solyc07g043680 (Supplementary file 1 and Figure 2—figure supplement 1). Expression of these three genes was trichome enriched (Figure 2D), and thus they were selected for further analysis.
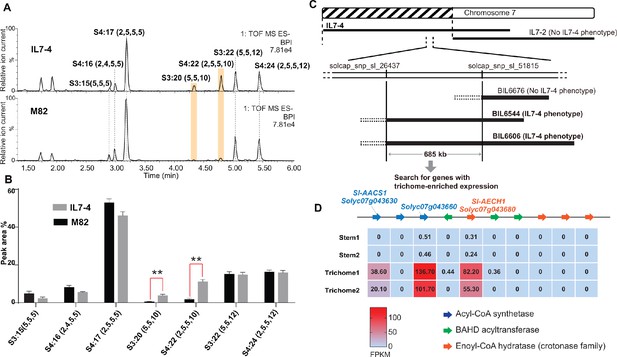
Mapping of a genetic locus related to acylsugar variations in tomato interspecific introgression lines.
(A) Electrospray ionization negative (ESI-) mode, base-peak intensity (BPI) LC/MS chromatogram of trichome metabolites from cultivated tomato S. lycopersicum M82 and introgression line IL7-4. The orange bars highlight two acylsugars that have higher abundance in IL7-4 than in M82. For the acylsucrose nomenclature, ‘S’ refers to a sucrose backbone, ‘3:22’ means three acyl chains with twenty-two carbons in total. The length of each acyl chain is shown in the parentheses. (B) Peak area percentage of seven major trichome acylsugars in M82 and IL7-4. The sum of the peak area percentage of each acylsugar is equal to 100% in each sample. The data is shown for three plants ± SEM. **p<0.01, Welch two-sample t test. Figure 2—source data 1 includes values for the analysis. (C) Mapping the genetic locus contributing to the IL7-4 acylsugar phenotype using selected backcross inbred lines (BILs) that have recombination break points within the introgression region of IL7-4. (D) Narrowing down candidate genes in the locus using trichome/stem RNA-seq datasets generated from previous study (Ning et al., 2015). A region with duplicated genes of three types – acyl-CoA synthetase (ACS), BAHD acyltransferase, and enoyl-CoA hydratase (ECH) – is shown. The red-blue color gradient provides a visual marker to rank the expression levels represented by Fragments Per Kilobase of transcript per Million mapped reads (FPKM). Coexpression analysis of tomato ACS, ECH, and BAHD acyltransferase family genes is shown in Figure 2—figure supplement 1.
-
Figure 2—source data 1
Data used to make Figure 2B.
Peak area percentage of seven major trichome acylsugars in M82 and IL7-4.
- https://cdn.elifesciences.org/articles/56717/elife-56717-fig2-data1-v2.xlsx
The three candidate genes were tested for involvement in tomato acylsugar biosynthesis by making loss of function mutations using the CRISPR-Cas9 gene editing system. Two guide RNAs (gRNAs) were designed to target one or two exons of each gene to assist site-specific DNA cleavage by hCas (Brooks et al., 2014; Figure 3—figure supplement 1, A–C). In the self-crossed T1 progeny of stably transformed M82 plants, at least two homozygous mutants were obtained in Solyc07g043630, Solyc07g043660, and Solyc07g043680 (Figure 3—figure supplement 1, A–C), and these were analyzed for leaf trichome acylsugar changes. Altered acylsugar profiles were observed in the ACS-annotated Solyc07g043630 or ECH-annotated Solyc07g043680 mutants (Figure 3, A and B), but not in the ACS-annotated Solyc07g043660 mutant (Figure 3—figure supplement 1D). Despite carrying mutations in distinctly annotated genes (ACS or ECH), the two mutants exhibited the same phenotype – no detectable medium acyl chain (C10 or C12) containing acylsugars (Figure 3, A and B). We renamed Solyc07g043630 as acylsugar acyl-CoA synthetase 1 (Sl-AACS1) and Solyc07g043680 as acylsugar enoyl-CoA hydratase 1 (Sl-AECH1) based on this analysis.
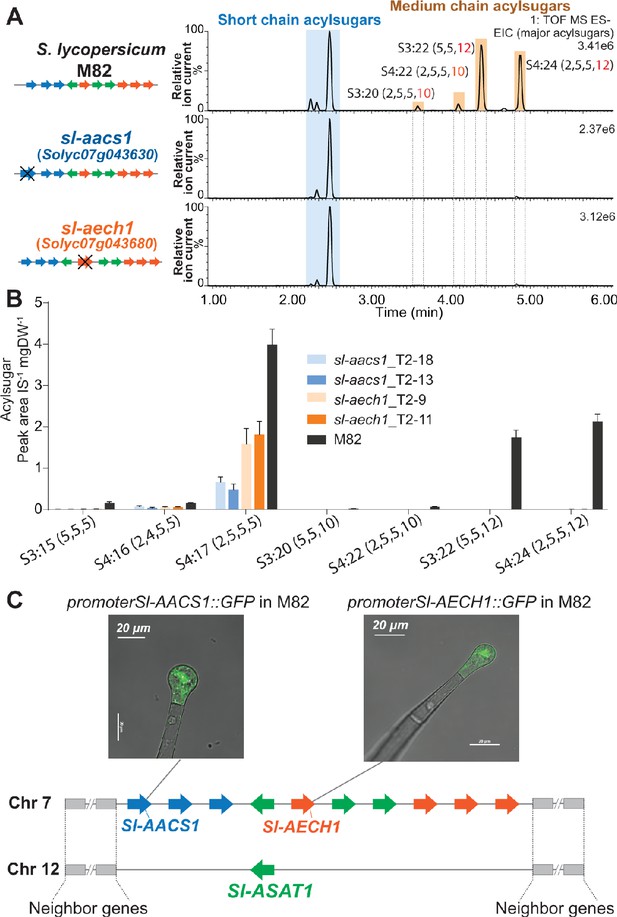
CRISPR/Cas9-mediated gene knockout of tomato Sl-AACS1 or Sl-AECH1 eliminates detectable medium chain containing acylsugars.
(A) Combined LC/MS extracted ion chromatograms of trichome metabolites from CRISPR mutants sl-aacs1 and sl-aech1. The medium chain acylsugars that are not detected in the two mutants are denoted by pairs of vertical dotted lines. Figure 3—figure supplement 1 describes the design of the gRNAs and details of the gene edits. (B) Quantification of seven major trichome acylsugars in sl-aacs1 and sl-aech1 mutants. Two independent T2 generation transgenic lines for each mutant were used for analysis. The peak area/internal standard (IS) normalized by leaf dry weight (DW) is shown from six plants ± SEM. Figure 3—source data 1 includes values for the analysis. (C) Confocal fluorescence images showing that GFP fluorescence driven by Sl-AACS1 or Sl-AECH1 is located in the tip cells of type I/IV trichomes. Their tissue specific expressions are similar to Sl-ASAT1 (Fan et al., 2016), which locates in a chromosome 12 region that is syntenic to the locus containing Sl-AACS1 and Sl-AECH1. Figure 3—figure supplement 2 provides the detailed information of the syntenic region. Sl-AACS1, Sl-AECH1, and Sl-ASAT1 are the only gene models with demonstrated functions in acylsugar biosynthesis.
-
Figure 3—source data 1
Data used to make Figure 3B.
Quantification of seven major trichome acylsugars in the CRISPR mutants sl-aacs1 and sl-aech1, as well as the parent M82.
- https://cdn.elifesciences.org/articles/56717/elife-56717-fig3-data1-v2.xlsx
Further genomic analysis revealed that Sl-AACS1 and Sl-AECH1 belong to a syntenic region shared with a locus on chromosome 12, where Sl-ASAT1 is located (Figure 3C and Figure 3—figure supplement 2). Sl-ASAT1 is specifically expressed in trichome tip cells and encodes the enzyme catalyzing the first step of tomato acylsucrose biosynthesis (Fan et al., 2016). This led us to test the cell-type expression pattern of Sl-AACS1 and Sl-AECH1. Like Sl-ASAT1, the promoters of both genes drove GFP expression in the trichome tip cells of stably transformed M82 plants (Figure 3C). This supports our hypothesis that Sl-AACS1 and Sl-AECH1 are involved in tomato trichome acylsugar biosynthesis. Taken together, we identified a metabolic gene cluster involved in medium chain acylsugar biosynthesis, which is composed of two cell-type specific genes.
In vitro analysis of Sl-AACS1 and Sl-AECH1 implicates their roles in medium chain acyl-CoA metabolism
ACS and ECH are established to function in multiple cell compartments for the metabolism of acyl-CoA (Buchanan et al., 2015), the acyl donor substrates for ASAT enzymes. We sought to understand the organelle targeting of Sl-AACS1 and Sl-AECH1, to advance our knowledge of acylsugar machinery at the subcellular level. We constructed expression cassettes of Sl-AACS1, Sl-AECH1 and Solyc07g043660 with C-terminal cyan fluorescent protein (CFP), hypothesizing that the targeting peptides reside at the N-terminus of precursor proteins. When co-expressed in tobacco leaf epidermal cells, three CFP-tagged recombinant proteins co-localized with the mitochondrial marker MT-RFP (Nelson et al., 2007; Figure 4A and Figure 4—figure supplement 1A). To rule out the possibility of peroxisomal localization, we fused Sl-AACS1, Sl-AECH1, or Solyc07g043660 with N-terminus fused yellow fluorescent protein (YFP), considering that potential peroxisomal targeting peptides are usually located on the C-terminus (Brocard and Hartig, 2006). The expressed YFP-recombinant proteins were not co-localized with the peroxisomal marker RFP-PTS (Nelson et al., 2007; Figure 4—figure supplement 1B). Instead, they appeared distributed in the cytosol (Figure 4—figure supplement 1B), presumably because the N-terminal YFP blocked the mitochondria targeting signal. Taken together, protein expression and co-localization analyses suggest that Sl-AACS1, Sl-AECH1, and Solyc07g043660 encode enzymes targeted to mitochondria.
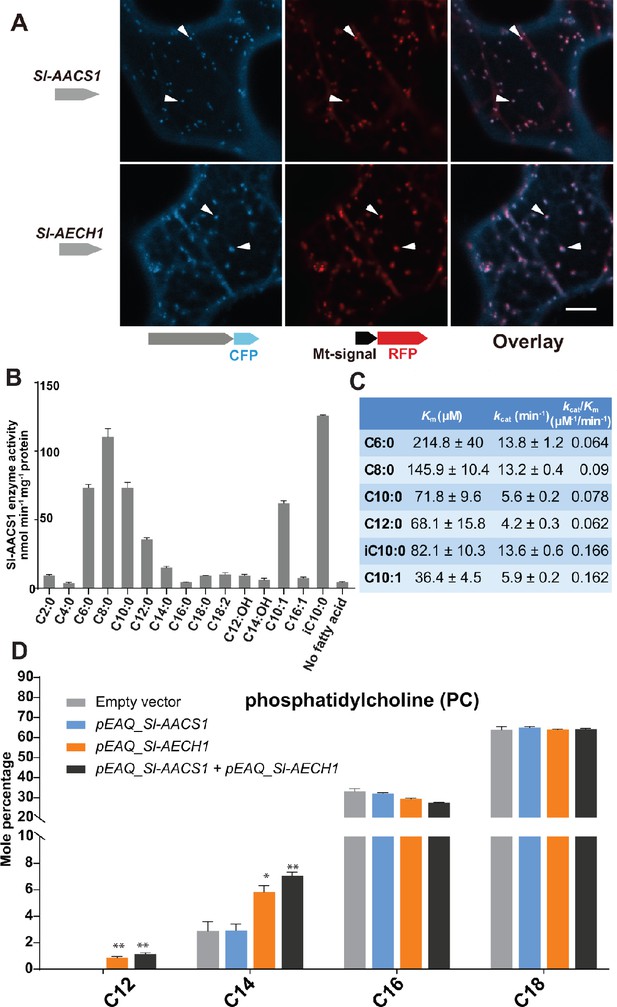
Functional analysis of Sl-AACS1 and Sl-AECH1 in N. benthamiana and recombinant Sl-AACS1 enzyme analysis.
(A) Confocal images of co-expression analysis in tobacco leaf epidermal cells using C-terminal CFP-tagged either Sl-AACS1 or Sl-AECH1 and the mitochondrial marker MT-RFP. Arrowheads point to mitochondria that are indicated by MT-RFP fluorescent signals. Scale bar equals 10 μm. Figure 4—figure supplement 1B describes that the expressed YFP-recombinant proteins were not co-localized with the peroxisomal marker RFP-PTS (B) Aliphatic fatty acids of different chain lengths were used as the substrates to test Sl-AACS1 acyl-CoA synthetase activity. Mean amount of acyl-CoAs generated (nmol min−1 mg−1 proteins) was used to represent enzyme activities. The results are from three measurements ± SEM. Figure 4—source data 1 includes values for the measurements. (C) Enzyme activity of Sl-AACS1 for six fatty acid substrates. (D) Identification of membrane lipid phosphatidylcholine (PC), which contains medium acyl chains, following transient expression of Sl-AECH1 in N. benthamiana leaves. The results from expressing Sl-AACS1 and co-expressing both Sl-AECH1 and Sl-AACS1 are also shown. Mole percentage (Mol %) of the acyl chains from membrane lipids with carbon number 12, 14, 16, and 18 are shown for three biological replicates ± SEM. *p<0.05, **p<0.01. Welch two-sample t test was performed comparing with the empty vector control. Figure 4—source data 2 includes values for the lipid analysis. Acyl groups of the same chain lengths with saturated and unsaturated bonds were combined in the calculation. Figure 4—figure supplement 2 shows that the putative Sl-AECH1 orthologs from S. pennellii and S. quitoense generated medium chain lipids in the infiltrated leaves.
-
Figure 4—source data 1
Data used to make Figure 4B.
Aliphatic fatty acids of different chain lengths were used as the substrates to test Sl-AACS1 acyl-CoA synthetase activity.
- https://cdn.elifesciences.org/articles/56717/elife-56717-fig4-data1-v2.xlsx
-
Figure 4—source data 2
Data used to make Figure 4D and Figure 4—figure supplement 1C.
N. benthamiana leaf membrane lipid acyl chain composition.
- https://cdn.elifesciences.org/articles/56717/elife-56717-fig4-data2-v2.xlsx
Sl-AACS1 belongs to a group of enzymes that activate diverse carboxylic acid substrates to produce acyl-CoAs. We hypothesized that Sl-AACS1 uses medium chain fatty acids as substrates, because ablation of Sl-AACS1 eliminated acylsugars with medium acyl chains. To characterize the in vitro activity of Sl-AACS1, we purified recombinant His-tagged proteins from Escherichia coli. Enzyme assays were performed by supplying fatty acid substrates with even carbon numbers from C2 through C18 (Figure 4B). The results showed that Sl-AACS1 utilized fatty acid substrates with lengths ranging from C6 to C12, including those with a terminal branched carbon (iC10:0) or an unsaturated bond (trans-2-decenoic acid, C10:1) (Figure 4, B and C). However, no activity was observed with the 3-hydroxylated C12 and C14 fatty acids as substrates (Figure 4B). These results support our hypothesis that Sl-AACS1 produces medium chain acyl-CoAs, which are in vivo substrates for acylsugar biosynthesis.
To test whether Sl-AACS1 and Sl-AECH1 can produce medium chain acyl-CoAs in planta, we transiently expressed these genes in Nicotiana benthamiana leaves using Agrobacterium-mediated infiltration (Sainsbury et al., 2009). It is challenging to directly measure plant acyl-CoAs, due to their low concentration and separate organellar pools. We used an alternative approach and characterized membrane lipids, which are produced from acyl-CoA intermediates. We took advantage of the observation that N. benthamiana membrane lipids do not accumulate detectable acyl chains of 12 carbons or shorter. N. benthamiana leaves were infiltrated with constructs containing Sl-AACS1 or Sl-AECH1 individually, or together (Figure 4D). In contrast to the empty vector control, infiltration of Sl-AECH1 led to detectable levels of C12 acyl chains in the leaf membrane lipid phosphatidylcholine (PC) (Figure 4D). We also observed increased C14 acyl chains in PC, phosphatidylglycerol (PG), sulfoquinovosyl diacylglycerol (SQDG), and digalactosyldiacylglycerol (DGDG) in Sl-AECH1 infiltrated plants (Figure 4D and Figure 4—figure supplement 1C). These results suggest that Sl-AECH1 participates in generation of medium chain acyl-CoAs in planta, which are channeled into lipid biosynthesis. No medium chain acylsugars were detected, presumably due to the lack of core acylsugar biosynthetic machinery in N. benthamiana mesophyll cells.
We asked whether the closest known homologs of Sl-AECH1 from Solanum species can generate medium chain lipids when transiently expressed in N. benthamiana. Two SQDGs with C12 chains were monitored by LC/MS as peaks diagnostic of lipids containing medium chain fatty acids (Figure 4—figure supplement 2, A and B). The results showed that only the putative Sl-AECH1 orthologs Sopen07g023250 (Sp-AECH1) and Sq_c37194 (Sq-AECH1) – from S. pennellii and S. quitoense respectively – generated medium chain lipids in the infiltrated leaves (Figure 4—figure supplement 2C). This confirms that not all ECHs can produce medium chain lipids and suggests that the function of Sl-AECH1 evolved recently, presumably as a result of neofunctionalization after gene duplication (Figure 4—figure supplement 2C).
AACS1 and AECH1 are evolutionarily conserved in the Solanum
Medium chain acylsugars were documented in Solanum species besides cultivated tomato, including S. pennellii (Leong et al., 2019), S. nigrum (Moghe et al., 2017), as well as the more distantly related S. quitoense (Leong et al., 2020; Hurney, 2018) and S. lanceolatum (Herrera-Salgado et al., 2005). We hypothesized that evolution of AACS1 and AECH1 contributed to medium chain acylsugar biosynthesis in Solanum. As a test, we analyzed the genomes of Solanum species other than cultivated tomato. Indeed, the acylsugar related synteny containing ACS and ECH was found in both S. pennellii and S. melongena (eggplant), suggesting that the cluster assembly evolved before divergence of the tomato and eggplant lineage (Figure 5A).
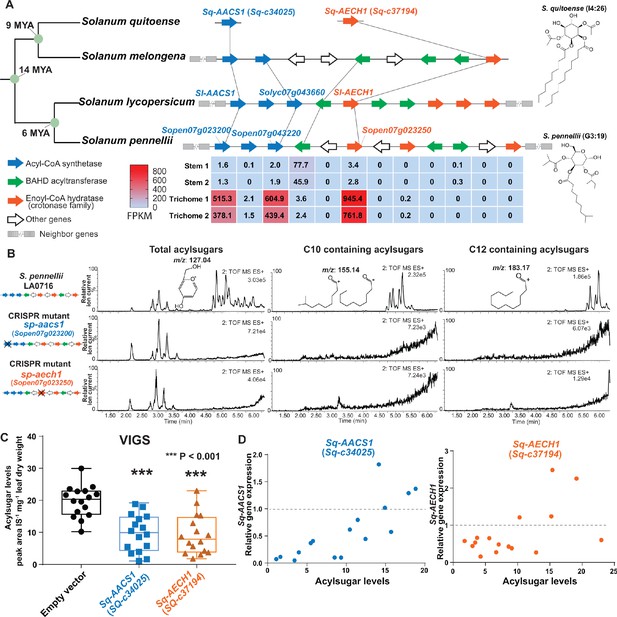
AACS1 and AECH1 are evolutionarily conserved in Solanum plants.
(A) A conserved syntenic genomic region containing AACS1 and AECH1 was found in three selected Solanum species. Nodes representing estimated dates since the last common ancestors (Särkinen et al., 2013) shown on the left. The closest homologs of AACS1 and AECH1 in Solanum quitoense are shown without genomic context because the genes were identified from RNA-seq and genome sequences are not available. The lines connect genes representing putative orthologs across the four species. The trichome/stem RNA-seq data of two biological S. pennellii replicates are summarized (Supplementary file 2) for genes in the syntenic region. The red-blue color gradient provides a visual marker to rank the expression levels in FPKM. Structures of representative medium chain acylsugars from S. quitoense (acylinositol, I4:26) (Hurney, 2018) and S. pennellii (acylglucose, G3:19) (Leong et al., 2019) are on the right. Figure 5—figure supplement 1 shows that stable Sp-AACS1 transformation of the M82 CRISPR mutant sl-aacs1 restores C12 containing acylsugars (B) CRISPR/Cas9-mediated gene knockout of Sp-AACS1 or Sp-AECH1 in S. pennellii produce no detectable medium chain containing acylsugars. The ESI+ mode LC/MS extracted ion chromatograms of trichome metabolites are shown for each mutant. The m/z 127.01 (left panel) corresponds to the glucopyranose ring fragment that both acylsucroses and acylglucoses generate under high collision energy positive-ion mode. The m/z 155.14 (center panel) and 183.17 (right panel) correspond to the acylium ions from acylsugars with chain length of C10 and C12, respectively. Figure 5—figure supplement 2A–C describes the design of the gRNAs and the detailed information of gene edits. (C) Silencing Sq-AACS1 (Sq-c34025) or Sq-AECH1 (Sq-c37194) in S. quitoense using VIGS leads to reduction of total acylsugars. The peak area/internal standard (IS) normalized by leaf dry weight was shown from sixteen plants ± SEM. ***p<0.001, Welch two-sample t test. Figure 5—figure supplement 2E and F describes the VIGS experimental design and the representative LC/MS extracted ion chromatograms of S. quitoense major acylsugars. (D) Reduced gene expression of Sq-AACS1 or Sq-AECH1 correlates with decreased acylsugar levels in S. quitoense. The qRT-PCR gene expression data are plotted with acylsugar levels of the same leaf as described in Figure 5—figure supplement 2E. Figure 5—source data 1 includes raw data for the S. quitoense VIGS experiments.
-
Figure 5—source data 1
Data used to make Figure 5C and D.
Silencing Sq-AACS1 or Sq-AECH1 in S. quitoense using VIGS leads to reduction of total acylsugars.
- https://cdn.elifesciences.org/articles/56717/elife-56717-fig5-data1-v2.xlsx
We applied gene expression and genetic approaches to test the in vivo functions of ACS and ECH in selected Solanum species. To explore the expression pattern of S. pennellii ACS and ECH cluster genes, we performed RNA-seq analysis on trichomes and shaved stems to identify acylsugar biosynthetic candidates (Supplementary file 2). The expression pattern of S. pennellii cluster genes is strikingly similar to S. lycopersicum: one ECH and two ACS genes are highly enriched in trichomes, including the orthologs of Sl-AACS1 and Sl-AECH1. Sp-AACS1 function (Sopen07g023200) was first tested by asking whether it can reverse the cultivated tomato sl-aacs1 mutant acylsugar phenotype. Indeed, Sp-AACS1 restored C12 containing acylsugars in the stably transformed sl-aacs1 plants (Figure 5—figure supplement 1). To directly test Sp-AACS1 and Sp-AECH1 function, we used CRISPR-Cas9 to make single mutants in S. pennellii LA0716. No medium chain acylsugars were detected in T0 generation mutants with edits for each gene (Figure 5B and Figure 5—figure supplement 2, A and C). Similar to the ACS-annotated Solyc07g043660 cultivated tomato mutant (Figure 3—figure supplement 1D), deletion of S. pennellii ortholog Sopen07g023220 has no observed effects on S. pennellii trichome acylsugars (Figure 5—figure supplement 2D).
The medium chain acylsugar producer S. quitoense (Hurney, 2018) was used for AACS1 and AECH1 functional analysis because of its phylogenetic distance from the tomato clade - it is in the Solanum Leptostemonum clade (including eggplant) - and the fact that it produces medium chain acylsugars. We found trichome-enriched putative orthologs of AACS1 and AECH1 in the transcriptome dataset of S. quitoense (Moghe et al., 2017), and tested their in vivo function through virus-induced gene silencing (VIGS) (Figure 5—figure supplement 2E). Silencing either gene led to decreased total acylsugars (Figure 5C and Figure 5—figure supplement 2F), which correlated with the degree of expression reduction in each sample (Figure 5D). These results are consistent with the hypothesis that Sq-AACS1 and Sq-AECH1 are involved in medium chain acylsugar biosynthesis, because all acylsugars in S. quitoense carry two medium chains (Leong et al., 2020; Hurney, 2018). The importance of AACS1 and AECH1 in medium chain acylsugar biosynthesis in distinct Solanum clades inspired us to explore the evolutionary origins of the gene cluster.
Evolution of the gene cluster correlates with the distribution of medium chain acylsugars across Solanaceae
We sought to understand how the acylsugar gene cluster evolved and whether it correlates with the distribution of medium chain acylsugars across the Solanaceae family. Taking advantage of the available genome sequences of 13 species from Solanaceae and sister families, we analyzed the regions that are syntenic with the tomato acylsugar gene cluster (Figure 6—figure supplement 1). This synteny was found in all these plants, including the most distantly related species analyzed, Coffea canephora (coffee, Rubiaceae) (Figure 6—figure supplement 1). BAHD acyltransferases were the only genes observed in the syntenic regions both inside and outside the Solanaceae, in contrast to ECH and ACS, which are restricted to the family (Figure 6A and Figure 6—figure supplement 1). Within the syntenic regions of the species analyzed, ECH homologs, including pseudogenes, are present in all Solanaceae except for Capsicum species, while ACS is more phylogenetically restricted, being found only in Nicotiana and Solanum (Figure 6A and Figure 6—figure supplement 1).
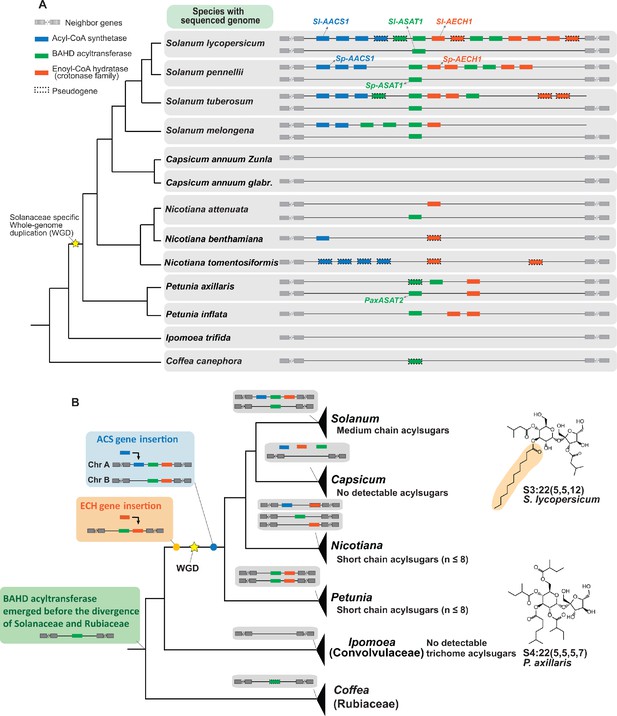
Evolution of the acylsugar gene cluster is associated with acylsugar acyl chain diversity across the Solanaceae family.
(A) The acylsugar gene cluster syntenic regions of 11 Solanaceae species and two outgroup species Ipomea trifida (Convolvulaceae) and Coffea canephora (Rubiaceae). This is a simplified version adapted from Figure 6—figure supplement 1. Only genes from the three families – ACS (blue), BAHD acyltransferase (green), and ECH (orange) – are shown. For information about the syntenic region size in each species refer to Figure 6—figure supplement 1 and Supplementary file 4. (B) The evolutionary history of the acylsugar gene cluster and its relation to the acylsugar phenotypic diversity. The evolution of BAHD acyltransferase genes is inferred based on Figure 6—figure supplement 2 and Figure 6—figure supplement 5. ECH genes based on Figure 6—figure supplement 3. ACS genes based on Figure 6—figure supplement 4 and Figure 6—figure supplement 6. The temporal order for the emergence for the three types of genes are shown in the colored boxes on the left: green box (BAHD acyltransferase), orange box (ECH), blue box (ACS). The yellow star represents the Solanaceae-specific WGD. Structures of representative short and medium chain acylsugars were shown on the right. Figure 6—figure supplement 8 describes distribution of acylsugar acyl chains with different lengths in species across the Solanaceae family.
We then performed phylogenetic analysis to reconstruct the evolutionary history of the ACS, ECH, and BAHD acyltransferase genes in the syntenic region (Figure 6). This analysis revealed a model for the temporal order of emergence for the three types of genes, leading to their presence in the syntenic regions in extant Solanaceae plants (Figure 6B). We propose that the BAHD acyltransferase was the first of three genes that emerged before the divergence between Solanaceae and Rubiaceae, and was likely lost in Convolvulaceae. This hypothesis is based on the discovery of a BAHD acyltransferase pseudogene in the syntenic region of C. canephora (Figure 6A and Figure 6—figure supplement 1), which is one of the closest Coffea sequences sister to the ASAT clade (Figure 6—figure supplement 2 and Figure 6—figure supplement 5). In our model, ECH was likely inserted into the syntenic region before the Solanaceae-specific whole genome duplication (WGD) event (Figure 6B and Figure 6—figure supplement 3).
We propose that ACS was inserted into the synteny through segmental duplication (Bailey et al., 2002; Figure 6—figure supplement 4). However, whether ACS insertion happened before or after the Solanaceae-specific WGD event cannot be resolved by the phylogenetic analysis (Figure 6—figure supplement 4). If the insertion happened before WGD, one ACS gene loss on chromosome 12 in Solanum – as well as two independent gene losses on chromosomes 7 and 12 in both Petunia (Figure 6—figure supplement 4) and in Salpiglossis sinuata (Figure 6—figure supplement 6) – should have happened. However, if the insertion happened after WGD, then only one gene loss in Petunia and Salpiglossis needs to be invoked (Figure 6—figure supplement 6). The latter scenario is more likely based on the principle of parsimony.
Our ancestral state reconstruction inference supports the notion that the medium chain acylsugars co-emerged with the ACS/ECH genes in the syntenic regions in the common ancestor of Solanum (Figure 6—figure supplement 7). This leads us to propose that the emergence of both ACS and ECH genes in the synteny was a prerequisite for the rise of medium chain acylsugars in Solanaceae species (Figure 6B). Consistent with the hypothesis, only short chain acylsugars were observed in Petunia (Liu et al., 2017; Figure 6—figure supplement 8), which correlates with the absence of ACS homolog (Figure 6B). In contrast, medium chain acylsugars were detected throughout Solanum (Figure 6—figure supplement 8), supported by the observation that both ACS and ECH homologs are present in extant Solanum species (Figure 6B). Interestingly, although Nicotiana species collectively have both ACS and ECH genes (Figure 6B), they do not produce medium chain acylsugars (Figure 6—figure supplement 8) presumably due to gene losses. For example, the ECH homologs are pseudogenes in N. benthamiana and N. tomentosiformis (Figure 6A). These results show that the presence of both functional ACS and ECH genes are associated with the accumulation of medium chain acylsugars, supporting our hypothesis above.
Although no medium chain acylsugars were detected in Nicotiana species examined, the ACS and ECH genes may have been present in the syntenic region prior to divergence of Solanum and Nicotiana. This suggests that one or more species that diverged from the common ancestor of Solanum and Nicotiana could have medium chain acylsugars. To test this hypothesis, we extended the phenotypic analysis to six such Solanaceae genera (Figure 6—figure supplement 8). Indeed, we found that species in Jaltomata, Physalis, Iochroma, Atropa, and Hyoscyamus, which diverged from the common ancestor with Nicotiana but before Solanum, have medium chain acylsugars (Figure 6—figure supplement 8).
Discussion
This study identified a S. lycopersicum chromosome 7 synteny of ACS, ECH, and BAHD acyltransferase genes including two involved in medium chain acylsugar biosynthesis. The discovery of this locus was prompted by our observation of increased C10 containing acylsugars in tomato recombinant lines carrying this region from the wild tomato S. pennellii chromosome 7. In vitro biochemistry revealed that Sl-AACS1 produces acyl-CoAs using C6-C12 fatty acids as substrates. The function of AACS1 and AECH1 in cultivated and wild tomato medium chain acylsugar biosynthesis was confirmed by genome editing, and extended to the phylogenetically distant S. quitoense using VIGS. The trichome tip cell-specific expression of these genes is similar to that of previously characterized acylsugar pathway genes (Fan et al., 2019).
There are increasing examples of plant specialized metabolic innovation evolving from gene duplication and neofunctionalization of primary metabolic enzymes (Maeda, 2019; Milo and Last, 2012; Moghe and Last, 2015; Zi et al., 2014). Recruitment of Sl-AACS1 and Sl-AECH1 from fatty acid metabolism provides new examples of ‘hijacking’ primary metabolic genes into acylsugar biosynthesis, in addition to an isopropylmalate synthase (Sl-IPMS3) and an invertase (Sp-ASFF1) (Leong et al., 2019; Ning et al., 2015). We hypothesize that both AACS1 and AECH1 participate in generation of medium chain acyl-CoAs, the acyl donor substrates for ASAT-catalyzed acylsugar biosynthesis. Indeed, Sl-AACS1 exhibits in vitro function consistent with this hypothesis, efficiently utilizing medium chain fatty acids as substrates to synthesize acyl-CoAs. Strikingly, Sl-AECH1 perturbs membrane lipid composition when transiently expressed in N. benthamiana leaves, generating unusual C12-chain membrane lipids.
These results suggest that evolution of trichome tip cell-specific gene expression potentiated the co-option of AACS1 and AECH1 in medium chain acylsugar biosynthesis. Analogous to trichomes producing medium chain acylsugars, seeds of phylogenetically diverse plants accumulate medium chain fatty acid storage lipids (Ohlrogge et al., 2018). In contrast, fatty acids with unusual structures, including those of medium chain lengths, are rarely found in membrane lipids, presumably because these would perturb membrane bilayer structure and function (Millar et al., 2000). For example, seed embryo-specific expression of three neofunctionalized enzyme variant genes in Cuphea species – an acyl-ACP thioesterase (Dehesh et al., 1996), a 3-ketoacyl-ACP synthase (Dehesh et al., 1998), and a diacylglycerol acyltransferase (Iskandarov et al., 2017) – lead to production of medium chain seed storage lipids (Voelker and Kinney, 2001). Trichome tip cell restricted expression of AACS1 and AECH1 represents an analogous example of diverting neofunctionalized fatty acid enzymes from general metabolism into cell-specific specialized metabolism. It is notable that we obtained evidence that Sl-AACS1 and Sl-AECH1 are targeted to mitochondria. Because the other characterized acylsugar biosynthetic enzymes – ASATs and Sl-IPMS3 – appear to be cytoplasmic, these results suggest that medium chain acylsugar substrates are intracellularly transported within the trichome tip cell. It is worth noting that Sl-AACS1 seems to show higher activity with C8 fatty acid than with C10 or C12 (Figure 4, B and C), while no C8 containing acylsugars were described in tomato trichomes (Ghosh et al., 2014). This suggests that C8 fatty acids are not synthesized in trichomes.
Beyond employing functional approaches, this study demonstrates the value of a combined comparative genomic and metabolomic analysis in reconstructing the evolutionary history of a gene cluster: in this case over tens of millions of years. We propose that the acylsugar gene cluster started with a ‘founder’ BAHD acyltransferase gene, followed by sequential insertion of ECH and ACS (Figure 6B). This de novo assembly process is analogous to evolution of the antimicrobial triterpenoid avenacin cluster in oat (Qi et al., 2006; Qi et al., 2004). There are two noteworthy features of our approach. First, reconstructing acylsugar gene cluster evolution in a phylogenetic context allows us to deduce cluster composition in extant species (Figure 6B). Second, it links cluster genotype with acylsugar phenotype and allows inference of acylsugar diversity across the Solanaceae (Figure 6 and Figure 6—figure supplement 8).
The current architecture of the Solanaceae acylsugar synteny merely represents a snapshot of a genomic neighborhood that is dynamic, which echoes a recent study of triterpene biosynthetic gene clusters in the Brassicaceae (Liu et al., 2019; Peters, 2020). De novo assembly of the gene cluster was accompanied by gene duplication, transposition, pseudogenization, and deletion in different genera. In the case of non-acylsugar producer Capsicum, although phylogenetic analysis revealed putative Sl-AACS1 and Sl-AECH1 orthologous genes, they are not harbored in the syntenic region, probably due to translocation or assembly quality issues (Figure 6A and Figure 6—figure supplement 1). In Nicotiana, the ECH genes became pseudogenized (Figure 6B), which is associated with lack of detectable plant medium chain acylsugars (Figure 6—figure supplement 8). In tomatoes, the trichome expressed Solyc07g043660 derives from a recent tandem duplication (Figure 6—figure supplement 4), yet its deletion has no effect on trichome acylsugars (Figure 3—figure supplement 1D). A parsimonious explanation is that Solyc07g043660 is experiencing functional divergence, which may eventually lead to pseudogenization as observed for other genes in the syntenic region.
In this study, we identified an acylsugar SMGC in the context of a multiple chromosome syntenic region. This synteny resulted from WGD, and the acylsugar-related genes are co-expressed, and involved in the same metabolic pathway. This resembles the tomato steroidal alkaloid gene cluster consisting of eight genes that are dispersed into two syntenic chromosome regions (Itkin et al., 2013). In fact, this tomato alkaloid SMGC is located next to the acylsugar cluster (Figure 3—figure supplement 2), which is reminiscent of two physically adjacent SMGCs in the fungus Aspergillus (Wiemann et al., 2013). Tomato steroidal alkaloids and acylsugars both serve defensive roles in plants, but are biosynthetically and structurally distinct and are stored in different tissues. This raises intriguing questions. Did the separation of acylsugar and alkaloid SMGCs into two chromosomes occur contemporaneously and by the same mechanism? Did this colocalization confer selective advantage through additive or synergistic effects of multiple classes of defensive metabolites? Answering these questions requires continued mining and functional validation of metabolic gene clusters across broader plant species and analysis of impacts of clustering in evolutionary and ecological contexts.
Materials and methods
| Reagent type (species) or resource | Designation | Source or reference | Identifiers | Additional information |
|---|---|---|---|---|
| Gene (Solanum lycopersicum M82) | Sl-AACS1 | This paper | GeneBank: MT078737 | Characterized and named in the results |
| Gene (Solanum lycopersicum M82) | Sl-AECH1 | This paper | GeneBank: MT078736 | Characterized and named in the results |
| Gene (Solanum pennellii LA0716) | Sp-AACS1 | This paper | GeneBank: MT078735 | Characterized and named in the results |
| Gene (Solanum pennellii LA0716) | Sp-AECH1 | This paper | GeneBank: MT078734 | Characterized and named in the results |
| Gene (Solanum quitoense) | Sq-AACS1 | This paper | GeneBank: MT078732 | Characterized and named in the results |
| Gene (Solanum quitoense) | Sq-AECH1 | This paper | GeneBank: MT078731 | Characterized and named in the results |
| Gene (Solanum quitoense) | Sq_c35719 | This paper | GeneBank: MT078733 | Characterized and named in the results |
| Software, algorithm | Trimmomatic | http://www.usadellab.org/cms/index.php?page=trimmomatic | RRID:SCR_011848 | |
| Software, algorithm | TopHat | http://ccb.jhu.edu/software/tophat/index.shtml | RRID:SCR_013035 | |
| Software, algorithm | Cufflinks | http://cole-trapnell-lab.github.io/cufflinks/cuffmerge/ | RRID:SCR_014597 | |
| Software, algorithm | MCScanX-transposed | http://chibba.pgml.uga.edu/mcscan2/transposed/ | ||
| Software, algorithm | RAxML | https://github.com/stamatak/standard-RAxML | RRID:SCR_006086 | |
| Software, algorithm | Mesquite | https://www.mesquiteproject.org/ | RRID:SCR_017994 |
Plant materials and trichome metabolite extraction
Request a detailed protocolThe seeds of cultivated tomato Solanum lycopersicum M82 were obtained from the C.M. Rick Tomato Genetic Resource Center (https://tgrc.ucdavis.edu/), RRID:SCR_014954. Tomato introgression lines (ILs) and tomato backcross inbred lines (BILs) were from Dr. Dani Zamir (Hebrew University of Jerusalem). The tomato seeds were treated with ½ strength bleach for 30 min and washed with de-ionized water three or more times before placing on wet filter paper in a Petri dish. After germination, the seedlings were transferred to peat-based propagation mix (Sun Gro Horticulture) and transferred to a growth chamber for two or three weeks under 16 hr photoperiod, 28°C day and 22°C night temperatures, 50% relative humidity, and 300 μmol m−2 s−1 photosynthetic photon flux density. The youngest fully developed leaf was submerged in 1 mL extraction solution in a 1.5 mL screw cap tube and agitated gently for 2 min. The extraction solution contains acetonitrile/isopropanol/water (3:3:2) with 0.1% formic acid and 10 μM propyl-4-hydroxybenzoate as internal standard. The interactive protocol of acylsugar extraction is available in Protocols.io at http://dx.doi.org/10.17504/protocols.io.xj2fkqe.
DNA construct assembly and tomato transformation
Request a detailed protocolAssembly of the CRISPR-Cas9 constructs was as described (Brooks et al., 2014; Leong et al., 2019). Two guide RNAs (gRNAs) were designed targeting one or two exons of each gene to be knocked out by the CRISPR-Cas9 system. The gRNAs were obtained from gene blocks (gBlocks) synthesized by IDT (Integrated DNA Technologies, location) (Supplementary file 3). For each CRISPR construct, two gBlocks and four plasmids from Addgene, pICH47742::2 × 35 S-5′UTR-hCas9 (STOP)-NOST (Addgene no. 49771), pICH41780 (Addgene no. 48019), pAGM4723 (Addgene no. 48015), pICSL11024 (Addgene no. 51144), were mixed for DNA assembly using the Golden Gate assembly kit (NEB).
For in planta tissue specific reporter gene analysis, 1.8 kb upstream of the annotated translational start site of Sl-AACS1 and Sl-AECH were amplified using the primer pairs SlAACS1-pro_F/R and SlAECH1-pro_F/R (Supplementary file 3). The amplicon was inserted into the entry vector pENTR/D-TOPO, followed by cloning into the GATEWAY vector pKGWFS7. For ectopically expressing Sp-AACS1 in the cultivated tomato CRISPR mutant sl-aacs1, Sp-AACS1 gene including 1.8 kb upstream of the translational start site of Sp-AACS1 was amplified using the primer pair SpAACS1-pro-gene_F/R (Supplementary file 3). The amplicon was inserted into the entry vector pENTR/D-TOPO, followed by cloning into the GATEWAY vector pK7WG.
The plant transformation of S. lycopersicum M82 and S. pennellii LA0716 was performed using the Agrobacterium tumefaciens strain AGL0 following published protocols (Leong et al., 2019; McCormick, 1997). The primers used for genotyping the S. lycopersicum M82 transgenic plants harboring pK7WG or pKGWFS7 construct are listed in Supplementary file 3. For genotyping the S. lycopersicum M82 CRISPR mutants in the T1 generation, the sequencing primers listed in Supplementary file 3 were used to amplify the genomic regions harboring the gRNAs and the resultant PCR products were sent for Sanger sequencing. For genotyping the S. pennellii LA0716 CRISPR mutants in the T0 generation, the sequencing primers listed in Supplementary file 3 were used to amplify the genomic regions harboring the gRNAs. The resulting PCR products were cloned into the pGEM-T easy vector (Promega) and transformed into E. coli. Plasmids from at least six individual E. coli colonies containing the amplified products were extracted and verified by Sanger sequencing.
Protein subcellular targeting in tobacco mesophyll cells
Request a detailed protocolFor protein subcellular targeting analysis, the open reading frame (ORF) of Sl-AACS1, Sl-AECH1, and Solyc07g043660 were amplified using the primers listed in Supplementary file 3. These amplicons were inserted into pENTR/D-TOPO respectively, followed by subcloning into the GATEWAY vectors pEarleyGate102 (no. CD3-684) and pEarleyGate104 (no. CD3-686), which were obtained from Arabidopsis Biological Resource Center (ABRC). For the pEarleyGate102 constructs, the CFP was fused to the C-terminal of the tested proteins. For the pEarleyGate104 constructs, the YFP was fused to the protein N-terminus. Transient expressing the tested proteins was performed following an established protocol (Batoko et al., 2000) with minor modifications. In brief, cultures of A. tumefaciens (strain GV3101) harboring the expression vectors were washed and resuspended with the infiltration buffer (20 mM acetosyringone, 50 mM MES pH 5.7, 0.5% glucose [w/v] and 2 mM Na3PO4) to reach OD600nm = 0.05. Four-week-old tobacco (Nicotiana tabacum cv. Petit Havana) plants grew in 21°C and 8 hr short-day conditions were infiltrated, and then maintained in the same growth condition for three days before being sampled for imaging. The GV3101 cultures containing the mitochondria marker MT-RFP (Nelson et al., 2007) were co-infiltrated to provide the control signals for mitochondrial targeting. In separate experiments, the GV3101 cultures containing the peroxisome marker RFP-PTS (Nelson et al., 2007) were co-infiltrated to provide the control signals for peroxisomal targeting.
Confocal microscopy
Request a detailed protocolA Nikon A1Rsi laser scanning confocal microscope and Nikon NIS-Elements Advanced Research software were used for image acquisition and handling. For visualizing GFP fluorescence in trichomes of the tomato transformants, the excitation wavelength at 488 nm and a 505- to 525 nm emission filter were used for the acquisition. For visualizing signals of fluorescence proteins in the tobacco mesophyll cells, CFP, YFP and RFP, respectively, were detected by excitation lasers of 443 nm, 513 nm, 561 nm and emission filters of 467–502 nm, 521–554 nm, 580–630 nm.
N. benthamiana transient gene expression and membrane lipid analysis
Request a detailed protocolFor N. benthamiana transient expression of Sl-AACS1, Sl-AECH1, and homologs of AECH1, the ORFs of these genes were amplified using primers listed in Supplementary file 3, followed by subcloning into pEAQ-HT vector using the Gibson assembly kit (NEB). Linearization of pEAQ-HT vector was performed by XhoI and AgeI restriction enzyme double digestion. A. tumefaciens (strain GV3101) harboring the pEAQ-HT constructs were grown in LB medium containing 50 µg/mL kanamycin, 30 µg/mL gentamicin, and 50 µg/ml rifampicin at 30 °C. The cells were collected by centrifugation at 5000 g for 5 min and washed once with the resuspension buffer (10 mM MES buffer pH 5.6, 10 mM MgCl2, 150 µM acetosyringone). The cell pellet was resuspended in the resuspension buffer to reach OD600nm = 0.5 for each strain and was incubated at room temperature for 1 hr prior to infiltration. Leaves of 4 to 5 week-old N. benthamiana grown under 16 hr photoperiod were used for infiltration. Five days post infiltration, the infiltrated leaves were harvested, ground in liquid nitrogen, and stored at −80 °C for later analysis.
The membrane lipid analysis was performed as previously described (Wang and Benning, 2011). In brief, the N. benthamiana leaf polar lipids were extracted in the organic solvent containing methanol, chloroform, and formic acid (20:10:1, v/v/v), separated by thin layer chromatography (TLC), converted to fatty acyl methylesters (FAMEs), and analyzed by gas-liquid chromatography (GLC) coupled with flame ionization. The TLC plates (TLC Silica gel 60, EMD Chemical) were activated by ammonium sulfate before being used for lipid separation. Iodine vapor was applied to TLC plates after lipid separation for brief reversible staining. Different lipid groups on the TLC plates were marked with a pencil and were scraped for analysis. For LC/MS analysis, lipids were extracted using the buffer containing acetonitrile/isopropanol/water (3:3:2) with 0.1% formic acid and 10 μM propyl-4-hydroxybenzoate as the internal standard.
Protein expression and ACS enzyme assay
Request a detailed protocolTo express His-tagged recombinant protein Sl-AACS1, the full-length Sl-AACS ORF sequence was amplified using the primer pair SlAACS1-pET28_F/R (Supplementary file 3) and was cloned into pET28b (EMD Millipore) using the Gibson assembly kit (NEB). The pET28b vector was linearized by digesting with BamHI and XhoI to create overhangs compatible for Gibson assembly. The pET28b constructs were transformed into BL21 Rosetta cells (EMD Millipore). The protein expression was induced by adding 0.05 mM isopropyl β-D-1-thiogalactopyranoside to the cultures when the OD600nm = 0.5. The E. coli cultures were further grown overnight at 16 °C, 120 rpm. The His-tagged proteins were purified by Ni-affinity gravity-flow chromatography using the Ni-NTA agarose (Qiagen) following the product manual.
Measurement of acyl-CoA synthetase activity was performed using minor modifications of the coupled enzyme assay described by Schneider et al., 2005. A multimode plate reader (PerkinElmer, mode EnVision 2104) compatible with the 96-well UV microplate was used for the assays. The fatty acid substrates were dissolved in 5% Triton X-100 (v/v) to make 5 mM stock solutions. The enzyme assay premix was prepared containing 0.1 M Tris-HCl (pH 7.5), 2 mM dithiothreitol, 5 mM ATP, 10 mM MgCl2, 0.5 mM CoA, 0.8 mM NADH, 250 µM fatty acid substrate, 1 mM phosphoenolpyruvate, 20 units myokinase (Sigma-Aldrich, catalog no. M3003), 10 units pyruvate kinase (Roche, 10128155001), 10 units lactate dehydrogenase (Roche, 10127230001), and was aliquoted 95 µL each to the 96-well microplate. The reaction was started by adding 5 µL (1–2 µg) proteins. The chamber of the plate reader was set to 30 °C and the OD at 340 nm was recorded every 5 min for 40 min. Oxidation of NADH, which is monitored by the decrease of OD340nm, was calculated using the NADH extinction coefficient 6.22 cm2 µmol−1. Every two moles of oxidized NADH is equivalent to one mole of acyl-CoA product generated in the reaction. To measure the parameters of enzyme kinetics, the fatty acid substrate concentration was varied from 0 to 500 µM, with NADH set at 1 mM. The fatty acid substrates, sodium acetate (C2:0), sodium butyrate (C4:0), sodium hexanoate (C6:0), sodium octanoate (C8:0), sodium decanoate (C10:0), sodium laurate (C12:0), sodium myristate (C14:0), sodium palmitate (C16:0), and sodium stearate (C18:0), were purchased from Sigma-Aldrich. Trans-2-decenoic acid (C10:1), 8-methylnonanoic acid (iC10:0), 3-hydroxy lauric acid (C12:OH), and 3-hydroxy myristic acid (C14:OH) were purchased from Cayman Chemical.
RNA extraction, sequencing, and differential gene expression analysis
Request a detailed protocolTotal RNA was extracted from trichomes isolated from stems and shaved stems of 7-week-old S. pennellii LA0716 plants using the RNAeasy Plant Mini kit (Qiagen) and digested with DNase I. A total of four RNA samples extracted from two tissues with two replicates were used for RNA sequencing. The sequencing libraries were prepared using the KAPA Stranded RNA-Seq Library Preparation Kit. Libraries went through quality control and quantification using a combination of Qubit dsDNA high sensitivity (HS), Applied Analytical Fragment Analyzer HS DNA and Kapa Illumina Library Quantification qPCR assays. The libraries were pooled and loaded onto one lane of an Illumina HiSeq 4000 flow cell. Sequencing was done in a 2 × 150 bp paired end format using HiSeq 4000 SBS reagents. Base calling was done by Illumina Real Time Analysis (RTA) v2.7.6 and output of RTA was demultiplexed and converted to FastQ format with Illumina Bcl2fastq v2.19.1.
The paired end reads were filtered and trimmed using Trimmomatic v0.32 (Bolger et al., 2014a) with the setting (LLUMINACLIP: TruSeq3-PE.fa:2:30:10 LEADING:3 TRAILING:3 SLIDINGWINDOW:4:30), and then mapped to the S. pennellii LA0716 genome v2.0 (Bolger et al., 2014a) using TopHat v1.4 (Trapnell et al., 2009) with the following parameters: -p (threads) 8, -i (minimum intron length) 50, -I (maximum intron length) 5000, and -g (maximum hits) 20. The FPKM (Fragments Per Kilobase of transcript per Million mapped reads) values for the genes were analyzed via Cufflinks v2.2 (Trapnell et al., 2010).For differential expression analysis, the HTseq package (Anders et al., 2015) in Python was used to get raw read counts, then Edge R version 3.22.5 (McCarthy et al., 2012) was used to compare read counts between trichome-only RNA and shaved stem RNA using a generalized linear model (glmQLFit).
VIGS and qRT-PCR
Request a detailed protocolFor VIGS analysis of Sq-AACS1 and Sq-AECH1 in S. quitoense, the fragments of these two genes, as well as the phytoene desaturase (PDS) gene fragment, were amplified using the primers listed in Supplementary file 3, cloned into pTRV2-LIC (ABRC no. CD3-1044) using the ligation-independent cloning method (Dong et al., 2007), and transformed into A. tumefaciens (strain GV3101). The VIGS experiments were performed as described (Leong et al., 2020). In brief, the Agrobacterium strains harboring pTRV2 constructs, the empty pTRV2, or pTRV1 were grown overnight in separate LB cultures containing 50 µg/mL kanamycin, 10 µg/mL gentamicin, and 50 µg/ml rifampicin at 30 °C. The cultures were re-inoculated in the induction media (50 mM MES pH5.6, 0.5% glucose [w/v], 2 mM NaH2PO4, 200 µM acetosyringone) for overnight growth. The cells were harvested, washed, and resuspended in the buffer containing 10 mM MES, pH 5.6, 10 mM MgCl2, and 200 µM acetosyringone with the OD600nm = 1. Different cultures containing pTRV2 constructs were mixed with equal volume of pTRV1 cultures prior to infiltration. The 2- to 3-week-old young S. quitoense seedlings grown under 16 hr photoperiod at 24°C were used for infiltration: the two fully expanded cotyledons were infiltrated. Approximately three weeks post inoculation, the fourth true leaf of each infiltrated plant was cut in half for acylsugar quantification and gene expression analysis, respectively. The onset of the albino phenotype of the control group infiltrated with the PDS construct was used as a visual marker to determine the harvest time and leaf selection for the experimental groups. At least fourteen plants were analyzed for each construct. The trichome acylsugars were extracted using the solution containing acetonitrile/isopropanol/water (3:3:2) with 0.1% formic acid and 1 μM telmisartan as internal standard, following the protocol at http://dx.doi.org/10.17504/protocols.io.xj2fkqe.
The leaf RNA was extracted using RNeasy Plant Mini kits (Qiagen) and digested with DNase I. The first-strand cDNA was synthesized by Superscript II (Thermofisher Scientific) using total RNA as templates. Quantitative real-time PCR was performed to analyze the Sq-AACS1 or Sq-AECH1 mRNA in S. quitoense leaves using the primers listed in Supplementary file 3. EF1α was used as a control gene. A QuantStudio 7 Flex Real-Time PCR System with Fast SYBR Green Master Mix (Applied Biosystems) was used for the analysis. The relative quantification method (2-ΔΔCt) was used to evaluate the relative transcripts levels.
LC/MS analysis
Request a detailed protocolTrichome acylsugars extracted from tomato IL and BILs were analyzed using a Shimadzu LC-20AD HPLC system connected to a Waters LCT Premier ToF mass spectrometer. Ten microliter samples were injected into a fused core Ascentis Express C18 column (2.1 mm ×10 cm, 2.7 μm particle size; Sigma-Aldrich) for reverse-phase separation with column temperature set at 40°C. The starting condition was 90% solvent A (0.15% formic acid in water) and 10% solvent B (acetonitrile) with flow rate set to 0.4 mL/min. A 7 min linear elution gradient was used: ramp to 40% B at 1 min, then to 100% B at 5 min, hold at 100% B to 6 min, return to 90% A at 6.01 min and hold until 7 min.
For analyzing trichome acylsugars extracted from S. pennellii transgenic plants and membrane lipids from N. benthamiana, a Shimadzu LC-20AD HPLC system connected to a Waters Xevo G2-XS QToF mass spectrometer was used. The starting conditions were 95% solvent A (10 mM ammonium formate, pH 2.8) and 5% solvent B (acetonitrile) with flow rate set to 0.3 mL/min. A 7 min linear elution gradient used for acylsugar analysis was: ramp to 40% B at 1 min, then to 100% B at 5 min, hold at 100% B to 6 min, return to 95% A at 6.01 min and hold until 7 min. A 12 min linear elution gradient used for the lipid analysis was: ramp to 40% B at 1 min, then to 100% B at 5 min, hold at 100% B to 11 min, return to 95% A at 11.01 min and hold until 12 min.
For analyzing trichome acylsugars extracted from other plants, a Waters Acquity UPLC was coupled to a Waters Xevo G2-XS QToF mass spectrometer. The starting condition was 95% solvent A (10 mM ammonium formate, pH 2.8) and 5% solvent B (acetonitrile) with flow rate set to 0.3 mL/min. A 7 min linear elution gradient was: ramp to 40% B at 1 min, then to 100% B at 5 min, hold at 100% B to 6 min, return to 95% A at 6.01 min and held until 7 min. A 14 min linear elution gradient was: ramp to 35% B at 1 min, then to 85% B at 12 min, then to 100% B at 12.01 min, hold at 100% B to 13 min, return to 95% A at 13.01 min and held until 14 min.
For Waters LCT Premier ToF mass spectrometer, the MS settings of electrospray ionization in negative mode were: 2.5 kV capillary voltage, 100°C source temperature, 350°C desolvation temperature, 350 liters/h desolvation nitrogen gas flow rate, 10 V cone voltage, and mass range m/z 50 to 1500 with spectra accumulated at 0.1 s/function. Three collision energies (10, 40, and 80 eV) were used in separate acquisition functions to generate both molecular ion adducts and fragments. For Waters Xevo G2-XS QToF mass spectrometer, the MS settings of the negative ion-mode electrospray ionization were as follows: 2.00 kV capillary voltage, 100°C source temperature, 350°C desolvation temperature, 600 liters/h desolvation nitrogen gas flow rate, 35 V cone voltage, mass range of m/z 50 to 1000 with spectra accumulated at 0.1 s/function. Three collision energies (0, 15, and 35 eV) were used in separate acquisition functions. The MS settings for positive ion-mode electrospray ionization were: 3.00 kV capillary voltage, 100°C source temperature, 350°C desolvation temperature, 600 liters/h desolvation nitrogen gas flow rate, 35 V cone voltage, mass range of m/z 50 to 1000 with spectra accumulated at 0.1 s/function. Three collision energies (0, 15, and 45 eV) were used in separate acquisition functions. The Waters QuanLynx software was used to integrate peak areas of the selected ion relative to the internal standard. For quantification purpose, data collected with the lowest collision energy was used in the analysis.
Gene coexpression analysis
Request a detailed protocolThe publicly available tomato RNA-seq datasets and the methods used for normalizing FPKM, gene expression correlation analysis were described in a recent study (Moore et al., 2020). 926 RNA-seq Sequence Read Archive (SRA) files for tomato from 47 studies were downloaded from National Center for Biotechnology Information (NCBI; https://www.ncbi.nlm.nih.gov/) (Table S6 in Moore et al., 2020). Reads were filtered using Trimmomatic (Bolger et al., 2014b) based on the sequence quality with default settings, and mapped to the tomato NCBI S. lycopersicum genome 2.5 using TopHat (Trapnell et al., 2009). Read files with <70% mapped reads were discarded. Fragments per kilobase of transcript per million mapped reads (FPKM) were calculated using Cufflinks (Trapnell et al., 2010). The pipeline for FPKM calling used in this study was put in https://github.com/ShiuLab/RNAseq_pipeline (Uygun et al., 2020; copy archived at https://github.com/elifesciences-publications/RNAseq_pipeline). The median FPKM of multiple replicates was used for each sample, resulting in FPKM values in 372 samples. To draw the heatmap of gene expression profiles, FPKM values of a gene across all the samples were scaled, where the maximum FPKM was scaled to 1, while the minimum value was 0.
Synteny scan
Request a detailed protocolProtein sequences of annotated genes and the corresponding annotation files in General Feature Format (GFF) of 11 Solanaceae species, Ipomoea trifida, and Coffea canephora were downloaded from National Center for Biotechnology Information (NCBI, https://www.ncbi.nlm.nih.gov/genome/) or Solanaceae Genomics Network (SGN, https://solgenomics.net/). The GFF files contain the coordinates of annotated genes on assembled chromosomes or scaffolds. The sources and version numbers of sequences and GFF files used are: S. lycopersicum ITAG3.2 (SGN) and V2.5 (NCBI), S. pennellii SPENNV200 (NCBI) and v2.0 (SGN), S. tuberosum V3.4 (SGN), S. melongena r2.5.1 (SGN), Capsicum annuum L. zunla-1 V2.0 (SGN), C. annuum_var. glabriusculum V2.0 (SGN), Nicotiana attenuata NIATTr2 (SGN), N. tomentosiformis V01 (NCBI), N. benthamiana V1.0.1 (SGN), Petunia axillaris V1.6.2 (SGN), P. inflata V1.0.1 (SGN), I. trifida V1.0 (NCBI), and C. canephora Vx (SGN).
To hypothesize the evolutionary history of genes in the acylsugar gene cluster, putative pseudogenes, which are homologs to protein-coding genes but with predicted premature stops/frameshifts and/or protein sequence truncation, were also identified for each species as described (Wang et al., 2018). Protein sequences from Arabidopsis thaliana, Oryza sativa, and S. lycopersicum were used as queries in the searches against the genomic regions of target species using TBLASTN (Altschul et al., 1990). The intergenic genomic sequences were identified as potential pseudogenes using the pipeline from as previously described (Campbell et al., 2014; Zou et al., 2009). If one of the six-frame translated sequences of the intergenic genomic sequences had significant similarity to annotated protein sequences, and had premature stops/frameshifts and/or were truncated (<30% of functional paralogs), the gene was defined as a pseudogene.
Genome-wide syntenic analysis was conducted using annotated protein-coding genes and putative pseudogenes from all the species with MCScanX-transposed (Wang et al., 2013) as described (Wang et al., 2018). The MCScanX-based analysis did not lead to a syntenic block of acylsugar gene cluster on chromosome 7 of S. melongena r2.5.1, which can be due to true absence, issues with genome assembly, or lack of coverage. To verify this, protein sequences of S. lycopersicum genes in genomic blocks on chromosome 7 were searched against an updated S. melongena genome from The Eggplant Genome Project (http://ddlab.dbt.univr.it/eggplant/) that led to the identification of the synteny.
Phylogenetic tree building
Request a detailed protocolHomologous genes of Sl-AACS1 (ACS), Sl-AECH1 (ECH), and Solyc07g043670 (BAHD acyltransferase) were obtained through BLAST (Altschul et al., 1990) search from the genomes of 11 Solanaceae species, Ipomoea trifida, and Coffea canephora with an Expect value threshold of 1e-5. To simply the phylogenetic tree, sequences which are distantly related to the target genes were removed, and the remained sequences were used to rebuild the phylogenetic trees. The amino acid sequences were aligned using MUSCLE (Edgar, 2004) with the default parameters. The phylogenetic trees were built using the maximum likelihood method with 1000 bootstrap replicates. The trees were generated using RAxML/8.0.6 (Stamatakis, 2014) with the following parameters: -f a -x 12345 p 12345 -# 1000 m PROTGAMMAAUTO --auto-prot=bic, and were shown with midpoint rooting. The final sequence alignments used to generate the phylogenetic trees were provided in Supplementary file 5.
Ancestral trait reconstruction
Request a detailed protocolAncestral trait state reconstruction was conducted using the maximum likelihood model Mk1 in Mesquite 3.6 (Massidon and Maddison, 2018). Four traits were inferred for their ancestral states. They are the presence of medium chain acylsugars, presence of ACS genes in the synteny, presence of ECH genes in the synteny, and presence of both ACS and ECH genes in the synteny. The phylogeny of Solanaceae species was based on a previous study (Särkinen et al., 2013).
Acylsugar acyl chain composition analysis by GC-MS
Request a detailed protocolAcyl chains were characterized from the corresponding fatty acid ethyl esters following transesterification of acylsugar extractions as previously reported (Ning et al., 2015). Plants were grown for 4–8 weeks and approximately ten leaves were extracted for 3 min in 10 mL of 1:1 isopropanol:acetonitrile with 0.01% formic acid. Extractions were dried to completeness using a rotary evaporator and then 300 µL of 21% (v/v) sodium ethoxide in ethanol (Sigma) was added and incubated for 30 min with gentle rocking and vortexing every five minutes and 400 µL hexane was added and vortexed for 30 s. To the hexane layer, 500 µL of saturated sodium chloride in water was added and vortexed to facilitate a phase separation. After phase separation, the top hexane layer was transferred to a new tube. The phase separation by addition of 500 µL hexane was repeated twice, with the final hexane layer transferred to a 2 mL glass vial with a glass insert.
The fatty acid ethyl esters were analyzed using an Agilent 5975 single quadrupole GC-MS equipped with a 30 m, 0.25 mm internal diameter fused silica column with a 0.25 µm film thickness VF5 stationary phase (Agilent). Injection of 1 µL of each hexane extract was performed using splitless mode. The gas chromatography program was as follows: inlet temperature, 250°C; initial column temperature, 70°C held for 2 min; ramped at 20 °C/min until 270°C, then held at 270°C 3 min. The helium carrier gas was used with 70 eV electron ionization. Acyl chain type was determined through NIST Version 2.3 library matches of the mass spectra of the corresponding ethyl ester and relative abundances were determined through integrating the corresponding peak area over the total acyl chain peak area.
Data availability
The RNA-seq reads were deposited in the National Center for Biotechnology Information Sequence Read Archive under the accession number PRJNA605501. Sequence data used in this study are in the GenBank/EMBL data libraries under these accession numbers: Sl-AACS1(MT078737), Sl-AECH1(MT078736), Sp-AACS1(MT078735), Sp-AECH1(MT078734), Sq-AACS1(MT078732), Sq-AECH1(MT078731), Sq_c35719 (MT078733). The following materials require a material transfer agreement: pEAQ-HT, pK7WG, pKGWFS7, pEarleyGate102, pEarleyGate104, pTRV2-LIC, pICH47742::2x35S-5'UTR-hCas9(STOP)-NOST, pICH41780, pAGM4723, and pICSL11024.
-
NCBI BioProjectID PRJNA605501. Solanum pennellii stem and trichome transcriptome.
References
-
Basic local alignment search toolJournal of Molecular Biology 215:403–410.https://doi.org/10.1016/S0022-2836(05)80360-2
-
The genome of the stress-tolerant wild tomato species Solanum pennelliiNature Genetics 46:1034–1038.https://doi.org/10.1038/ng.3046
-
Trimmomatic: a flexible trimmer for illumina sequence dataBioinformatics 30:2114–2120.https://doi.org/10.1093/bioinformatics/btu170
-
Peroxisome targeting signal 1: is it really a simple tripeptide?Biochimica Et Biophysica Acta (BBA) - Molecular Cell Research 1763:1565–1573.https://doi.org/10.1016/j.bbamcr.2006.08.022
-
BookBiochemistry and Molecular Biology of PlantsRockville, MD: American Society of Plant Physiologists.
-
An effective strategy for exploring unknown metabolic pathways by genome miningJournal of the American Chemical Society 135:5885–5894.https://doi.org/10.1021/ja401535g
-
Acyltransferases in plants: a good time to be BAHDCurrent Opinion in Plant Biology 9:331–340.https://doi.org/10.1016/j.pbi.2006.03.016
-
KAS IV: a 3-ketoacyl-ACP synthase from Cuphea sp. is a medium chain specific condensing enzymeThe Plant Journal : For Cell and Molecular Biology 15:383–390.https://doi.org/10.1046/j.1365-313x.1998.00218.x
-
MUSCLE: multiple sequence alignment with high accuracy and high throughputNucleic Acids Research 32:1792–1797.https://doi.org/10.1093/nar/gkh340
-
An introgression line population of lycopersicon pennellii in the cultivated tomato enables the identification and fine mapping of yield-associated QTLGenetics 141:1147–1162.
-
Tip of the trichome: evolution of acylsugar metabolic diversity in SolanaceaeCurrent Opinion in Plant Biology 49:8–16.https://doi.org/10.1016/j.pbi.2019.03.005
-
Myo-inositol-derived glycolipids with anti-inflammatory activity from Solanum lanceolatumJournal of Natural Products 68:1031–1036.https://doi.org/10.1021/np050054s
-
BookStrategies for Profiling and Discovery of Acylsugar Specialized MetabolitesMichigan State University.
-
Enzyme promiscuity: a mechanistic and evolutionary perspectiveAnnual Review of Biochemistry 79:471–505.https://doi.org/10.1146/annurev-biochem-030409-143718
-
Promiscuity, impersonation and accommodation: evolution of plant specialized metabolismCurrent Opinion in Structural Biology 47:105–112.https://doi.org/10.1016/j.sbi.2017.07.005
-
Differential expression analysis of multifactor RNA-Seq experiments with respect to biological variationNucleic Acids Research 40:4288–4297.https://doi.org/10.1093/nar/gks042
-
BookTransformation of tomato with Agrobacterium tumefaciensIn: Lindsey K, editors. Plant Tissue Culture Manual. Dordrecht: Netherlands: Springer . pp. 311–319.https://doi.org/10.1007/978-94-009-0103-2_17
-
All fatty acids are not equal: discrimination in plant membrane lipidsTrends in Plant Science 5:95–101.https://doi.org/10.1016/S1360-1385(00)01566-1
-
Plant defense against herbivores: chemical aspectsAnnual Review of Plant Biology 63:431–450.https://doi.org/10.1146/annurev-arplant-042110-103854
-
Metabolite-Enzyme coevolution: from single enzymes to metabolic pathways and networksAnnual Review of Biochemistry 87:187–216.https://doi.org/10.1146/annurev-biochem-062917-012023
-
Plant metabolic clusters – from genetics to genomicsThe New Phytologist 211:771–789.https://doi.org/10.1111/nph.13981
-
Gene clustering in plant specialized metabolismCurrent Opinion in Biotechnology 26:91–99.https://doi.org/10.1016/j.copbio.2013.10.009
-
Evolution of gene duplication in plantsPlant Physiology 171:2294–2316.https://doi.org/10.1104/pp.16.00523
-
Convergent evolution in plant specialized metabolismAnnual Review of Plant Biology 62:549–566.https://doi.org/10.1146/annurev-arplant-042110-103814
-
The birth, evolution and death of metabolic gene clusters in fungiNature Reviews Microbiology 16:731–744.https://doi.org/10.1038/s41579-018-0075-3
-
Genome-Wide prediction of metabolic enzymes, pathways, and gene clusters in plantsPlant Physiology 173:2041–2059.https://doi.org/10.1104/pp.16.01942
-
Glandular trichomes: micro-organs with model status?New Phytologist 225:2251–2266.https://doi.org/10.1111/nph.16283
-
TopHat: discovering splice junctions with RNA-SeqBioinformatics 25:1105–1111.https://doi.org/10.1093/bioinformatics/btp120
-
Variations in the biosynthesis of seed-storage lipidsAnnual Review of Plant Physiology and Plant Molecular Biology 52:335–361.https://doi.org/10.1146/annurev.arplant.52.1.335
-
Factors influencing gene family size variation among related species in a plant family, SolanaceaeGenome Biology and Evolution 10:2596–2613.https://doi.org/10.1093/gbe/evy193
-
Arabidopsis thaliana polar glycerolipid profiling by thin layer chromatography (TLC) Coupled with Gas-Liquid chromatography (GLC)Journal of Visualized Experiments 18:2518.https://doi.org/10.3791/2518
-
The evolutionary paths towards complexity: a metabolic perspectiveNew Phytologist 201:1141–1149.https://doi.org/10.1111/nph.12416
-
To gibberellins and beyond! surveying the evolution of (di)terpenoid metabolismAnnual Review of Plant Biology 65:259–286.https://doi.org/10.1146/annurev-arplant-050213-035705
Article and author information
Author details
Funding
National Science Foundation (1546617)
- Shin-Han Shiu
- Robert L Last
National Science Foundation (1655386)
- Shin-Han Shiu
U.S. Department of Energy (BER DE-SC0018409)
- Shin-Han Shiu
National Science Foundation (1727362)
- Federica Brandizzi
National Institutes of Health (GM110523)
- Bryan J Leong
- Robert L Last
National Science Foundation (1757043)
- Rachel Combs
- Robert L Last
National Science Foundation (1811055)
- Craig A Schenck
The funders had no role in study design, data collection and interpretation, or the decision to submit the work for publication.
Acknowledgements
We thank the CM Rick Tomato Genetics Resource Center (University of California Davis, CA USA) for providing tomato seeds, Zamir lab in Hebrew University of Jerusalem for providing tomato ILs and BILs seeds. We acknowledge Dr. Kun Wang and Dr. Christoph Benning for their helpful guidance in lipid analysis. We thank Krystle Wiegert-Rininger and Cornelius Barry for their help in RNA sequencing and Dr. Kent Chapman from University of North Texas for helpful discussions. We acknowledge Kathleen Imre and Sara Haller for their help with tomato transformation. We thank the MSU Center for Advanced Microscopy and RTSF Mass Spectrometry and Metabolomics Core Facilities for their support with LC/MS analysis.
Copyright
© 2020, Fan et al.
This article is distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use and redistribution provided that the original author and source are credited.
Metrics
-
- 5,402
- views
-
- 936
- downloads
-
- 68
- citations
Views, downloads and citations are aggregated across all versions of this paper published by eLife.
Citations by DOI
-
- 68
- citations for umbrella DOI https://doi.org/10.7554/eLife.56717






