TMAO, a seafood-derived molecule, produces diuresis and reduces mortality in heart failure rats
Figures
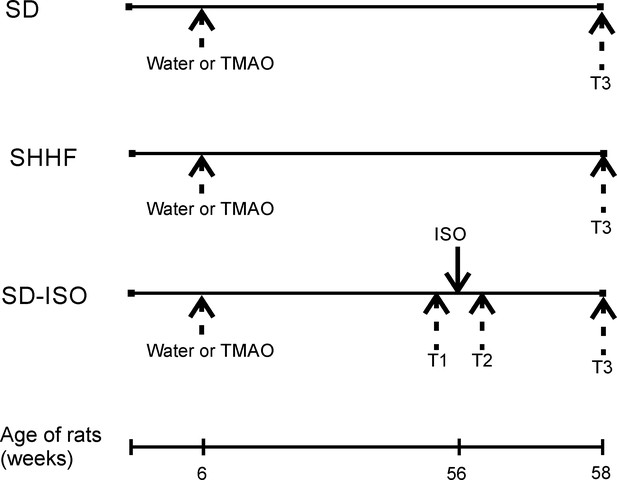
Schematic illustration of experimental series.
6-week-old rats started drinking either water (control) or a TMAO solution. SHHF - Spontaneously Hypertensive Heart Failure (SHHF/MccGmiCrl-Leprcp/Crl) SHHF, SD – Sprague-Dawley rats, SD-ISO - SD rats treated with ISO at the age of 56 week. ISO - administration of isoprenaline at a dose of 100 mg/kg s.c. T1 - metabolic and echocardiographic measurements, T2 - echocardiographic measurements, T3 - metabolic, echocardiographic and direct hemodynamic measurements.
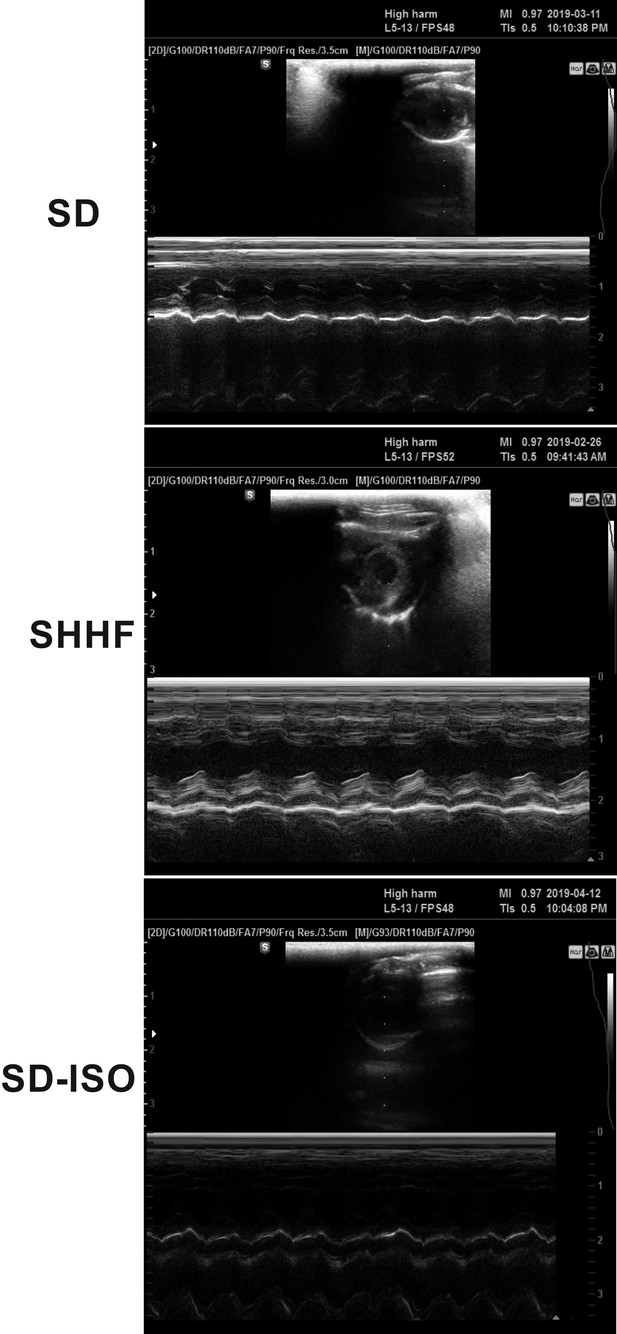
Echocardiography in 58-wk-old rats.
SHHF - Spontaneously Hypertensive Heart Failure (SHHF/MccGmiCrl-Leprcp/Crl), SD – Sprague-Dawley rats, SD-ISO: SD rats treated with ISO at the age of 56 week. ISO: administration of isoprenaline at a dose of 100 mg/kg s.c. SD: Left ventricular systolic function is normal/preserved. Left and right ventricular diameter is normal. Left ventricular wall thickness is normal. Left atrial diameter is normal. SHHF: Septal hypokinesis. Left ventricular free wall is hypertrophic. Endocardium is hyperechogenic. SD-ISO: Septal hypokinesis. Left ventricular end-systolic diameter is increased. Left atrial enlargement.
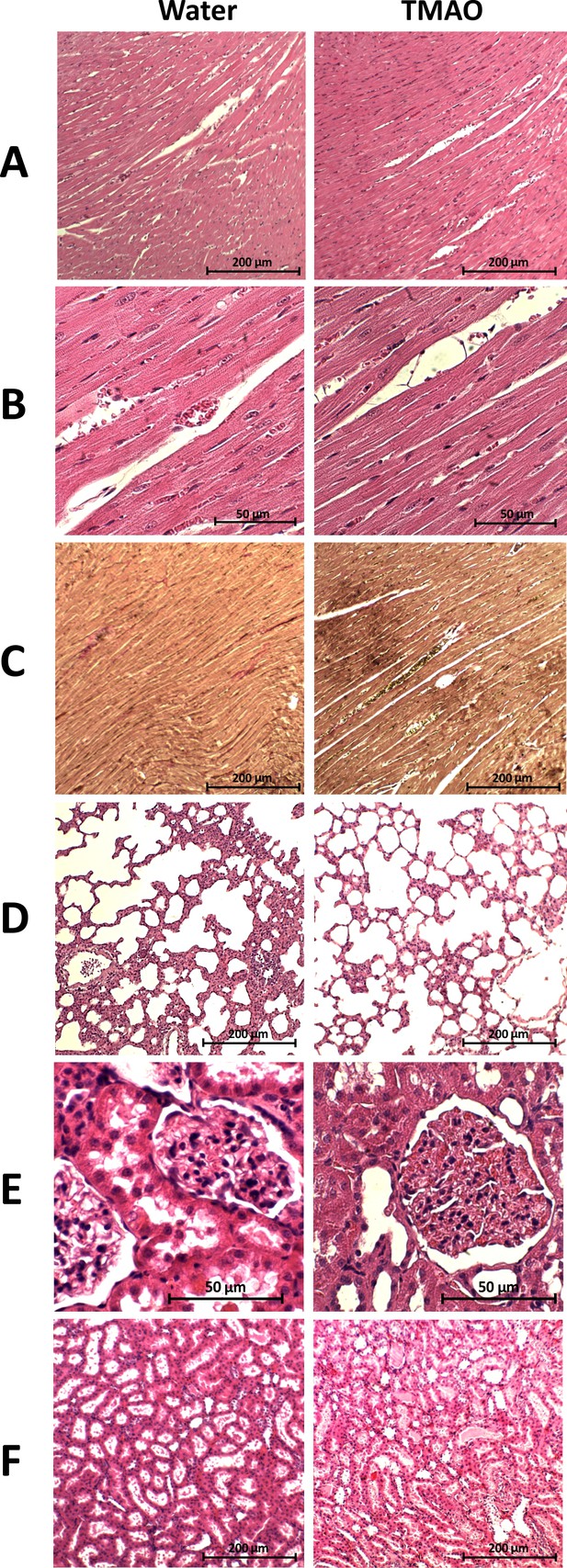
Histopathological picture of the heart, lungs and kidneys in the Sprague-Dawley rats drinking either water (control group) or TMAO solution.
A - myocardium; hematoxylin-eosin (HE) staining at magnification x10; B - myocardium; HE staining at magnification x40; C - myocardium; van Gieson staining at magnification x10; D - lungs; HE staining at magnification x10; E – kidney - renal cortex, renal bodies; HE staining at magnification x40; F - kidney - renal medulla; hematoxylin-eosin staining at magnification x10.
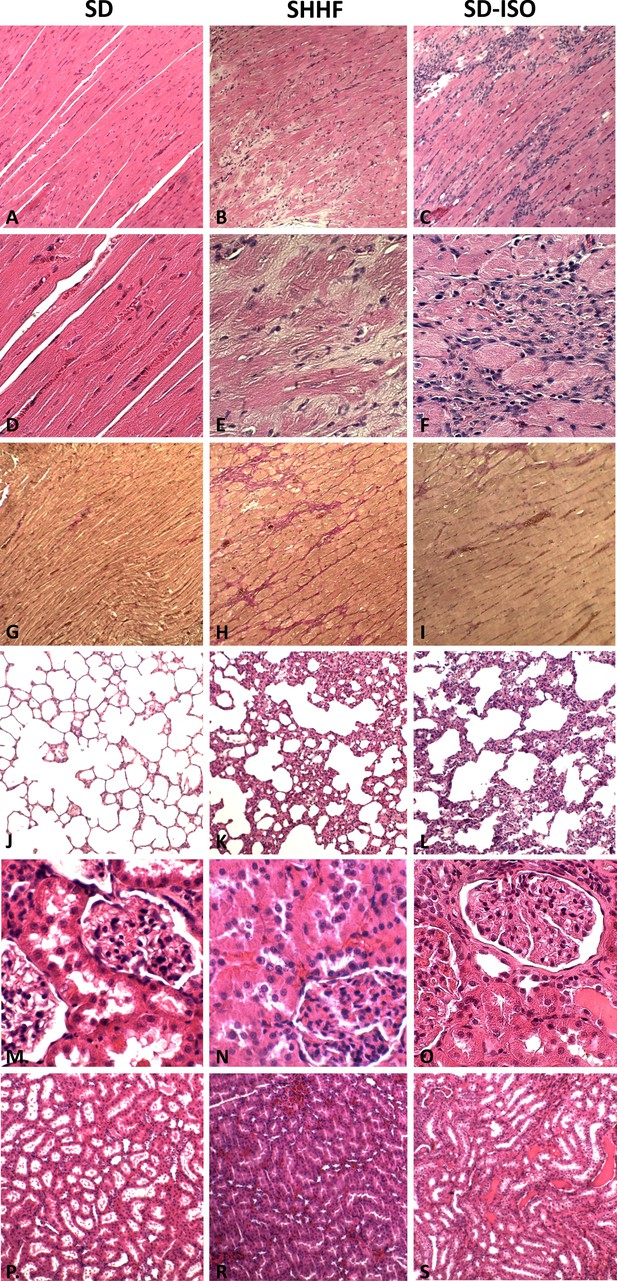
Comparison of the histopathological picture between SD, SHHF and ISO-SD.
A, B, C - myocardium; hematoxylin-eosin staining at magnification x10; D, E, F - myocardium; hematoxylin-eosin staining at magnification x40; G, H, I- myocardium; van Gieson staining at magnification x10; J, K, L - lungs; hematoxylin-eosin staining at magnification x10; M, N, O – kidney - renal cortex, renal bodies; hematoxylin-eosin staining at magnification x40; P, T, S – kidney - renal medulla; hematoxylin-eosin staining at magnification x10.
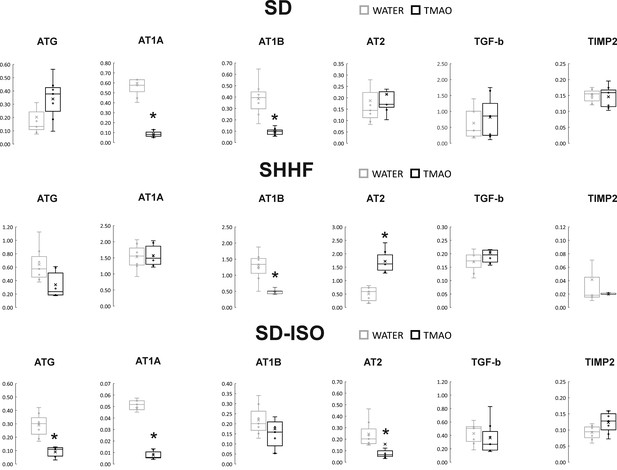
Real-time RT-PCR analysis, heart.
Box plot comparing the expression profiles of ATG (angiotensinogen), AT1A (angiotensin II receptor type 1a), AT1B (angiotensin II receptor type 1b), AT2 (angiotensin II receptor type 2), TGF-b (transforming growth factor-beta), TIMP2 (metalloproteinase inhibitor 2) in hearts of SD – Sprague-Dawley rats, SHHF - Spontaneously Hypertensive Heart Failure (SHHF/MccGmiCrl-Leprcp/Crl), SD-ISO - SD rats treated with ISO at the age of 56 week, drinking either water (control groups) or TMAO solution, (n = 6–7 for each series). × - mean value, * indicates significant difference compared with the control group. *p<0.05 by t-test or Mann-Whitney U test.
-
Figure 4—source data 1
Real-time RT-PCR analysis, expression profiles in heart.
- https://cdn.elifesciences.org/articles/57028/elife-57028-fig4-data1-v2.xlsx

Real-time RT-PCR analysis, kidneys.
Box plot comparing the expression profiles of ATG (angiotensinogen), AT1A (angiotensin II receptor type 1a), AT1B (angiotensin II receptor type 1b), AT2 (angiotensin II receptor type 2), renin in kidneys of SD – Sprague-Dawley rats, SHHF - Spontaneously Hypertensive Heart Failure (SHHF/MccGmiCrl-Leprcp/Crl), SD-ISO - SD rats treated with ISO at the age of 56 week, drinking either water (control groups) or TMAO solution, (n = 6–7 for each series). × - mean value,* indicates significant difference compared with the control group. *p<0.05 by t-test or Mann-Whitney U test.
-
Figure 5—source data 1
Real-time RT-PCR analysis, expression profiles in kidneys.
- https://cdn.elifesciences.org/articles/57028/elife-57028-fig5-data1-v2.xlsx
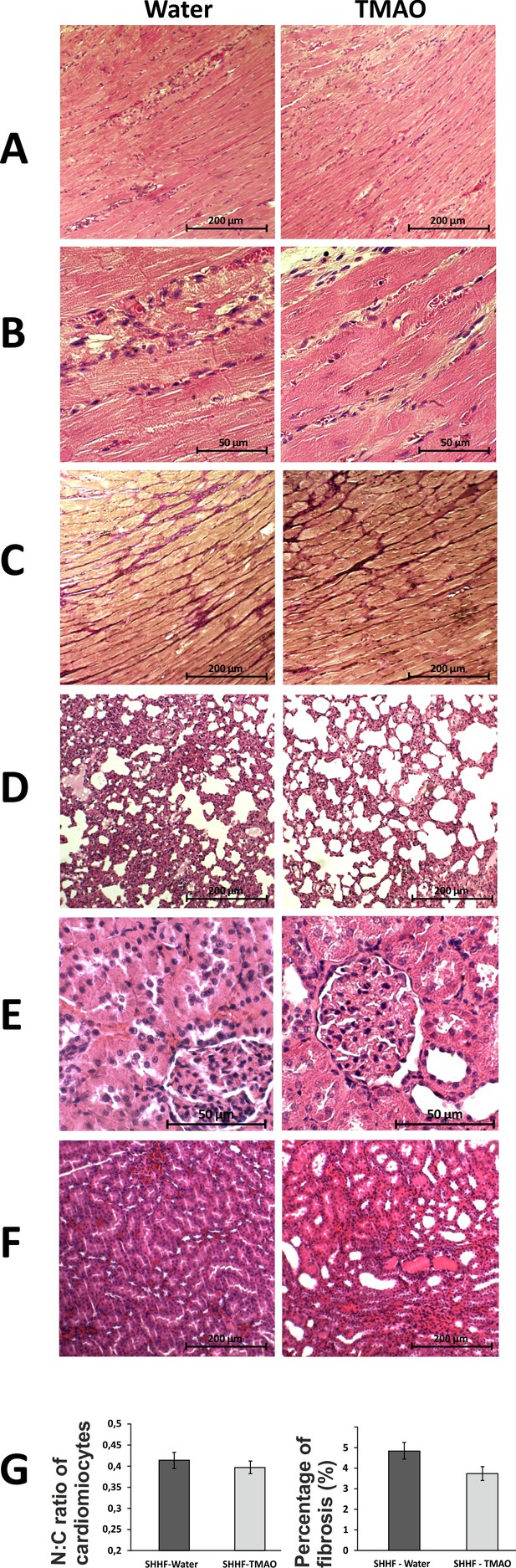
Histopathological picture of the heart, lungs and kidneys in Spontaneously Hypertensive Heart Failure (SHHF/MccGmiCrl-Leprcp/Crl) drinking either water (control group) or TMAO solution.
A - myocardium; hematoxylin-eosin staining at magnification x10; B - myocardium; hematoxylin-eosin staining at magnification x40; C - myocardium; van Gieson staining at magnification x10; D - lungs; hematoxylin-eosin staining at magnification x10; E – kidney - renal cortex, renal bodies; hematoxylin-eosin staining at magnification x40; F – kidney - renal medulla; hematoxylin-eosin staining at magnification x10; G – Percentage of myocardial fibrosis [%], N:C ratio of cardiomyocytes, values are means,± SE.
-
Figure 6—source data 1
Percentage of myocardial fibrosis in SHHF rats.
- https://cdn.elifesciences.org/articles/57028/elife-57028-fig6-data1-v2.xlsx
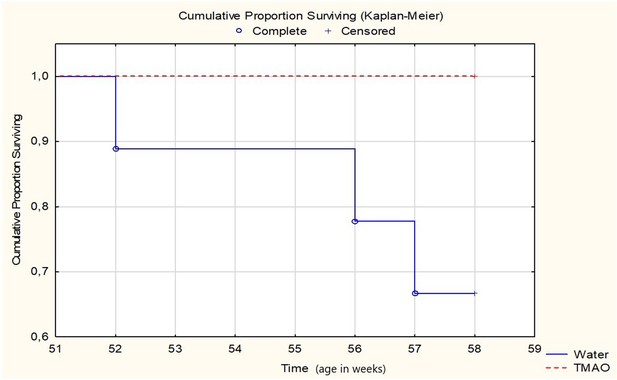
Survival Kaplan-Meier curves for SHHF - Spontaneously Hypertensive Heart Failure (SHHF/MccGmiCrl-Leprcp/Crl) rats drinking either water (control group, n = 9) or TMAO solution (n = 9).
Log-Rank test p=0.06555.
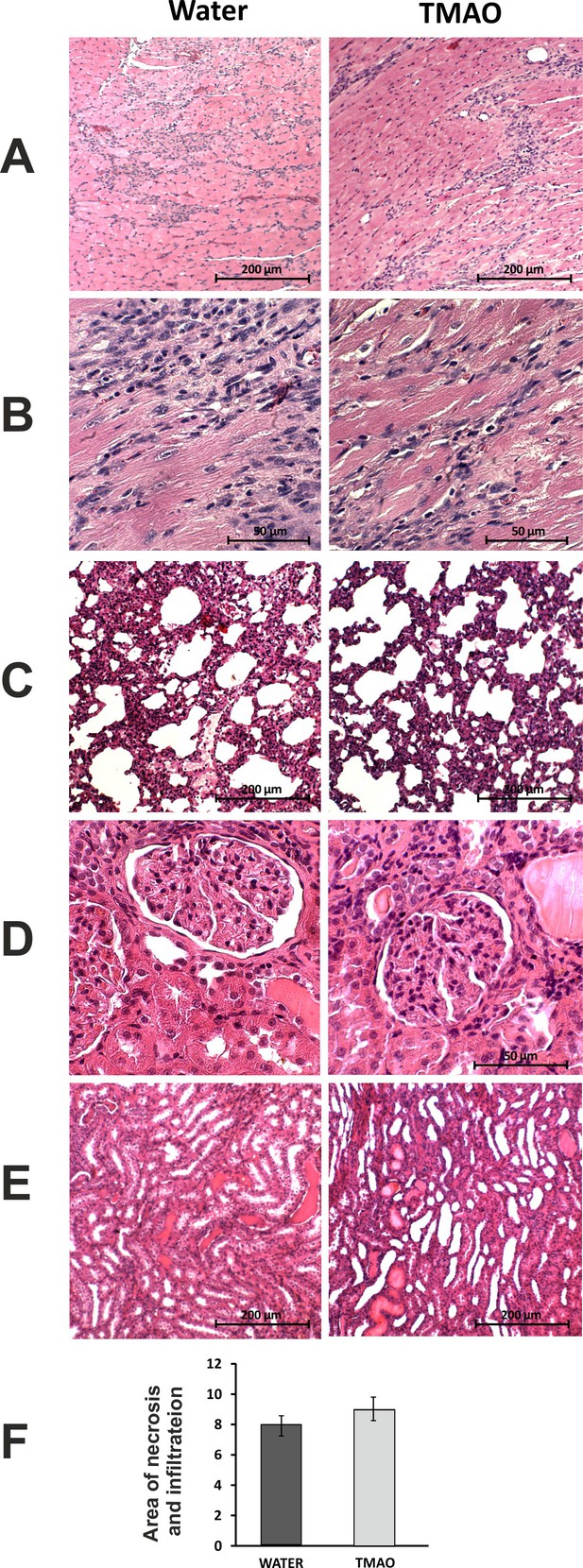
Histopathological picture of the heart, lungs and kidneys in Sprague-Dawley rats treated with isoprenaline at the age of 56 weeks, and drinking either water (control group) or TMAO solution.
A - myocardium; hematoxylin-eosin (HE) staining at magnification x10; B - myocardium; HE staining at magnification x40; C - lungs; HE staining at magnification x10; D – kidney - renal cortex, renal bodies; HE staining at magnification x40; E – kidney - renal medulla; HE staining at magnification x10; F – Percentage of necrotic and inflammatory area in myocardium [%], values are means,± SE.
-
Figure 8—source data 1
Percentage of necrotic and inflammatory area in myocardium of SD-ISO rats.
- https://cdn.elifesciences.org/articles/57028/elife-57028-fig8-data1-v2.xlsx
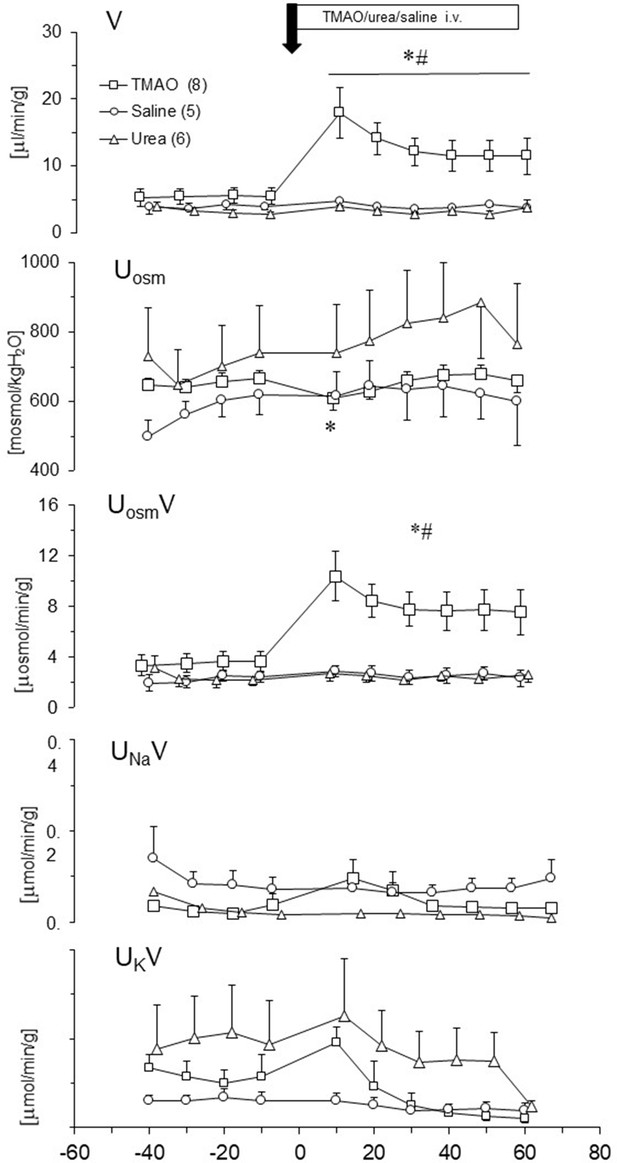
Effects of TMAO (n = 8), urea (n = 6) and saline (n = 5) on renal excretion in anesthetized Sprague-Dawley rats.
The priming dose (indicated by arrow) of TMAO and urea were 2.8 mmol/kg b.w. (bolus), followed by continuous infusion at a rate of 2.8 mmol/kg b.w./60 min. V – urine flow; Uosm – urine osmolality; UosmV, UNaV, UKV – total solute, sodium and potassium excretion, respectively. Values are means ± SE. * - p<0.05 vs pretreatment values, # - p<0.02 TMAO vs saline, TMAO vs urea.
-
Figure 9—source data 1
Effects of TMAO, urea and saline on renal excretion.
- https://cdn.elifesciences.org/articles/57028/elife-57028-fig9-data1-v2.xls
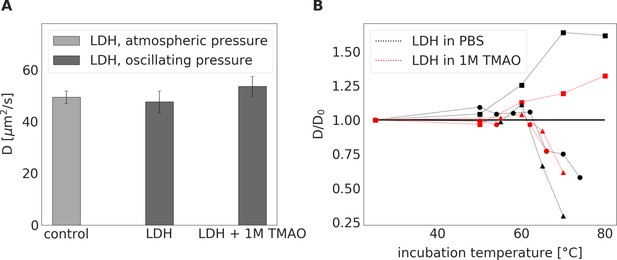
LDH incubated at a constant atmospheric pressure for 24 hrs or LDH exposed to oscillating pressure for 24 hrs with or without TMAO.
(A) - Diffusion coefficient of LDH incubated either in PBS buffer at a constant atmospheric pressure and room temperature (serving as a control) or exposed to pressure oscillation for 24 hrs either in PBS buffer or in 1 M TMAO solution in PBS buffer at room temperature. Irrespective of the presence of 1M TMAO, 24-hour incubation under oscillating pressure did not cause dissociation, denaturation, or aggregation of LDH. ( B) – Relative diffusion coefficient (diffusion coefficient divided by its value in PBS buffer at room temperature ) of LDH exposed to elevated temperatures for 15 min either in PBS buffer (black symbols) or 1 M TMAO solution in PBS (red symbols). Symbol shapes differentiate between three independent measurement series (series 1 (3nM of LDH) – squares, series 2 (30nM of LDH) – circles, series 3(300nM) – triangles). In series 1, we observed an increase in the relative diffusion coefficient, suggesting degradation of LHD tetramer structure to monomers. In series 2 and 3, an increase in diffusion coefficient was followed by a decrease in diffusion coefficient suggesting the degradation of LDH followed by the aggregation of the LDH monomers. The presence of TMAO shifted the threshold of change in relative diffusion coefficient towards higher temperatures.
-
Figure 10—source data 1
LDH incubated at a constant atmospheric pressure for 24 hrs or LDH exposed to oscillating pressure for 24 hrs with or without TMAO.
- https://cdn.elifesciences.org/articles/57028/elife-57028-fig10-data1-v2.xlsx
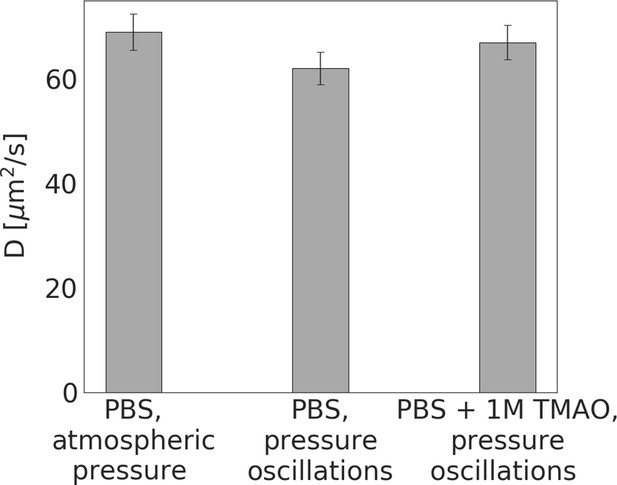
Diffusion coefficients of LDH measured by FCS.
First bar is a control; the two following correspond to samples incubated for 24 hr in the high-pressure oscillation system without and with 1M TMAO in the solution.
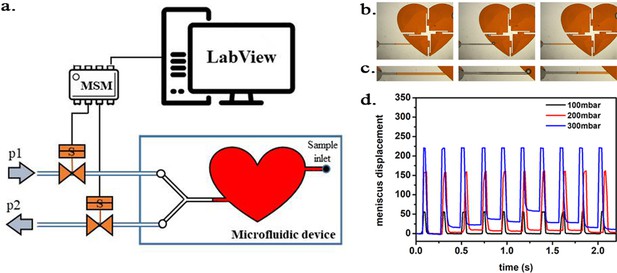
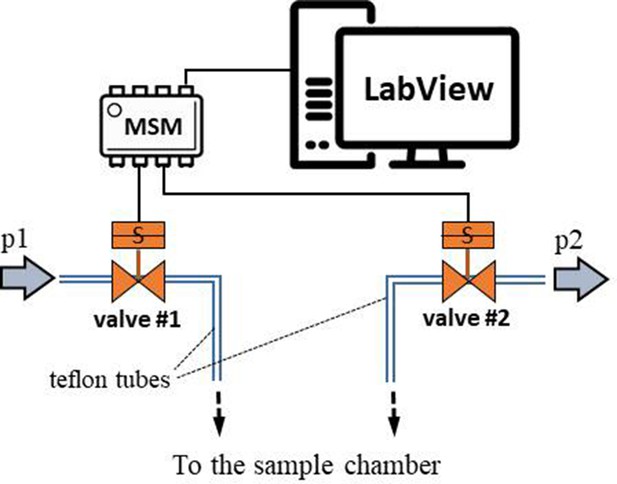
Scheme of the experimental setup.
(a) Scheme of the experimental setup with the heart-shaped polydimethylsiloxane (PDMS) microfluidic device. (b) time-sequence snapshots of the device in operations. From left to right: the device is filled with liquid and is at rest (for better visualisation we used here red-dyed water instead of the transparent protein solution). The microchannel connecting the pressure system to the microfluidic chamber is filled halfway. Applying high pressure (valve # one open) pushes the liquid meniscus towards the chamber. Closing valve # one and opening valve # 2 (low pressure) the liquid meniscus pulls back (even further than its original position). (c) Close-up of the moving liquid meniscus, (d) oscillation profile generated from the position of the liquid meniscus in the microchannel as a function of time for various pressure differences Δp=p1 p2.
-
Figure 12—source data 1
Oscillation profile generated from the position of the liquid meniscus in the microchannel.
- https://cdn.elifesciences.org/articles/57028/elife-57028-fig12-data1-v2.xlsx
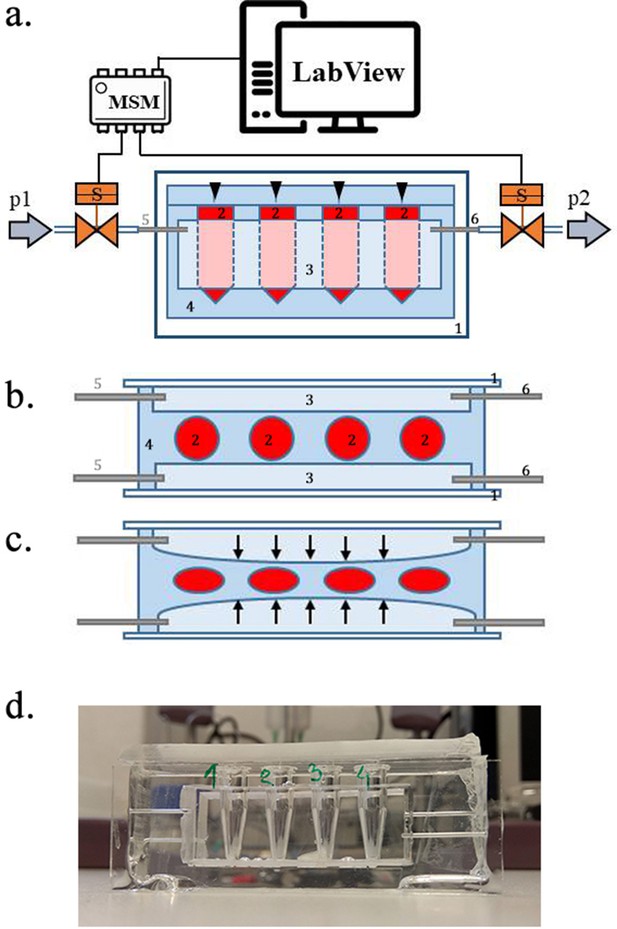
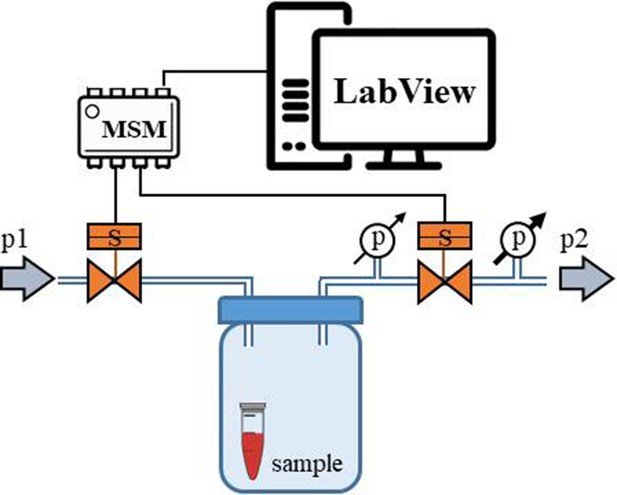
Scheme of the experimental setup.
(a) Scheme of the set-up with the pressure oscillator connected to the Eppendorf chip with side view and b) top view of the ‘Eppendorf chip’.Numbers indicate the glass slide (1), the sample chambers (2), the cuboidal cavity (3), the PDMS and the inlet (5) and outlet (6)Stubbs et al., 2019 steel capillaries, respectively. (b–c) Schematic representation of the chip’s operation upon applied pressure: the cavity (3) expands towards the sample chambers (2) squeezing and deforming them. (d) Photo of the constructed device prior to filling and connecting to the pressure controller.
Tables
Metabolic, renal and cardiovascular parameters in 58-week-old normotensive SD rats.
Sprague-Dawley rats maintained on either water (SD-control, n = 6–10) or TMAO solution (SD-TMAO, n = 7–10). Creatinine clearance calculated as urine creatinine x urine output (ml/min)/plasma creatinine. LVEDV - left ventricle end diastolic volume, LVESV - left ventricle end systolic volume, SV – stroke volume, EF - ejection fraction, IVSs(d) - intraventricular septum diameter during systole and diastole, respectively, LVEDP - pressure in the left ventricle during the end of diastole measured directly with a catheter, +dP/dt - maximal slope of systolic ventricular pressure increment, - dP/dt - maximal slope of diastolic ventricular pressure decrement. Values are means,± SD. P values by t-test or Mann-Whitney U test.
| Group/Parameter | SD-control | SD-TMAO | P | |
|---|---|---|---|---|
| Survival, energy and water balance | ||||
| Survival from the study onset (%, n) | 100% (10/10) | 100% (10/10) | - | |
| Body mass (g) | 446.4 ± 40.63 | 452.86 ± 37.11 | 0.36 | |
| 24 hr food intake (g) | 19.99 ± 2.83 | 21.49 ± 2.64 | 0.12 | |
| 24 hr water intake (g) | 31.56 ± 10.02 | 36.66 ± 5.18 | 0.09 | |
| 24 hr urine output (g) | 18.66 ± 2.35 | 22.66 ± 6.49 | 0.04 | |
| Tibia length (cm) | 4.31 ± 0.1 | 4.25 ± 0.15 | 0.15 | |
| TMAO | ||||
| Plasma TMAO (µmol/L) | 6.55 ± 0.65 | 39.73 ± 20.6 | <0.001 | |
| 24 hr TMAO urine excretion (µmoles) | 5.96 ± 1.49 | 103.05 ± 56.7 | <0.001 | |
| Heart mass | ||||
| Heart mass (g) | 1.44 ± 0.08 | 1.46 ± 0.14 | 0.38 | |
| Arterial blood pressure and heart rate | ||||
| Systolic (mmHg) | 129.72 ± 8.56 | 127.07 ± 5.84 | 0.87 | |
| Diastolic (mmHg) | 80.56 ± 13.3 | 86.61 ± 9.74 | 0.15 | |
| HR (beats/min) | 333.9 ± 45 | 364.4 ± 8.6 | 0.04 | |
| Echocardiographic parameters | ||||
| LVEDV (mL) | 0.47 ± 0.15 | 0.57 ± 0.1 | 0.06 | |
| LVESV (mL) | 0.12 ± 0.04 | 0.13 ± 0.03 | 0.44 | |
| IVSs (cm) | 0.35 ± 0.03 | 0.35 ± 0.03 | 0.42 | |
| IVSd (cm) | 0.24 ± 0.03 | 0.25 ± 0.03 | 0.19 | |
| SV (mL) | 0.36 ± 0.11 | 0.44 ± 0.1 | 0.04 | |
| EF (%) | 75.63 ± 3.02 | 77.13 ± 6.01 | 0.27 | |
| Left ventricle hemodynamic parameters (direct measurements) | ||||
| LVEDP (mmHg) | 4.12 ± 0.78 | 4.25 ± 0.92 | 0.45 | |
| dP/dt (mmHg/ms) | 6.54 ± 1.02 | 7.31 ± 1.23 | 0.12 | |
| -dP/dt (mmHg/ms) | 5.00 ± 0.69 | 5.26 ± 0.45 | 0.21 | |
| Plasma NT-proBNP | ||||
| NT-proBNP (pg/mL) | 24.79 ± 8.1 | 18.61 ± 8.17 | 0.22 | |
| Electrolyte balance | ||||
| Serum sodium (mmol/L) | 138.86 ± 2.27 | 138.44 ± 0.50 | 1.0 | |
| 24 hr sodium urine excretion (mmoles) | 1.76 ± 0.23 | 2.11 ± 0.08 | 0.003 | |
| Serum potassium (mmol/L) | 5.27 ± 0.89 | 5.13 ± 0.17 | 0.71 | |
| 24 hr potassium urine excretion (mmoles) | 2.83 ± 0.58 | 3.00 ± 0.12 | 0.23 | |
| Serum creatinine clearance (mL/min) | 1.15 ± 0.18 | 1.16 ± 0.07 | 0.67 | |
| Hormones | ||||
| Angiotensin II (pg/mL) | 244.93 ± 35.55 | 250.07 ± 64.95 | 0.43 | |
| Aldosterone (pg/mL) | 897.05 ± 95.34 | 925.61 ± 75.29 | 0.27 | |
| Vasopressin (ng/mL) | 0.92 ± 0.98 | 1.78 ± 0.59 | 0.02 | |
-
Table 1—source data 1
Metabolic, renal and cardiovascular parameters in 58-week-old normotensiveSD rats.
- https://cdn.elifesciences.org/articles/57028/elife-57028-table1-data1-v2.xlsx
Metabolic, renal and cardiovascular parameters in 58-week-old SHHF rats.
Spontaneously Hypertensive Heart Failure (SHHF/MccGmiCrl-Leprcp/Crl) rats maintained on either water (SHHF-control, n = 5–6) or TMAO solution (SHHF-TMAO, n = 7–9).Creatinine clearance calculated as urine creatinine x urine output (ml/min)/plasma creatinine. LVEDV - left ventricle end diastolic volume, LVESV - left ventricle end systolic volume, SV – stroke volume, EF - ejection fraction, IVSs(d) - intraventricular septum diameter during systole and diastole, respectively, LVEDP - pressure in the left ventricle during the end of diastole measured directly with a catheter, +dP/dt - maximal slope of systolic ventricular pressure increment, - dP/dt - maximal slope of diastolic ventricular pressure decrement. Values are means,± SD. P values by t-test or Mann-Whitney U test.
| Group/ Parameter | SHHF-control | SHHF-TMAO | P | |
|---|---|---|---|---|
| Survival, Energy and water balance | ||||
| Survival from the onset of the study (%, n) | 66% (6/9) | 100% (9/9) | 0.07 # | |
| Body mass (g) | 475.2 ± 17.1 | 476.3 ± 12.1 | 0.43 | |
| 24 hr food intake (g) | 23.2 ± 3.2 | 24.2 ± 2.3 | 0.26 | |
| 24 hr water intake (mL) | 37.5 ± 7.5 | 41.1 ± 6.6 | 0.17 | |
| 24 hr urine output (mL) | 14.8 ± 2.8 | 17.9 ± 2.5 | 0.02 | |
| Tibia length (cm) | 3.95 ± 0.21 | 3.99 ± 0.11 | 0.34 | |
| TMAO | ||||
| Plasma TMAO (µmol/L) | 6.71 ± 1.49 | 20.32 ± 7.21 | <0.001 | |
| 24 hr TMAO urine excretion (µmoles) | 9.97 ± 3.46 | 126.8 ± 32.8 | <0.001 | |
| Heart mass | ||||
| Heart mass (g) | 1.87 ± 0.31 | 1.72 ± 0.3 | 0.19 | |
| Arterial blood pressure and heart rate | ||||
| Systolic (mmHg) | 136.2 ± 12.8 | 126.8 ± 12.7 | 0.11 | |
| Diastolic (mmHg) | 98.6 ± 7.3 | 87.6 ± 5.6 | 0.004 | |
| HR (beats/min) | 314 ± 61 | 302 ± 20 | 0.31 | |
| Echocardiographic parameters | ||||
| LVEDV (mL) | 0.37 ± 0.19 | 0.52 ± 0.20 | 0.11 | |
| LVESV (mL) | 0.14 ± 0.08 | 0.15 ± 0.1 | 0.41 | |
| IVSs (cm) | 0.41 ± 0.01 | 0.35 ± 0.09 | 0.21 | |
| IVSd (cm) | 0.29 ± 0.05 | 0.27 ± 0.07 | 0.28 | |
| SV (mL) | 0.24 ± 0.12 | 0.36 ± 0.12 | 0.053 | |
| EF (%) | 64 ± 8.5 | 71 ± 6.1 | 0.06 | |
| Left ventricle hemodynamic parameters (direct measurements) | ||||
| LVEDP (mmHg) | 3.10 ± 0.78 | 3.41 ± 1.95 | 0.87 | |
| dP/dt (mmHg/ms) | 5.88 ± 0.92 | 5.50 ± 0.98 | 0.41 | |
| -dP/dt (mmHg/ms) | 2.35 ± 0.28 | 2.55 ± 0.67 | 0.27 | |
| Plasma NT-proBNP | ||||
| NT-proBNP (pg/mL) | 52.26 ± 0.15.0 | 42.80 ± 9.5 | 0.09 | |
| Electrolyte balance | ||||
| Serum sodium (mmol/L) | 142.4 ± 3.31 | 138.9 ± 2.98 | 0.04 | |
| 24 hr sodium urine excretion (mmoles) | 1.42 ± 0.28 | 1.93 ± 0.33 | 0.005 | |
| Serum potassium (mmol/L) | 4.73 ± 0.33 | 4.49 ± 0.28 | 0.09 | |
| 24 hr potassium urine excretion (mmoles) | 2.89 ± 0.42 | 3.40 ± 0.54 | 0.04 | |
| Serum creatinine clearance (mL/min) | 0.42 ± 0.17 | 0.53 ± 0.05 | 0.06 | |
| Hormones | ||||
| Angiotensin II (pg/mL) | 325.7 ± 39.8 | 276.7 ± 38.3 | 0.02 | |
| Aldosterone (pg/mL) | 816.8 ± 300.4 | 758.4 ± 142.8 | 0.32 | |
| Vasopressin (ng/mL) | 3.02 ± 1.24 | 3.11 ± 1.03 | 0.45 | |
| Cytokines | ||||
| TNF-a (pg/mL) | 34.56 ± 24.69 | 24.98 ± 7.92 | 0.19 | |
| IL-10 (pg/mL) | 15.91 ± 4.66 | 28.17 ± 14.39 | 0.036 | |
-
Table 2—source data 1
Metabolic, renal and cardiovascular parameters in SHHF rats.
- https://cdn.elifesciences.org/articles/57028/elife-57028-table2-data1-v2.xlsx
Metabolic, renal and cardiovascular parameters in 58-week-old SD-ISO rats.
Sprague-Dawley rats treated with isoprenaline at the age of 56 weeks. Rats maintained on either water (ISO-control, n = 5–9) or TMAO solution (ISO-TMAO, n = 7–10). T1 - metabolic and echocardiographic measurements, T2 - echocardiographic measurements, T3 - metabolic, echocardiographic and direct hemodynamic measurements (see also the study design, Figure 1). Creatinine clearance calculated as urine creatinine x urine output (ml/min)/plasma creatinine. LVEDV - left ventricle end diastolic volume, LVESV - left ventricle end systolic volume, SV – stroke volume, EF - ejection fraction, IVSs(d), intraventricular septum diameter during systole and diastole, respectively. LVEDP - pressure in the left ventricle during the end of diastole measured directly with a catheter, +dP/dt - maximal slope of systolic ventricular pressure increment, - dP/dt - maximal slope of diastolic ventricular pressure decrement. Values are means,± SD. P values by t-test or Mann-Whitney U test, except # - by log-rank test.
| Group/ Parameter | ISO-control | ISO-TMAO | P | |
|---|---|---|---|---|
| Survival, Energy and water balance | ||||
| Survival from the study onset (%, n) | 90% (9/10) | 100% (10/10) | 0.32# | |
| Body mass (g) | T1 | 434.44 ± 22.93 | 432.69 ± 37.59 | 0.45 |
| T3 | 438.67 ± 23.99 | 428.71 ± 37.14 | 0.25 | |
| 24 hr food intake (g) | T1 | 21.54 ± 1.43 | 21.84 ± 1.94 | 0.51 |
| T3 | 21.24 ± 2.44 | 22.08 ± 1.91 | 0.21 | |
| 24 hr water intake (mL) | T1 | 34.99 ± 3.10 | 34.82 ± 3.89 | 0.53 |
| T3 | 34.77 ± 5.47 | 35.59 ± 1.92 | 0.34 | |
| 24 hr urine output (g) | T1 | 19.28 ± 2.73 | 20.03 ± 4.42 | 0.34 |
| T3 | 19.68 ± 4.29 | 20.23 ± 2.6 | 0.37 | |
| Tibia length (cm) | T3 | 4.33 ± 0.06 | 4.34 ± 0.1 | 0.35 |
| TMAO | ||||
| Plasma TMAO (µmol/L) | T3 | 5.95 ± 2.35 | 32.51 ± 11.43 | <0.001 |
| 24 hr TMAO urine excretion (µmoles) | T3 | 5.74 ± 1.64 | 119.12 ± 65.96 | <0.001 |
| Heart mass | ||||
| Heart mass (g) | T3 | 1.46 ± 0.13 | 1.43 ± 0.19 | 0.39 |
| Arterial blood pressure and heart rate | ||||
| Systolic (mmHg) | T3 | 142.48 ± 10.63 | 130.92 ± 11.67 | 0.026 |
| Diastolic (mmHg) | T3 | 97.10 ± 9.95 | 87.59 ± 10.47 | 0.037 |
| HR (beats/min) | T3 | 356.56 ± 23.39 | 344.19 ± 49.69 | 0.26 |
| Echocardiographic parameters | ||||
| LVEDV (mL) | T1 | 0.44 ± 0.22 | 0.33 ± 0.17 | 0.15 |
| T2 | 0.54 ± 0.22 | 0.53 ± 0.23 | 0.47 | |
| T3 | 0.51 ± 0.11 | 0.53 ± 0.34 | 0.21 | |
| LVESV (mL) | T1 | 0.13 ± 0.09 | 0.11 ± 0.07 | 0.31 |
| T2 | 0.22 ± 0.13 | 0.13 ± 0.05 | 0.054 | |
| T3 | 0.12 ± 0.05 | 0.08 ± 0.02 | 0.01 | |
| IVSs (cm) | T1 | 0.33 ± 0.03 | 0.32 ± 0.05 | 0.29 |
| T2 | 0.33 ± 0.06 | 0.35 ± 0.09 | 0.29 | |
| T3 | 0.36 ± 0.04 | 0.32 ± 0.05 | 0.07 | |
| IVSd (cm) | T1 | 0.24 ± 0.04 | 0.23 ± 0.04 | 0.28 |
| T2 | 0.26 ± 0.03 | 0.25 ± 0.04 | 0.41 | |
| T3 | 0.24 ± 0.04 | 0.25 ± 0.03 | 0.31 | |
| SV (mL) | T1 | 0.29 ± 0.15 | 0.26 ± 0.12 | 0.40 |
| T2 | 0.35 ± 0.17 | 0.47 ± 0.22 | 0.09 | |
| T3 | 0.38 ± 0.09 | 0.34 ± 0.07 | 0.69 | |
| EF (%) | T1 | 71.22 ± 6.12 | 72.11 ± 14.51 | 0.45 |
| T2 | 73.11 ± 8.88 | 70.11 ± 12.84 | 0.28 | |
| T3 | 78.67 ± 7.18 | 77.89 ± 11.94 | 0.43 | |
| Left ventricle hemodynamic parameters (direct measurements) | ||||
| LVEDP (mmHg) | T3 | 6.73 ± 2.55 | 4.46 ± 0.74 | 0.03 |
| dP/dt (mmHg/ms) | T3 | 9.20 ± 1.54 | 6.55 ± 1.18 | 0.004 |
| -dP/dt (mmHg/ms) | T3 | 5.19 ± 0.38 | 4.76 ± 0.65 | 0.09 |
| Plasma NT-proBNP | ||||
| NT-proBNP (pg/mL) | T3 | 64.49 ± 43.59 | 22.01 ± 22.83 | 0.02 |
| Electrolyte balance | ||||
| Serum sodium (mmol/L) | T3 | 136.88 ± 3.56 | 137.89 ± 1.62 | 0.23 |
| 24 hr sodium urine excretion (mmoles) | T3 | 1.93 ± 0.27 | 2.09 ± 0.42 | 0.18 |
| Serum potassium (mmol/L) | T3 | 5.53 ± 0.89 | 5.02 ± 0.77 | 0.11 |
| 24 hr potassium urine excretion (mmoles) | T3 | 2.56 ± 0.35 | 2.92 ± 0.35 | 0.03 |
| Serum creatinine clearance (mL/min) | T3 | 1.26 ± 0.23 | 1.24 ± 0.26 | 0.43 |
| Hormones | ||||
| Angiotensin II (pg/mL) | T3 | 286.4 ± 24.4 | 272.6 ± 39.5 | 0.35 |
| Aldosterone (pg/mL) | T3 | 938.6 ± 114.6 | 1032.4 ± 120.6 | 0.07 |
| Vasopressin (ng/mL) | T3 | 0.98 ± 0.55 | 1.28 ± 0.66 | 0.18 |
-
Table 3—source data 1
Metabolic, renal and cardiovascular parameters in SD-ISO rats.
- https://cdn.elifesciences.org/articles/57028/elife-57028-table3-data1-v2.xlsx
| Reagent type (species) or resource | Designation | Source or reference | Identifiers | Additional information |
|---|---|---|---|---|
| Strain, strain background (Rattus norvegicus, male) | SHHF/MccGmiCrl-Leprcp/Crl | Charles River Laboratories (USA) | RRID:RGD_2313221 | |
| Commercial assay or kit | NT-proBNP | FineTest | cat. no. ER0309 | |
| Commercial assay or kit | aldosterone | Cayman Chemicals | cat. no. 501090 | |
| Commercial assay or kit | vasopressin | Biorbyt | cat. no. orb410987 | |
| Commercial assay or kit | angiotensin II | FineTest | cat. no. ER1637 | |
| Commercial assay or kit | TNFα | R and D System | cat. no. RTA00 | |
| Commercial assay or kit | IL-10 | R and D System | cat. no. R1000 | |
| Commercial assay or kit | angiotensinogen | Bio-Rad | Unique Assay ID: qRnoCED0051666 | |
| Commercial assay or kit | angiotensin II receptor type 1a | Bio-Rad | Unique Assay ID: qRnoCID0052626 | |
| Commercial assay or kit | angiotensin II receptor type 1b | Bio-Rad | Unique Assay ID: qRnoCED0005729 | |
| Commercial assay or kit | angiotensin II receptor type 2 | Bio-Rad | Unique Assay ID: qRnoCED0007551 | |
| Commercial assay or kit | transforming growth factor-beta | Bio-Rad | Unique Assay ID: qRnoCED0007638 | |
| Commercial assay or kit | renin | Bio-Rad | Unique Assay ID: qRnoCID0008721 | |
| Commercial assay or kit | metalloproteinase inhibitor 2 | Bio-Rad | Unique Assay ID: qRnoCID0001559 | |
| Commercial assay or kit | Beta-actin | Bio-Rad | Unique Assay ID: qRnoCED0018219 | |
| Commercial assay or kit | TMAO | abcr GmbH | cat. no. AB 109058 | |
| Commercial assay or kit | isoprenaline hydrochloride | Sigma-Aldrich | cat. no. I5627 | |
| Software, algorithm | CFX Manager | Bio-Rad | RRID:SCR_017251 | |
| Software, algorithm | SymPhoTime 64 | PicoQuant | RRID:SCR_016263 | |
| Software, algorithm | AcqKnowledge Software | Biopac Systems, Inc | RRID:SCR_014279 |