Spreading of a mycobacterial cell-surface lipid into host epithelial membranes promotes infectivity
Figures
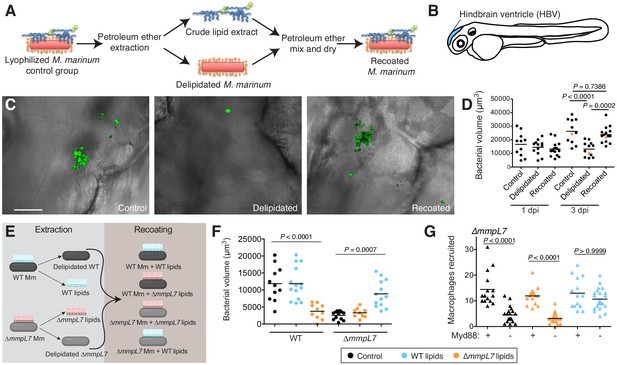
Lipid removal and recoating reveals that pre-infection PDIM reservoirs are required for M.marinum infection of zebrafish.
(A) Model of lipid removal and recoating of M. marinum. (B) Model of zebrafish larva showing the hindbrain ventricle (HBV) injection site. (C) Representative images of the experiment in D (orange dots), wasabi (green) fluorescent protein expressing M. marinum in the HBV at 3 dpi are shown, scale bar = 50 μm. (D) Mean bacterial volume after HBV infection of wildtype fish with ~100 control, delipidated, or recoated M. marinum. (E) Model of lipid-swap experiment. (F) Mean bacterial volume at 3 dpi after HBV infection of wildtype fish with ~100 WT or ∆mmpL7 M. marinum treated as follows: non-extracted control (black), extracted and recoated with WT lipids (blue), or extracted and recoated with ∆mmpL7 lipids (orange). (G) Mean macrophage recruitment at 3 hpi of the HBV of wildtype or Myd88-depleted fish with ~100 ∆mmpL7 M. marinum as treated in F. (D), (F), and (G) representative of at least three separate experiments. Ordinary one-way ANOVA with (D) Sidak's multiple comparisons test for the comparison’s shown and (F) Tukey’s multiple comparisons test with selected adjusted P values shown. (G) Kruskal-Wallis ANOVA for unequal variances with Dunn’s multiple comparisons test with selected adjusted P values shown.
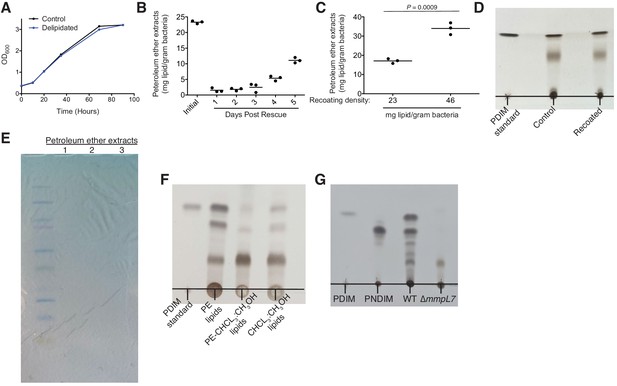
Optimization of petroleum ether extraction and recoating.
(A) Mean OD600 values of control or delipidated M. marinum recovered in complete growth medium. (B) Mean mg of lipid extracted per gram of dry bacteria from initial bacteria and from delipidated bacteria following designated recovery time in culture. (C) Mean mg of lipid extracted per gram of dry bacteria following recoating with designated lipid densities. Two-tailed, unpaired t test. (D) Thin-layer chromatography (TLC) showing no difference between extracts from untreated or recoated bacteria. PDIM standard, petroleum ether extracts from untreated bacteria, and petroleum ether extracts from bacteria that were recoated with lipids from petroleum ether extracts (recoated). TLC was ran once in 98:2 petroleum ether:ethyl acetate. (E) 1 mg of extract from three separate petroleum ether extracts was dissolved in DMSO and separated by SDS-PAGE and stained with Colloidal Coomassie Brilliant Blue for 24 hr. Ladder is precision plus protein kaleidoscope. (F) TLC showing PDIM’s complete removal following petroleum ether extraction. PDIM standard, petroleum ether extract (PE lipids), chloroform:methanol extract of the bacterial pellet previously extracted with petroleum ether (PE-CHCL3:CH3OH lipids), and chloroform:methanol extract of a fresh bacterial pellet (CHCL3:CH3OH lipids). TLC was ran twice in 98:2 petroleum ether:ethyl acetate. (G) TLC of PDIM standard, PNDIM standard, and petroleum ether extracts of wildtype (WT) and ∆mmpL7 M. marinum. TLC was ran twice in 98:2 petroleum ether:ethyl acetate. (A)-(D) and (F)-(G) representative of three separate experiments.
-
Figure 1—figure supplement 1—source data 1
- https://cdn.elifesciences.org/articles/60648/elife-60648-fig1-figsupp1-data1-v2.xlsx
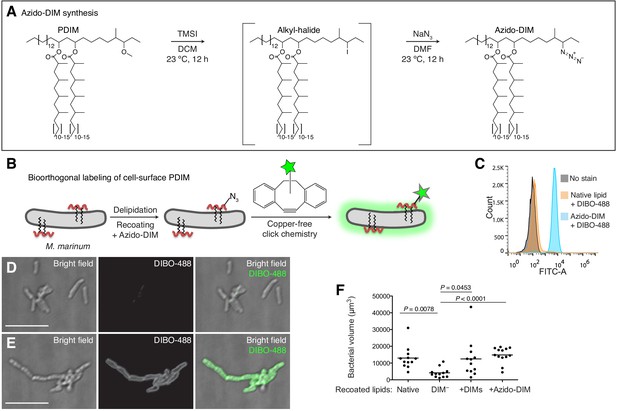
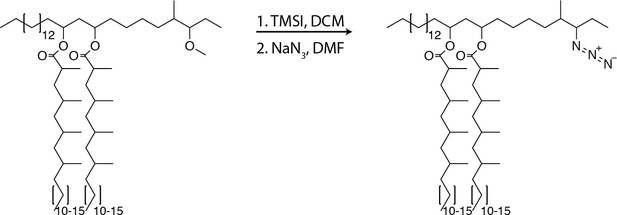
Synthesis and application of a chemically tractable, biologically active PDIM variant, azido-DIM.
(A) Synthesis of azido-DIM. (B) Model of delipidation and recoating of bacteria with or without azido-DIM followed by treatment with an azide-reactive cyclooctyne, DIBO-488. (C) Flow cytometry analysis of M. marinum recoated with or without azido-DIM treated with DIBO-488. Image of (D) native lipid control or (E) azido-DIM recoated bacteria treated with DIBO-488, scale bar = 8 μm. (F) Mean bacterial volume 3 days following HBV infection of wildtype fish with ~100 delipidated M. marinum recoated with Native, DIM-depleted (DIM–), DIM– plus native DIMs (+DIMs), or DIM– plus azido-DIM (+Azido-DIM) lipids. Kruskal-Wallis ANOVA for unequal variances with Dunn’s multiple comparisons test with selected adjusted P values shown. (C), (F) representative of three separate experiments.

Thin-layer chromatography of Native, DIM-depleted (DIM—), DIM— plus native DIMs (+DIMs), or DIM— plus azido-DIM (+Azido-DIM) lipids prior to recoating onto delipidated bacteria.
TLC was ran once in 98:2 petroleum ether:ethyl acetate. Representative of three separate experiments.
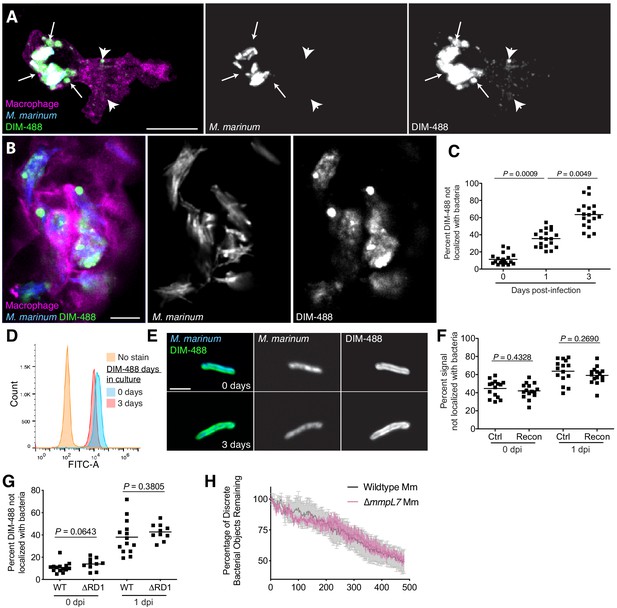
PDIM spreads into macrophage membranes.
Images of M. marinum expressing a cytosolic blue-fluorescent protein recoated with DIBO-488 labeled azido-DIM (DIM-488) at (A) 3 hpi and (B) 3 dpi of ~100 M. marinum in the HBV of transgenic fish whose macrophages express a fluorescent protein. Scale bar = 10 μm. Arrows, DIM-488 spread in vicinity of infecting bacteria, arrowheads, DIM-488 spread throughout macrophage. (C) Mean percent DIM-488 not localized with bacteria following HBV infection of wildtype fish with ~100 M. marinum. Kruskal-Wallis ANOVA for unequal variances with Dunn’s multiple comparisons test with selected adjusted P values shown. (D) Flow cytometry analysis of M. marinum expressing a cytosolic blue-fluorescent protein recoated with DIM-488 following 0 or 3 days in culture. Representative of two separate experiments. (E) Representative images of bacteria from D, scale bar = 3 μm. (F) Mean percent fluorescent signal not localized with bacteria following HBV infection of wildtype fish with ~100 control or recoated M. marinum labeled with periodate-hydroxylamine chemistry. Two-tailed, unpaired t test. (G) Mean percent DIM-488 not localized with bacteria following HBV infection of wildtype fish with ~100 wildtype or ∆RD1 M. marinum. Two-tailed Mann Whitney test for 0 dpi and two-tailed, unpaired t test for one dpi (H) Mean (+/- SEM) percentage of discrete bacterial objects remaining following HBV infection of wildtype fish with ~100 wildtype or ∆mmpL7 M. marinum. Representative of two separate experiments. (C), (F) and (G) representative of three separate experiments.
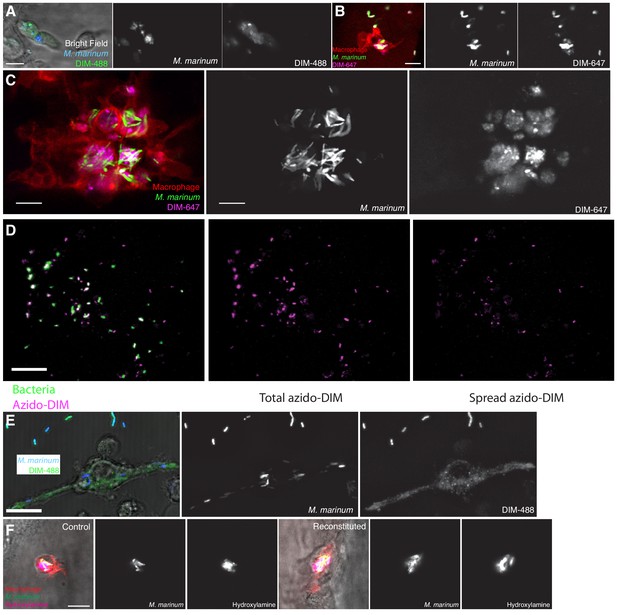
Further characterization of PDIM spread.
(A) Image of M. marinum expressing a cytosolic blue-fluorescent protein recoated with DIBO-488 (green) labeled azido-DIM at 3hpi of ~100 M. marinum in the HBV of wildtype fish. Scale bar = 10 μm. Images of M. marinum expressing a cytosolic wasabi fluorescent protein recoated with DIBO-647 labeled azido-DIM (DIM-647) at (B) 3 hpi and (C) 3 dpi of ~100 M. marinum in the HBV of transgenic fish whose macrophages express the red-fluorescent protein tdTomato. Scale bar = 10 μm. (D) Example of calculating percent spread: A surface rendering of the cytosolic expressing protein of the fluorescent bacteria (green) is subtracted from the total azido-DIM signal (magenta) to give the spread azido-DIM signal. The volume of the spread azido-DIM signal is then divided by the volume of the total azido-DIM signal to calculate percent spread. Scale bar = 50 μm. (E) Image of M. marinum expressing a blue-fluorescent protein recoated with DIM-488 24 hpi of THP-1 macro- phages, MOI = 5, and scale bar = 10 μm. (F) Images of control or recoated M. marinum expressing a cytosolic green-fluorescent protein labeled with periodate-hydroxylamine chemistry at 3 hpi of ~100 M. marinum into the HBV of transgenic fish whose macrophages express a fluorescent protein, scale bar = 10 μm.
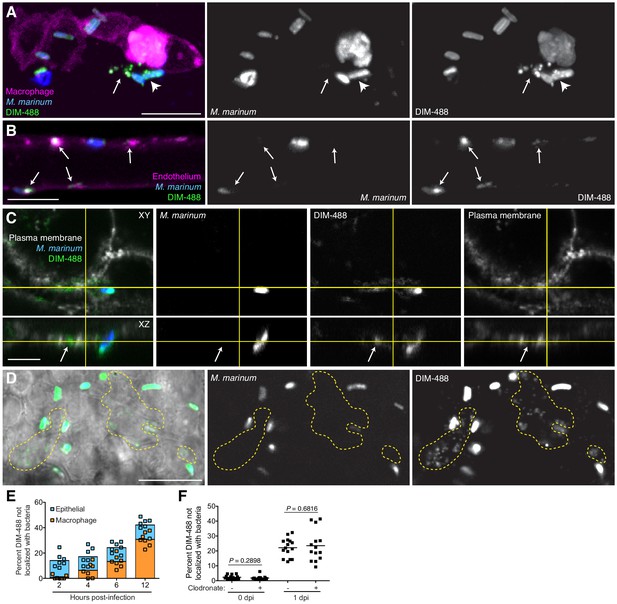
PDIM spreads into epithelial membranes.
(A) Image of M. marinum expressing a cytosolic blue-fluorescent protein recoated with DIBO-488 labeled azido-DIM (DIM-488) highlighting DIM-488 spread from bacteria (arrowhead) to epithelial cells (arrows) at 3 hpi of ~100 M. marinum in the HBV, scale bar = 10 μm. (B) Image of DIM-488 labeled M. marinum at 1 day post-intravenous infection of transgenic fish whose endothelium express a red-fluorescent protein. Arrows, DIM-488 spread onto endothelium, scale bar = 5 μm. (C) Image of A549 epithelial cells whose plasma membranes are labeled with Alexa-fluor 594 wheat germ agglutinin at one day post infection with DIM-488 labeled M. marinum at an MOI of 5. Arrows, DIM-488 spread into plasma membrane, scale bar = 5 μm. (D) Image highlighting DIM-488 spread onto epithelial surfaces (yellow-dashed outline) at 2 hpi of ~100 M. marinum in the HBV, scale bar = 10 μm. (E) Mean percent DIM-488 in macrophage or epithelial cells not localized with bacteria following HBV infection with ~100 M. marinum. Representative of two separate experiments. (F) Mean percent DIM-488 not localized with bacteria following HBV infection of lipo-PBS or lipo-clodronate treated fish with ~100 M. marinum. Two-tailed Mann Whitney test for 0 dpi and two-tailed, unpaired t test for one dpi. Representative of three separate experiments.
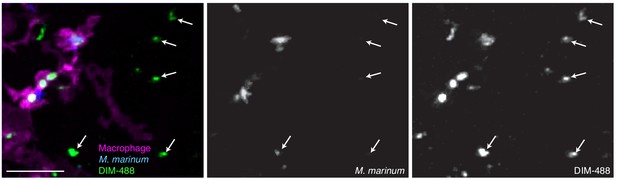
Image of M. marinum expressing a cytosolic blue-fluorescent protein recoated with DIBO-488 labeled azido-DIM (DIM-488) highlighting DIM-488 spread on epithelial cells (arrows) at 24 hpi of ~100 M. marinum in the HBV of transgenic fish whose macrophages express a fluorescent protein, scale bar = 30 μm.
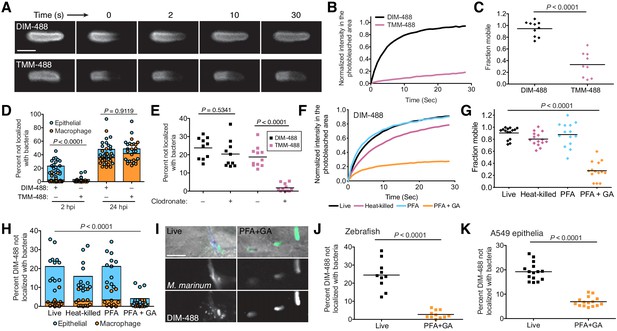
PDIM’s mobility promotes spread into epithelial cell membranes.
(A) Representative FRAP images of DIM-488 and TMM-488 labeled M. marinum, scale bar = 2 μm. (B) Fluorescent recovery curves after photobleaching of DIM-488 or TMM-488 labeled M. marinum, lines represent the average signal from n = 10 cells. (C) Mean fraction mobile which is the plateau following fitting of data generated in B to a non-linear regression with a one-phase association. (D) Mean percent DIM-488 or TMM-488 in macrophage or epithelial cells not localized with bacteria following HBV infection with ~100 M. marinum. (E) Mean percent DIM-488 or TMM-488 not localized with bacteria 24 hr following HBV infection of lipo-PBS or lipo-clodronate treated fish with ~100 M. marinum. (F) Fluorescent recovery curves after photobleaching of live, heat-killed, 4% paraformaldehyde (PFA) fixed, or 4% paraformaldehyde plus 1% glutaraldehyde (PFA+GA) fixed DIM-488 labeled M. marinum, lines represent the average signal from n = 14–15 cells. (G) Mean fraction mobile which is the plateau following fitting of data generated in F to a non-linear regression with a one-phase association. (H) Mean percent DIM-488 in macrophage or epithelial cells not localized with bacteria 2 hr following HBV infection with ~100 M. marinum treated as in F. (I) Images of live or PFA+GA treated DIM-488 labeled M. marinum at 2 hpi of the HBV with ~100 bacteria, scale bar = 5 μm. Mean percent DIM-488 not localized with bacteria 24 hr following (J) infection of lipo-clodronate treated fish or (K) A549 epithelial cells with live or PFA+GA fixed DIM-488 labeled M. marinum. (C), (J), and (K) two-tailed, unpaired t test. (E), (G), and (H) ordinary one-way ANOVA with Tukey’s multiple comparisons test with selected adjusted P values shown. (B)-(H) and (J)-(K) representative of three separate experiments.
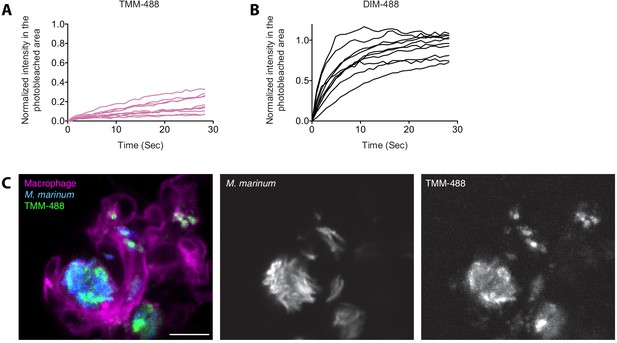
Individual FRAP curves of (A) TMM-488 and (B) DIM-488 labeled bacteria.
(C) Image of M. marinum expressing a cytosolic blue-fluorescent protein metabolically labeled with 6-azido trehalose followed by labeling with DIBO-488 (TMM-488) highlighting TMM-488 spread across macrophage membranes at 24 hpi of ~100 M. marinum in the HBV of transgenic fish whose macrophages express a fluorescent protein.
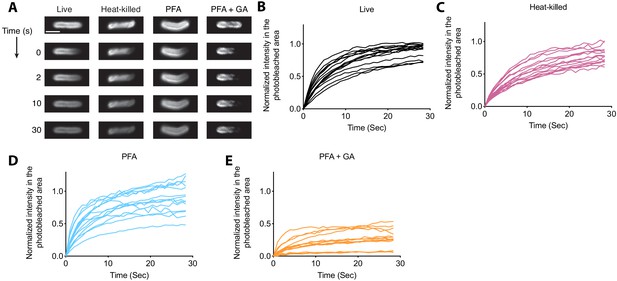
FRAP analysis of DIM-488 labeled bacteria.
(A) Representative FRAP images of live, heat-killed, PFA, and PFA+GA treated DIM-488 labeled M. marinum, scale bar = 2 μm. (B)-(E) Individual fluorescent recovery curves of bacteria described in A. Representative of three separate experiments.
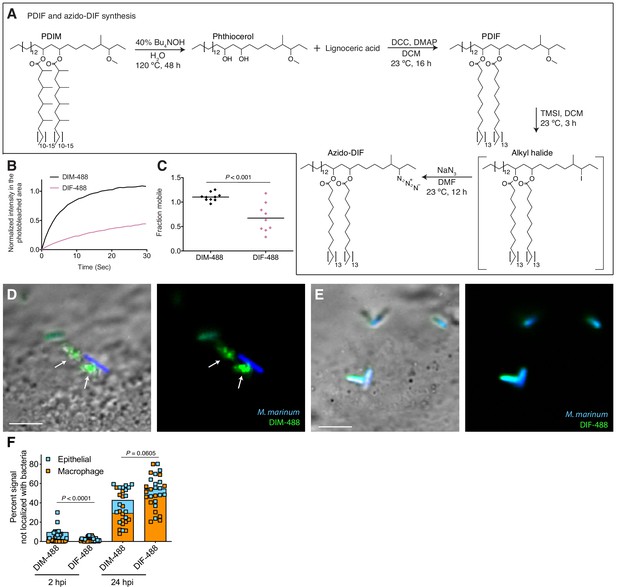
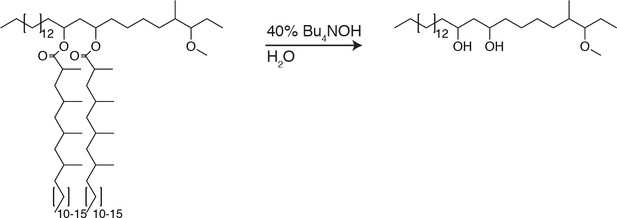
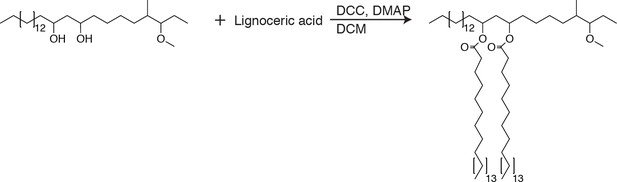
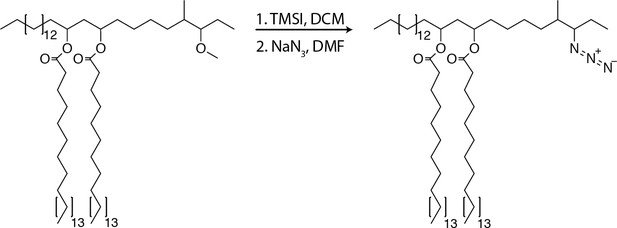
PDIM’s methyl-branched mycocerosic acids promote mobility and spreading.
(A) Phthiocerol di-fatty acid (PDIF) and azido-DIF synthesis. (B) Fluorescent recovery curves after photobleaching of DIM-488 or DIF-488 labeled M. marinum, lines represent the average signal from n = 9–10 cells. (C) Mean fraction mobile which is the plateau following fitting of data generated in B to a non-linear regression with a one-phase association. Two-tailed, unpaired t test. Images of M. marinum expressing a blue-fluorescent protein recoated with (D) DIM-488 or (E) DIF-488 at 2 hpi into the HBV of wildtype fish, arrows indicate spread signal, scale bar = 5 μm. (F) Mean percent DIM-488 or DIF-488 in macrophage or epithelial cells no longer localized with bacteria following HBV infection with ~100 M. marinum. Two-tailed Mann Whitney test for 2 hpi and two-tailed, unpaired t test for 24 hpi. (B), (C), and (F) representative of three separate experiments.
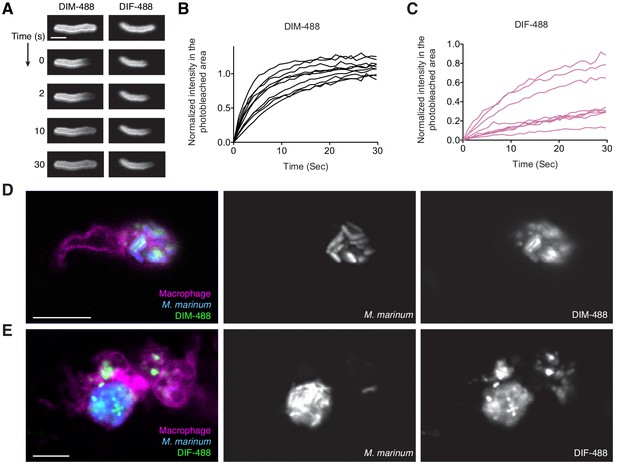
Analysis of DIM-488 and DIF-488 labeled M.marinum.
(A) Representative FRAP images of DIM-488 and DIF-488 labeled M. marinum, scale bar = 2 μm. Individual fluorescent recovery curves of (B) DIM-488 and (C) DIF-488 labeled M. marinum. Images of M. marinum expressing a cytosolic blue-fluorescent protein recoated with green (D) DIM-488 or (E) DIF-488 at 24 hpi of ~100 M. marinum in the HBV of transgenic fish whose macrophages express a red-fluorescent protein. Scale bar = 10 μm. (B) and (C) representative of three separate experiments.
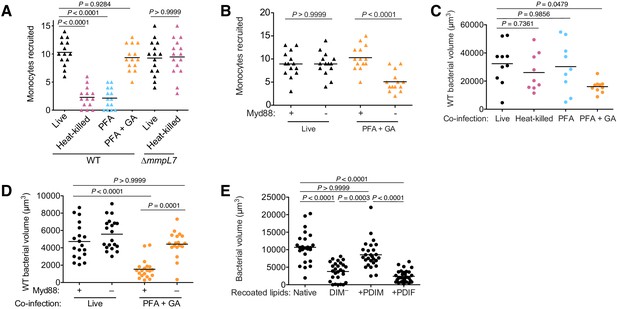
PDIM spreading into epithelial membranes is required to evade microbicidal monocytes.
(A) Mean monocyte recruitment at 3 hpi of the HBV with ~100 live, heat-killed, PFA treated, or PFA+GA treated wildtype (WT) or ~100 live or heat-killed ∆mmpL7 M. marinum. (B) Mean monocyte recruitment at 3 hpi of the HBV of wildtype or Myd88-depleted fish with ~100 live or PFA+GA treated wildtype M. marinum. Mean volume of wildtype M. marinum following co-infection with (C) wildtype M. marinum treated as in A or (D) live or PFA+GA treated wildtype M. marinum in wildtype or Myd88-depleted fish. (E) Mean bacterial volume 3 days following HBV infection of wildtype fish with ~100 M. marinum recoated with Native, DIM-depleted (DIM–), DIM– plus PDIM (+PDIM), or DIM– plus PDIF (+PDIF) lipids. (A)-(C) ordinary one-way ANOVA with Tukey’s multiple comparisons test with selected adjusted P values shown. (D) and (E) Kruskal-Wallis ANOVA for unequal variances with Dunn’s multiple comparisons test with selected adjusted P values shown. (A)-(E) representative of three separate experiments.
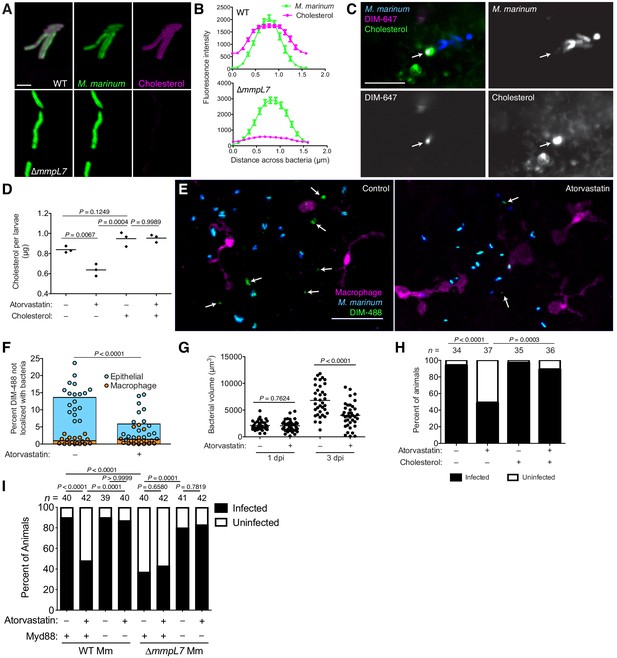
Host cholesterol promotes PDIM spread and mycobacterial infectivity.
(A) Images of wildtype (WT) and ∆mmpL7 M. marinum expressing a green-fluorescent protein following 48 hr incubation with alkyne-cholesterol reacted with AlexaFlour647 Azide, scale bar = 3 μm. (B) Mean (± SEM) fluorescent intensity of line profiles drawn perpendicular to poles of WT and ∆mmpL7 M. marinum labeled as in A. (C) Image of A549 epithelial cells labeled with BODIPY-cholesterol at 1 dpi with DIM-647 labeled M. marinum at an MOI of 5. Arrows, spread DIM-647 co-localizing with BODIPY-cholesterol, scale bar = 10 μm. (D) Mean cholesterol content of 3 dpf zebrafish following a 24 hr treatment with atorvastatin, water-soluble cholesterol, or both. Ordinary one-way ANOVA with Tukey’s multiple comparisons test with selected adjusted P values shown. (E) Images of control or atorvastatin treated transgenic fish whose macrophages express tdTomato at 2 hpi with ~100 M. marinum expressing a cytosolic blue-fluorescent protein recoated with DIBO-488 labeled azido-DIM (DIM-488), scale bar = 40 μm. Arrows, DIM-488 spread onto epithelial cells. (F) Mean percent DIM-488 in macrophage or epithelial cells not localized with bacteria at 2 h following HBV infection with ~100 M. marinum in control or atorvastatin treated fish. Two-tailed, unpaired t test. (G) Mean bacterial volume following HBV infection of control or atorvastatin treated fish with ~100 M. marinum. Two-tailed Mann Whitney test for 2 hpi and two-tailed, unpaired t test for 24 hpi. (H) Percentage of infected or uninfected fish at 3 dpi into the HBV with 1–3 wildtype M. marinum with or without atorvastatin and water-soluble cholesterol. (I) Percentage of infected or uninfected wildtype or Myd88-depleted fish at 3 dpi into the HBV with 1–3 wildtype or ∆mmpL7 M. marinum with or without atorvastatin. (H) and (I) Fisher’s exact test with Bonferroni’s correction for multiple comparisons. (B)-(D) and (F)-(I) representative of three separate experiments.
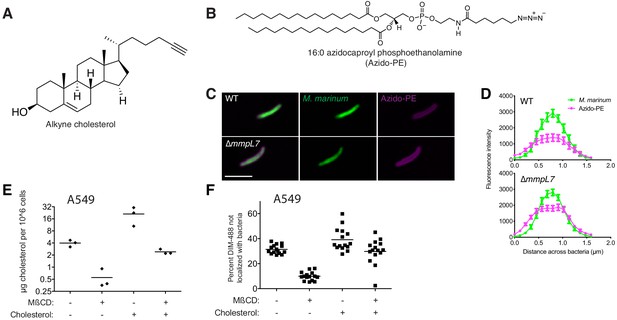
Cholesterol mediates DIM-488 spread on A549 epithelial cells.
(A) Structure of alkyne-cholesterol. (B) Structure of 16:0 azidocaproyl phosphoethanolamine (Azido-PE). (C) Images of wildtype (WT) and ∆mmpL7 M. marinum expressing a green-fluorescent protein following 48 h incubation with azido-PE reacted with DIBO-647, scale bar = 3 μm. (D) Mean (± SEM) fluorescent intensity of line profiles drawn perpendicular to poles of WT and ∆mmpL7 M. marinum labeled as in C. Representative of two separate experiments. (E) Mean cholesterol content of A549 cells treated with methyl-ß cyclodextrin (MßCD), water-soluble cholesterol, or both. (F) Mean percent DIM-488 spread at 24 hpi of A549 epithelial cells treated with MßCD, water-soluble cholesterol, or both, MOI = 5. (E) and (F) representative of three separate experiments.
-
Figure 8—figure supplement 1—source data 1
- https://cdn.elifesciences.org/articles/60648/elife-60648-fig8-figsupp1-data1-v2.xlsx
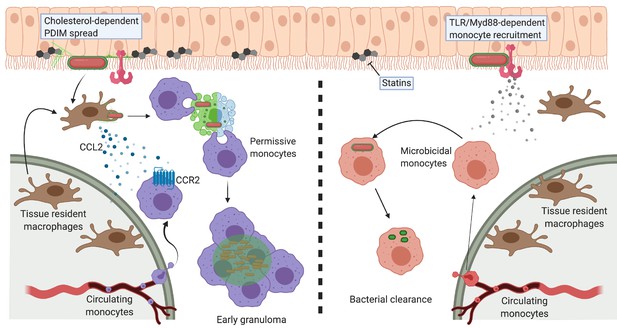
Model of PDIM spreading to promote virulence in the zebrafish hindbrain ventricle.
Cholesterol-dependent PDIM spread into epithelial membranes prevents TLR/Myd88 detection at the site of infection. Bacteria then drive resident macrophages to produce CCL2 to recruit CCR2-positive permissive monocytes which go on to form early granulomas. PDIM continues to spread into host membranes throughout this process. In low cholesterol settings (statin treatment) PDIM does not spread as readily into epithelial membranes. TLR/Myd88-dependent recruitment of microbicidal monocytes occurs, which can then clear infecting mycobacteria. Figure created with BioRender.com.
Videos
PDIM dynamics.
Real-time video of M. marinum expressing blue-fluorescent protein recoated with DIBO-488 labeled azido-DIM at 3 hpi of the HBV with ~100 M. marinum.
Tables
| Reagent type (species) or resource | Designation | Source or reference | Identifiers | Additional information |
|---|---|---|---|---|
| Strain, strain background (Mycobacterium marinum M strain) | wildtype M. marinum | Takaki et al., 2013. | derivatives of ATCC #BAA-535 | Fluorescent strains: pMS12:tdTomato, pMS12:EBFP2, or pMS12:wasabi |
| Strain, strain background (M. marinum M strain) | ∆mmpL7 M. marinum | Cambier et al., 2014b. | NA | Fluorescent strains: pMS12:tdTomato, pMS12:wasabi |
| Strain, strain background (Escherichia coli) | Top10 | Urbanek et al., 2014. | NA | Transformed with pBAD:tret from Thermoproteus tenax |
| Genetic reagent (Zebrafish, Danio rerio) | Wildtype AB | Stanford University | ZFIN ID: ZDB-GENO -960809–7 | |
| Genetic reagent (Zebrafish, Danio rerio) | Tg(mfap4:tdTomato) | Walton et al., 2015. | ZFIN ID: ZDB-TGCONSTRCT -160122–3 | |
| Genetic reagent (Zebrafish, Danio rerio) | Tg(flk1:mcherry) | Wang et al., 2010. | ZFIN ID: ZDB-TGCONSTRCT -110127–23 | |
| Chemical compound, drug | Iodotrimethylsilane | ACROS | Cat#10530964 CAS:16029-98-4 | |
| Chemical compound, drug | Sodium azide | Sigma-Aldrich | Cat#S8032 CAS: 26628-22-8 | |
| Chemical compound, drug | Tetrabutylammonium hydroxide solution | Sigma-Aldrich | Cat#178780 CAS: 2052-49-5 | |
| Chemical compound, drug | Lignoceric acid | Sigma-Aldrich | Cat# L6641 CAS: 557-59-5 | |
| Chemical compound, drug | Click-IT Alexa Fluor 488 DIBO Alkyne | Thermo-Fisher | Cat# C10405 Discontinued* | |
| Chemical compound, drug | Click-IT Alexa Fluor 647 DIBO Alkyne | Thermo-Fisher | Cat# C10408 Discontinued* | |
| Chemical compound, drug | Alexa Fluor 647 Azide | Thermo-Fisher | Cat# A10277 | |
| Chemical compound, drug | Alkyne-Cholesterol | Click Chemistry Tools | Cat# 1409 | |
| Chemical compound, drug | 6-azido-6-deoxy-glucose | Sigma-Aldrich | Cat# 712760 CAS: 20847-05-6 | |
| Chemical compound, drug | UDP-glucose | Sigma-Aldrich | Cat# U4625 CAS: 28053-08-9 | |
| Chemical compound, drug | Alexa-647 hydroxylamine | Thermo-Fisher | Cat# A30632 | Chemical |
| Compound, drug | Methyl-ß-cyclodextrin | Sigma-Aldrich | Cat# C4555 CAS: 128446-36-6 | |
| Chemical compound, drug | Cholesterol-water soluble | Sigma-Aldrich | Cat# C4951 | |
| Chemical compound, drug | Atorvastatin | Sigma-Aldrich | Cat# PHR1422 | |
| Chemical compound, drug | Methylene blue | Sigma-Aldrich | Cat# M9140 CAS: 122965-43-9 | |
| Chemical compound, drug | Tango Buffer (10x) | Thermo-Fisher | Cat#BY5 | |
| Chemical compound, drug | Phenol Red Sodium Salt | Sigma-Aldrich | Cat#P4758 CAS: 34487-61-1 | |
| Chemical compound, drug | PMA (Phorbol 12-myristate 13-acetate) | Sigma-Aldrich | Cat#P1585 CAS:16561-29-8 | |
| Chemical compound, drug | Hygromycin B | Thermo-Fisher | Cat#10687010 | |
| Chemical compound, drug | Glutaraldehyde solution (70% in water) | Sigma-Aldrich | Cat#G7776 CAS: 111-30-8 | |
| Chemical compound, drug | 16% paraformaldehyde solution | Thermo-Fisher | Cat#28908 | Chemical |
| Compound, drug | BTTP | Click Chemistry Tools | Cat#1414 CAS: 1334179-85-9 | |
| Chemical compound, drug | 16:0 azidocaproyl phosphoethanolamine (Azido-PE) | Avanti Polar Lipids | Cat#870126 CAS: 2252461-34-8 | |
| Chemical compound, drug | BODIPY-Cholesterol | Cayman Chemical | Cat#24618 CAS: 878557-19-8 | |
| Chemical compound, drug | Alexa-Fluor 594 Wheat Germ Agglutinin | Fisher | Cat#W11262 | |
| Cell line (Homo sapiens) | A549 human alveolar epithelial cells | ATCC | CCL-185 | |
| Cell line (Homo sapiens) | THP-1 human monocytic cell line | ATCC | TIB-202 | |
| Sequence-based reagent | myD88 morpholino sequence: GTTAAACACTGACCCTGTGGATCAT | Bates et al., 2007. | ZFIN ID: ZDB-MRPHLNO -080325–4 | |
| Commercial assay or kit | Total Cholesterol and Cholesterol Ester Colorimetric/Fluorometric Assay Kit | Bio-Vision | Cat# K603 | |
| Software, algorithm | NIS-Elements | Nikon | ||
| Software, algorithm | Imaris | Bitplane | ||
| Software, algorithm | Prism | GraphPad |
Additional files
-
Supplementary file 1
Summary of P values and statistical tests.
Gaussian distribution was determined using the D'Agostino-Pearson normality test. The result of this test guided subsequent analyses.
- https://cdn.elifesciences.org/articles/60648/elife-60648-supp1-v2.xlsx
-
Supplementary file 2
NMR spectra.
- https://cdn.elifesciences.org/articles/60648/elife-60648-supp2-v2.docx
-
Transparent reporting form
- https://cdn.elifesciences.org/articles/60648/elife-60648-transrepform-v2.docx